Performance, Carcass Yield, Muscle Amino Acid Profile, and Levels of Brain Neurotransmitters in Aged Laying Hens Fed Diets Supplemented with Guanidinoacetic Acid
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Birds and Management
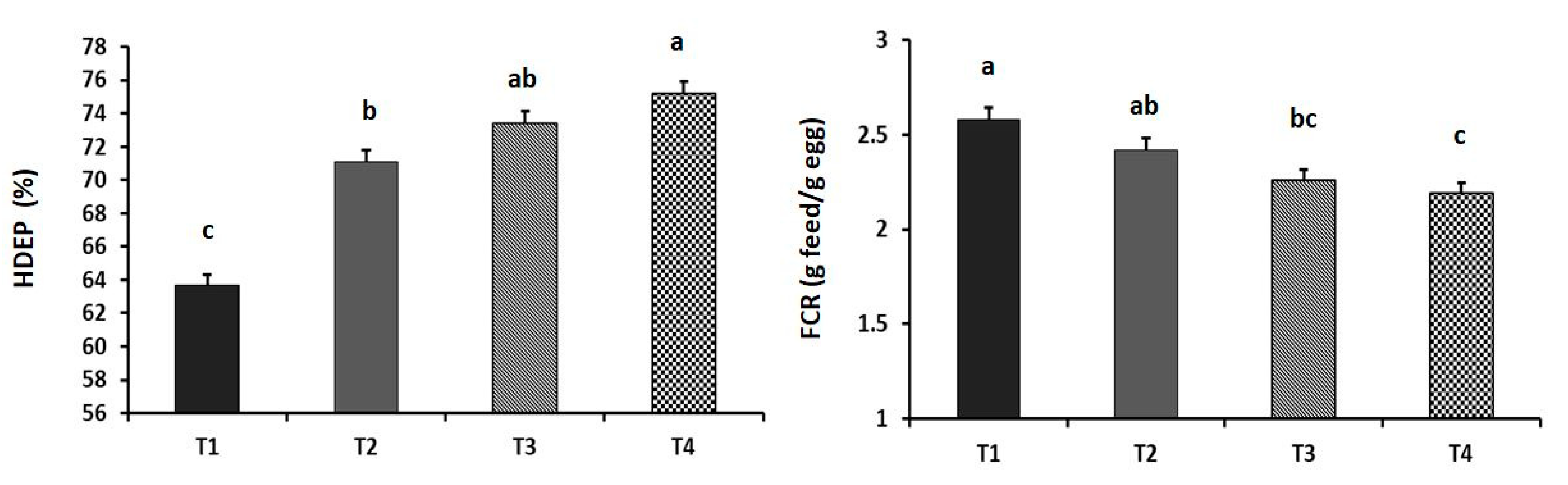
2.2. Laying Performance
2.3. Blood Sampling and Serum Biochemical Parameters
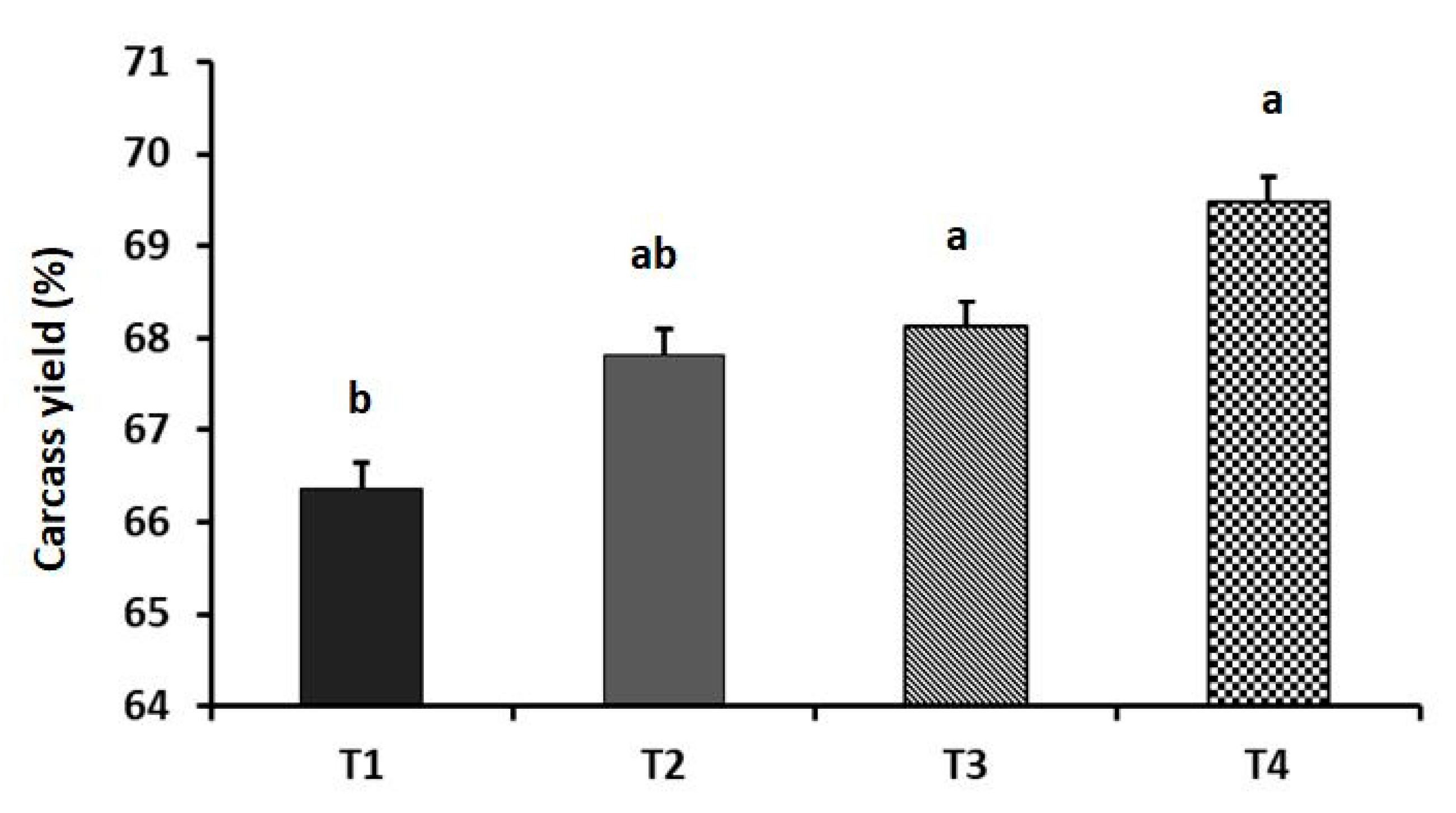
2.4. Carcass Yield and Muscle Amino Acid Profile
2.5. Nitric Oxide and Monoamine Concentrations in Brain Tissues
2.6. Statistical Analyses
- Yij = the dependent variable;
- μ = the population mean;
- Ti = the fixed effect of GA dietary supplements (i = T1, T2, T3, and T4);
- eij = random error, assumed to be normally and independently distributed.
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abudabos, A.M.; Saleh, F.; Lemme, A.; Zakaria, H.A. The relationship between guanidino acetic acid and metabolisable energy level of diets on performance of broiler chickens. Ital. J. Anim. Sci. 2014, 13, 3269. [Google Scholar] [CrossRef]
- Michiels, J.; Maertens, L.; Buyse, J.; Lemme, A.; Rademacher, M.; DIerick, N.A.; Desmet, S. Supplementation of guanininoacetic acid to broiler diets: Effects on performance, carcass characteristics, meat quality and energy metabolism. Poult. Sci. 2012, 91, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Wyss, M.; Kaddurah-Daouk, R. Creatine and creatinine metabolism. Physiol. Rev. 2000, 80, 1107–1213. [Google Scholar] [CrossRef]
- Lemme, A.; Ringel, J.; Sterk, A.; Young, J.F. Supplemental guanidino acetic acid affect energy metabolism of broiler. In Proceedings of the 16th European Symposium on Poultry Nutrition, Strasbourg, France, 26–30 August 2007; pp. 339–342. [Google Scholar]
- Mousavi, S.N.; Afsar, A.; Lotfollahian, H. Effects of guanidinoacetic acid supplementation to broiler diets with varying energy contents. J. Appl. Poult. Res. 2013, 22, 47–54. [Google Scholar] [CrossRef]
- Meyer, L.E.; Machado, L.B.; Santiago, A.P.S.; da-Silva, W.S.; de Felice, F.G.; Holub, O.; Oliveira, M.F.; Galina, A. Mitochondrial creatine kinase activity prevents reactive oxygen species generation antioxidant role of mitochondrial kinase-dependent ADP re-cycling activity. J. Biol. Chem. 2006, 281, 37361.e71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murakami, A.E.; Rodrigueiro, R.J.B.; Santos, T.C.; Ospina-Rojas, I.C.; Rademacher, M. Effects of dietary supplementation of meat-type quail breeders with guanidinoacetic acid on their reproductive parameters and progeny performance. Poult. Sci. 2014, 93, 2237–2244. [Google Scholar] [CrossRef]
- Khakran, G.; Chamani, M.; Foroudi, F.; Sadeghi, A.A.; Afshar, M.A. Effect of guanidine acetic acid addition to corn-soybean meal based diets on productive performance, blood biochemical parameters and reproductive hormones of laying hens. Kafkas. Univ. Vet. Fak. Derg. 2018, 24, 99–105. [Google Scholar]
- Smith, R.N.; Agharkar, A.S.; Gonzales, E.B. A review of creatine supplementation in age-related diseases: More than a supplement for athletes. F1000Research 2014, 3, 222. [Google Scholar] [CrossRef] [Green Version]
- Rae, C.D.; Bröer, S. Creatine as a booster for human brain function. How might it work? Neurochem. Int. 2015, 89, 249–259. [Google Scholar] [CrossRef]
- Chung, Y.L.; Alexanderson, H.; Pipitone, N. Creatine supplements in patients with idiopathic inflammatory myopathies who are clinically weak after conventional pharmacologic treatment: Six-month, double-blind, randomized, placebo-controlled trial. Arthritis Rheumatol. 2007, 57, 694–702. [Google Scholar] [CrossRef]
- Beal, F.M. Neuroprotective effects of creatine. Amino. Acids. 2011, 40, 1305–1313. [Google Scholar] [CrossRef]
- Abasht, B.; Mutryn, M.F.; Michalek, R.D.; Lee, W.R. Oxidative stress and metabolic perturbations in wooden breast disorder in chickens. PLoS ONE 2016, 11, e0153750. [Google Scholar] [CrossRef] [Green Version]
- Mueller, S.; Kreuzer, M.; Messikommer, R.E.; Gangnat, I.D. Carcass and Meat Quality of Spent Hens from Dual Purpose vs Layer Genotypes When Fed a By-Product Diet. In Proceedings of the International Congress of Meat Science and Technology (62nd ICoMST), Bangkok, Thailand, 14–16 August 2016; pp. 12–15. [Google Scholar]
- Bhattacharyya, D.; SInhamahapatra, M.; Biswas, S. Preparation of sausage from spent duck–an acceptability study. Int. J. Food Sci. Technol. 2007, 42, 24–29. [Google Scholar] [CrossRef]
- El-Tarabany, M.S.; Ahmed-Farid, O.A. Effect of age at the onset of natural molting on carcass traits, muscle oxidative stability, and amino acid and fatty acid profiles in commercial laying hens. LWT 2021, 147, 111627. [Google Scholar] [CrossRef]
- de Souza, K.M.R.; Araujo, R.B.; dos Santos, A.L.; Rodrigues, C.E.C.; de Faria, D.E.; Trindade, M.A. Adding value to the meat of spent laying hens manufacturing sausages with a healthy appeal. Rev. Bras. D Ciência. Avícola. 2011, 13, 57–63. [Google Scholar] [CrossRef] [Green Version]
- National Research Council. Nutrient Requirements of Domestic Animals. In Nutrient Requirements of Poultry, 9th ed.; National Academy of Science: Washington, DC, USA, 1994. [Google Scholar]
- JAKIM (Department of Islamic Development Malaysia). Malaysian Protocol for the Halal Meat and Poultry Productions; JAKIM: Putrajaya, Malaysia, 2011; pp. 1–30. [Google Scholar]
- Hughes, M.C.; Kerry, J.P.; Arendt, E.K.; Kenneally, P.M.; McSweeney, P.L.H.; O’Neill, E.E. Characterization of proteolysis during the ripening of semi-dry fermented sausages. Meat Sci. 2002, 62, 205–216. [Google Scholar] [CrossRef]
- Arafa, N.M.S.; Salem, S.M.A.; Farid, O.A.H.A. Influence of echinacea extract pre- or postnatal supplementation on immune and oxidative status of growing rabbits. Ital. J. Anim. Sci. 2010, 9, 338–343. [Google Scholar]
- Pagel, P.; Blome, J.; Wolf, H.U. High-performance liquid chromatographic separation and measurement of various biogenic compounds possibly involved in the pathomechanism of Parkinson’s disease. J. Chromatogr. B 2000, 746, 297–304. [Google Scholar] [CrossRef]
- Papadoyannis, I.; Samanidou, V.; Nitsos, C.C. Simultaneous determination of nitrite and nitrate in drinking water and human serum by high performance anionexchange chromatography and UV detection. J. Liq. Chromatogr. Relat. Technol. 1999, 22, 2023–2041. [Google Scholar] [CrossRef]
- Dilger, R.N.; Bryant-Angeloni, K.; Payne, R.L.; Lemme, A.; Parsons, C.M. Dietary guanidino acetic acid is an efficacious replacement for arginine for young chicks. Poult. Sci. 2013, 92, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Morris, S.M., Jr. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 336, 1–17. [Google Scholar] [CrossRef] [PubMed]
- McCann, S.M.; Mastronardi, C.; Walczewska, A.; Karanth, S.; Rettori, V.; Yu, W.H. The role of nitric oxide in reproduction. Braz. J. Med. Biol. Res. 1999, 32, 1367–1379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basiouni, G.F.; Najib, H.; Zaki, M.M.; Al-Ankari, A.S. Influence of extra supplementation with arginine and lysine on overall Performance, ovarian activities and humoral immune response in local Saudi hens. Int. J. Poult. Sci. 2006, 5, 441–448. [Google Scholar]
- Ringel, J.; Lemme, A.; Knox, A.; McNab, J.; Redshaw, M.S. Effects of graded levels of creatine and guanidino acetic acid in vegetable-based diets on performance and biochemical parameters in muscle tissue. In Proceedings of the 16th European Symposium on Poultry Nutrition, Strasbourg, France, 26–30 August 2007; pp. 387–390. [Google Scholar]
- Tossenberger, J.; Rademacher, M.; Németh, K.; Halas, V.; Lemme, A. Digestibility and metabolism of dietary guanidino acetic acid fed to broilers. Poult. Sci. 2016, 95, 2058–2067. [Google Scholar] [CrossRef]
- Esser, A.F.G.; Gonçalves, D.R.M.; Rorig, A.; Cristo, A.B.; Perini, R.; Fernandes, J.I.M. Effects of guanidionoacetic acid and arginine supplementation to vegetable diets fed to broiler chickens subjected to heat stress before slaughter. Rev. Bras. D Ciência. Avícola. 2017, 19, 429–436. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Wang, M.; Kong, Y.; Ma, H.; Zou, S. Comparison of the novel compounds creatine and pyruvate on lipid and protein metabolism in broiler chickens. Animals 2011, 5, 1082–1089. [Google Scholar]
- Córdova-Noboa, H.A.; Oviedo-Rondón, E.O.; Sarsour, A.H.; Barnes, J.; Ferzola, P.; Rademacher-Heilshorn, M.; Braun, U. Performance, meat quality, and pectoral myopathies of broilers fed either corn or sorghum based diets supplemented with guanidinoacetic acid. Poult. Sci. 2018, 97, 2479–2493. [Google Scholar] [CrossRef]
- Majdeddin, M.; Golian, A.; Kermanshahi, H.; De Smet, S.; Michiels, J. Guanidinoacetic acid supplementation in broiler chickens fed corn-soybean diets affects performance in the finisher period and energy metabolites in breast muscle independent of diet nutrient density. Br. Poult. Sci. 2018, 59, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Reddy, G.B.; Mallika, E.N.; Reddy, B.O.; Azad, S.A.K.; Reddy, D.M. Comparison on meat quality characteristics of spent breder, layer and broiler birds. Int. J. Sci. Environ. Technol. 2016, 5, 2590–2595. [Google Scholar]
- Loetscher, Y.; Albiker, D.; Stephan, R.; Kreuzer, M.; Messikommer, R.E. Differences between spent hens of different genotype in performance, meat yield and suitability of the meat for sausage production. Animal 2015, 9, 347–355. [Google Scholar] [CrossRef]
- Tessari, P.; Lante, A.; Mosca, G. Essential amino acids: Master regulators of nutrition and environmental footprint? Sci. Rep. 2016, 6, 26074. [Google Scholar] [CrossRef] [Green Version]
- Haščík, P.; Trembecká, L.; Bobko, M.; Čuboň, J.; Kačániová, M.; Tkáčová, J. Amino acid profile of broiler chickens meat fed diets supplemented with bee pollen and propolis. J. Apic. Res. 2016, 55, 324–334. [Google Scholar] [CrossRef]
- Straková, E.; Suchý, P.A.; Vitula, F.R.; Večerek, V.L. Differences in the amino acid composition of muscles from pheasant and broiler chickens. Arch. Anim. Breed. 2006, 49, 508–514. [Google Scholar] [CrossRef] [Green Version]
- Ostojic, S.M.; Niess, B.; Stojanovic, M.D.; Idrizovic, K. Serum creatine, creatinine and total homocysteine concentration-time profiles after a single oral dose of guanidinoacetic acid in humans. J. Funct. Foods 2014, 6, 598–605. [Google Scholar] [CrossRef]
- Haščí k, P.; Pavelková, A.; Tkáčová, J.; Čuboň, J.; Bobko, M.; Kačániová, M.; Arpášová, H.; Čech, M. The amino acid profile after addition of humic acids and phytobiotics into diet of broiler chicken. Potravin. Slovak. J. Food Sci. 2019, 13, 884–890. [Google Scholar] [CrossRef] [Green Version]
- Stead, L.M.; Au, K.P.; Jacobs, R.L.; Brosnan, M.E.; Brosnan, J.T. Methylation demand and homocysteine metabolism: Effects of dietary provision of creatine and guanidinoacetate. Am. J. Physiol. Endocrinol. Metab. 2001, 281, E1095–E1100. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Safety and efficacy of guanidinoacetic acid as feed additive for chickens for fattening. EFSA J. 2009, 988, 1–30. [Google Scholar]
- Metwally, A.E.; Ibrahim, D.; Khater, S.I. Effects of supplementing broiler diets with CreAMINO® on broiler performance, carcass traits and the expression of muscle growth related genes. Res. Opin. Anim. Vet. Sci. 2015, 5, 435–442. [Google Scholar]
- Teixeira, K.A.; Mascarenhas, A.G.; de Carvalho Mello, H.H.; Arnhold, E.; da Silva Assunção, P.; Carvalho, D.P.; Lopes, S.G. Effect of diets with different levels of guanidinoacetic acid on newly weaned piglets. Semin. Ciências Agrárias 2017, 38, 3887–3896. [Google Scholar] [CrossRef] [Green Version]
- Ostojic, S.M. Guanidinoacetic acid as a performance-enhancing agent. Amino Acids 2016, 48, 1867–1875. [Google Scholar] [CrossRef] [PubMed]
- Raybould, H.E. Visceral perception: Sensory transduction in visceral afferents and nutrients. Gut 2002, 51, i11–i14. [Google Scholar] [CrossRef] [PubMed]
- Saadoun, A.; Cabrera, M.C. Hypophagic and dipsogenic effect of the 5-HT1A receptor agonist 8-OH-DPAT in broiler chickens. J. Anim. Physiol. Anim. Nutr. 2008, 92, 597–604. [Google Scholar] [CrossRef]
- Saadoun, A.; Cabrera, M.C. Effect of the 5-HT1A receptor agonist 8-OH-DPAT on food and water intake in chickens. Physiol. Behav. 2002, 75, 271–275. [Google Scholar] [CrossRef]
- Reis, L.C.; Marinho, V.R. Influence of 5-HT1A agonist on the feeding behavior of Coturnix Japonica (galliformes: Aves). Braz. J. Biol. 2005, 65, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Santiago-López, D.; Bautista-Martínez, J.A.; Reyes-Hernandez, C.I.; Aguilar-Martínez, M.; Rivas-Arancibia, S. Oxidative stress, progressive damage in the substantia nigra and plasma dopamine oxidation, in rats chronically exposed to ozone. Toxicol. Lett. 2010, 197, 193–200. [Google Scholar] [CrossRef]
- Sartsoongnoen, N.; Kosonsiriluk, S.; Prakobsaeng, N.; Songserm, T.; Rozenboim, I.; El Halawani, M.; Chaiseha, Y. The dopaminergic system in the brain of the native Thai chicken, Gallusdomesticus: Localization and differential expression across the reproductive cycle. Gen. Comp. Endocrinol. 2008, 159, 107–115. [Google Scholar] [CrossRef]
- Sun, Z.; Reiner, A. Localization of dopamine D1A and D1B receptor mRNAs in the forebrain and midbrain of the domestic chick. J. Chem. Neuroanat. 2000, 19, 211–224. [Google Scholar] [CrossRef]
- Li, C.; Tong, H.; Yan, Q.; Tang, S.; Han, X.; Xiao, W.; Tan, Z. L-Theanine improves immunity by altering TH2/TH1 cytokine balance, brain neurotransmitters, and expression of phospholipase C in rat hearts. Med. Sci. Monit. 2016, 22, 662–669. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.F.; Gao, F.; Zhang, W.H.; Song, S.X.; Xu, X.L.; Zhou, G.H. Effects of dietary glutamine and gamma-aminobutyric acid on performance, carcass characteristics and serum parameters in broilers under circular heat stress. Anim. Feed. Sci. Technol. 2011, 168, 51–60. [Google Scholar] [CrossRef]
- Neu, A.; Neuhoff, H.; Trube, G.; Fehr, S.; Ullrich, K.; Roeper, J.; Isbrandt, D. Activation of GABA (A) receptors by guanidinoacetate: A novel pathophysiological mechanism. Neurobiol. Dis. 2002, 11, 298–307. [Google Scholar] [CrossRef] [Green Version]
- Ostojic, S.M.; Stojanovic, M.D. Guanidinoacetic acid loading affects plasma γ-aminobutyric acid in healthy men. Eur. J. Nutr. 2015, 54, 855–858. [Google Scholar] [CrossRef] [PubMed]
- De Deyn, P.P.; Macdonald, R.L. Guanidino compounds that are increased in cerebrospinal fluid and brain of uremic patients inhibit GABA and glycine responses on mouse neurons in cell culture. Ann. Neurol. 1990, 28, 627–633. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| g/kg DM | |
|---|---|
| Ingredients | |
| Yellow maize | 602.0 |
| Soybean meal (44%) | 260.0 |
| Limestone | 87.0 |
| Dicalcium phosphate | 17.0 |
| Sodium bicarbonate | 2.3 |
| DL-methionine | 1.3 |
| Vitamin and trace mineral mix | 3.0 |
| NaCl | 2.4 |
| Maize oil | 25.0 |
| Calculated analysis | |
| ME (KJ/kg) | 12,029 |
| Crude protein | 166.0 |
| Calcium | 37.7 |
| Available phosphorus | 4.5 |
| Lysine | 8.5 |
| Leucine | 12.8 |
| Isoleucine | 6.7 |
| Arginine | 9.4 |
| Methionine | 3.9 |
| Methionine + cysteine | 6.3 |
| Tryptophan | 2.2 |
| Threonine | 6.1 |
| Phenylalanine | 7.8 |
| Histidine | 4.3 |
| Valine | 7.7 |
| Item | Experimental Groups | Contrast | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 T1 | 2 T2 | 3 T3 | 4 T4 | 5 RSD | Linear | Quadratic | Cubic | |
| Lysine | 84.4 a | 77.5 ab | 73.9 b | 72.3 b | 6.04 | 0.013 | 0.245 | 0.308 |
| Leucine | 45.8 c | 59.3 b | 60.3 b | 71.1 a | 4.49 | 0.003 | 0.227 | 0.026 |
| Isoleucine | 30.4 b | 38.7 a | 38.7 a | 39.1 a | 2.44 | 0.047 | 0.087 | 0.178 |
| Phenylalanine | 15.7 c | 19.2 b | 21.1 a | 25.7 a | 1.68 | 0.001 | 0.045 | 0.018 |
| Valine | 33.3 | 41.9 | 42.7 | 43.7 | 3.71 | 0.073 | 0.257 | 0.416 |
| Threonine | 26.4 b | 33.9 ab | 30.2 ab | 37.7 a | 3.60 | 0.015 | 0.150 | 0.102 |
| Methionine | 13.9 c | 17.8 b | 17.1 bc | 21.4 a | 1.52 | 0.001 | 0.824 | 0.055 |
| Serine | 21.8 | 28.5 | 27.8 | 28.8 | 2.86 | 0.098 | 0.390 | 0.374 |
| Aspartic acid | 62.6 b | 66.2 ab | 71.4 ab | 85.6 a | 8.44 | 0.010 | 0.707 | 0.156 |
| Glutamine | 67.9 c | 85.5 b | 91.2 b | 122.9 a | 9.60 | 0.016 | 0.180 | 0.220 |
| Proline | 13.3 b | 16.5 ab | 17.1 ab | 20.3 a | 1.47 | 0.003 | 0.832 | 0.034 |
| Alanine | 35.5 b | 44.6 ab | 38.7 ab | 47.9 a | 3.34 | 0.019 | 0.229 | 0.076 |
| Arginine | 54.3 b | 51.5 b | 65.6 a | 65.2 a | 4.48 | 0.001 | 0.225 | 0.013 |
| Histidine | 32.5 b | 36.9 ab | 40.6 a | 42.1 a | 3.16 | 0.012 | 0.296 | 0.030 |
| Glycine | 28.3 | 35.5 | 35.9 | 36.7 | 2.08 | 0.087 | 0.303 | 0.219 |
| Tyrosine | 25.3 b | 35.1 a | 31.9 ab | 37.8 a | 3.27 | 0.004 | 0.118 | 0.087 |
| Taurine | 69.9 c | 85.8 bc | 93.7 ab | 109.7 a | 6.46 | 0.016 | 0.319 | 0.022 |
| Item | Experimental Groups | Contrast | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 T1 | 2 T2 | 3 T3 | 4 T4 | 5 RSD | Linear | Quadratic | Cubic | |
| Lysine | 52.7 a | 53.7 a | 44.8 b | 36.7 c | 2.37 | 0.003 | 0.036 | 0.001 |
| Leucine | 35.3 b | 43.5 ab | 44.5 ab | 53.2 a | 2.97 | 0.026 | 0.417 | 0.193 |
| Isoleucine | 28.3 b | 35.7 a | 33.3 ab | 37.1 a | 2.06 | 0.001 | 0.139 | 0.014 |
| Phenylalanine | 16.5 b | 22.1 ab | 20.8 b | 27.2 a | 1.41 | 0.020 | 0.076 | 0.029 |
| Valine | 23.8 b | 29.9 ab | 33.2 ab | 37.8 a | 3.52 | 0.035 | 0.676 | 0.491 |
| Threonine | 20.5 b | 26.9 a | 23.3 ab | 27.9 a | 2.18 | 0.028 | 0.369 | 0.123 |
| Methionine | 9.6 c | 11.6 bc | 12.2 ab | 14.5 a | 1.79 | 0.009 | 0.561 | 0.292 |
| Serine | 17.1 c | 21.9 b | 24.2 ab | 27.1 a | 2.13 | 0.012 | 0.255 | 0.394 |
| Aspartic acid | 44.1 b | 56.4 a | 59.6 a | 56.1 a | 4.75 | 0.010 | 0.031 | 0.753 |
| Glutamine | 68.8 c | 86.2 b | 105.9 a | 111.5 a | 6.84 | 0.001 | 0.065 | 0.233 |
| Proline | 13.2 c | 16.7 bc | 18.4 b | 25.6 a | 1.67 | 0.004 | 0.039 | 0.062 |
| Alanine | 28.5 c | 35.8 bc | 37.5 ab | 45.2 a | 2.36 | 0.008 | 0.630 | 0.142 |
| Arginine | 35.6 c | 34.2 c | 40.9 b | 46.8 a | 2.32 | 0.022 | 0.025 | 0.032 |
| Histidine | 18.1 b | 21.9 b | 19.7 b | 29.9 a | 1.64 | 0.019 | 0.020 | 0.004 |
| Glycine | 22.4 b | 30.4 a | 30.3 a | 36.9 a | 2.37 | 0.036 | 0.586 | 0.020 |
| Tyrosine | 13.8 b | 17.9 b | 17.4 b | 23.2 a | 1.70 | 0.024 | 0.414 | 0.031 |
| Taurine | 56.4 b | 74.3 ab | 67.6 b | 92.2 a | 7.87 | 0.025 | 0.454 | 0.009 |
| Parameter | Experimental Groups | Contrast | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 T1 | 2 T2 | 3 T3 | 4 T4 | 5 RSD | Linear | Quadratic | Cubic | |
| Nitric oxide (μmol/g) | 25.8 | 24.9 | 26.6 | 31.4 | 2.68 | 0.080 | 0.132 | 0.216 |
| Dopamine (μg/g) | 1.02 b | 1.03 b | 1.27 a | 1.48 a | 0.101 | 0.011 | 0.131 | 0.006 |
| Serotonin (μg/g) | 0.76 a | 0.57 b | 0.61 b | 0.49 b | 0.065 | 0.010 | 0.019 | 0.042 |
| 6 GABA (μg/g) | 8.12 a | 7.33 ab | 6.32 bc | 5.41 c | 0.689 | 0.027 | 0.017 | 0.001 |
| Glutamate (μg/g) | 4.16 | 3.92 | 3.39 | 3.44 | 0.592 | 0.592 | 0.321 | 0.460 |
| Aspartic acid (μg/g) | 4.47 a | 3.63 ab | 3.35 b | 3.62 ab | 0.494 | 0.019 | 0.011 | 0.086 |
| Histidine (μg/g) | 0.46 | 0.43 | 0.52 | 0.71 | 0.041 | 0.102 | 0.243 | 0.016 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed-Farid, O.A.; Salah, A.S.; Nassan, M.A.; El-Tarabany, M.S. Performance, Carcass Yield, Muscle Amino Acid Profile, and Levels of Brain Neurotransmitters in Aged Laying Hens Fed Diets Supplemented with Guanidinoacetic Acid. Animals 2021, 11, 3091. https://doi.org/10.3390/ani11113091
Ahmed-Farid OA, Salah AS, Nassan MA, El-Tarabany MS. Performance, Carcass Yield, Muscle Amino Acid Profile, and Levels of Brain Neurotransmitters in Aged Laying Hens Fed Diets Supplemented with Guanidinoacetic Acid. Animals. 2021; 11(11):3091. https://doi.org/10.3390/ani11113091
Chicago/Turabian StyleAhmed-Farid, Omar A., Ayman S. Salah, Mohamed Abdo Nassan, and Mahmoud S. El-Tarabany. 2021. "Performance, Carcass Yield, Muscle Amino Acid Profile, and Levels of Brain Neurotransmitters in Aged Laying Hens Fed Diets Supplemented with Guanidinoacetic Acid" Animals 11, no. 11: 3091. https://doi.org/10.3390/ani11113091
APA StyleAhmed-Farid, O. A., Salah, A. S., Nassan, M. A., & El-Tarabany, M. S. (2021). Performance, Carcass Yield, Muscle Amino Acid Profile, and Levels of Brain Neurotransmitters in Aged Laying Hens Fed Diets Supplemented with Guanidinoacetic Acid. Animals, 11(11), 3091. https://doi.org/10.3390/ani11113091






