Respiratory Disease Risk of Zoo-Housed Bonobos Is Associated with Sex and Betweenness Centrality in the Proximity Network
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection and Study Sample
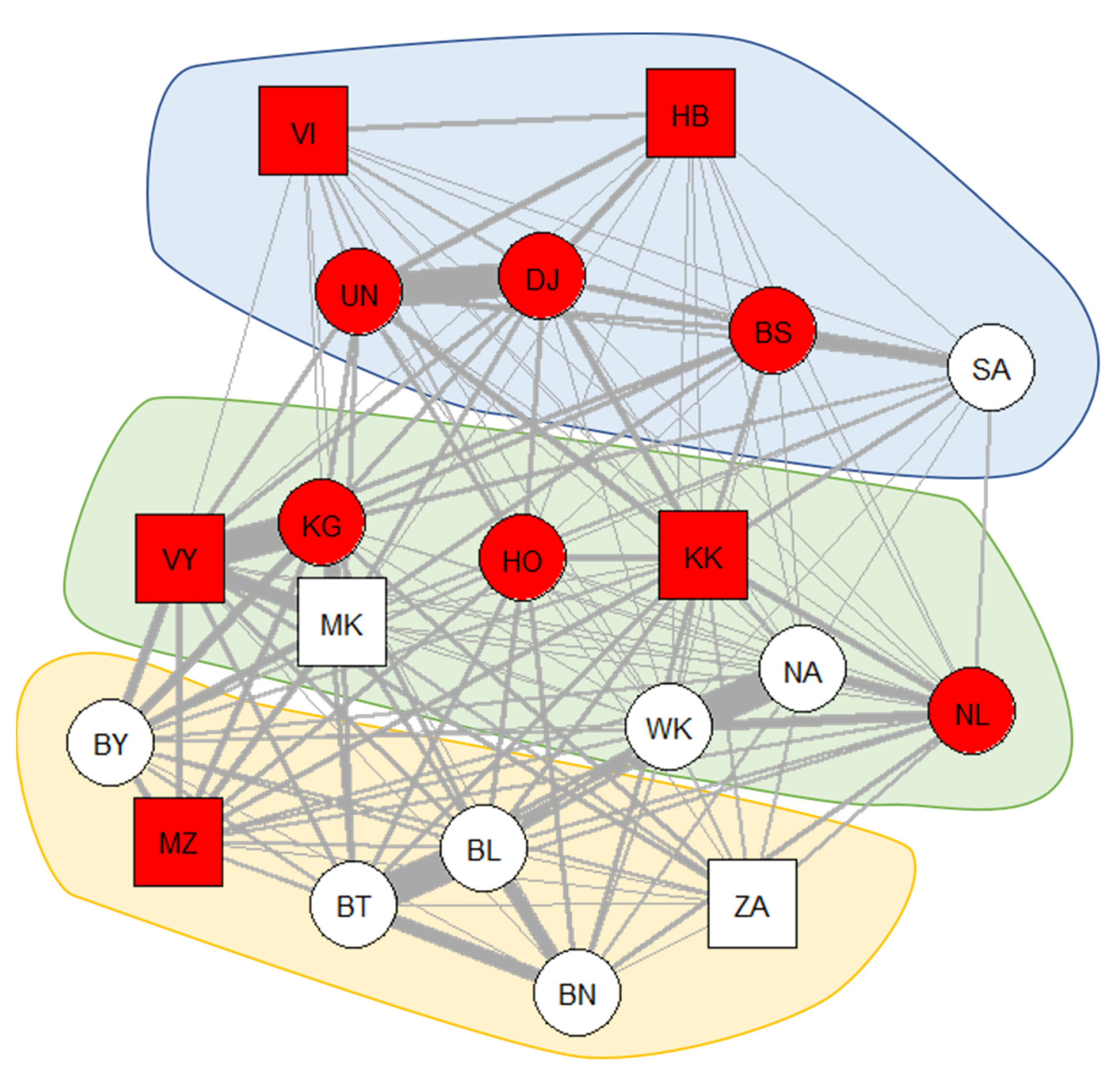
2.2. Social Network Analysis
2.3. Statistical Analyses
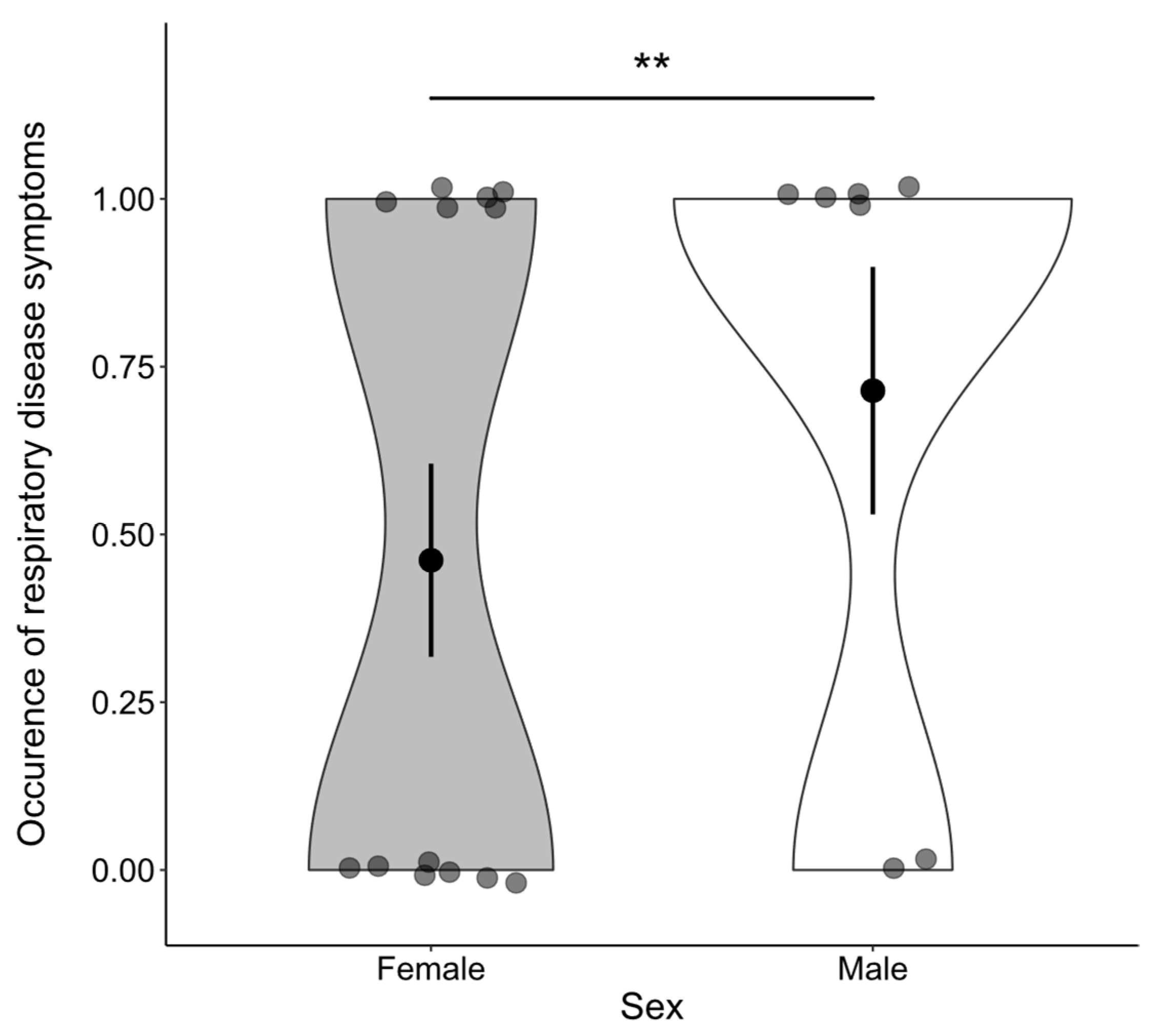
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hamilton, W.D. Geometry for the Selfish Herd. J. Theor. Biol. 1971, 31, 295–311. [Google Scholar] [CrossRef]
- Alexander, R.D. The Evolution of Social Behavior. Annu. Rev. Ecol. Syst. 1974, 5, 325–383. [Google Scholar] [CrossRef]
- Wrangham, R.W. An Ecological Model of Female-Bonded Primate Groups. Behaviour 1980, 75, 262–300. [Google Scholar] [CrossRef]
- Silk, J.B. The Adaptive Value of Sociality in Mammalian Groups. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362, 539–559. [Google Scholar] [CrossRef]
- Freeland, W.J. Pathogens and the Evolution of Primate Sociality. Biotropica 1976, 8, 12–24. [Google Scholar] [CrossRef]
- Freeland, W.J. Primate Social Groups as Biological Islands. Ecology 1979, 60, 719–728. [Google Scholar] [CrossRef]
- Kappeler, P.M.; Cremer, S.; Nunn, C.L. Sociality and Health: Impacts of Sociality on Disease Susceptibility and Transmission in Animal and Human Societies. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140116. [Google Scholar] [CrossRef] [PubMed]
- Altizer, S.; Nunn, C.L.; Thrall, P.H.; Gittleman, J.L.; Antonovics, J.; Cunningham, A.A.; Dobson, A.P.; Ezenwa, V.; Jones, K.E.; Pedersen, A.B.; et al. Social Organization and Parasite Risk in Mammals: Integrating Theory and Empirical Studies. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 517–547. [Google Scholar] [CrossRef]
- Lloyd-Smith, J.O.; Schreiber, S.J.; Kopp, P.E.; Getz, W.M. Superspreading and the Effect of Individual Variation on Disease Emergence. Nature 2005, 438, 355–359. [Google Scholar] [CrossRef] [PubMed]
- May, R.M. Network Structure and the Biology of Populations. Trends Ecol. Evol. 2006, 21, 394–399. [Google Scholar] [CrossRef]
- Cross, P.C.; Drewe, J.; Patrek, V.; Pearce, G.; Samuel, M.D.; Delahay, R.J. Wildlife Population Structure and Parasite Transmission: Implications for Disease Management. In Management of Disease in Wild Mammals; Delahay, R.J., Smith, G.C., Hutchings, M.R., Eds.; Springer: Tokyo, Japan, 2009; pp. 9–29. ISBN 978-4-431-77134-0. [Google Scholar]
- White, L.A.; Forester, J.D.; Craft, M.E. Using Contact Networks to Explore Mechanisms of Parasite Transmission in Wildlife. Biol. Rev. 2017, 92, 389–409. [Google Scholar] [CrossRef] [PubMed]
- Croft, D.P.; James, R.; Krause, J. Exploring Animal Social Networks; Princeton University Press: Princeton, NJ, USA, 2008; ISBN 978-0-691-12752-1. [Google Scholar]
- Krause, J.; Lusseau, D.; James, R. Animal Social Networks: An Introduction. Behav. Ecol. Sociobiol. 2009, 63, 967–973. [Google Scholar] [CrossRef]
- Sih, A.; Hanser, S.F.; McHugh, K.A. Social Network Theory: New Insights and Issues for Behavioral Ecologists. Behav. Ecol. Sociobiol. 2009, 63, 975–988. [Google Scholar] [CrossRef]
- Brent, L.J.N. Friends of Friends: Are Indirect Connections in Social Networks Important to Animal Behaviour? Anim. Behav. 2015, 103, 211–222. [Google Scholar] [CrossRef]
- Silk, M.J.; Croft, D.P.; Delahay, R.J.; Hodgson, D.J.; Boots, M.; Weber, N.; McDonald, R.A. Using Social Network Measures in Wildlife Disease Ecology, Epidemiology, and Management. BioScience 2017, 67, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, S.S.; Bull, C.M.; James, R.; Murray, K. Network Structure and Parasite Transmission in a Group Living Lizard, the Gidgee Skink, Egernia stokesii. Behav. Ecol. Sociobiol. 2009, 63, 1045–1056. [Google Scholar] [CrossRef]
- Drewe, J.A. Who Infects Whom? Social Networks and Tuberculosis Transmission in Wild Meerkats. Proc. R. Soc. B Biol. Sci. 2010, 277, 633–642. [Google Scholar] [CrossRef]
- Williams, A.E.; Worsley-Tonks, K.E.L.; Ezenwa, V.O. Drivers and Consequences of Variation in Individual Social Connectivity. Anim. Behav. 2017, 133, 1–9. [Google Scholar] [CrossRef]
- Fenner, A.L.; Godfrey, S.S.; Bull, C.M. Using Social Networks to Deduce Whether Residents or Dispersers Spread Parasites in a Lizard Population. J. Anim. Ecol. 2011, 80, 835–843. [Google Scholar] [CrossRef]
- Bull, C.M.; Godfrey, S.S.; Gordon, D.M. Social Networks and the Spread of Salmonella in a Sleepy Lizard Population. Mol. Ecol. 2012, 21, 4386–4392. [Google Scholar] [CrossRef]
- Whitehead, H. SOCPROG Programs: Analysing Animal Social Structures. Behav. Ecol. Sociobiol. 2009, 63, 765–778. [Google Scholar] [CrossRef]
- Freeman, L.C.; Borgatti, S.P.; White, D.R. Centrality in Valued Graphs: A Measure of Betweenness Based on Network Flow. Soc. Netw. 1991, 13, 141–154. [Google Scholar] [CrossRef]
- Wilson-Aggarwal, J.K.; Ozella, L.; Tizzoni, M.; Cattuto, C.; Swan, G.J.F.; Moundai, T.; Silk, M.J.; Zingeser, J.A.; McDonald, R.A. High-Resolution Contact Networks of Free-Ranging Domestic Dogs Canis familiaris and Implications for Transmission of Infection. PLoS Negl. Trop. Dis. 2019, 13, e0007565. [Google Scholar] [CrossRef]
- Weber, N.; Carter, S.P.; Dall, S.R.X.; Delahay, R.J.; McDonald, J.L.; Bearhop, S.; McDonald, R.A. Badger Social Networks Correlate with Tuberculosis Infection. Curr. Biol. 2013, 23, R915–R916. [Google Scholar] [CrossRef]
- Corner, L.A.L.; Pfeiffer, D.U.; Morris, R.S. Social-Network Analysis of Mycobacterium bovis Transmission among Captive Brushtail Possums (Trichosurus vulpecula). Prev. Vet. Med. 2003, 59, 147–167. [Google Scholar] [CrossRef]
- Wolfe, N.D.; Escalante, A.A.; Karesh, W.B.; Kilbourn, A.; Spielman, A.; Lal, A.A. Wild Primate Populations in Emerging Infectious Disease Research: The Missing Link? Emerg. Infect. Dis. 1998, 4, 149–158. [Google Scholar] [CrossRef]
- Wallis, J.; Lee, D.R. Primate Conservation: The Prevention of Disease Transmission. Int. J. Primatol. 1999, 20, 803–826. [Google Scholar] [CrossRef]
- Chapman, C.A.; Gillespie, T.R.; Goldberg, T.L. Primates and the Ecology of Their Infectious Diseases: How Will Anthropogenic Change Affect Host-Parasite Interactions? Evol. Anthropol. Issues News Rev. 2005, 14, 134–144. [Google Scholar] [CrossRef]
- Leendertz, F.H.; Pauli, G.; Maetz-Rensing, K.; Boardman, W.; Nunn, C.; Ellerbrok, H.; Jensen, S.A.; Junglen, S.; Christophe, B. Pathogens as Drivers of Population Declines: The Importance of Systematic Monitoring in Great Apes and Other Threatened Mammals. Biol. Conserv. 2006, 131, 325–337. [Google Scholar] [CrossRef]
- Formenty, P.; Boesch, C.; Wyers, M.; Steiner, C.; Donati, F.; Dind, F.; Walker, F.; Le Guenno, B. Ebola Virus Outbreak among Wild Chimpanzees Living in a Rain Forest of Côte d’Ivoire. J. Infect. Dis. 1999, 179, S120–S126. [Google Scholar] [CrossRef]
- Huijbregts, B.; Wachter, P.D.; Obiang, L.S.N.; Akou, M.E. Ebola and the Decline of Gorilla Gorilla gorilla and Chimpanzee Pan troglodytes Populations in Minkebe Forest, North-Eastern Gabon. Oryx 2003, 37, 437–443. [Google Scholar] [CrossRef]
- Walsh, P.D.; Abernethy, K.A.; Bermejo, M.; Beyers, R.; De Wachter, P.; Akou, M.E.; Huijbregts, B.; Mambounga, D.I.; Toham, A.K.; Kilbourn, A.M.; et al. Catastrophic Ape Decline in Western Equatorial Africa. Nature 2003, 422, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Bermejo, M.; Rodríguez-Teijeiro, J.D.; Illera, G.; Barroso, A.; Vilà, C.; Walsh, P.D. Ebola Outbreak Killed 5000 Gorillas. Science 2006, 314, 1564. [Google Scholar] [CrossRef] [PubMed]
- Caillaud, D.; Levréro, F.; Cristescu, R.; Gatti, S.; Dewas, M.; Douadi, M.; Gautier-Hion, A.; Raymond, M.; Ménard, N. Gorilla Susceptibility to Ebola Virus: The Cost of Sociality. Curr. Biol. 2006, 13, R489–R491. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, N.C.C.d.A.; Cunha, M.S.; Guerra, J.M.; Réssio, R.A.; Cirqueira, C.d.S.; Iglezias, S.D.; de Carvalho, J.; Araujo, E.L.L.; Catão-Dias, J.L.; Díaz-Delgado, J. Outbreak of Yellow Fever among Nonhuman Primates, Espirito Santo, Brazil, 2017. Emerg. Infect. Dis. 2017, 23, 2038–2041. [Google Scholar] [CrossRef]
- Mares-Guia, M.A.M.d.M.; Horta, M.A.; Romano, A.; Rodrigues, C.D.S.; Mendonça, M.C.L.; dos Santos, C.C.; Torres, M.C.; Araujo, E.S.M.; Fabri, A.; de Souza, E.R.; et al. Yellow Fever Epizootics in Non-Human Primates, Southeast and Northeast Brazil (2017 and 2018). Parasites Vectors 2020, 13, 90. [Google Scholar] [CrossRef]
- Kalema-Zikusoka, G.; Kock, R.A.; Macfie, E.J. Scabies in Free-Ranging Mountain Gorillas (Gorilla beringei beringei) in Bwindi Impenetrable National Park, Uganda. Vet. Rec. 2002, 150, 12–15. [Google Scholar] [CrossRef]
- Ferber, D. Human Diseases Threaten Great Apes. Science 2000, 289, 1277–1278. [Google Scholar] [CrossRef]
- Kaur, T.; Singh, J.; Tong, S.; Humphrey, C.; Clevenger, D.; Tan, W.; Szekely, B.; Wang, Y.; Li, Y.; Muse, E.A.; et al. Descriptive Epidemiology of Fatal Respiratory Outbreaks and Detection of a Human-Related Metapneumovirus in Wild Chimpanzees (Pan troglodytes) at Mahale Mountains National Park, Western Tanzania. Am. J. Primatol. 2008, 70, 755–765. [Google Scholar] [CrossRef]
- Köndgen, S.; Kühl, H.; N’Goran, P.K.; Walsh, P.D.; Schenk, S.; Ernst, N.; Biek, R.; Formenty, P.; Mätz-Rensing, K.; Schweiger, B.; et al. Pandemic Human Viruses Cause Decline of Endangered Great Apes. Curr. Biol. 2008, 18, 260–264. [Google Scholar] [CrossRef]
- Williams, J.M.; Lonsdorf, E.V.; Wilson, M.L.; Schumacher-Stankey, J.; Goodall, J.; Pusey, A.E. Causes of Death in the Kasekela Chimpanzees of Gombe National Park, Tanzania. Am. J. Primatol. 2008, 70, 766–777. [Google Scholar] [CrossRef] [PubMed]
- Palacios, G.; Lowenstine, L.J.; Cranfield, M.R.; Gilardi, K.V.K.; Spelman, L.; Lukasik-Braum, M.; Kinani, J.-F.; Mudakikwa, A.; Nyirakaragire, E.; Bussetti, A.V.; et al. Human Metapneumovirus Infection in Wild Mountain Gorillas, Rwanda. Emerg. Infect. Dis. 2011, 17, 711–713. [Google Scholar] [CrossRef] [PubMed]
- Grützmacher, K.S.; Köndgen, S.; Keil, V.; Todd, A.; Feistner, A.; Herbinger, I.; Petrzelkova, K.; Fuh, T.; Leendertz, S.A.; Calvignac-Spencer, S.; et al. Codetection of Respiratory Syncytial Virus in Habituated Wild Western Lowland Gorillas and Humans during a Respiratory Disease Outbreak. EcoHealth 2016, 13, 499–510. [Google Scholar] [CrossRef]
- Scully, E.J.; Basnet, S.; Wrangham, R.W.; Muller, M.N.; Otali, E.; Hyeroba, D.; Grindle, K.A.; Pappas, T.E.; Thompson, M.E.; Machanda, Z.; et al. Lethal Respiratory Disease Associated with Human Rhinovirus C in Wild Chimpanzees, Uganda, 2013. Emerg. Infect. Dis. 2018, 24, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Negrey, J.D.; Reddy, R.B.; Scully, E.J.; Phillips-Garcia, S.; Owens, L.A.; Langergraber, K.E.; Mitani, J.C.; Thompson, M.E.; Wrangham, R.W.; Muller, M.N.; et al. Simultaneous Outbreaks of Respiratory Disease in Wild Chimpanzees Caused by Distinct Viruses of Human Origin. Emerg. Microbes Infect. 2019, 8, 139–149. [Google Scholar] [CrossRef]
- Yoshida, T.; Takemoto, H.; Sakamaki, T.; Tokuyama, N.; Hart, J.; Hart, T.; Dupain, J.; Cobden, A.; Mulavwa, M.; Hashimoto, C.; et al. Prevalence of Antibodies against Human Respiratory Viruses Potentially Involving Anthropozoonoses in Wild Bonobos. Primates 2021, 62, 897–903. [Google Scholar] [CrossRef]
- Rimbach, R.; Bisanzio, D.; Galvis, N.; Link, A.; Di Fiore, A.; Gillespie, T.R. Brown Spider Monkeys (Ateles hybridus): A Model for Differentiating the Role of Social Networks and Physical Contact on Parasite Transmission Dynamics. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140110. [Google Scholar] [CrossRef]
- Balasubramaniam, K.N.; Beisner, B.A.; Hubbard, J.A.; Vandeleest, J.J.; Atwill, E.R.; McCowan, B. Affiliation and Disease Risk: Social Networks Mediate Gut Microbial Transmission among Rhesus Macaques. Anim. Behav. 2019, 151, 131–143. [Google Scholar] [CrossRef] [PubMed]
- MacIntosh, A.J.J.; Jacobs, A.; Garcia, C.; Shimizu, K.; Mouri, K.; Huffman, M.A.; Hernandez, A.D. Monkeys in the Middle: Parasite Transmission through the Social Network of a Wild Primate. PLoS ONE 2012, 7, e51144. [Google Scholar] [CrossRef]
- Friant, S.; Ziegler, T.E.; Goldberg, T.L. Primate Reinfection with Gastrointestinal Parasites: Behavioural and Physiological Predictors of Parasite Acquisition. Anim. Behav. 2016, 117, 105–113. [Google Scholar] [CrossRef]
- Deere, J.R.; Schaber, K.L.; Foerster, S.; Gilby, I.C.; Feldblum, J.T.; VanderWaal, K.; Wolf, T.M.; Travis, D.A.; Raphael, J.; Lipende, I.; et al. Gregariousness Is Associated with Parasite Species Richness in a Community of Wild Chimpanzees. Behav. Ecol. Sociobiol. 2021, 75, 87. [Google Scholar] [CrossRef] [PubMed]
- Spelman, L.H.; Gilardi, K.V.K.; Lukasik-Braum, M.; Kinani, J.-F.; Nyirakaragire, E.; Lowenstine, L.J.; Cranfield, M.R. Respiratory Disease in Mountain Gorillas (Gorilla beringei beringei) in Rwanda, 1990–2010: Outbreaks, Clinical Course, and Medical Management. J. Zoo Wildl. Med. 2013, 44, 1027–1035. [Google Scholar] [CrossRef]
- Thompson, M.E.; Machanda, Z.P.; Scully, E.J.; Enigk, D.K.; Otali, E.; Muller, M.N.; Goldberg, T.L.; Chapman, C.A.; Wrangham, R.W. Risk Factors for Respiratory Illness in a Community of Wild Chimpanzees (Pan troglodytes schweinfurthii). R. Soc. Open Sci. 2018, 5, 180840. [Google Scholar] [CrossRef] [PubMed]
- Lonsdorf, E.V.; Gillespie, T.R.; Wolf, T.M.; Lipende, I.; Raphael, J.; Bakuza, J.; Murray, C.M.; Wilson, M.L.; Kamenya, S.; Mjungu, D.; et al. Socioecological Correlates of Clinical Signs in Two Communities of Wild Chimpanzees (Pan troglodytes) at Gombe National Park, Tanzania. Am. J. Primatol. 2018, 80, e22562. [Google Scholar] [CrossRef]
- Morrison, R.E.; Mushimiyimana, Y.; Stoinski, T.S.; Eckardt, W. Rapid Transmission of Respiratory Infections within but Not between Mountain Gorilla Groups. Sci. Rep. 2021, 11, 19622. [Google Scholar] [CrossRef] [PubMed]
- Sandel, A.A.; Rushmore, J.; Negrey, J.D.; Mitani, J.C.; Lyons, D.M.; Caillaud, D. Social Network Predicts Exposure to Respiratory Infection in a Wild Chimpanzee Group. EcoHealth 2020, 17, 437–448. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex Differences in Immune Responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef]
- Krasnov, B.R.; Bordes, F.; Khokhlova, I.S.; Morand, S. Gender-Biased Parasitism in Small Mammals: Patterns, Mechanisms, Consequences. Mammalia 2012, 76, 1–13. [Google Scholar] [CrossRef]
- Metcalf, C.J.E.; Roth, O.; Graham, A.L. Why Leveraging Sex Differences in Immune Trade-Offs May Illuminate the Evolution of Senescence. Funct. Ecol. 2020, 34, 129–140. [Google Scholar] [CrossRef]
- Poulin, R. Sexual Inequalities in Helminth Infections: A Cost of Being a Male? Am. Nat. 1996, 147, 287–295. [Google Scholar] [CrossRef]
- Poulin, R. Helminth Growth in Vertebrate Hosts: Does Host Sex Matter? Int. J. Parasitol. 1996, 26, 1311–1315. [Google Scholar] [CrossRef]
- Schalk, G.; Forbes, M.R. Male Biases in Parasitism of Mammals: Effects of Study Type, Host Age, and Parasite Taxon. Oikos 1997, 78, 67–74. [Google Scholar] [CrossRef]
- Fischer, J.; Jung, N.; Robinson, N.; Lehmann, C. Sex Differences in Immune Responses to Infectious Diseases. Infection 2015, 43, 399–403. [Google Scholar] [CrossRef]
- Kelly, C.D.; Stoehr, A.M.; Nunn, C.; Smyth, K.N.; Prokop, Z.M. Sexual Dimorphism in Immunity across Animals: A Meta-Analysis. Ecol. Lett. 2018, 21, 1885–1894. [Google Scholar] [CrossRef] [PubMed]
- Zuk, M. The Sicker Sex. PLoS Pathog. 2009, 5, e1000267. [Google Scholar] [CrossRef]
- Klein, S.L. The Effects of Hormones on Sex Differences in Infection: From Genes to Behavior. Neurosci. Biobehav. Rev. 2000, 24, 627–638. [Google Scholar] [CrossRef]
- East, M.L.; Wibbelt, G.; Lieckfeldt, D.; Ludwig, A.; Goller, K.; Wilhelm, K.; Schares, G.; Thierer, D.; Hofer, H. A Hepatozoon Species Genetically Distinct from H. canis Infecting Spotted Hyenas in the Serengeti Ecosystem, Tanzania. J. Wildl. Dis. 2008, 44, 45–52. [Google Scholar] [CrossRef]
- Ebersole, J.L.; Steffen, M.J.; Gonzalez-Martinez, J.; Novak, M.J. Effects of Age and Oral Disease on Systemic Inflammatory and Immune Parameters in Nonhuman Primates. Clin. Vaccine Immunol. 2008, 15, 1067–1075. [Google Scholar] [CrossRef]
- Fallon, P.G.; Gibbons, J.; Vervenne, R.A.; Richardson, E.J.; Fulford, A.J.C.; Kiarie, S.; Sturrock, R.F.; Coulson, P.S.; Deelder, A.M.; Langermans, J.A.M.; et al. Juvenile Rhesus Monkeys Have Lower Type 2 Cytokine Responses than Adults after Primary Infection with Schistosoma mansoni. J. Infect. Dis. 2003, 187, 939–945. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Simon, A.K.; Hollander, G.A.; McMichael, A. Evolution of the Immune System in Humans from Infancy to Old Age. Proc. R. Soc. B Biol. Sci. 2015, 282, 20143085. [Google Scholar] [CrossRef]
- Asquith, M.; Haberthur, K.; Brown, M.; Engelmann, F.; Murphy, A.; Al-Mahdi, Z.; Messaoudi, I. Age-Dependent Changes in Innate Immune Phenotype and Function in Rhesus Macaques (Macaca mulatta). Pathobiol. Aging Age-Relat. Dis. 2012, 2, 18052. [Google Scholar] [CrossRef] [PubMed]
- Cheynel, L.; Lemaître, J.-F.; Gaillard, J.-M.; Rey, B.; Bourgoin, G.; Ferté, H.; Jégo, M.; Débias, F.; Pellerin, M.; Jacob, L.; et al. Immunosenescence Patterns Differ between Populations but Not between Sexes in a Long-Lived Mammal. Sci. Rep. 2017, 7, 13700. [Google Scholar] [CrossRef]
- Cichoń, M.; Sendecka, J.; Gustafsson, L. Age-Related Decline in Humoral Immune Function in Collared Flycatchers. J. Evol. Biol. 2003, 16, 1205–1210. [Google Scholar] [CrossRef] [PubMed]
- Nussey, D.H.; Watt, K.; Pilkington, J.G.; Zamoyska, R.; McNeilly, T.N. Age-Related Variation in Immunity in a Wild Mammal Population. Aging Cell 2012, 11, 178–180. [Google Scholar] [CrossRef]
- Ujvari, B.; Madsen, T. Age, Parasites, and Condition Affect Humoral Immune Response in Tropical Pythons. Behav. Ecol. 2006, 17, 20–24. [Google Scholar] [CrossRef]
- Wilson, K.; Bjørnstad, O.N.; Dobson, A.P.; Merler, S.; Poglayen, G.; Randolph, S.E.; Read, A.F.; Skorping, A. Heterogeneities in Macroparasite Infections: Patterns and Processes. In The Ecology of Wildlife Diseases; Oxford University Press: Oxford, UK, 2002; pp. 6–44. [Google Scholar]
- Ryu, H.; Hill, D.A.; Sakamaki, T.; Garai, C.; Tokuyama, N.; Furuichi, T. Occurrence and Transmission of Flu-like Illness among Neighboring Bonobo Groups at Wamba. Primates 2020, 61, 775–784. [Google Scholar] [CrossRef]
- Davies, T.J.; Pedersen, A.B. Phylogeny and Geography Predict Pathogen Community Similarity in Wild Primates and Humans. Proc. R. Soc. B Biol. Sci. 2008, 275, 1695–1701. [Google Scholar] [CrossRef]
- Dunay, E.; Apakupakul, K.; Leard, S.; Palmer, J.L.; Deem, S.L. Pathogen Transmission from Humans to Great Apes Is a Growing Threat to Primate Conservation. EcoHealth 2018, 15, 148–162. [Google Scholar] [CrossRef]
- Grützmacher, K.S.; Keil, V.; Metzger, S.; Wittiger, L.; Herbinger, I.; Calvignac-Spencer, S.; Mätz-Rensing, K.; Haggis, O.; Savary, L.; Köndgen, S.; et al. Human Respiratory Syncytial Virus and Streptococcus Pneumoniae Infection in Wild Bonobos. EcoHealth 2018, 15, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Patrono, L.V.; Samuni, L.; Corman, V.M.; Nourifar, L.; Röthemeier, C.; Wittig, R.M.; Drosten, C.; Calvignac-Spencer, S.; Leendertz, F.H. Human Coronavirus OC43 Outbreak in Wild Chimpanzees, Côte D’Ivoire, 2016. Emerg. Microbes Infect. 2018, 7, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Slater, O.M.; Terio, K.A.; Zhang, Y.; Erdman, D.D.; Schneider, E.; Kuypers, J.M.; Wolinsky, S.M.; Kunstman, K.J.; Kunstman, J.; Kinsel, M.J.; et al. Human Metapneumovirus Infection in Chimpanzees, United States. Emerg. Infect. Dis. 2014, 20, 2115–2118. [Google Scholar] [CrossRef]
- Szentiks, C.A.; Köndgen, S.; Silinski, S.; Speck, S.; Leendertz, F.H. Lethal Pneumonia in a Captive Juvenile Chimpanzee (Pan troglodytes) Due to Human-Transmitted Human Respiratory Syncytial Virus (HRSV) and Infection with Streptococcus pneumoniae. J. Med. Primatol. 2009, 38, 236–240. [Google Scholar] [CrossRef]
- Unwin, S.; Chatterton, J.; Chantrey, J. Management of Severe Respiratory Tract Disease Caused by Human Respiratory Syncytial Virus and Streptococcus pneumoniae in Captive Chimpanzees (Pan troglodytes). J. Zoo Wildl. Med. 2013, 44, 105–115. [Google Scholar] [CrossRef]
- Lowenstine, L.J.; McManamon, R.; Terio, K.A. Comparative Pathology of Aging Great Apes: Bonobos, Chimpanzees, Gorillas, and Orangutans. Vet. Pathol. 2016, 53, 250–276. [Google Scholar] [CrossRef] [PubMed]
- Furuichi, T.; Idani, G.; Ihobe, H.; Kuroda, S.; Kitamura, K.; Mori, A.; Enomoto, T.; Okayasu, N.; Hashimoto, C.; Kano, T. Population Dynamics of Wild Bonobos (Pan paniscus) at Wamba. Int. J. Primatol. 1998, 19, 1029–1043. [Google Scholar] [CrossRef]
- Kano, T. The Last Ape: Pygmy Chimpanzee Behavior and Ecology; Stanford University Press: Stanford, CA, USA, 1992. [Google Scholar]
- Xu, Z.; MacIntosh, A.J.J.; Castellano-Navarro, A.; Macanás-Martínez, E.; Suzumura, T.; Duboscq, J. Linking Parasitism to Network Centrality and the Impact of Sampling Bias in Its Interpretation. bioRxiv 2021. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, W.; Chan, P.-T.; Yen, H.-L.; Tang, J.W.-T.; Li, Y. Close Contact Behavior in Indoor Environment and Transmission of Respiratory Infection. Indoor Air 2020, 30, 645–661. [Google Scholar] [CrossRef] [PubMed]
- Altmann, J. Observational Study of Behavior: Sampling Methods. Behaviour 1974, 49, 227–266. [Google Scholar] [CrossRef] [PubMed]
- Sosa, S.; Puga-Gonzalez, I.; Hu, F.; Pansanel, J.; Xie, X.; Sueur, C. A Multilevel Statistical Toolkit to Study Animal Social Networks: The Animal Network Toolkit Software (ANTs) R Package. Sci. Rep. 2020, 10, 12507. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Sosa, S.; Sueur, C.; Puga-Gonzalez, I. Network Measures in Animal Social Network Analysis: Their Strengths, Limits, Interpretations and Uses. Methods Ecol. Evol. 2021, 12, 10–21. [Google Scholar] [CrossRef]
- Webber, Q.M.R.; Schneider, D.C.; Vander Wal, E. Is Less More? A Commentary on the Practice of ‘Metric Hacking’ in Animal Social Network Analysis. Anim. Behav. 2020, 168, 109–120. [Google Scholar] [CrossRef]
- VanderWaal, K.L.; Enns, E.A.; Picasso, C.; Packer, C.; Craft, M.E. Evaluating Empirical Contact Networks as Potential Transmission Pathways for Infectious Diseases. J. R. Soc. Interface 2016, 13, 20160166. [Google Scholar] [CrossRef] [PubMed]
- Farine, D.R. A Guide to Null Models for Animal Social Network Analysis. Methods Ecol. Evol. 2017, 8, 1309–1320. [Google Scholar] [CrossRef] [PubMed]
- Franks, D.W.; Weiss, M.N.; Silk, M.J.; Perryman, R.J.Y.; Croft, D.P. Calculating Effect Sizes in Animal Social Network Analysis. Methods Ecol. Evol. 2021, 12, 33–41. [Google Scholar] [CrossRef]
- Kawazoe, T.; Sosa, S. Social Networks Predict Immigration Success in Wild Japanese Macaques. Primates 2019, 60, 213–222. [Google Scholar] [CrossRef]
- Csardi, G.; Nepusz, T. The Igraph Software Package for Complex Network Research. 2006. Available online: https://www.semanticscholar.org/paper/The-igraph-software-package-for-complex-network-Cs%C3%A1rdi-Nepusz/1d2744b83519657f5f2610698a8ddd177ced4f5c (accessed on 16 December 2021).
- Harcourt, A.H. Social Relationships among Adult Female Mountain Gorillas. Anim. Behav. 1979, 27, 251–264. [Google Scholar] [CrossRef]
- Voelkl, B.; Kasper, C.; Schwab, C. Network Measures for Dyadic Interactions: Stability and Reliability. Am. J. Primatol. 2011, 73, 731–740. [Google Scholar] [CrossRef]
- Barber, I.; Dingemanse, N.J. Parasitism and the Evolutionary Ecology of Animal Personality. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 4077–4088. [Google Scholar] [CrossRef]
- Santicchia, F.; Romeo, C.; Ferrari, N.; Matthysen, E.; Vanlauwe, L.; Wauters, L.A.; Martinoli, A. The Price of Being Bold? Relationship between Personality and Endoparasitic Infection in a Tree Squirrel. Mamm. Biol. 2019, 97, 1–8. [Google Scholar] [CrossRef]
- Paquette, C.; Garant, D.; Savage, J.; Réale, D.; Bergeron, P. Individual and Environmental Determinants of Cuterebra Bot Fly Parasitism in the Eastern Chipmunk (Tamias striatus). Oecologia 2020, 193, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Vanden Broecke, B.; Bernaerts, L.; Ribas, A.; Sluydts, V.; Mnyone, L.; Matthysen, E.; Leirs, H. Linking Behavior, Co-Infection Patterns, and Viral Infection Risk with the Whole Gastrointestinal Helminth Community Structure in Mastomys natalensis. Front. Vet. Sci. 2021, 8, 912. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torfs, J.R.R.; Eens, M.; Laméris, D.W.; Staes, N. Respiratory Disease Risk of Zoo-Housed Bonobos Is Associated with Sex and Betweenness Centrality in the Proximity Network. Animals 2021, 11, 3597. https://doi.org/10.3390/ani11123597
Torfs JRR, Eens M, Laméris DW, Staes N. Respiratory Disease Risk of Zoo-Housed Bonobos Is Associated with Sex and Betweenness Centrality in the Proximity Network. Animals. 2021; 11(12):3597. https://doi.org/10.3390/ani11123597
Chicago/Turabian StyleTorfs, Jonas R. R., Marcel Eens, Daan W. Laméris, and Nicky Staes. 2021. "Respiratory Disease Risk of Zoo-Housed Bonobos Is Associated with Sex and Betweenness Centrality in the Proximity Network" Animals 11, no. 12: 3597. https://doi.org/10.3390/ani11123597
APA StyleTorfs, J. R. R., Eens, M., Laméris, D. W., & Staes, N. (2021). Respiratory Disease Risk of Zoo-Housed Bonobos Is Associated with Sex and Betweenness Centrality in the Proximity Network. Animals, 11(12), 3597. https://doi.org/10.3390/ani11123597






