Litopenaeus vannamei BMAL1 Is a Critical Mediator Regulating the Expression of Glucose Transporters and Can Be Suppressed by Constant Darkness
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Gene Expression (BMAL1, GLUT1 and SGLT1) Daily over 24-h in the Intestine and Hepatopancreas
2.2. Protein Sequence and Domain Structure Analysis
2.3. In Vitro Dual-Luciferase Reporter Assays
2.4. In Vivo Knockdown by dsRNA Injection
2.5. Eight-Week Light/Dark Treatment
2.6. Quantitative Reverse Transcription PCR (RT-qPCR)
2.7. Statistical Analysis
3. Results
3.1. Daily Profile of BMAL1, GLUT1 and SGLT1 Expression in the Hepatopancreas and Intestine
3.2. The Sequence Characteristics of BMAL1
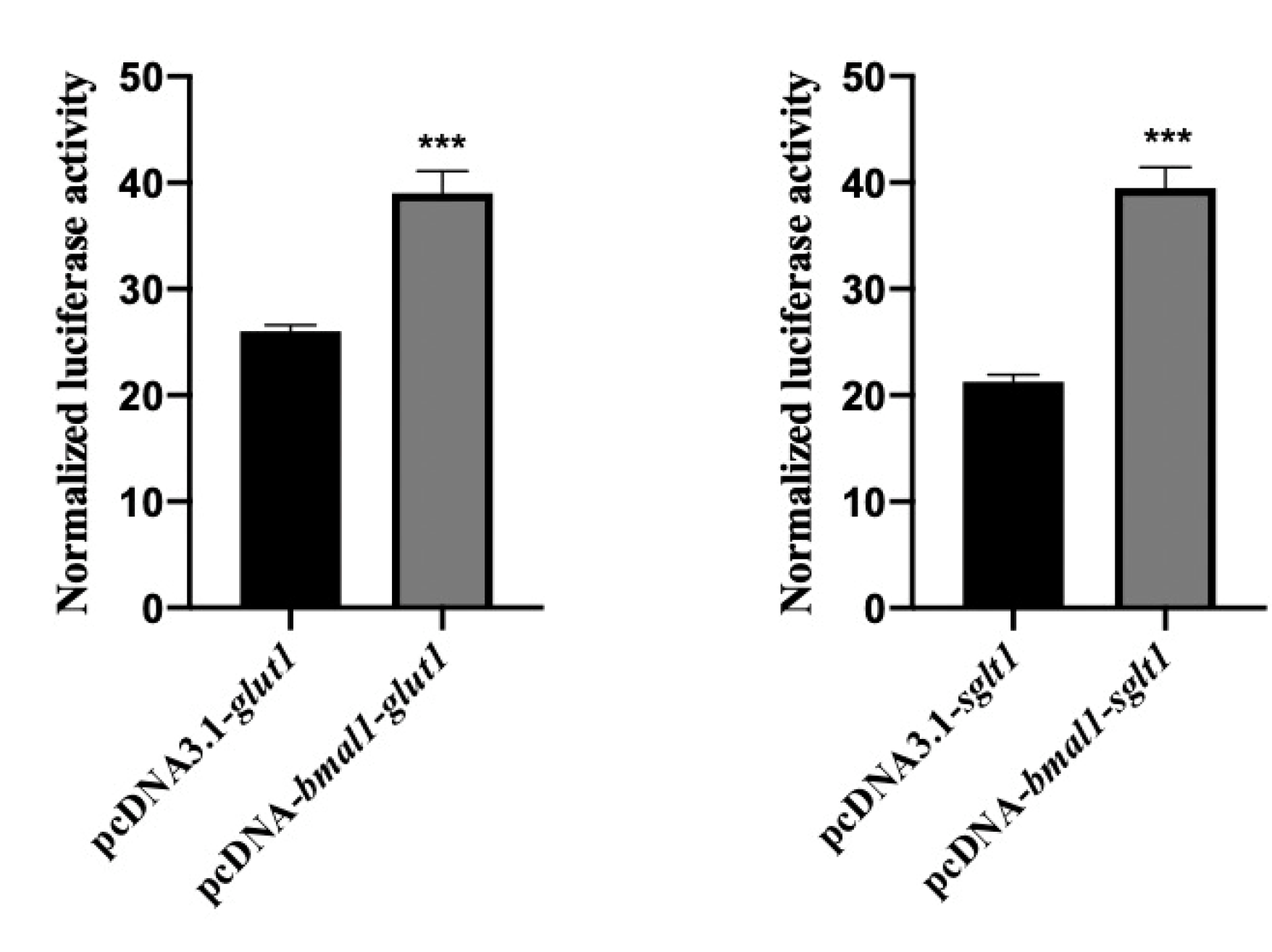
3.3. Overexpression of BMAL1 Significantly Activated the Gene Expression of GLUT1 and SGLT1 In Vitro
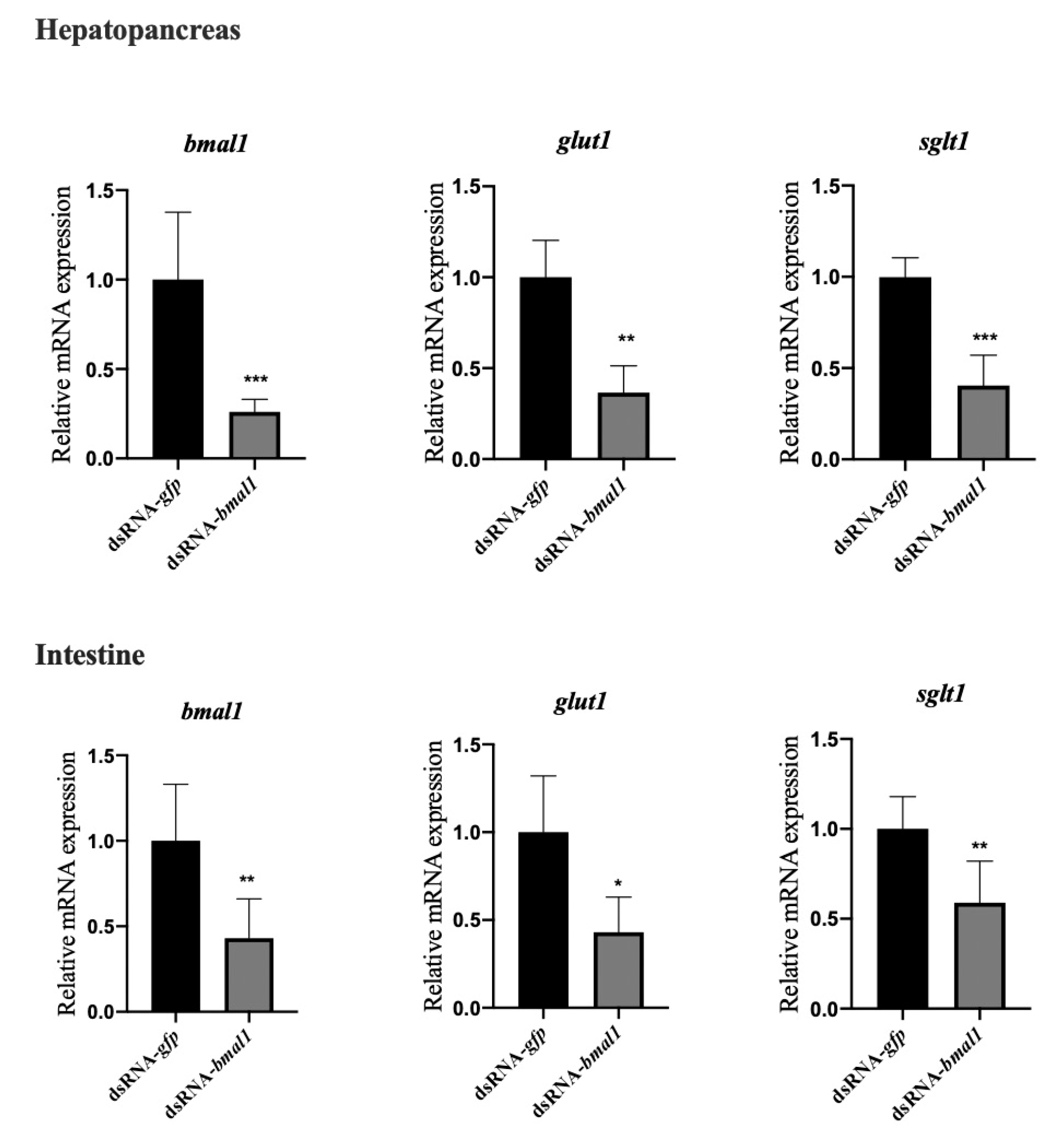
3.4. Knockdown of BMAL1 Decreased the mRNA Expression of Glucose Transporters (GLUT1, SGLT1) in the Hepatopancreas and Intestine of L. vannamei
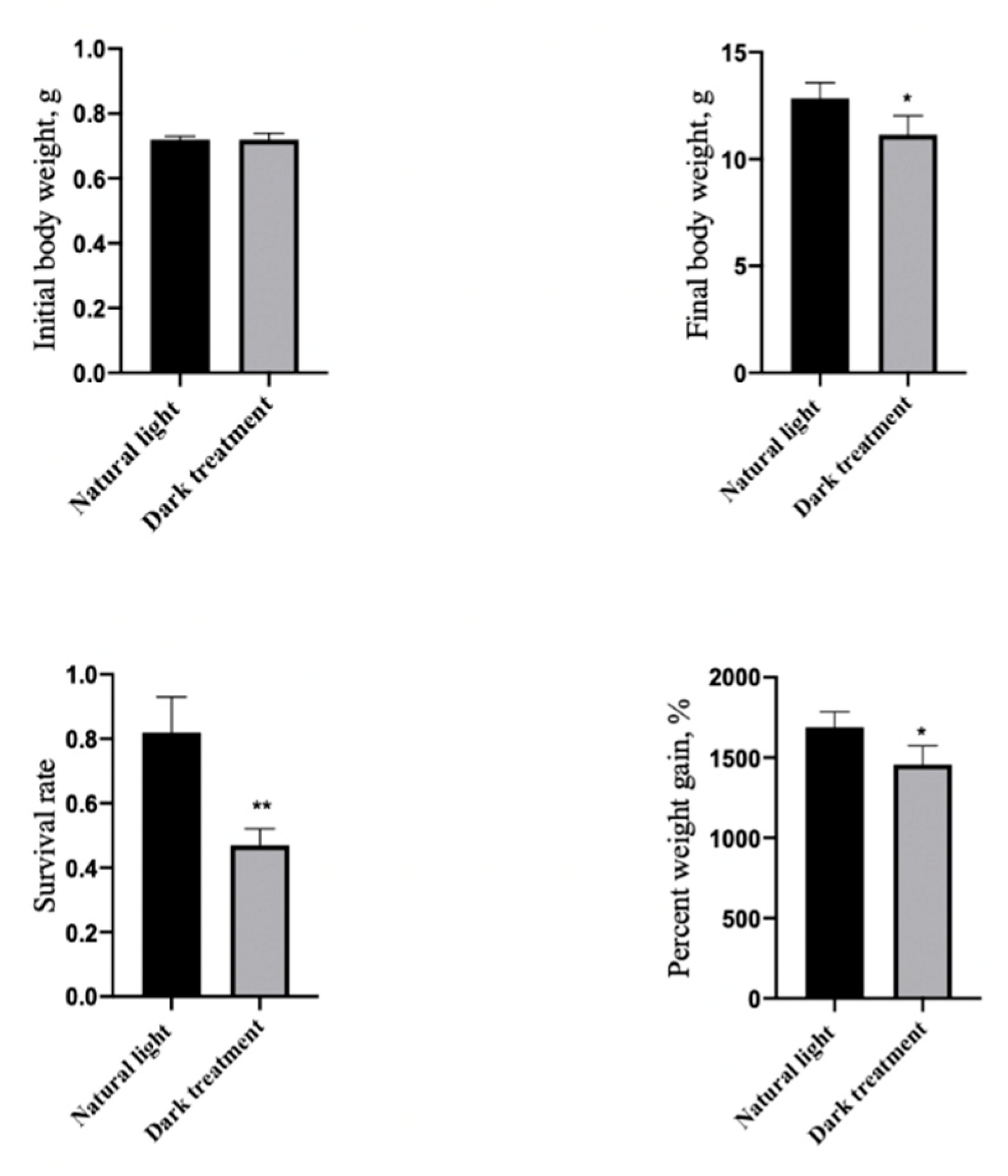
3.5. Effects of Dark Treatment on Growth Performance and Survival Rate in L. vannamei
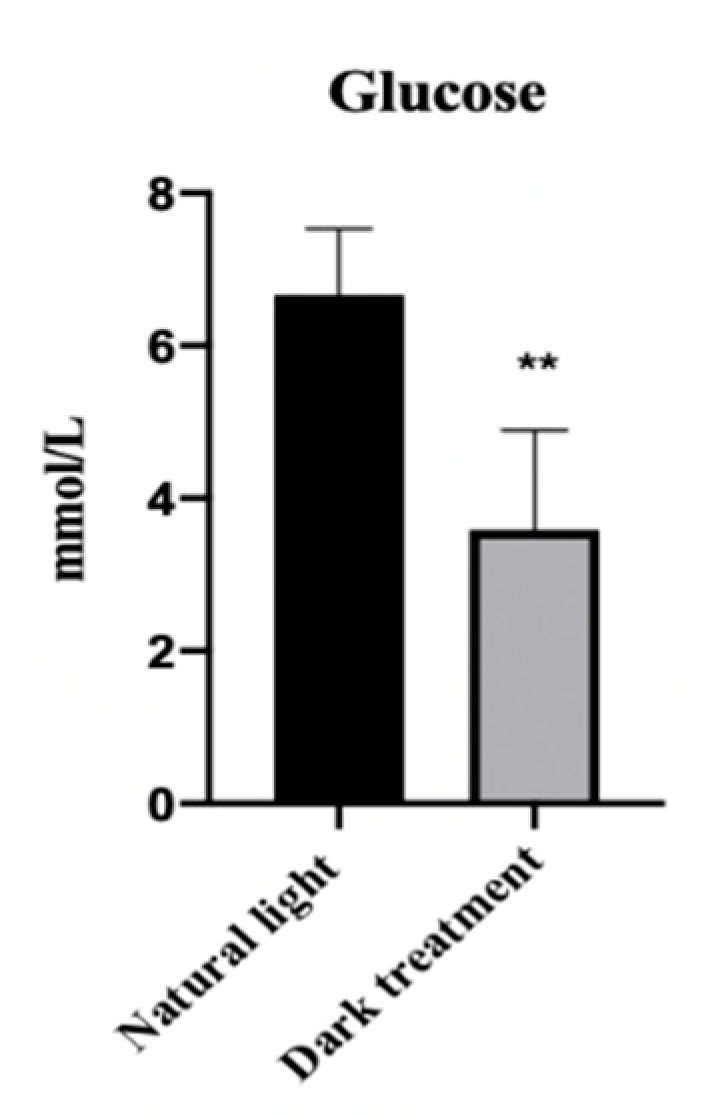
3.6. Effects of Dark Treatment on Serum Glucose Concentration in L. vannamei
3.7. Effects of Dark Treatment on the mRNA Expression of BMAL1, GLUT1 and SGLT1 in the Hepatopancreas and Intestine of L. vannamei
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pilorz, V.; Helfrich-Forster, C.; Oster, H. The role of the circadian clock system in physiology. Pflugers Arch. 2018, 470, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, A.; Stearns, A.T.; Ashley, S.W.; Rhoads, D.B.; Tavakkolizadeh, A. PER1 modulates SGLT1 transcription in vitro independent of E-box status. Dig. Dis. Sci. 2012, 57, 1525–1536. [Google Scholar] [CrossRef] [PubMed]
- Buijs, F.N.; Leon-Mercado, L.; Guzman-Ruiz, M.; Guerrero-Vargas, N.N.; Romo-Nava, F.; Buijs, R.M. The circadian system, a regulatory feedback network of periphery and brain. Physiology (Bethesda) 2016, 31, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Strauss, J.; Dircksen, H. Circadian clocks in crustaceans, identified neuronal and cellular systems. Front. Biosci. Landmark. Ed. 2010, 15, 1040. [Google Scholar] [CrossRef]
- Aguzzi, J.; Company, J.B.; Sarda, F.; Sanchez-Pardo, J.; Garcia, J.A.; Rotllant, G. Is the glucose concentration in the haemolymph a suitable indicator of circadian rhythmicity in Nephrops norvegicus (Decapoda, Nephropidae)? Crustaceana 2004, 77, 213–229. [Google Scholar]
- Wright, E.M.; Sala-Rabanal, M.; Ghezzi, G.M.; Loo, D.D.F. Sugar Absorption, 6th ed.; Physiology of the Gastrointestinal Tract; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1051–1062. [Google Scholar]
- Navale, A.M.; Paranjape, A.N. Glucose transporters, physiological and pathological roles. Biophys. Rev. 2016, 8, 5–9. [Google Scholar] [CrossRef]
- Balakrishnan, A.; Stearns, A.T.; Rounds, J.; Irani, J.; Giuffrida, M.; Rhoads, D.B.; Ashley, S.W.; Tavakkolizadeh, A. Diurnal rhythmicity in glucose uptake is mediated by temporal periodicity in the expression of the sodium-glucose cotransporter (SGLT1). Surgery 2008, 143, 813–818. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pacha, J.; Sumova, A. Circadian regulation of epithelial functions in the intestine. Acta. Physiol. (Oxf). 2013, 208, 11–24. [Google Scholar] [CrossRef]
- Buhr, E.D.; Takahashi, J.S. Molecular components of the mammalian circadian clock. Handb. Exp. Pharmacol. 2013, 217, 3–27. [Google Scholar]
- Menet, J.S.; Pescatore, S.; Rosbash, M. CLOCK:BMAL1 is a pioneer-like transcription factor. Genes Dev. 2014, 28, 8–13. [Google Scholar] [CrossRef]
- Lee, J.; Kim, M.S.; Li, R.; Liu, V.Y.; Fu, L.; Moore, D.D.; Ma, K.; Yechoor, V.K. Loss of Bmal1 leads to uncoupling and impaired glucose-stimulated insulin secretion in beta-cells. Islets 2011, 3, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Whitney, S.; Matthew, S.; Mowdawalla, C.; Mota, S.; Louis, R.; Pan, X.Y. BMAL1 controls glucose uptake through paired-homeodomain transcription factor 4 in caco-2 cells. Am. J. Physiol. Cell Physiol. 2019, 317, 492–501. [Google Scholar]
- Rudic, R.D.; McNamara, P.; Curtis, A.M.; Boston, R.C.; Panda, S.; Hogenesch, J.B.; Fitzgerald, G.A. BMAL1 and CLOCK, two essential components of the circadian clock, are involved in glucose homeostasis. PLoS Biol. 2004, 2, e377. [Google Scholar] [CrossRef] [PubMed]
- Mata-Sotres, J.A.; Martinez-Rodriguez, G.; Perez-Sanchez, J.; Sanchez-Vazquez, F.J.; Yufera, M. Daily rhythms of clock gene expression and feeding behavior during the larval development in gilthead seabream, Sparus aurata. Chronobiol. Int. 2015, 32, 1061–1074. [Google Scholar] [CrossRef]
- Zhang, P.; Yu, C.; Sun, L. Japanese flounder (Paralichthys olivaceus) Bmal1 is involved in the regulation of inflammatory response and bacterial infection. Aquaculture 2020, 525, 735330. [Google Scholar] [CrossRef]
- Farhud, D.; Aryan, Z. Circadian rhythm, lifestyle and health: A narrative review. Iran. J. Public. Health 2018, 47, 1068–1076. [Google Scholar] [PubMed]
- Loh, D.H.; Navarro, J.; Hagopian, A.; Wang, L.M.; Deboer, T.; Colwell, C.S. Rapid changes in the light/dark cycle disrupt memory of conditioned fear in mice. PLoS ONE 2010, 5, e12546. [Google Scholar] [CrossRef]
- Sanudin, N.; Tuzan, A.D.; Yong, A.S.K. Feeding activity and growth performance of shrimp post larvae Litopenaeus vannamei under light and dark condition. J. Agr. Sci. 2014, 6, 103. [Google Scholar] [CrossRef]
- Hoang, T.; Marchiesis, B.; Lee, S.Y.; Keenan, C.P.; Marsden, G.E. Influences of light intensity and photoperiod on moulting and growth of Penaeus merguiensis cultured under laboratory conditions. Aquaculture 2003, 216, 343–354. [Google Scholar] [CrossRef]
- Patino, M.A.; Rodriguez-Illamola, A.; Conde-Sieira, M.; Soengas, J.L.; Miguez, J.M. Daily rhythmic expression patterns of clock1a, bmal1, and per1 genes in retina and hypothalamus of the rainbow trout, Oncorhynchus mykiss. Chronobiol. Int. 2011, 28, 381–389. [Google Scholar] [CrossRef]
- Heigwer, F.; Port, F.; Boutros, M. RNA interference (RNAi) screening in Drosophila. Genetics 2018, 208, 853–874. [Google Scholar] [CrossRef]
- Miraglia, L.J.; King, F.J.; Damoiseaux, R. Seeing the light: Luminescent reporter gene assays. Comb. Chem. High. Throughput Screen 2011, 14, 648–657. [Google Scholar] [CrossRef]
- Basic Local Alignment Search Tool. Available online: http://www.ncbi.nlm.nih.gov/blast (accessed on 13 July 2021).
- SIB Swiss Institute of Bioinformatics|Expasy. Available online: http://www.expasy.org (accessed on 1 October 2021).
- SMART: Main Page. Available online: http://smart.embl-heidelberg.de/ (accessed on 7 August 2021).
- Balakrishnan, A.; Tavakkolizadeh, A.; Rhoads, D.B. Circadian clock genes and implications for intestinal nutrient uptake. J. Nutr. Biochem. 2012, 23, 417–422. [Google Scholar] [CrossRef][Green Version]
- Cagampang, F.R.; Bruce, K.D. The role of the circadian clock system in nutrition and metabolism. Br. J. Nutr. 2012, 108, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Scheepers, A.; Joost, H.G.; Schurmann, A. The glucose transporter families SGLT and GLUT: Molecular basis of normal and aberrant function. J. Parenter. Enteral. Nutr. 2004, 28, 364–371. [Google Scholar] [CrossRef]
- Li, S.; Zhai, J.; Chu, W.; Geng, X.; Chen, Z.J.; Du, Y. Altered circadian clock as a novel therapeutic target for constant darkness-induced insulin resistance and hyperandrogenism of polycystic ovary syndrome. Transl. Res. 2020, 219, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Masri, S.; Orozco-Solis, R.; Aguilar-Arnal, L.; Cervantes, M.; Sassone-Corsi, P. Coupling circadian rhythms of metabolism and chromatin remodelling. Diabetes Obes. Metab. 2015, 17 Suppl 1, 17–22. [Google Scholar] [CrossRef]
- Ravi, R.; Manisseri, M.K. The effect of different pH and photoperiod regimes on the survival rate and development period of the larvae of Portunus pelagicus (Decapoda, Brachyura, Portunidae). Iran. J. Fish. Sci. 2012, 12, 490–499. [Google Scholar]
- Walton, J.C.; Weil, Z.M.; Nelson, R.J. Influence of photoperiod on hormones, behavior, and immune function. Front. Neuroendocrinol. 2011, 32, 303–319. [Google Scholar] [CrossRef] [PubMed]
- Durrant, J.; Michaelides, E.B.; Rupasinghe, T.; Tull, D.; Green, M.P.; Jones, T.M. Constant illumination reduces circulating melatonin and impairs immune function in the cricket Teleogryllus commodus. PeerJ 2015, 3, e1075. [Google Scholar] [CrossRef]
- Vera, L.M.; Negrini, P.; Zagatti, C.; Frigato, E.; Sánchez-Vázquez, F.J.; Bertolucci, C. Light and feeding entrainment of the molecular circadian clock in a marine teleost (Sparus aurata). Chronobiol. Int. 2013, 30, 649–661. [Google Scholar] [CrossRef] [PubMed]
- Bilotta, J. Effects of abnormal lighting on the development of zebrafish visual behavior. Behav. Brain. Res. 2000, 116, 81–87. [Google Scholar] [CrossRef]
- O’Connor, J.J.; Fobert, E.F.; Bessonc, M.; Jacob, H.; Lecchini, D. Live fast, die young: Behavioural and physiological impacts of light pollution on a marine fish during larval recruitment. Mar. Pollut. Bull. 2019, 146, 908–914. [Google Scholar] [CrossRef]
- Maciel, F.E.; Geihs, M.A.; Vargas, M.A.; Cruz, B.P.; Ramos, B.P.; Vakkuri, O.; Meyer-Rochow, V.B.; Maia Nery, L.E.; Allodi, S. Daily variation of melatonin content in the optic lobes of the crab Neohelice granulata. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 2008, 149, 162–166. [Google Scholar] [CrossRef]
- Vargas, M.A.; Geish, M.A.; Maciel, F.E.; Cruz, B.P.; Filgueira, D.; Ferreira, G.; Nery, L.; Allodi, S. Influence of the dark/light rhythm on the effects of UV radiation in the eyestalk of the crab Neohelice granulata. Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 2010, 151, 343–350. [Google Scholar] [CrossRef]
- Martins, R.S.; Ana, G.; Silvia, Z.; Manuel, C.; Canário, A.V.M.; Nicolas, C. Photoperiodic modulation of circadian clock and reproductive axis gene expression in the pre-pubertal European Sea Bass brain. PLoS ONE 2015, 10, e0144158. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Hutchison, A.T.; Heilbronn, L.K. Carbohydrate intake and circadian synchronicity in the regulation of glucose homeostasis. Curr. Opin. Clin. Nutr. Metab. Care 2021, 24, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhou, B.; Yan, M.H.; Huang, R.; Wang, Y.G.; He, Z.S.; Yang, Y.G.; Dai, C.G.; Wang, Y.Q.; Zhang, F.; et al. Clock and. bmal1 regulate muscle insulin sensitivity via sirt1 in male mice. Endocrinology 2016, 6, 2259–2269. [Google Scholar]
- Wang, X.; Li, E.; Chen, L. A review of carbohydrate nutrition and metabolism in crustaceans. N. Am. J. Aquacult. 2016, 78, 178–187. [Google Scholar] [CrossRef]



| Primers | Sequences (5’ to 3’) |
|---|---|
| For RNA interference experiment | |
| GFP-F | ATGGTGAGCAAGGGCGAGGAG |
| GFP-R | TTACTTGTACAGCTCGTCCATGCC |
| T7-GFP-F | GGATCCTAATACGACTCACTATAGGATGGTGAGCAAGGGCGAGGAG |
| T7-GFP-R | GGATCCTAATACGACTCACTATAGGTTACTTGTACAGCTCGTCCATGCC |
| BMAL1-F | ATGTTTGATCTTGGCAACTATG |
| BMAL1-R | AAAGTTTGATACACCGATTCCG |
| T7-BMAL1-F | GGATCCTAATACGACTCACTATAGGATGTTTGATCTTGGCAACTATG |
| T7-BMAL1-R | GGATCCTAATACGACTCACTATAGGAAAGTTTGATACACCGATTCCG |
| For RT-PCR | |
| BMAL1-F | CCTTCAACCACAATAACAACCATAC |
| BMAL1-R | CCTCGTCTGAGTCGCTCGTGCCATC |
| GLUT1-F | CTTGGAGTTGGGTCGGTGATGGTTA |
| GLUT1-R | TCCACGGAATACTGCCAGGACCCAC |
| SGLT1-F | ATCGGCTTGGTCATAGGACTCATTC |
| SGLT1-R | GCAGCCGAAGTGGAGATAATGGACA |
| β-actin-F | CCACGAGACCACCTACAAC |
| β-actin-R | AGCGAGGGCAGTGATTTC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiao, L.; Dai, T.; Sun, P.; Jin, M.; Zhou, Q. Litopenaeus vannamei BMAL1 Is a Critical Mediator Regulating the Expression of Glucose Transporters and Can Be Suppressed by Constant Darkness. Animals 2021, 11, 2893. https://doi.org/10.3390/ani11102893
Jiao L, Dai T, Sun P, Jin M, Zhou Q. Litopenaeus vannamei BMAL1 Is a Critical Mediator Regulating the Expression of Glucose Transporters and Can Be Suppressed by Constant Darkness. Animals. 2021; 11(10):2893. https://doi.org/10.3390/ani11102893
Chicago/Turabian StyleJiao, Lefei, Tianmeng Dai, Peng Sun, Min Jin, and Qicun Zhou. 2021. "Litopenaeus vannamei BMAL1 Is a Critical Mediator Regulating the Expression of Glucose Transporters and Can Be Suppressed by Constant Darkness" Animals 11, no. 10: 2893. https://doi.org/10.3390/ani11102893
APA StyleJiao, L., Dai, T., Sun, P., Jin, M., & Zhou, Q. (2021). Litopenaeus vannamei BMAL1 Is a Critical Mediator Regulating the Expression of Glucose Transporters and Can Be Suppressed by Constant Darkness. Animals, 11(10), 2893. https://doi.org/10.3390/ani11102893






