Tryptophan, Kynurenine and Kynurenic Acid Concentrations in Milk and Serum of Dairy Cows with Prototheca Mastitis
(This article belongs to the Section Animal Products)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Dairy Farms and Animals
2.2. Sampling Procedures
2.3. Study Group
2.4. Measurements of TRP and Its Metabolites Concentrations in Milk and Serum
2.5. Statistical Analysis
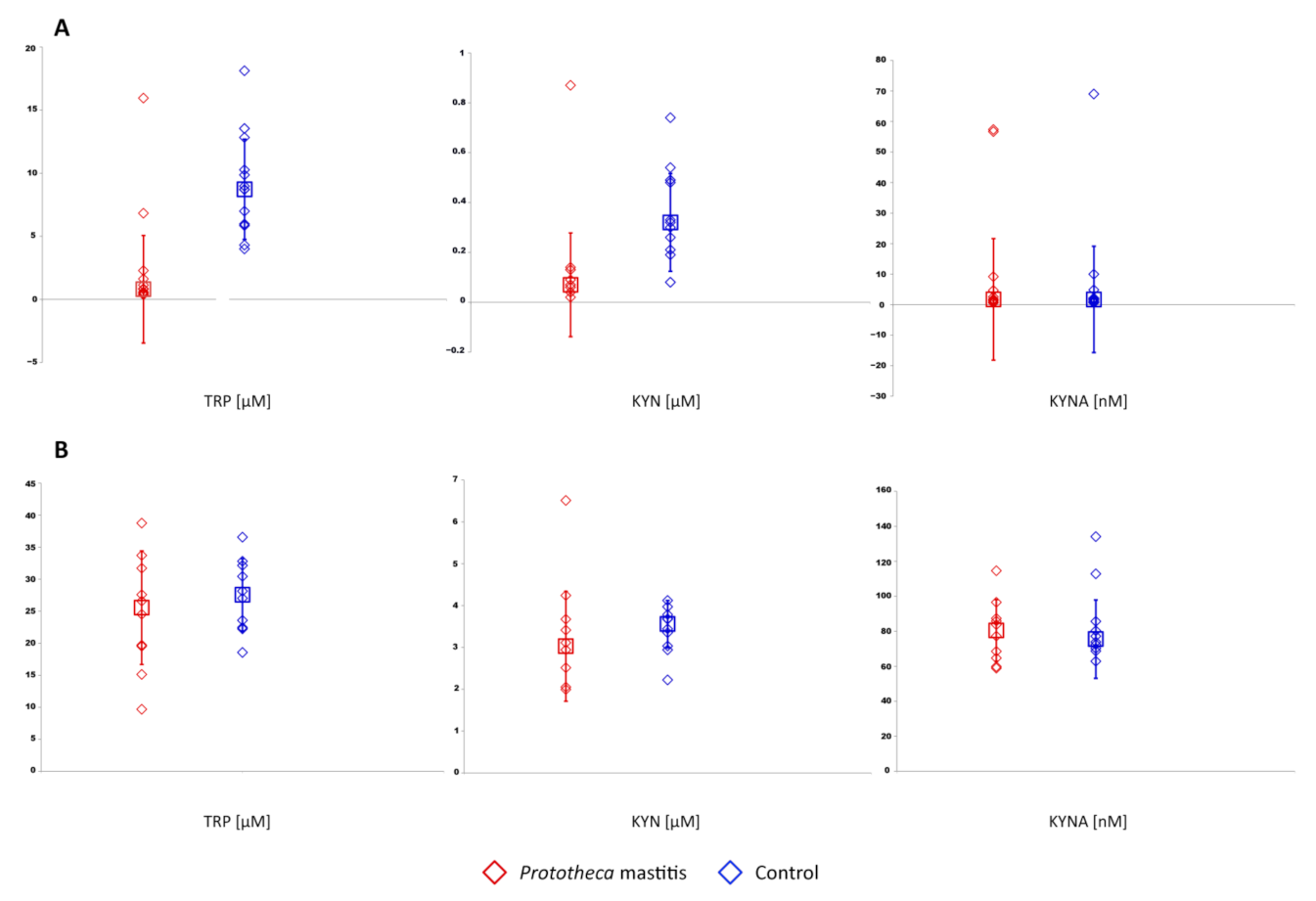
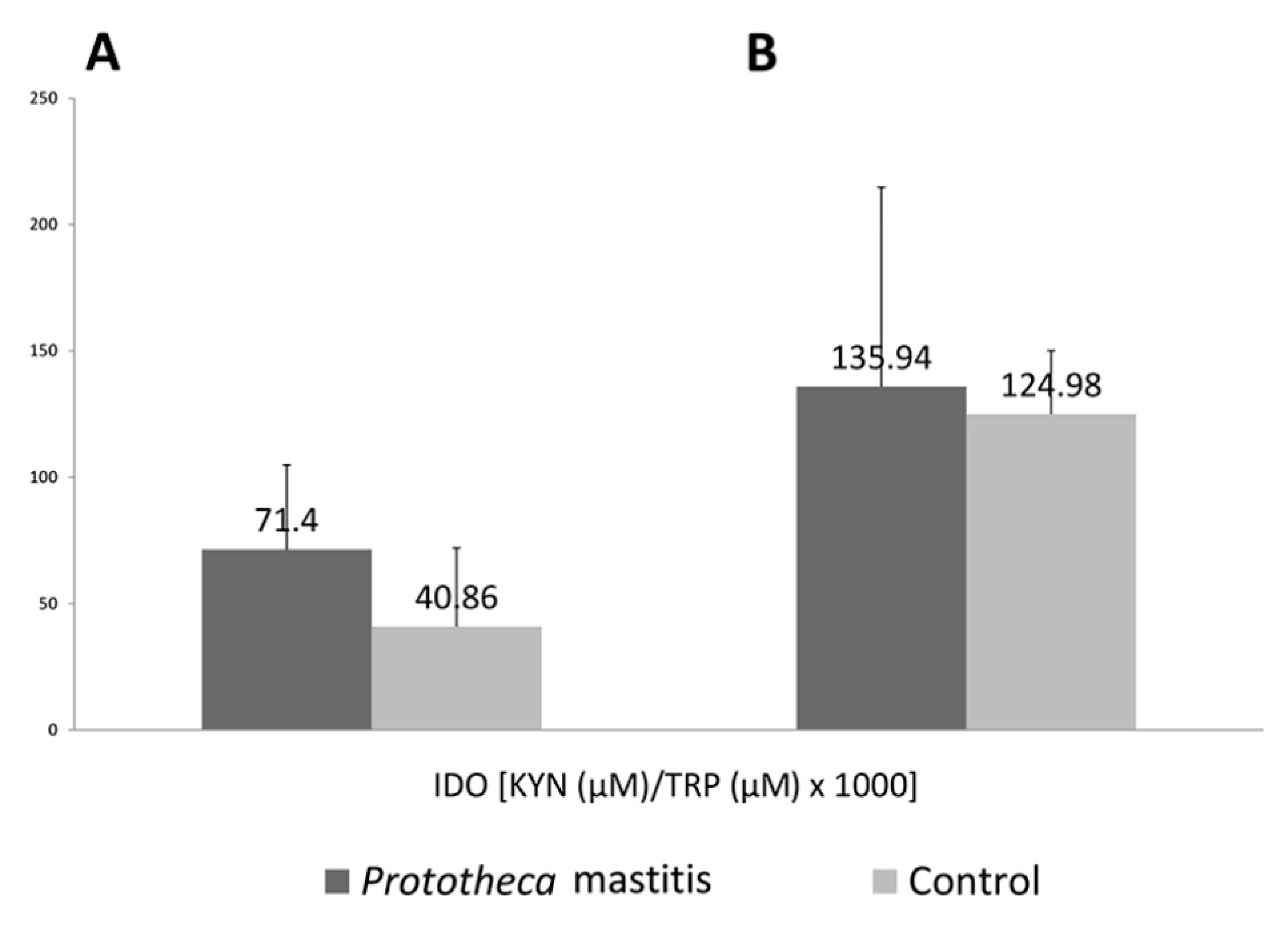
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moffett, J.R.; Namboodiri, A. Tryptophan and the immune response. Immunol. Cell Biol. 2003, 81, 247–265. [Google Scholar] [CrossRef] [PubMed]
- Le Floc’H, N.; Seve, B. Biological roles of tryptophan and its metabolism: Potential implications for pig feeding. Livest. Sci. 2007, 112, 23–32. [Google Scholar] [CrossRef]
- Bai, M.; Liu, H.; Xu, K.; Oso, A.O.; Wu, X.; Liu, G.; Tossou, M.C.B.; Al-Dhabi, N.A.; Duraipandiyan, V.; Xi, Q.; et al. A review of the immunomodulatory role of dietary tryptophan in livestock and poultry. Amino Acids 2017, 49, 67–74. [Google Scholar] [CrossRef]
- Cortamira, N.; Seve, O.B.; Lebreton, Y.; Ganier, P. Effect of dietary tryptophan on muscle, liver and whole-body protein-synthesis in weaned piglets-relationship to plasma-insulin. Br. J. Nutr. 1991, 66, 423–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, K.; Fang, J.; Yin, Y.L.; Feng, Z.M.; Tang, Z.R.; Wu, G. Tryptophan metabolism in animals: Important roles in nutrition and health. Front. Biosci. 2011, 3, 286–297. [Google Scholar]
- Ruan, Z.; Yang, Y.; Wen, Y.; Zhou, Y.; Fu, X.; Ding, S.; Liu, G.; Yao, K.; Wu, X.; Deng, Z.; et al. Metabolomic analysis of amino acid and fat metabolism in rats with l-tryptophan supplementation. Amino Acids 2014, 46, 2681–2691. [Google Scholar] [CrossRef]
- Melchior, D.; Sève, B.; Le Floch, N. Chronic lung inflammation affects plasma amino acid concentrations in pigs. J. Anim. Sci. 2004, 82, 1091–1099. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.Y. Amino acids: Metabolism, functions, and nutrition. Amino Acids 2009, 37, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Laeger, T.; Görs, S.; Metges, C.; Kuhla, B. Effect of feed restriction on metabolites in cerebrospinal fluid and plasma of dairy cows. J. Dairy Sci. 2012, 95, 1198–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kollmann, M.T.; Locher, M.; Hirche, F.; Eder, K.; Meyer, H.H.; Bruckmaier, R.M. Effects of tryptophan supplementation on plasma tryptophan and related hormone levels in heifers and dairy cows. Domest. Anim. Endocrinol. 2008, 34, 14–24. [Google Scholar] [CrossRef]
- Le Floc’H, N.; Melchior, D.; Sève, B. Dietary tryptophan helps to preserve tryptophan homeostasis in pigs suffering from lung inflammation1. J. Anim. Sci. 2008, 86, 3473–3479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esteban, S.; Nicolaus, C.; Garmundi, A.; Rial, R.V.; Rodriguez, A.B.; Ortega, E.; Ibars, C.B. Effect of orally administered l-tryptophan on serotonin, melatonin, and the innate immune response in the rat. Mol. Cell Biochem. 2004, 267, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Höglund, E.; Øverli, Ø.; Winberg, S. Tryptophan Metabolic Pathways and Brain Serotonergic Activity: A Comparative Review. Front. Endocrinol. 2019, 10, e158. [Google Scholar] [CrossRef]
- Fatokun, A.A.; Hunt, N.H.; Ball, H.J. Indoleamine 2,3-dioxygenase 2 (IDO1) and the kynurenine pathway: Characteristics and potential roles in health and disease. Amino Acids 2013, 45, 1319–1329. [Google Scholar] [CrossRef]
- Poormasjedi-Meibod, M.-S.; Jalili, R.B.; Hosseini-Tabatabaei, A.; Hartwell, R.; Ghahary, A. Immuno-Regulatory Function of Indoleamine 2,3 Dioxygenase through Modulation of Innate Immune Responses. PLoS ONE 2013, 8, e71044. [Google Scholar]
- Cervenka, I.; Agudelo, L.Z.; Ruas, J.L. Kynurenines: Tryptophan’s metabolites in exercise, inflammation, and mental health. Science 2017, 357, 6349. [Google Scholar] [CrossRef] [Green Version]
- Narui, K.; Noguchi, N.; Saito, A.; Kakimi, K.; Motomura, N.; Kubo, K.; Takamoto, S.; Sasatsu, M. Anti-infectious Activity of Tryptophan Metabolites in the l-Tryptophan-l-Kynurenine Pathway. Biol. Pharm. Bull. 2009, 32, 41–44. [Google Scholar] [CrossRef] [Green Version]
- Nino-Castro, A.; Abdullah, Z.; Popov, A.; Thabet, Y.; Beyer, M.; Knolle, P.A.; Domann, E.; Chakraborty, T.; Schmidt, S.V.; Schultze, J. The IDO1-induced kynurenines play a major role in the antimicrobial effect of human myeloid cells against Listeria monocytogenes. Innate Immun. 2014, 20, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Harden, J.L.; Lewis, S.M.; Lish, S.R.; Suarez-Farinas, M.; Gareau, D.; Lentini, T.; Johnson-Huang, L.M.; Krueger, J.G.; Lowes, M.A. The tryptophan metabolism enzyme l-kynureninase is a novel inflammatory factor in psoriasis and other inflammatory diseases. J. Allergy Clin. Immunol. 2016, 137, 1830–1840. [Google Scholar] [CrossRef] [Green Version]
- Bochniarz, M.; Kocki, T.; Dąbrowski, R.; Szczubiał, M.; Wawron, W.; Turski, W.A. Tryptophan, kynurenine, kynurenic acid concentrations and indoleamine 2,3-dioxygenase activity in serum and milk of dairy cows with subclinical mastitis caused by coagulase-negative staphylococci. Reprod. Domest. Anim. 2018, 53, 1491–1497. [Google Scholar] [CrossRef]
- Buzzini, P.; Turchetti, B.; Facelli, R.; Baudino, R.; Cavarero, F.; Mattalia, L.; Mosso, P.; Martini, A. First large-scale isolation of Prototheca zopfii from milk produced by dairy herds in Italy. Mycopathologia 2004, 158, 427–430. [Google Scholar] [CrossRef] [PubMed]
- Bueno, V.F.; de Mesquita, A.J.; Neves, R.B.; de Souza, M.A.; Ribeiro, A.R.; Nicolau, E.S.; de Oliveira, A.N. Epidemiological and clinical aspects of the first outbreak of bovine mastitis caused by Prototheca zopfii in Goias State, Brazil. Mycopathologia 2006, 161, 141–145. [Google Scholar] [CrossRef]
- Osumi, T.; Kishimoto, Y.; Kano, R.; Maruyama, H.; Onozaki, M.; Makimura, K.; Ito, T.; Matsubara, K.; Hasegawa, A. Prototheca zopfii genotypes isolated from cow barns and bovine mastitis in Japan. Veter. Microbiol. 2008, 131, 419–423. [Google Scholar] [CrossRef]
- Pieper, L.; Godkin, A.; Roesler, U.; Polleichtner, A.; Slavić, D.; Leslie, K.; Kelton, D. Herd characteristics and cow-level factors associated with Prototheca mastitis on dairy farms in Ontario, Canada. J. Dairy Sci. 2012, 95, 5635–5644. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Ali, T.; Zhang, L.; Hou, R.; Zhang, S.; Ding, L.; Han, D.; Deng, Z.; Rahman, A. Characterization of Prototheca zopfii genotypes isolated from cases of bovine mastitis and cow barns in China. Mycopathologia 2016, 181, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Jagielski, T.; Roeske, K.; Bakuła, Z.; Piech, T.; Wlazło, Ł.; Bochniarz, M.; Woch, P.; Krukowski, H. A survey on the incidence of Prototheca mastitis in dairy herds in Lublin province, Poland. J. Dairy Sci. 2019, 102, 619–628. [Google Scholar] [CrossRef] [Green Version]
- Jagielski, T.; Krukowski, H.; Bochniarz, M.; Piech, T.; Roeske, K.; Bakuła, Z.; Wlazło, Ł.; Woch, P. Prevalence of Prototheca spp. on dairy farms in Poland—A cross-country study. Microb. Biotechnol. 2019, 12, 556–566. [Google Scholar] [CrossRef] [Green Version]
- Park, H.-S.; Moon, D.C.; Hyun, B.-H.; Lim, S.-K. Short communication: Occurrence and persistence of Prototheca zopfii in dairy herds of Korea. J. Dairy Sci. 2019, 102, 2539–2543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jagielski, T.; Bakuła, Z.; Gawor, J.; Maciszewski, K.; Dyląg, M.; Nowakowska, J.; Gromadka, R.; Karnkowska, A. The genus Prototheca (Trebouxiophyceae, Chlorophyta) revisited: Implications from molecular taxonomic studies. Algal Res. 2019, 43, e101639. [Google Scholar] [CrossRef]
- Marques, S.; Silva, E.; Kraft, C.; Carvalheira, J.; Videira, A.; Huss, V.A.R.; Thompson, G. Bovine Mastitis Associated with Prototheca blaschkeae. J. Clin. Microbiol. 2008, 46, 1941–1945. [Google Scholar] [CrossRef] [Green Version]
- Ricchi, M.; De Cicco, C.; Buzzini, P.; Cammi, G.; Arrigoni, N.; Cammi, M.; Garbarino, C. First outbreak of bovine mastitis caused by Prototheca blaschkeae. Veter. Microbiol. 2013, 162, 997–999. [Google Scholar] [CrossRef] [PubMed]
- Jagielski, T.; Lassa, H.; Ahrholdt, J.; Malinowski, E.; Roesler, U. Genotyping of bovine Prototheca mastitis isolates from Poland. Veter. Microbiol. 2011, 149, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Wawron, W.; Bochniarz, M.; Piech, T.; Łopuszyński, W.; Wysocki, J. Outbreak of protothecal mastitis in a herd of dairy cows in Poland. Bull. Veter. Inst. Pulawy 2013, 57, 335–339. [Google Scholar] [CrossRef] [Green Version]
- Buzzini, P.; Turchetti, B.; Branda, E.; Goretti, M.; Amici, M.; Lagneau, P.-E.; Scaccabarozzi, L.; Bronzo, V.; Moroni, P. Large-scale screening of the in vitro susceptibility of Prototheca zopfii towards polyene antibiotics. Med. Mycol. 2008, 46, 511–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jagielski, T.; Buzzini, P.; Lassa, H.; Malinowski, E.; Branda, E.; Turchetti, B.; Polleichtner, A.; Roesler, U.; Lagneau, P.-E.; Marques, S.; et al. Multicentre Etest evaluation of in vitro activity of conventional antifungal drugs against European bovine mastitis Prototheca spp. isolates. J. Antimicrob. Chemother. 2012, 67, 1945–1947. [Google Scholar] [CrossRef] [Green Version]
- Quinn, P.J.; Carter, M.E.; Markey, B.K.; Carter, G.R. Bacterial Causes of Bovine Mastitis. Veterinary Microbiology and Microbial Disease, 8th ed.; Mosby International Ltd.: London, UK, 2002; pp. 465–475. [Google Scholar]
- Jagielski, T.; Gawor, J.; Bakuła, Z.; Decewicz, P.; Maciszewski, K.; Karnkowska, A. cytb as a new genetic marker for differentiation of Prototheca species. J. Clin. Microbiol. 2018, 56, e00584-18. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Ghao, P.; Zhu, D. Optimization of Zn2+—containing mobile phase for simultaneous determination of kynurenine, kynurenic acid and tryptophan in human plasma by high performance liquid chromatography. J. Chromatogr. B 2010, 878, 603–608. [Google Scholar] [CrossRef]
- Chen, Y.; Xie, Z.; Xiao, C.; Zhang, M.; Li, Z.; Xie, J.; Zhang, Y.; Zhao, X.; Zeng, P.; Mo, L.; et al. Peripheral kynurenine/tryptophan ratio is not a reliable marker of systemic indoleamine 2,3-dioxygenase: A lesson drawn from patients on hemodialysis. Oncotarget 2017, 8, 25261–25269. [Google Scholar] [CrossRef] [Green Version]
- Plain, K.M.; de Silva, K.; Earl, J.; Begg, D.; Purdie, A.; Whittington, R.J. Indoleamine 2,3-Dioxygenase, Tryptophan Catabolism, and Mycobacterium avium subsp. paratuberculosis: A Model for Chronic Mycobacterial Infections. Infect. Immun. 2011, 79, 3821–3832. [Google Scholar] [CrossRef] [Green Version]
- Sorgdrager, F.J.H.; Naudé, P.J.W.; Kema, I.P.; Nollen, E.A.; De Deyn, P.P. Tryptophan metabolism in inflammaging: From biomarker to therapeutic target. Front. Immunol. 2019, 10, 2565. [Google Scholar] [CrossRef]
- Petzer, I.-M.; Karzis, J.; Donkin, E.F.; Webb, E.C.; Etter, E. Somatic cell count thresholds in composite and quarter milk samples as indicator of bovine intramammary infection status. Onderstepoort J. Veter. Res. 2017, 84, e1–e10. [Google Scholar] [CrossRef]
- MacKenzie, C.R.; Worku, D.; Däubener, W. Regulation of Ido-Mediated Bacteriostasis in Macrophages: Role of Antibiotics and Anti-Inflammatory Agents. Adv. Exp. Med. Biol. 2003, 527, 67–76. [Google Scholar]
- Bhutia, Y.D.; Babu, E.; Ganapathy, V. Interferon-γ induces a tryptophan-selective amino acid transporter in human colonic epithelial cells and mouse dendritic cells. Biochim. Biophys. Acta Biomembr. 2015, 1848, 453–462. [Google Scholar] [CrossRef] [Green Version]
- Dai, W.; Pan, H.; Kwok, O.; Dubey, J.P. Human indoleamine 2,3-dioxygenase inhibits Toxoplasma gondii growth in fibroblast cells. J. Interferon Res. 1994, 14, 313–317. [Google Scholar] [CrossRef]
- Murr, C.; Widner, B.; Gerlach, D.; Werner-Felmayer, G.; Dierich, M.P.; Wachter, H.; Fuchs, D. Streptococcal erythrogenic toxins induce tryptophan degradation in human peripheral blood mononuclear cells. Int. Arch. Allergy Immunol. 1997, 114, 224–228. [Google Scholar] [CrossRef] [PubMed]
- Däubener, W.; MacKenzie, C.R. IFN-gamma activated indoleamine 2,3-dioxygenase activity in human cells is an antiparasitic and an antibacterial effector mechanism. Adv. Exp. Med. Biol. 1999, 467, 517–524. [Google Scholar]
- Murr, C.; Gerlach, D.; Widner, B.; Dierich, M.P.; Fuchs, D. Neopterin production and tryptophan degradation in humans infected by Streptococcus pyogenes. Med. Microbiol. Immunol. 2001, 189, 161–163. [Google Scholar] [CrossRef]
- Bortolotti, P.; Hennart, B.; Thieffry, C.; Jausions, G.; Faure, E.; Grandjean, T.; Thepaut, M.; Dessein, R.; Allorge, D.; Guery, B.P.; et al. Tryptophan catabolism in Pseudomonas aeruginosa and potential for inter-kingdom relationship. BMC Microbiol. 2016, 16, 137–147. [Google Scholar] [CrossRef] [Green Version]
- Klein, M.S.; Almstetter, M.F.; Nürnberger, N.; Sigl, G.; Gronwald, W.; Wiedemann, S.; Dettmer, K.; Oefner, P.J. Correlations between milk and plasma levels of amino and carboxylic acids in dairy cows. J. Proteome Res. 2013, 12, 5223–5232. [Google Scholar] [CrossRef] [Green Version]
- Saito, K.; Markey, S.; Heyes, M. Effects of immune activation on quinolinic acid and neuroactive kynurenines in the mouse. Neuroscience 1992, 51, 25–39. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bochniarz, M.; Piech, T.; Kocki, T.; Iskra, M.; Krukowski, H.; Jagielski, T. Tryptophan, Kynurenine and Kynurenic Acid Concentrations in Milk and Serum of Dairy Cows with Prototheca Mastitis. Animals 2021, 11, 3608. https://doi.org/10.3390/ani11123608
Bochniarz M, Piech T, Kocki T, Iskra M, Krukowski H, Jagielski T. Tryptophan, Kynurenine and Kynurenic Acid Concentrations in Milk and Serum of Dairy Cows with Prototheca Mastitis. Animals. 2021; 11(12):3608. https://doi.org/10.3390/ani11123608
Chicago/Turabian StyleBochniarz, Mariola, Tomasz Piech, Tomasz Kocki, Mateusz Iskra, Henryk Krukowski, and Tomasz Jagielski. 2021. "Tryptophan, Kynurenine and Kynurenic Acid Concentrations in Milk and Serum of Dairy Cows with Prototheca Mastitis" Animals 11, no. 12: 3608. https://doi.org/10.3390/ani11123608
APA StyleBochniarz, M., Piech, T., Kocki, T., Iskra, M., Krukowski, H., & Jagielski, T. (2021). Tryptophan, Kynurenine and Kynurenic Acid Concentrations in Milk and Serum of Dairy Cows with Prototheca Mastitis. Animals, 11(12), 3608. https://doi.org/10.3390/ani11123608





