1. Introduction
Rodents are the most diverse order of mammals [
1], and a small portion of them (~7–10%) are considered agricultural and infrastructural pests [
2], as well as important reservoirs of a variety of zoonotic diseases [
3,
4]. Therefore, rodent management strategies are needed in order to prevent and control rodent populations in a wide range of human-dominated landscapes [
5].
Because rodents are prey to several predators (reptiles, birds and mammals), they can perceive risk of predation in response to auditory, visual, and olfactory cues emanating from their predators [
6,
7,
8,
9]. Recently, the use of perceived predation risk has been proposed as a management tool for ecologically based rodent management, under The Landscape of Fear (LOF) framework [
10,
11,
12], in which rodent populations could be managed by manipulating their perceived predation risk to deter them from target areas [
11]. Therefore, the presence of a rodent predator can be simulated by using direct cues, such as carnivore urine and feces or raptor calls [
12,
13,
14], inducing antipredator behaviors, such as modifications to space-use, foraging activity, social behavior and vigilance patterns [
13,
14,
15,
16].
Despite chemosensory cues being the most common signals used by small mammals to assess predation risk [
17], the importance of auditory cues should not be overlooked, as they can provide equally valuable information [
12]. In rodents, several laboratory and field experiments using playbacks of raptor calls successfully triggered antipredator responses in wild rodents [
12,
18,
19,
20].
In southern Chile, small rodents are commonly found in agroecosystems and peridomestic settings [
21,
22]. Some of these rodents are reservoirs of several zoonotic pathogens, including the Andes virus (ANDV), which causes hantavirus cardiopulmonary syndrome (HCPS) in humans [
23]. The main reservoir of ANDV is the long-tailed pygmy rice rat (
Oligoryzomys longicaudatus) [
24]. Furthermore, serologic evidence of ANDV has also been found to a lesser extent in other native species such as the long–haired mouse (
Abrothrix hirta) and the olive grass mouse (
A. olivacea), and even in invasive species such as
Rattus spp. [
22,
25]. Since HCPS and other rodent-borne zoonoses are a major concern for public health, there is a need to better understand the behavioral ecology of zoonotic hosts to find effective solutions to prevent human exposure to rodents and their pathogens.
The rodent species mentioned above are common prey to diverse raptors, such as the Austral pigmy owl (
Glaucidium nana) and the Rufous-legged owl (
Strix rufipes) [
26].
Glaucidium nana is a small habitat generalist raptor (body length ~ 200 mm) that hunts and/or nests within forests, shrublands, and around human settlements [
27]. Small mammals are important components of its diet, preying upon a wide variety of rodents, including
A. hirta,
A. olivaceus,
O. longicaudatus [
26,
28].
Strix rufipes is a medium-sized forest specialist owl (body length ~ 400 mm) that hunts and nests only within forests [
27]. It is a generalist feeder, but it preys mostly on small mammals and insects. The most consumed small mammal preys are scansorial and arboreal species, such as
O. longicaudatus, the arboreal rat (
Irenomys tarsalis) and the colocolo opossum (
Dromiciops gliroides) [
27,
29]. Hence, playback calls of these species may be expected to induce antipredator responses in these rodents. However, to our knowledge, this topic has not been addressed.
For these reasons, the main objective of this study was to assess how direct auditory predation risk cues (i.e., owl calls) affect the behavior (feeding, vigilance and movement patterns) of a rodent assemblage composed of native species (
Abrothrix spp. and
O. longicaudatus) and an introduced species (
Rattus rattus) inhabiting a temperate forest in southern Chile. We also wanted to test if there were response differences depending on the predator species (
G. nana vs.
S. rufipes), shrub cover thickness and moonlight (indirect cues of predation risk). Therefore, we tested the following predictions: (a) Rodents would increase vigilance and decrease total feeding time under risk of predation. In addition, rodent movement patterns would be affected by the presence of predator calls, limiting locomotion in higher risk settings. (b) Rodent behavioral responses would be stronger when moonlight is greater or shrub cover is scarce, due to a higher perceived risk. (c) Rodents would modulate their antipredator responses depending on the owl species because predator dietary preferences pose different perceived risks to each rodent species. (d) Native rodents would exhibit a more finely tuned response depending on the predator selected, as they have been exposed to natural selection driven by these predators, while
R. rattus was introduced in the past centuries. The exact date of introduction of
R. rattus to Chile is unknown, but likely dates back to the mid-1600s [
30], and to date it has colonized a wide range of natural areas [
31].
2. Materials and Methods
2.1. Study Area and Target Species
The study was conducted in a temperate forest located in Huelemolle, at the Villarrica lake basin (39°16′ S, 71°48′ W) in the Araucanía Region (southern Chile). The climate in this area is temperate-humid with a short dry season (<4 months) in summer (January–March) and an average yearly rainfall of 2000 mm [
32]. Mean minimum and maximum temperatures are, respectively, 10.4 °C and 25.3 °C in the warmest month (January) and 4.2 °C and 12.1 °C in the coldest month (July) [
32]. Forests are dominated by Patagonian oak (
Lophozonia obliqua) and coigue (
Nothofagus dombeyi), mainly associated with Chilean laurel (
Laurelia sempervirens), olivillo (
Aextoxicon punctatum), ulmo (
Eucryphia cordifolia) and lingue (
Persea lingue) [
33]. Several raptors inhabit the temperate forest of southern Chile [
31]. The most abundant owls in the study area are
G. nana and
S. rufipes (Ibarra, data unpublished). Other predators of rodents commonly found in this area are foxes (
Lycalopex griseus,
L. culpaeus) and the kodkod cat (
Leopardus guigna) [
34].
To determine the composition of the rodent community in the study area, we performed a live-trapping survey in six plots during April 2020. Each plot consisted of 42 Sherman live traps shaping a 6 × 7 grid set at 5 m intervals. Each plot was sampled during nine consecutive nights (trapping effort = 2268 traps/night). A total of 123 individuals were captured. Rodent assemblage was composed of four species: A. hirta, A. olivaceus, O. longicaudatus (native species) and R. rattus (exotic species). Both Abrothrix species are terrestrial and omnivorous. A. hirta (until 2014 considered a synonym of A. longipilis) is a medium-sized rodent (body length~130 mm), while A. olivaceus is smaller (body length~90 mm). O. longicaudatus is small (~90 mm), scansorial (i.e., ability or propensity to climb), and omnivorous. R. rattus is a large species (~200 mm), scansorial and omnivorous.
2.2. Field Methods
We conducted an experimental field study during the austral autumn (May 2019), when several rodent species reach their highest abundances in the year. The experiment had three treatments, two of which simulated the presence of owls (predation risk treatments): (1)
G. nana calls, and (2)
S. rufipes calls. The third was the control treatment (without owl vocalizations). For each treatment, we installed two grids (2 grids × 3 treatments = 6 grids), each one consisting of four foraging stations separated by 25 m (
Figure S1), similar to other studies on rodent predation risks, in terms of distance between stations and grid configuration [
8]. Therefore, each treatment had a total of eight foraging stations. To avoid overlapping owl calls between treatments, the distance between grids was at least 300 m. This distance was tested in the study area, and smaller distances (e.g., 200 m) have been used in other studies in temperate forests [
35]. All grids were similar in rodent abundance (mean = 20 individuals) and composition among them, according to the live-trapping survey described above. Each grid in the experiment was sampled for three consecutive nights, simultaneously sampling one grid per treatment (
Figure S2) to reduce possible temporal/environmental variations between days. Each grid was only used for one treatment type. We did not implement enclosures or a semi-captive experiment, therefore, all foraging stations could be visited by rodents around the area.
Each foraging station contained 30 g of rolled oats with vanilla extract placed on a plastic petri dish, located on the forest ground. An infrared motion-triggered camera trap (Bushnell Trophy Cam, 119537C, Bushnell Optics, Overland Park, Kansas) was installed at each foraging station to record videos of the rodents visiting the station. Each camera was mounted horizontally 1.5 m above the ground on a PVC pipe [
36]. Cameras were set to take 30-sec videos once an individual visited a foraging station and continued recording as long as at least one individual stayed in front of the sensor (0 sec intervals between videos) [
37]. Cameras were activated from dusk (6 pm) to dawn (7 am), since rodents in Chilean temperate forests are mostly nocturnal [
38], and set for 3 consecutive nights at each foraging station. Each camera recorded an area of 0.89 m
2. The medium sensitivity setting was applied to all cameras, and two layers of masking tape were added to the cameras to reduce flash brightness for close-range operation. Camera traps have been used to monitor small mammals, as they can readily detect and reliably identify small mammals to species [
36,
37]. Before the experiment, we conducted pilot trials of camera performance in the field to test their effectiveness in recording rodent presence and distinguish individual behaviors.
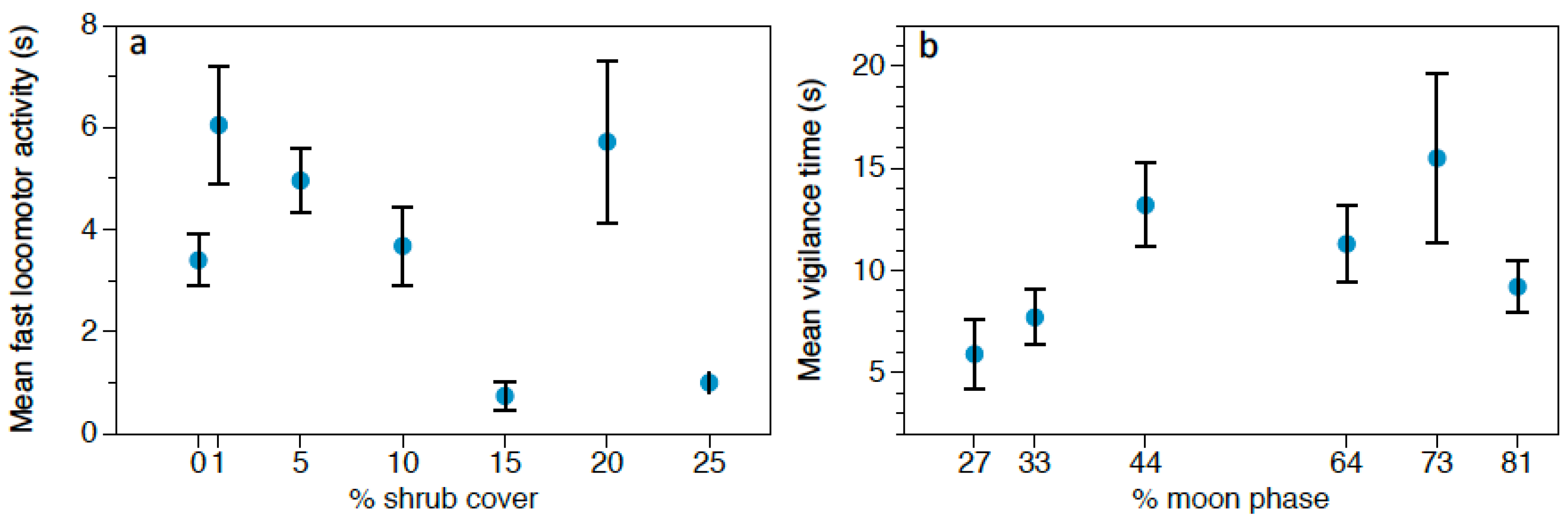
We also estimated vegetation cover of shrubs by averaging ocular estimates of shrub cover to the nearest 5% within a square frame of 1 m
2 centered at each foraging station [
39]. Each 1 m
2 frame is divided into 10 cm × 10 cm cells, creating proportional compartments to estimate shrub coverage. Moon phase (%, where full moon = 100%) for each night surveyed was also registered (
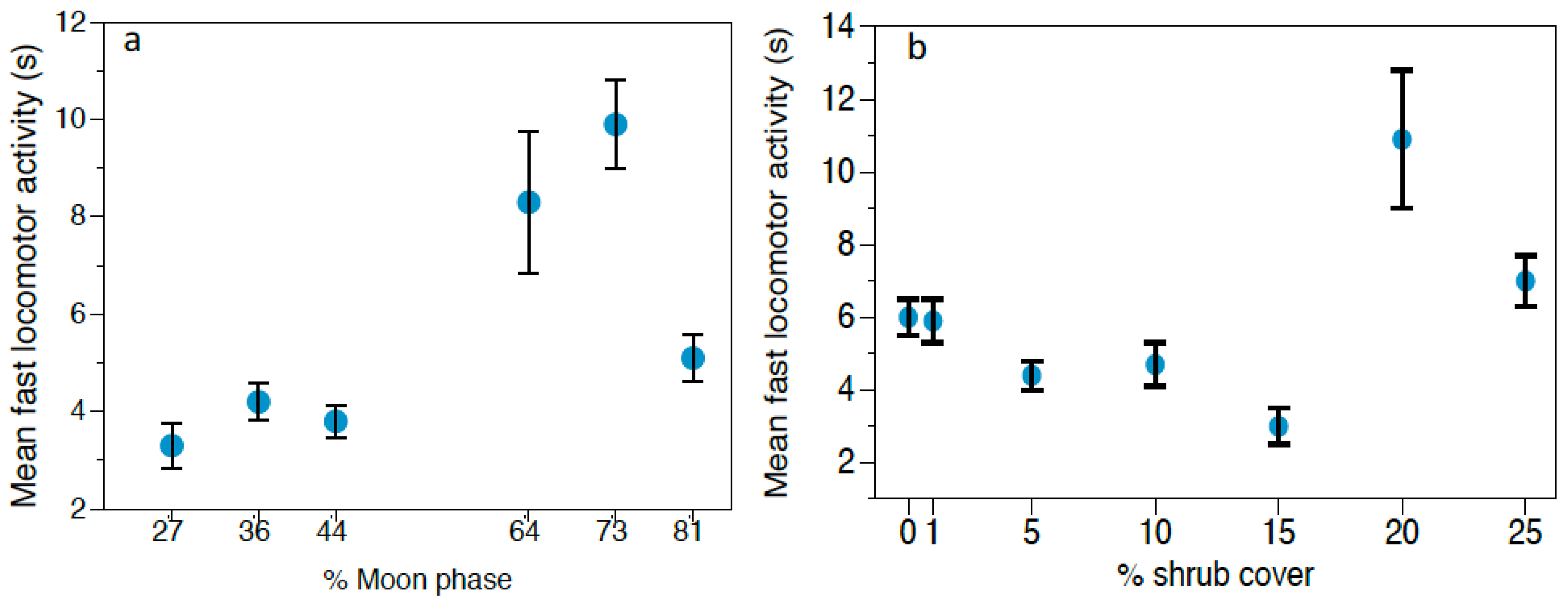
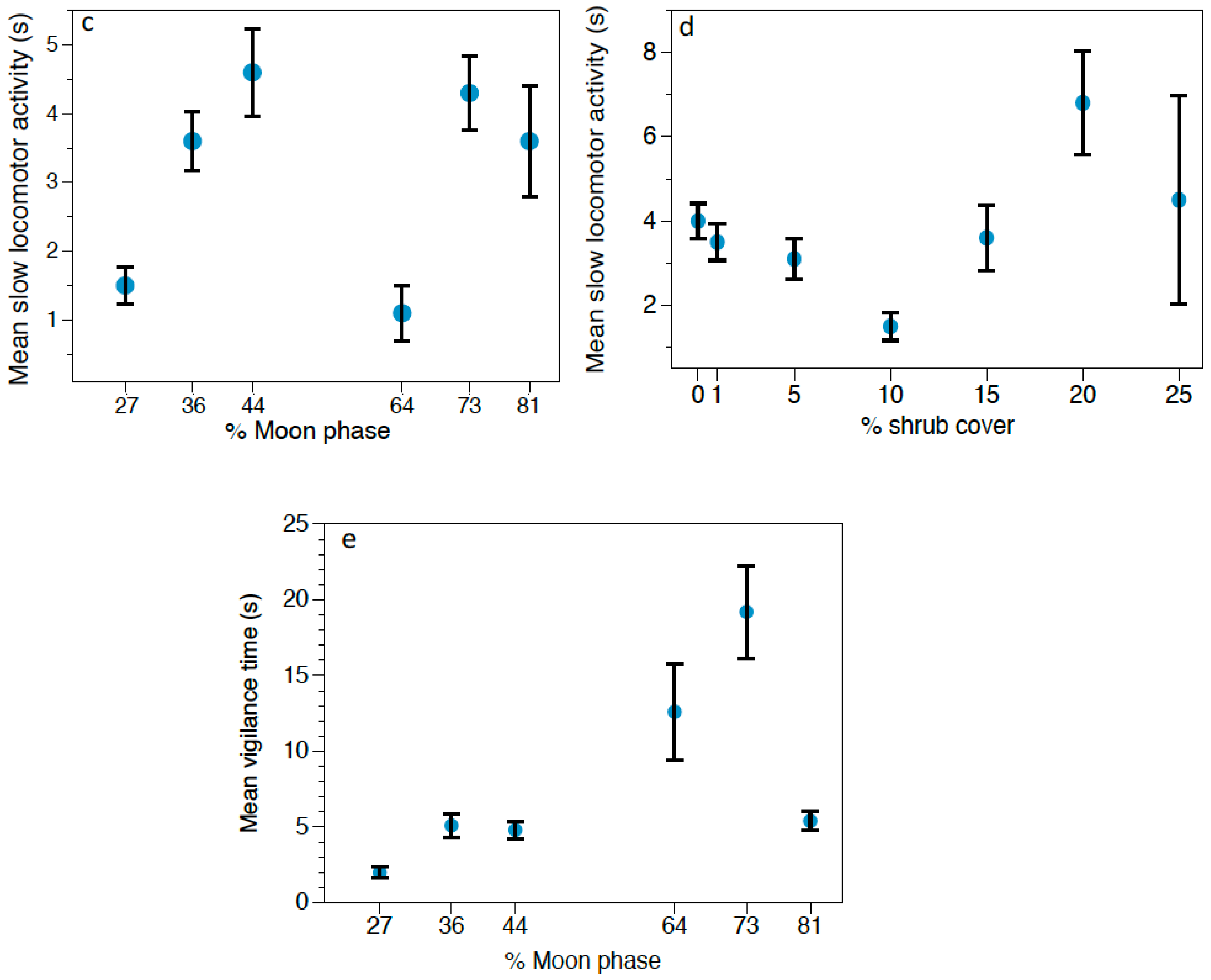
https://www.timeanddate.com/moon/phases/chile/villarrica (accessed on 6 February 2020)). These variables are commonly used as indirect cues of predation risk.
2.3. Vocalizations and Playbacks
To simulate the presence of owls, playback stimuli were broadcasted in both predation risk treatments (
G. nana calls and
S. rufipes calls). We used the most common owl calls available in recordings; in the case of
G. nana, we used the three most common types of vocalizations: mating call or partner contact, aggression call and juvenile vocalization. In the case of
S. rufipes, two types of vocalizations were emitted: a location call, used to delimit territory and maintain couple ties, and a territorial call. The sound system (JBL Charge 3 speaker and mp3 player, Sony NWZ-B183F (Tokyo, Japan)) was set up on a tree (2 m height) at the center of each grid (12.5 m apart from foraging stations). From dusk to dawn, playbacks were emitted randomly, alternating periods of one to three min, with silence periods of one to five min [
40].
2.4. Behavioral Analysis
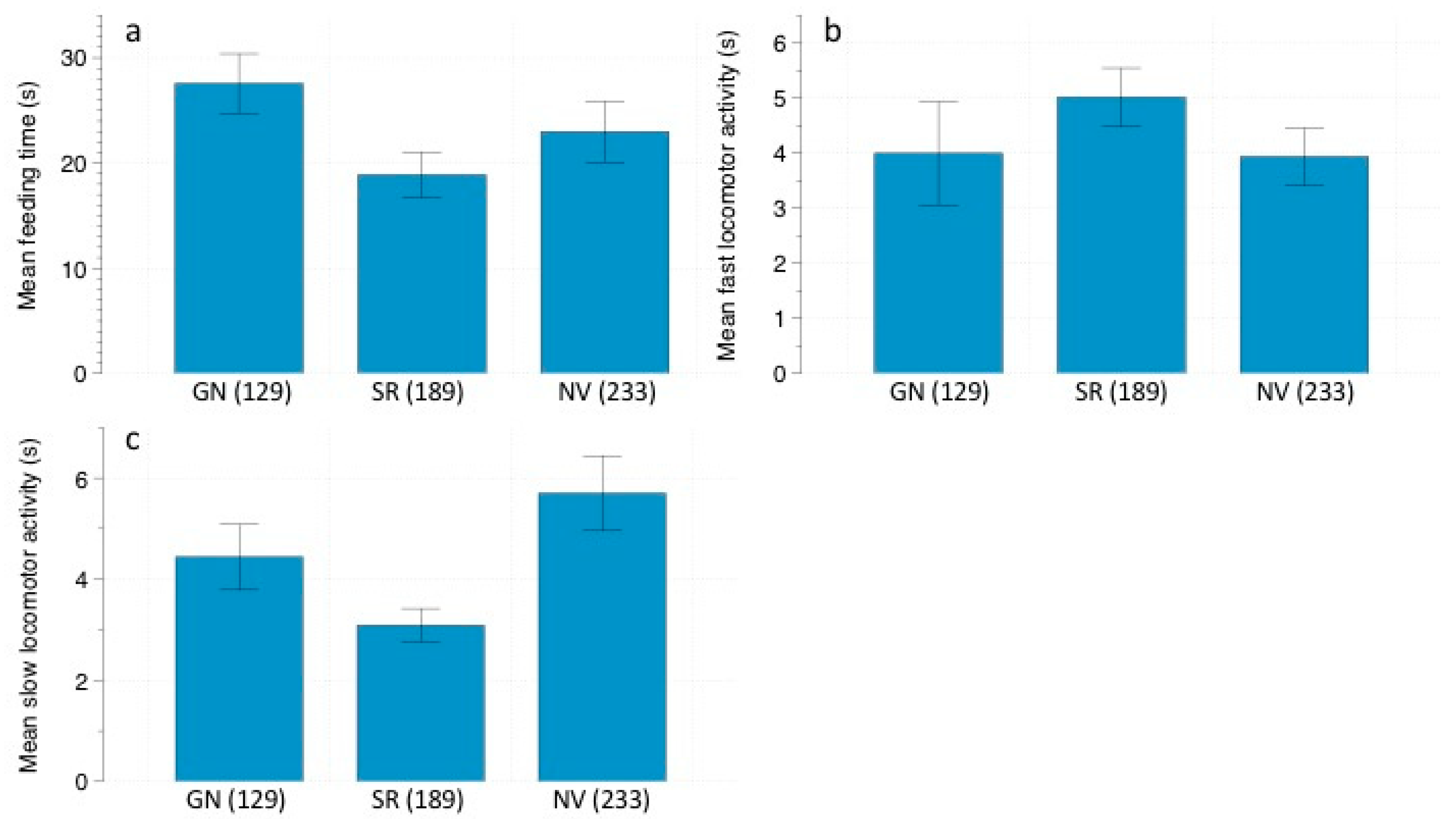
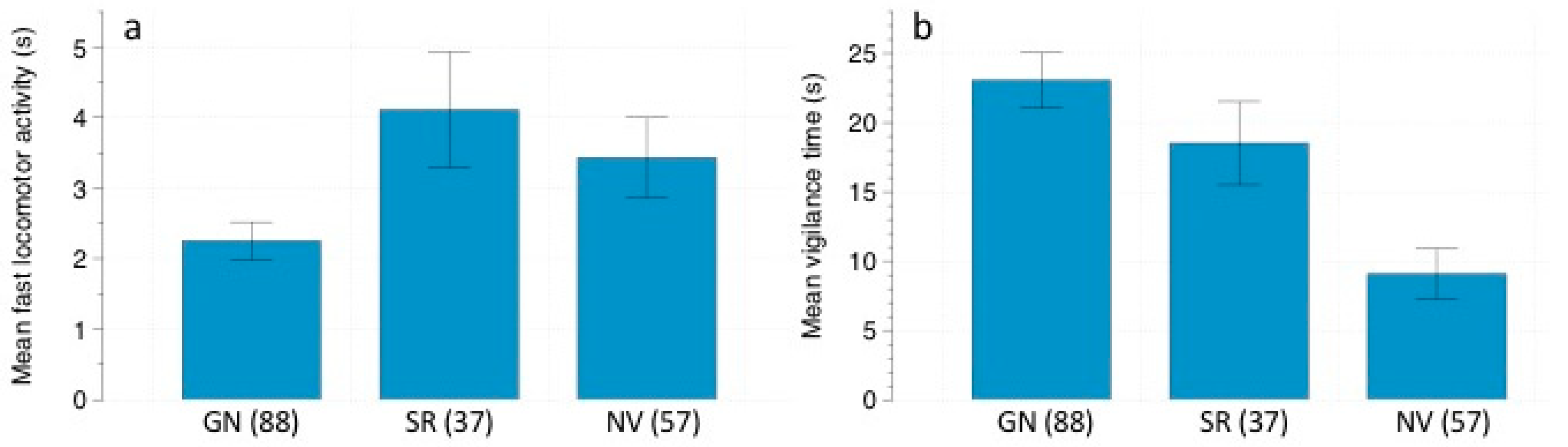
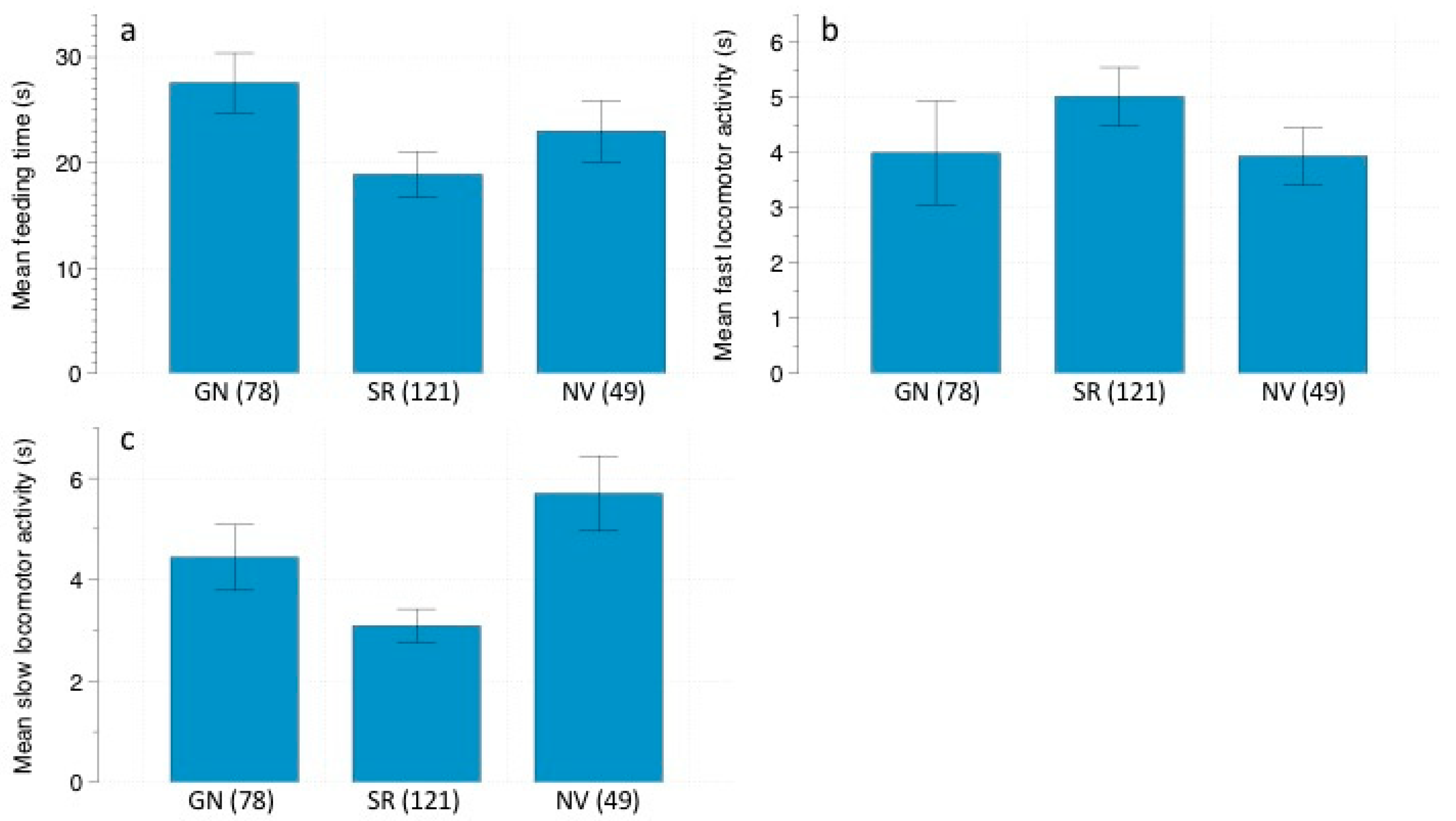
We analyzed the following rodent behaviors in the recorded videos: (1) feeding time: the total amount of time spent foraging at the stations; (2) fast locomotor activity: total amount of time spent running; (3) slow locomotor activity: total amount of time spent walking; (4) vigilance: total amount of time spent standing still, with head and ears pricked, looking around. These behaviors are usually used in studies of perceived predation risk [
40,
41].
Video recordings did not allow reliable identification of specific individuals. Therefore, all analyses were to the species level. For rodents of the genus
Abrothrix, it was difficult to identify both species in all recordings, therefore we combined their records as
Abrothrix spp. At each foraging station, we organized video recordings by “events” to distinguish between independent detections of each species. An event was considered as a video or several videos of the same species within a five-minute interval [
42]. Therefore, we grouped behavioral analyses for each event, and we summed the time dedicated to each behavior within each event. When more than one individual was recorded in the same video, we included the behavior of each individual as a different event.
2.5. Statistical Analysis
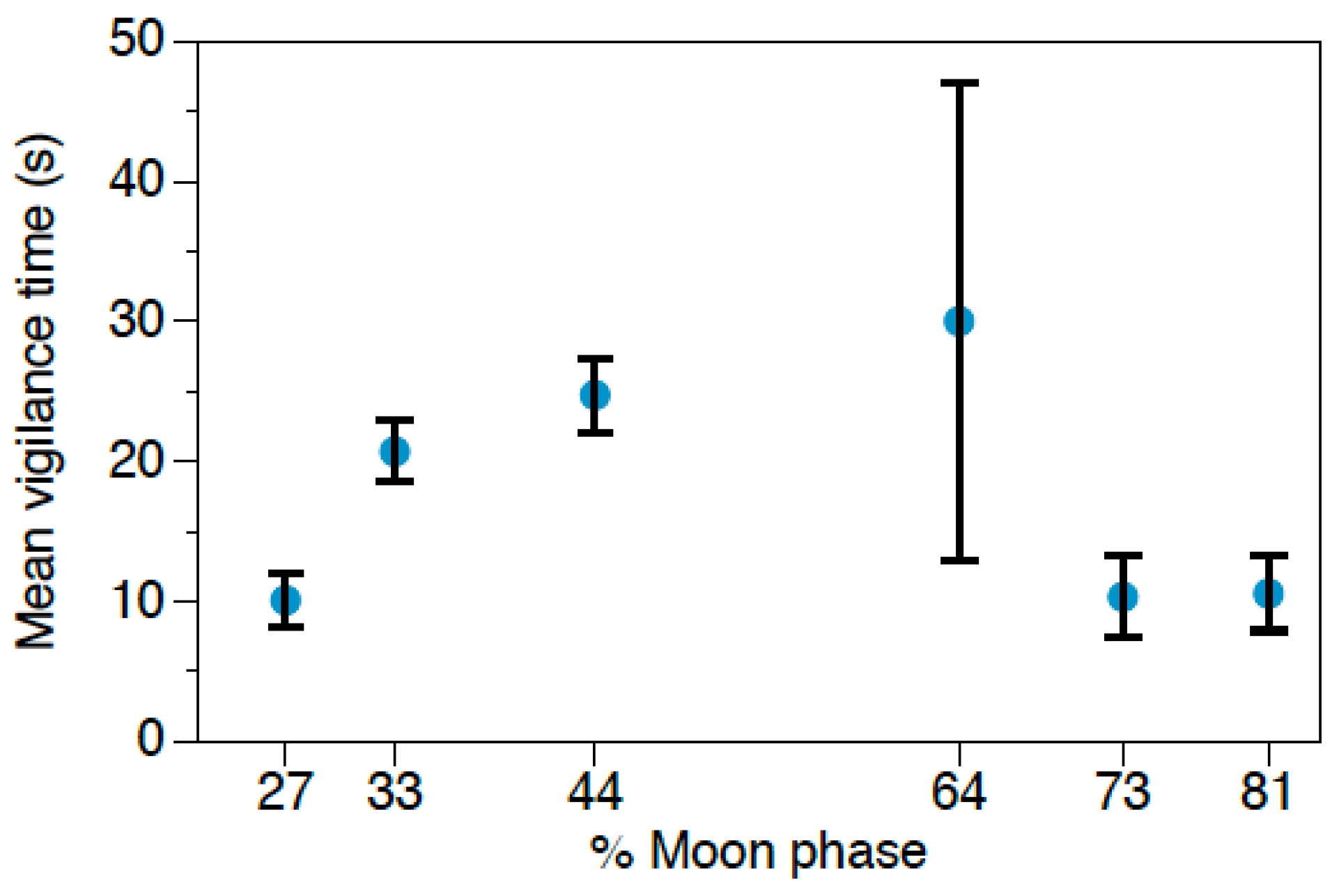
Behavioral responses to owl calls were analyzed using Generalized Linear Models (GLMs) with Gamma distribution and Log as the link function, since data did not fit a normal distribution. We performed three different GLMs, one for each rodent species (Abrothrix spp., O. longicaudatus, and R. rattus), considering the following response variables: feeding time, locomotor activity and vigilance. The predictor variables were: predation risk treatment (control/G. nana/S. rufipes), moon phase, and vegetation cover at each foraging station, considering the four stations in each grid as pseudoreplicates. Moreover, we set the total number of videos of each event as the offset to make data comparable and to avoid any possible bias caused by small-scale variations in rodent population densities. Results were considered significant at α < 0.05. Data are represented as mean ± standard error (SE). The software used to perform the statistical analysis was SPSS 23.0 for Windows (SPSS Inc., Chicago, IL, USA).
2.6. Ethics Statement
Rodent sampling was authorized by Servicio Agrícola y Ganadero (SAG; Chilean Fish and Wildlife Service) under permit No. 7479/2018. Bioethical approval (No. 18197-VET-UCH) was issued by the Faculty of Animal and Veterinary Sciences, University of Chile.