Effects of Sedation with Medetomidine and Dexmedetomidine on Doppler Measurements of Ovarian Artery Blood Flow in Bitches
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Murrell, J.C.; Hellebrekers, L.J. Medetomidine and dexmedetomidine: A review of cardiovascular effects and antinociceptive properties in the dog. Vet. Anaesth. Analg. 2005, 32, 117–127. [Google Scholar] [CrossRef]
- Gomez-Villamandos, R.J.; Palacios, C.; Benitez, A.; Granados, M.M.; Dominguez, J.M.; Lopez, I.; Ruiz, I.; Aguilera, E.; Santisteban, J.M. Dexmedetomidine or medetomidine premedication before propofol-desflurane anaesthesia in dogs. J. Vet. Pharmacol. Ther. 2006, 29, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Vainio, O.; Ojala, M. Medetomidine, an α 2-agonist, alleviates post-thoracotomy pain in dogs. Lab. Anim. 1994, 28, 369–375. [Google Scholar] [CrossRef]
- Vaisanen, M.; Raekallio, M.; Kuusela, E.; Huttunen, P.; Leppaluoto, J.; Kirves, P.; Vainio, O. Evaluation of the perioperative stress response in dogs administered medetomidine or acepromazine as part of the preanesthetic medication. Am. J. Vet. Res. 2002, 63, 969–975. [Google Scholar] [CrossRef]
- Valtolina, C.; Robben, J.H.; Uilenreef, J.; Murrell, J.C.; Aspegrén, J.; McKusick, B.C.; Hellebrekers, L.J. Clinical evaluation of the efficacy and safety of a constant rate infusion of dexmedetomidine for postoperative pain management in dogs. Vet. Anaesth. Analg. 2009, 36, 369–383. [Google Scholar] [CrossRef]
- Salonen, S.; Vuorilehto, L.; Vainio, O.; Anttila, M. Atipamezole increases medetomidine clearance in the dog: An agonist—antagonist interaction. J. Vet. Pharmacol. Ther. 1995, 18, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Kuusela, E.; Raekallio, M.; Anttila, M.; Falck, I.; Molsa, S.; Vainio, O. Clinical effects and pharmacokinetics of medetomidine and its enantiomers in dogs. J. Vet. Pharmacol. Ther. 2000, 23, 15–20. [Google Scholar] [CrossRef]
- Kuusela, E.; Vainio, O.; Kaistinen, A.; Kobylin, S.; Raekallio, M. Sedative, analgesic, and cardiovascular effects of levomedetomidine alone and in combination with dexmedetomidine in dogs. Am. J. Vet. Res. 2001, 62, 616–621. [Google Scholar] [CrossRef]
- Peeters, M.E.; Kirpensteijn, J. Comparison of surgical variables and short-term postoperative complications in healthy dogs undergoing ovariohysterectomy or ovariectomy. J. Am. Vet. Med. Assoc. 2011, 238, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.E.; Campbell, N.B.; Posner, L.P.; Swanson, C. The hemodynamic effects of medetomidine continuous rate infusions in the dog. Vet. Anaesth. Analg. 2010, 37, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, M.D. A review of the physiological effects of alpha2-agonists related to the clinical use of medetomidine in small animal practice. Can. Vet. J. 2003, 44, 885–897. [Google Scholar]
- Miño, N.; Espino, L.; Barreiro, A. Effects of medetomidine on doppler variables of major abdominal arteries in normal dogs. Vet. Res. Commun. 2008, 32, 175–186. [Google Scholar] [CrossRef]
- Lawrence, C.J.; Prinzen, F.W.; de Lange, S. The Effect of Dexmedetomidine on Nutrient Organ Blood Flow. Anest. Analg. 1996, 83, 1160–1165. [Google Scholar] [CrossRef]
- Kellihan, H.B.; Stepien, R.L. Pulmonary Hypertension in Dogs: Diagnosis and Therapy. Vet. Clin. N. Am. Small Anim. Pract. 2010, 40, 623–641. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.; Miyamoto, A. Evaluation of ovarian blood flow by colour Doppler ultrasound: Practical use for reproductive management in the cow. Vet. J. 2009, 181, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Herzog, K.; Bollwein, H. Application of Doppler Ultrasonography in Cattle Reproduction. Reprod. Domest. Anim. 2007, 42, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Blanco, P.G.; Rodríguez, R.; Rube, A.; Arias, D.O.; Tórtora, M.; Díaz, J.D.; Gobello, C. Doppler ultrasonographic assessment of maternal and fetal blood flow in abnormal canine pregnancy. Anim. Reprod. Sci. 2011, 126, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Blanco, P.G.; Arias, D.O.; Gobello, C. Doppler Ultrasound in Canine Pregnancy. J. Ultrasound Med. 2008, 27, 1745–1750. [Google Scholar] [CrossRef]
- Koster, K.; Poulsen Nautrup, C.; Gunzel-Apel, A. A Doppler ultrasonographic study of cyclic changes of ovarian perfusion in the Beagle bitch. Reproduction 2001, 122, 453–461. [Google Scholar] [CrossRef]
- Burrow, R.; Batchelor, D.; Cripps, P. Complications observed during and after ovariohysterectomy of 142 bitches at a veterinary teaching hospital. Vet. Rec. 2005, 157, 829–833. [Google Scholar] [CrossRef]
- Raszplewicz, J.; MacFarlane, P.; West, E. Comparison of sedation scores and propofol induction doses in dogs after intramuscular premedication with butorphanol and either dexmedetomidine or medetomidine. Vet. Anaesth. Analg. 2013, 40, 584–589. [Google Scholar] [CrossRef]
- Duque, F.J.; Dominguez-Roldan, J.M.; Martinez-Taboada, F.; Macías-García, B.; Rodríguez-Medina, P.; Barrera-Chacon, R. Effects of medetomidine and medetomidine-butorphanol on transcranial color-coded duplex ultrasonography in healthy dogs. Res. Vet. Sci. 2013, 95, 648–653. [Google Scholar] [CrossRef] [PubMed]
- Niswender, G.D.; Reimers, T.J.; Diekman, M.A.; Nett, T.M. Blood Flow: A Mediator of Ovarian Function1. Biol. Reprod. 1976, 14, 64–81. [Google Scholar] [CrossRef] [PubMed]
- Keegan, R.D.; Greene, S.A.; Bagley, R.S.; Moore, M.P.; Weil, A.B.; Short, C.E. Effects of medetomidine administration on intracranial pressure and cardiovascular variables of isoflurane-anesthetized dogs. Am. J. Vet. Res. 1995, 56, 193–198. [Google Scholar] [PubMed]
- Ko, J.C.; Bailey, J.E.; Pablo, L.S.; Heaton-Jones, T.G. Comparison of sedative and cardiorespiratory effects of medetomidine and medetomidine-butorphanol combination in dogs. Am. J. Vet. Res. 1996, 57, 535–540. [Google Scholar] [CrossRef]
- Tranquilli, W.J.; Thurmon, J.C.; Grimm, K.A. Veterinary Anaesthesia and Analgesia, 4th ed.; Blackwell Publishing: Ames, IA, USA, 2007; p. 1114. [Google Scholar]
- Brown, S.; Atkins, C.; Bagley, R.; Carr, A.; Cowgill, L.; Davidson, M.; Egner, B.; Elliott, J.; Henik, R.; Labato, M.; et al. Guidelines for the Identification, Evaluation, and Management of Systemic Hypertension in Dogs and Cats. J. Vet. Intern. Med. 2007, 21, 542–558. [Google Scholar] [CrossRef]
- Chalifoux, A.; Dallaire, A.; Blais, D.; Larivière, N.; Pelletier, N. Evaluation of the arterial blood pressure of dogs by two noninvasive methods. Can. J. Comp. Med. 1985, 49, 419–423. [Google Scholar] [PubMed]
- Vincent, I.C.; Michell, A.R. Relationship between blood pressure and stress-prone temperament in dogs. Physiol. Behav. 1996, 60, 135–138. [Google Scholar] [CrossRef]
- Vincent, I.C.; Michell, A.R.; Leahy, R.A. Non-invasive measurement of arterial blood pressure in dogs: A potential indicator for the identification of stress. Res. Vet. Sci. 1993, 54, 195–201. [Google Scholar] [CrossRef]
- Belew, A.M.; Barlett, T.; Brown, S.A. Evaluation of the White-Coat Effect in Cats. J. Vet. Intern. Med. 1999, 13, 134–142. [Google Scholar] [CrossRef]
- Pypendop, B.H.; Verstegen, J.P. Hemodynamic Effects of Medetomidine in the Dog: A Dose Titration Study. Vet. Surg. 1998, 27, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Vainio, O.; Palmu, L. Cardiovascular and respiratory effects of medetomidine in dogs and influence of anticholinergics. Acta Vet. Scand. 1989, 30, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Savola, J.M. Cardiovascular actions of medetomidine and their reversal by atipamezole. Acta Vet. Scand. Suppl. 1989, 85, 39–47. [Google Scholar] [PubMed]
- Ko, J.C.H.; Fox, S.M.; Mandsager, R.E. Effects of preemptive atropine administration on incidence of medetomidine-induced bradycardia in dogs. J. Am. Vet. Med. Assoc. 2001, 218, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.C.H.; Fox, S.M.; Mandsager, R.E. Sedative and cardiorespiratory effects of medetomidine, medetomidine-butorphanol, and medetomidine-ketamine in dogs. J. Am. Vet. Med. Assoc. 2000, 216, 1578–1583. [Google Scholar] [CrossRef]
- Pypendop, B.; Serteyn, D.; Verstegen, J. Hemodynamic effects of medetomidine-midazolam-butorphanol and medetomidine-midazolam-buprenorphine combinations and reversibility by atipamezole in dogs. Am. J. Vet. Res. 1996, 57, 724–730. [Google Scholar]
- Link, R.E.; Desai, K.; Hein, L.; Stevens, M.E.; Chruscinski, A.; Bernstein, D.; Barsh, G.B.; Kobilka, K.B. Cardiovascular Regulation in Mice Lacking alpha 2-Adrenergic Receptor Subtypes b and c. Science 1996, 273, 803–805. [Google Scholar] [CrossRef] [PubMed]
- Hieble, J.P.; Bondinell, W.; Ruffolo, R.R. alpha.- and beta.-Adrenoceptors: From the Gene to the Clinic. Part 1. Molecular Biology and Adrenoceptor Subclassification. J. Med. Chem. 1995, 38, 3415–3444. [Google Scholar] [CrossRef]
- Jedruch, J.; Gajewski, Z.; Ratajska-Michalczak, K. Uterine motor responses to an alpha 2-adrenergic agonist medetomidine hydrochloride in the bitches during the end of gestation and the post-partum period. Acta Vet. Scand. Suppl. 1989, 85, 129–134. [Google Scholar]
- Reynolds, L.P.; Killilea, S.D.; Redmer, D.A. Angiogenesis in the female reproductive system. FASEB J. 1992, 6, 886–892. [Google Scholar] [CrossRef]
- Campbell, S.; Bourne, T.H.; Waterstone, J.; Reynolds, K.M.; Crayford, T.J.; Jurkovic, D.; Okokon, E.V.; Collins, W.P. Transvaginal color blood flow imaging of the periovulatory follicle. Fertil. Steril. 1993, 60, 433–438. [Google Scholar] [CrossRef]
- Klagsbrun, M.; D’Amore, P.A. Regulators of Angiogenesis. Annu. Rev Physiol. 1991, 53, 217–239. [Google Scholar] [CrossRef] [PubMed]
- Araujo, R.R.; Ginther, O.J. Vascular perfusion of reproductive organs in pony mares and heifers during sedation with detomidine or xylazine. Am. J. Vet. Res. 2009, 70, 141–148. [Google Scholar] [CrossRef] [PubMed]
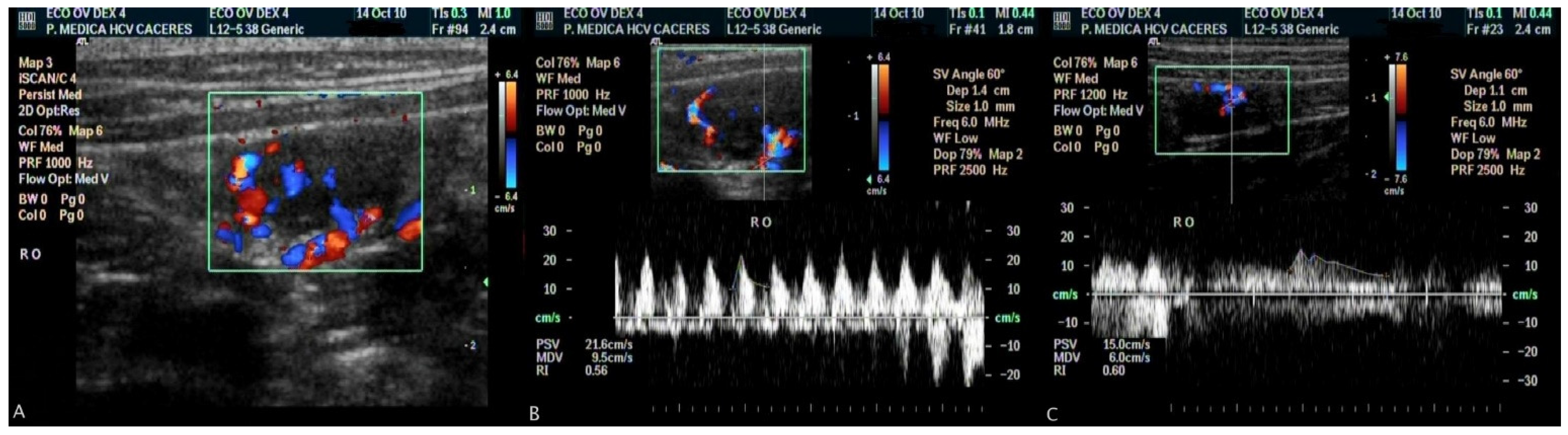
| Doppler Parameters | Before Administration | After Administration | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Group 1 | Group 2 | Group 1 | Group 2 | ||||||
| Mean | SEM | Mean | SEM | Mean | SEM | Mean | SEM | ||
| RO | PSV | 20.79 | 1.44 | 19.50 | 1.54 | 11.97 | 1.6 | 12.50 | 1.53 |
| EDV | 8.09 | 0.78 | 8.20 | 1.05 | 5.15 | 0.88 | 5.65 | 0.76 | |
| RI | 0.61 | 0.03 | 0.59 | 0.03 | 0.58 | 0.02 | 0.55 | 0.02 | |
| LO | PSV | 20.30 | 3.14 | 19.92 | 1.99 | 10.94 | 1.26 | 13.03 | 1.41 |
| EDV | 6.72 | 0.8 | 7.40 | 0.08 | 4.20 | 0.52 | 5.82 | 0.98 | |
| RI | 0.66 | 0.02 | 0.63 | 0.02 | 0.62 | 0.01 | 0.58 | 0.03 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicolás-Barceló, P.; Facchin, M.; Martínez-Taboada, F.; Barrera, R.; Cristóbal, J.I.; González, M.A.; Durán-Galea, Á.; Macías-García, B.; Duque, F.J. Effects of Sedation with Medetomidine and Dexmedetomidine on Doppler Measurements of Ovarian Artery Blood Flow in Bitches. Animals 2021, 11, 538. https://doi.org/10.3390/ani11020538
Nicolás-Barceló P, Facchin M, Martínez-Taboada F, Barrera R, Cristóbal JI, González MA, Durán-Galea Á, Macías-García B, Duque FJ. Effects of Sedation with Medetomidine and Dexmedetomidine on Doppler Measurements of Ovarian Artery Blood Flow in Bitches. Animals. 2021; 11(2):538. https://doi.org/10.3390/ani11020538
Chicago/Turabian StyleNicolás-Barceló, Paloma, Martina Facchin, Fernando Martínez-Taboada, Rafael Barrera, José Ignacio Cristóbal, Mario Alberto González, Ángela Durán-Galea, Beatriz Macías-García, and Francisco Javier Duque. 2021. "Effects of Sedation with Medetomidine and Dexmedetomidine on Doppler Measurements of Ovarian Artery Blood Flow in Bitches" Animals 11, no. 2: 538. https://doi.org/10.3390/ani11020538
APA StyleNicolás-Barceló, P., Facchin, M., Martínez-Taboada, F., Barrera, R., Cristóbal, J. I., González, M. A., Durán-Galea, Á., Macías-García, B., & Duque, F. J. (2021). Effects of Sedation with Medetomidine and Dexmedetomidine on Doppler Measurements of Ovarian Artery Blood Flow in Bitches. Animals, 11(2), 538. https://doi.org/10.3390/ani11020538







