Omics Insights into Animal Resilience and Stress Factors
Abstract
Simple Summary
Abstract
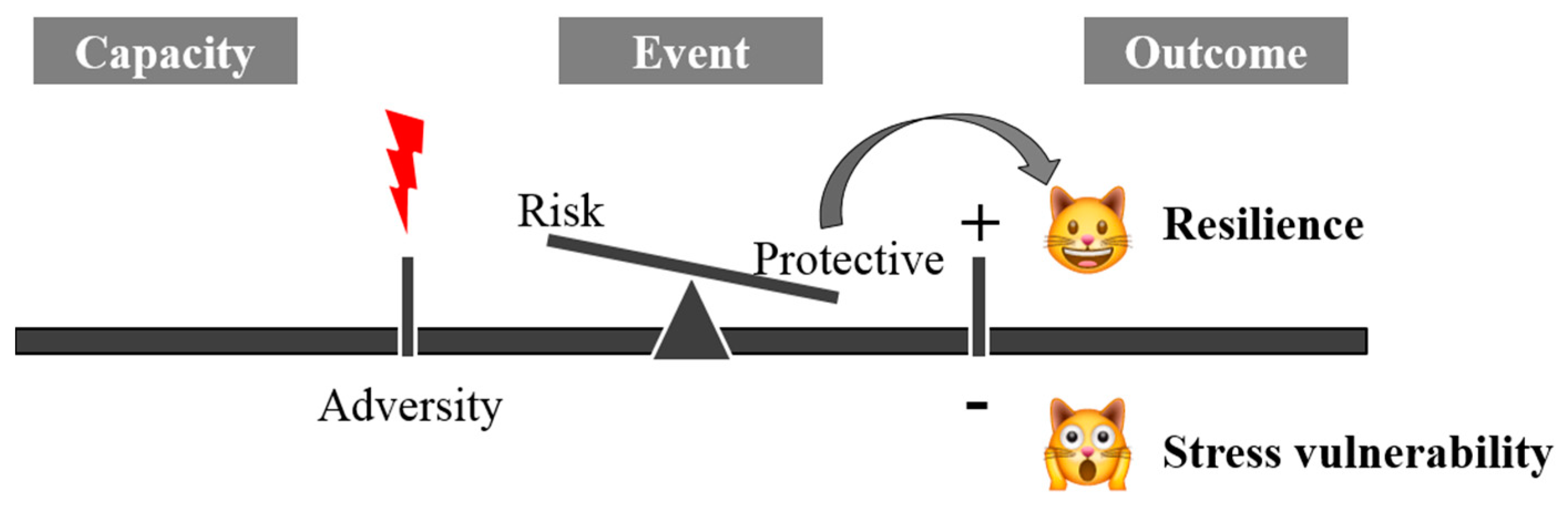
1. Understanding Resilience
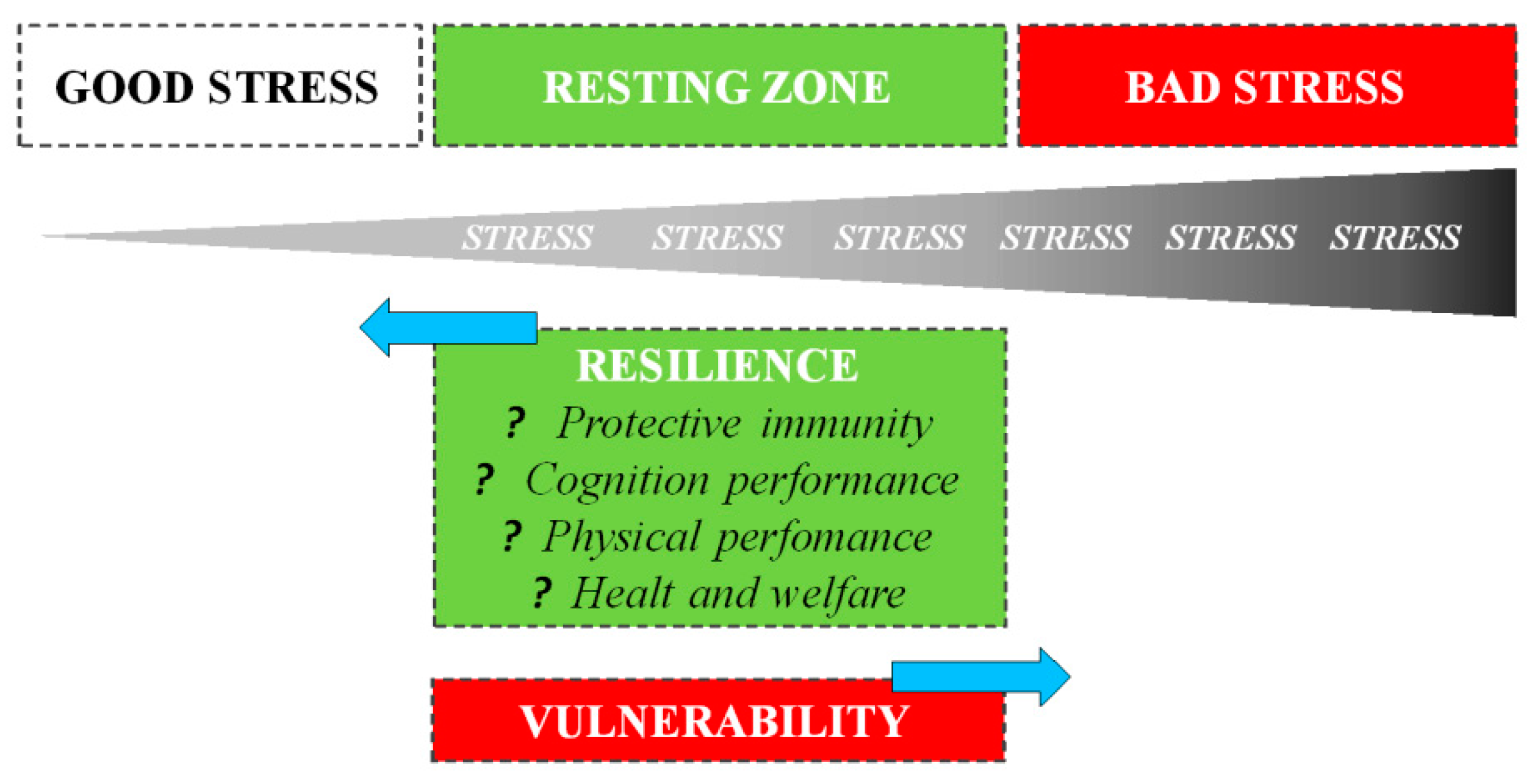
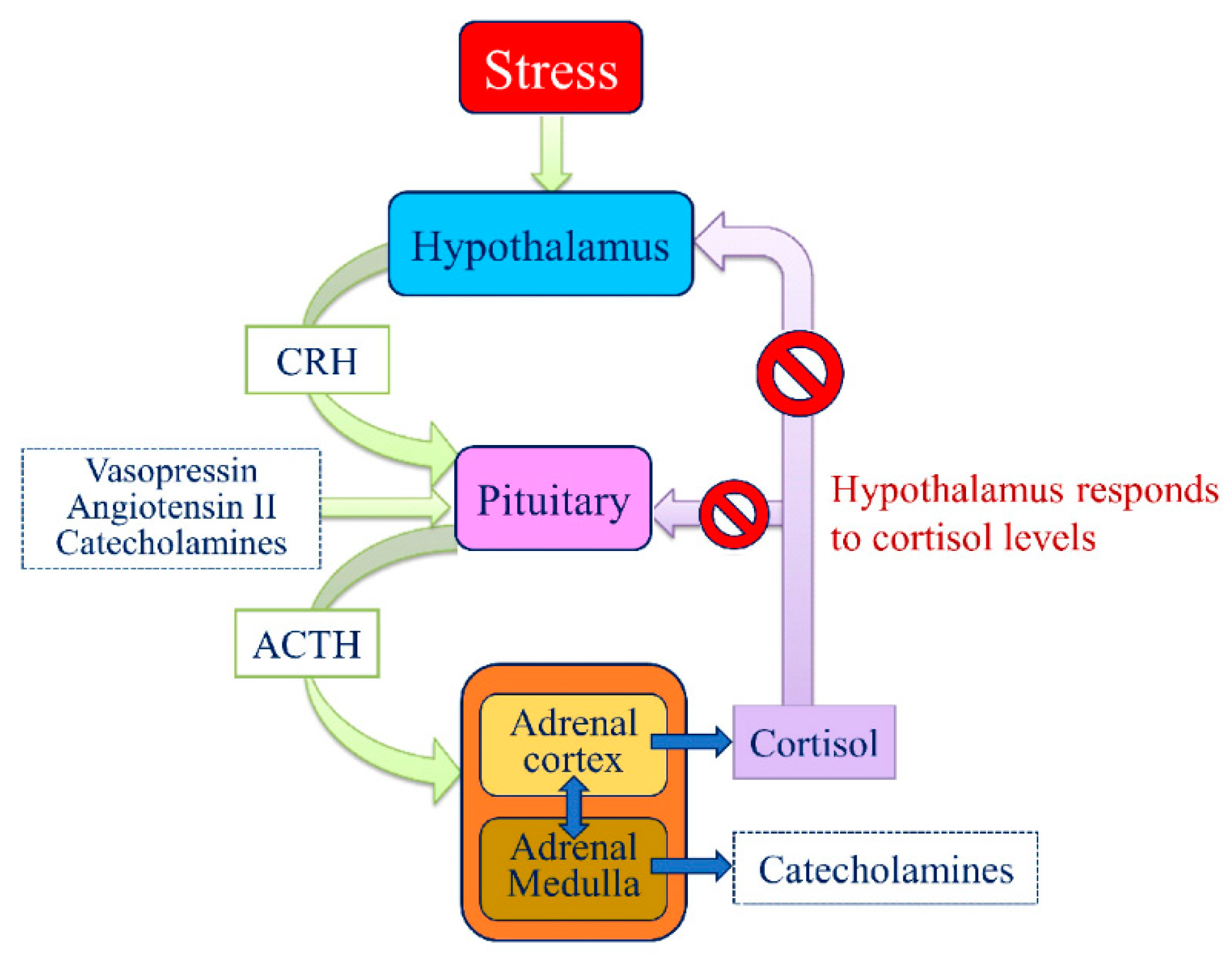
2. Stress Vulnerability and Resilience
3. Environmental Stressors and Gene Responses
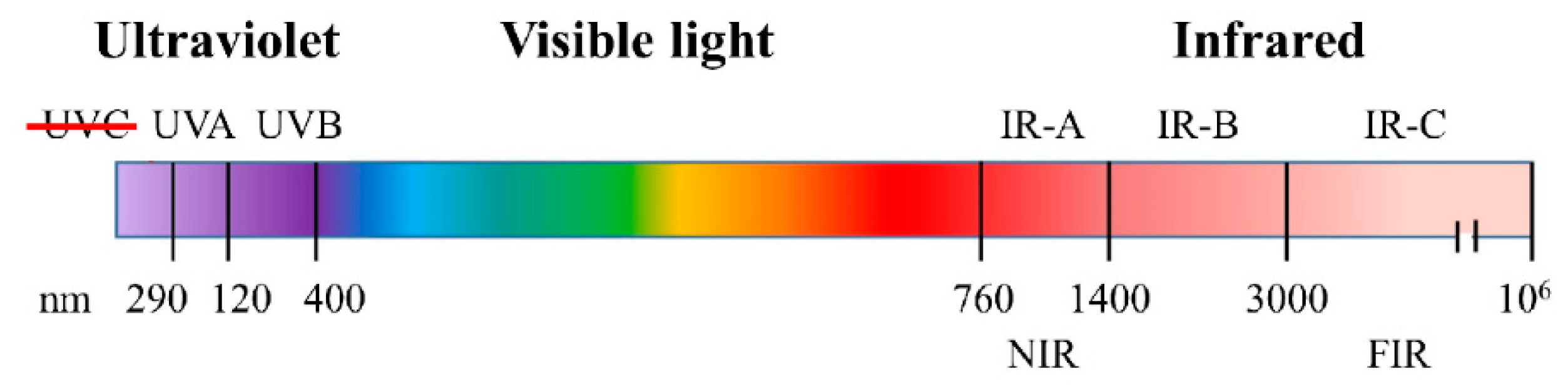
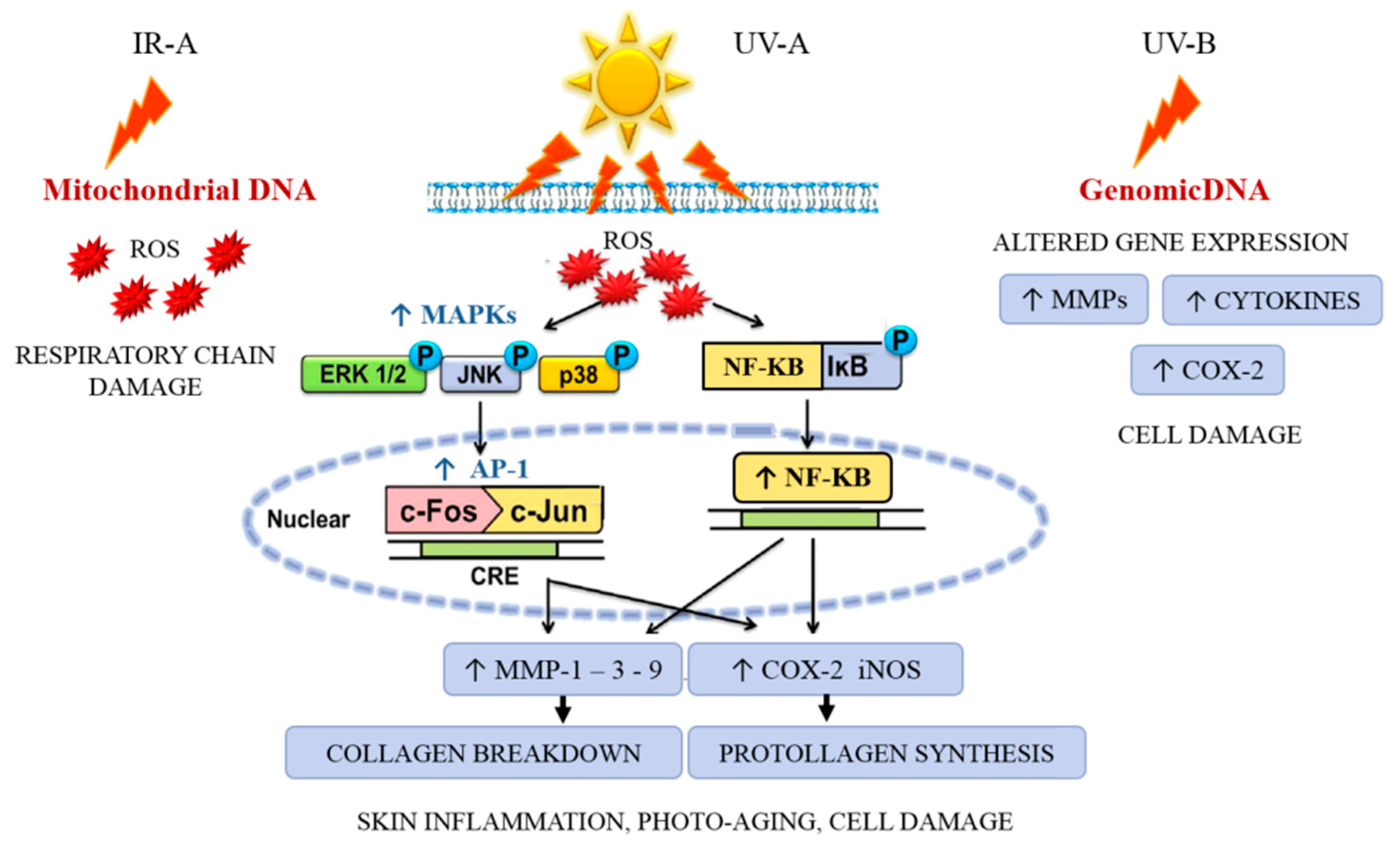
3.1. Sunlight
3.2. Hot and Cold Temperature
3.3. Lack of Oxygen
3.4. Effects of Environmental Enrichment: Feeding and Early Life Conditions
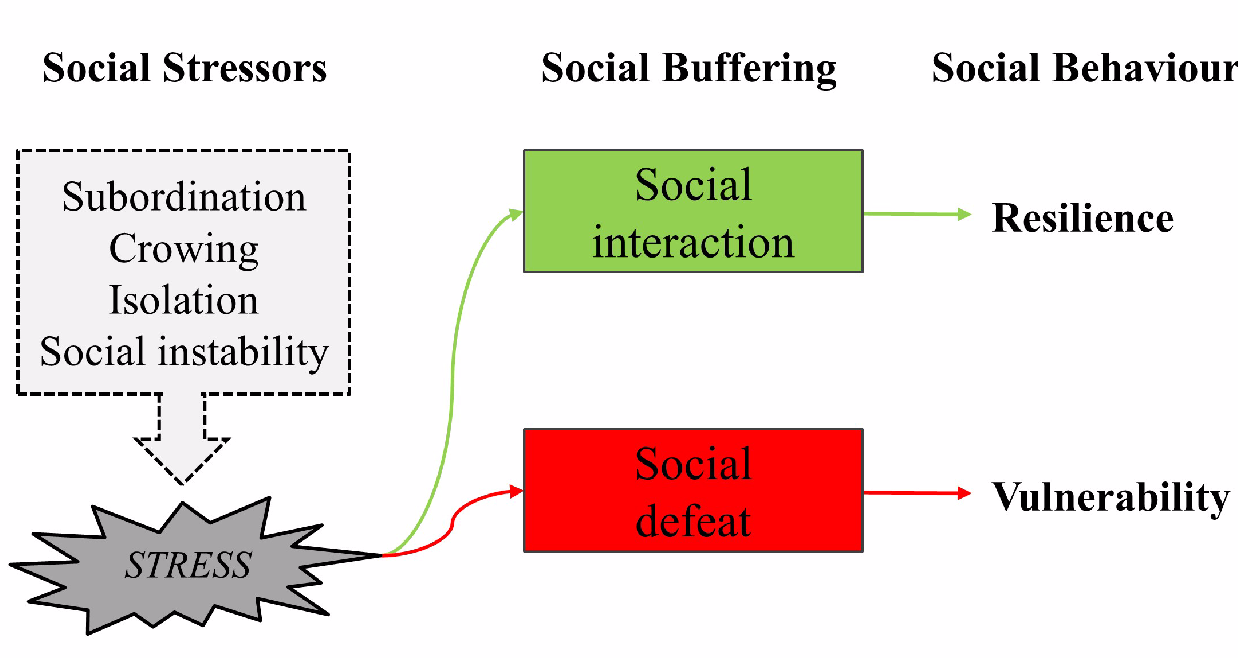
3.5. Social Interactions and Social Defeat
4. Resilience in Response to Epigenetics Remodeling
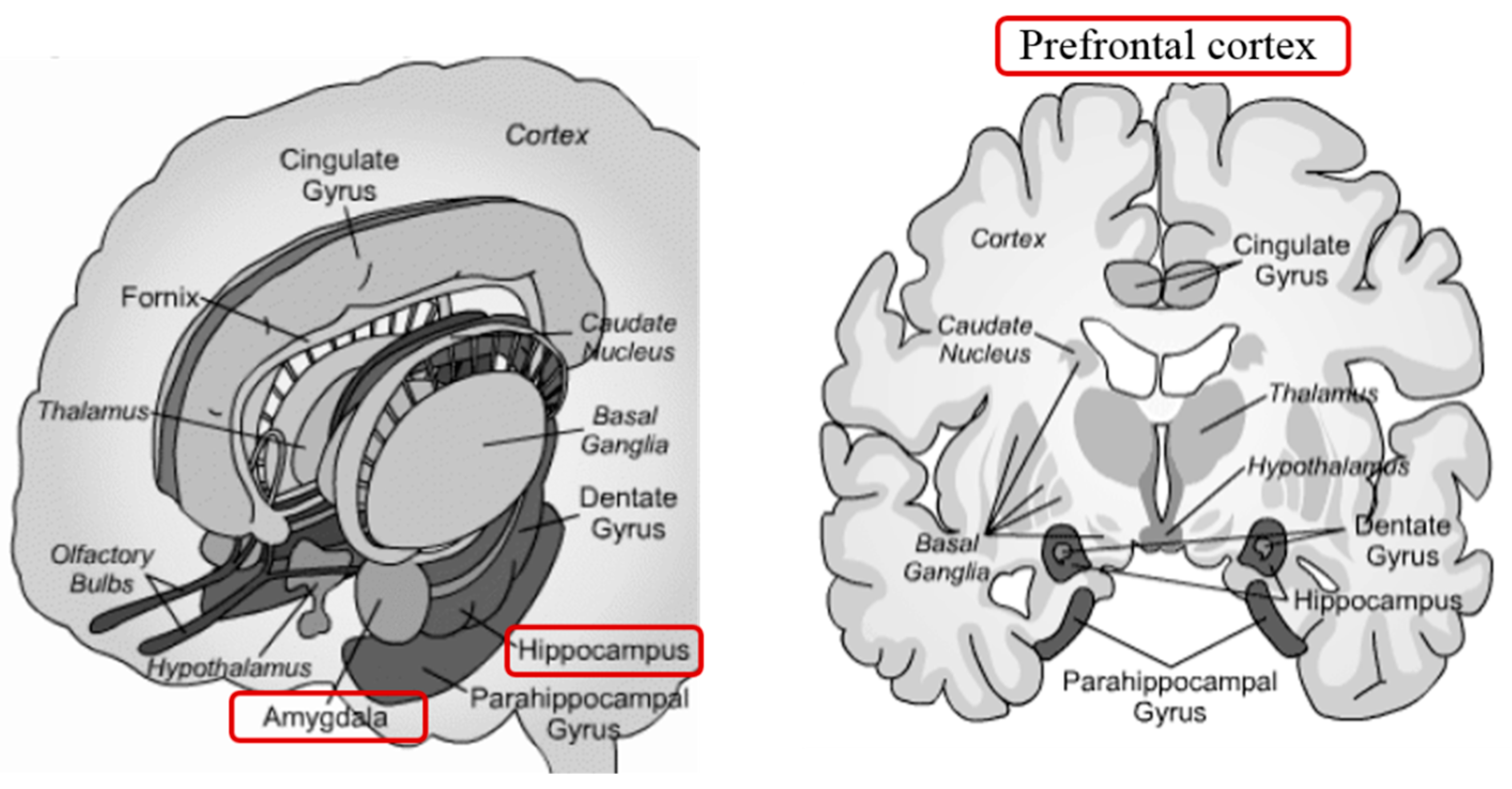
5. Neural Mechanisms Related to Stress Resilience
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rutter, M. Implications of Resilience Concepts for Scientific Understanding. Ann. N. Y. Acad. Sci. 2006, 1094, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Döring, T.F.; Vieweger, A.; Pautasso, M.; Vaarst, M.; Finckh, M.R.; Wolfe, M.S. Resilience as a Universal Criterion of Health. J. Sci. Food Agric. 2015, 95, 455–465. [Google Scholar] [CrossRef]
- Building Your Resilience. Available online: https://www.apa.org/topics/resilience (accessed on 10 October 2020).
- Dantzer, R.; Cohen, S.; Russo, S.J.; Dinan, T.G. Resilience and Immunity. Brain Behav. Immun. 2018, 74, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Colditz, I.G.; Hine, B.C. Resilience in Farm Animals: Biology, Management, Breeding and Implications for Animal Welfare. Anim. Prod. Sci. 2016, 56, 1961. [Google Scholar] [CrossRef]
- Fletcher, D.; Sarkar, M. Psychological Resilience: A Review and Critique of Definitions, Concepts, and Theory. Eur. Psychol. 2013, 18, 12–23. [Google Scholar] [CrossRef]
- Von Holst, D. The Concept of Stress and Its Relevance for Animal Behavior. In Advances in the Study of Behavior; Elsevier: Amsterdam, The Netherlands, 1998; Volume 27, pp. 1–131. ISBN 978-0-12-004527-3. [Google Scholar]
- Franklin, T.B.; Saab, B.J.; Mansuy, I.M. Neural Mechanisms of Stress Resilience and Vulnerability. Neuron 2012, 75, 747–761. [Google Scholar] [CrossRef]
- Michelena, P.; Sibbald, A.M.; Erhard, H.W.; McLeod, J.E. Effects of Group Size and Personality on Social Foraging: The Distribution of Sheep across Patches. Behav. Ecol. 2009, 20, 145–152. [Google Scholar] [CrossRef]
- Scharf, S.H.; Schmidt, M.V. Animal Models of Stress Vulnerability and Resilience in Translational Research. Curr. Psychiatry Rep. 2012, 14, 159–165. [Google Scholar] [CrossRef]
- Nederhof, E.; Schmidt, M.V. Mismatch or Cumulative Stress: Toward an Integrated Hypothesis of Programming Effects. Physiol. Behav. 2012, 106, 691–700. [Google Scholar] [CrossRef]
- Gluckman, P.D.; Hanson, M.A.; Buklijas, T.; Low, F.M.; Beedle, A.S. Epigenetic Mechanisms That Underpin Metabolic and Cardiovascular Diseases. Nat. Rev. Endocrinol. 2009, 5, 401–408. [Google Scholar] [CrossRef]
- Dhabhar, F.S. The Power of Positive Stress—A Complementary Commentary. Stress 2019, 22, 526–529. [Google Scholar] [CrossRef] [PubMed]
- Dhabhar, F.S. The Short-Term Stress Response—Mother Nature’s Mechanism for Enhancing Protection and Performance under Conditions of Threat, Challenge, and Opportunity. Front. Neuroendocrinol. 2018, 49, 175–192. [Google Scholar] [CrossRef] [PubMed]
- Hornor, G. Resilience. J. Pediatr. Health Care 2017, 31, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Dhabhar, F.S. Acute Stress Enhances While Chronic Stress Suppresses Skin Immunity: The Role of Stress Hormones and Leukocyte Trafficking. Ann. N. Y. Acad. Sci. 2006, 917, 876–893. [Google Scholar] [CrossRef]
- Dhabhar, F.S. Effects of Stress on Immune Function: The Good, the Bad, and the Beautiful. Immunol Res 2014, 58, 193–210. [Google Scholar] [CrossRef]
- McEWEN, B.S. Stress, Adaptation, and Disease: Allostasis and Allostatic Load. Ann. N. Y. Acad. Sci. 1998, 840, 33–44. [Google Scholar] [CrossRef]
- Daskalakis, N.P.; Bagot, R.C.; Parker, K.J.; Vinkers, C.H.; de Kloet, E.R. The Three-Hit Concept of Vulnerability and Resilience: Toward Understanding Adaptation to Early-Life Adversity Outcome. Psychoneuroendocrinology 2013, 38, 1858–1873. [Google Scholar] [CrossRef]
- Fagiolini, M.; Jensen, C.L.; Champagne, F.A. Epigenetic Influences on Brain Development and Plasticity. Curr. Opin. Neurobiol. 2009, 19, 207–212. [Google Scholar] [CrossRef]
- Zhang, Y.; Kutateladze, T. Diet and the Epigenome. Nat. Commun. 2018, 9, 3375. [Google Scholar] [CrossRef]
- Rutter, M. Resilience in the Face of Adversity: Protective Factors and Resistance to Psychiatric Disorder. Br. J. Psychiatry 1985, 147, 598–611. [Google Scholar] [CrossRef]
- Willett, W.C. Balancing Life-Style and Genomics Research for Disease Prevention. Science 2002, 296, 695–698. [Google Scholar] [CrossRef] [PubMed]
- Scheffer, M.; Bolhuis, J.E.; Borsboom, D.; Buchman, T.G.; Gijzel, S.M.W.; Goulson, D.; Kammenga, J.E.; Kemp, B.; van de Leemput, I.A.; Levin, S.; et al. Quantifying resilience of humans and other animals. Proc. Natl. Acad. Sci. USA 2018, 115, 11883–11890. [Google Scholar] [CrossRef] [PubMed]
- Stone, J.; Mitrofanis, J.; Johnstone, D.M.; Falsini, B.; Bisti, S.; Adam, P.; Nuevo, A.B.; George-Weinstein, M.; Mason, R.; Eells, J. Acquired Resilience: An Evolved System of Tissue Protection in Mammals. Dose-Response 2018, 16, 155932581880342. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-W.; Na, Y.; Park, N.; Kim, H.-S.; Ahn, S.M.; Kim, J.W.; Kim, H.-K.; Jang, Y.P. Amentoflavone Inhibits UVB-Induced Matrix Metalloproteinase-1 Expression Through the Modulation of AP-1 Components in Normal Human Fibroblasts. Appl. Biochem. Biotechnol. 2012, 166, 1137–1147. [Google Scholar] [CrossRef]
- Bickers, D.R.; Athar, M. Oxidative Stress in the Pathogenesis of Skin Disease. J. Investig. Dermatol. 2006, 126, 2565–2575. [Google Scholar] [CrossRef]
- Hong, M.J.; Ko, E.B.; Park, S.K.; Chang, M.S. Inhibitory effect of Astragalus membranaceus root on matrix metalloproteinase-1 collagenase expression and procollagen destruction in ultraviolet B-irradiated human dermal fibroblasts by suppressing nuclear factor kappa-B activity: Effect of A. membranaceus on NF-κB. J. Pharm. Pharmacol. 2013, 65, 142–148. [Google Scholar] [CrossRef]
- Hwang, Y.P.; Kim, H.G.; Choi, J.H.; Han, E.H.; Kwon, K.; Lee, Y.C.; Choi, J.M.; Chung, Y.C.; Jeong, T.C.; Jeong, H.G. Saponins from the roots of Platycodon grandiflorum suppress ultraviolet A-induced matrix metalloproteinase-1 expression via MAPKs and NF-κB/AP-1-dependent signaling in HaCaT cells. Food Chem. Toxicol. 2011, 49, 3374–3382. [Google Scholar] [CrossRef]
- Zhao, D.; Yu, Y.; Shen, Y.; Liu, Q.; Zhao, Z.; Sharma, R.; Reiter, R.J. Melatonin Synthesis and Function: Evolutionary History in Animals and Plants. Front. Endocrinol. 2019, 10, 249. [Google Scholar] [CrossRef]
- Jung, C.M.; Khalsa, S.B.S.; Scheer, F.A.J.L.; Cajochen, C.; Lockley, S.W.; Czeisler, C.A.; Wright, K.P. Acute Effects of Bright Light Exposure on Cortisol Levels. J. Biol. Rhythms 2010, 25, 208–216. [Google Scholar] [CrossRef]
- Mason, R.S.; Reichrath, J. Sunlight vitamin D and skin cancer. Anticancer Agents Med. Chem. 2013, 13, 83–97. [Google Scholar] [CrossRef]
- Tongkao-on, W.; Carter, S.; Reeve, V.E.; Dixon, K.M.; Gordon-Thomson, C.; Halliday, G.M.; Tuckey, R.C.; Mason, R.S. CYP11A1 in skin: An alternative route to photoprotection by vitamin D compounds. J. Steroid Biochem. Mol. Biol. 2015, 148, 72–78. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, B.Y.; Dixon, K.M.; Halliday, G.M.; Reeve, V.E.; Mason, R.S. The Vitamin D Saga: Breaking Dawn. IEMAMC 2015, 14, 137–151. [Google Scholar] [CrossRef]
- Ferraresi, C.; Hamblin, M.R.; Parizotto, N.A. Low-level laser (light) therapy (LLLT) on muscle tissue: Performance, fatigue and repair benefited by the power of light. Photonics Lasers Med. 2012, 1. [Google Scholar] [CrossRef]
- Penn, J.S.; Tolman, B.L.; Thum, L.A.; Koutz, C.A. Effect of light history on the rat retina: Timecourse of morphological adaptation and readaptation. Neurochem. Res. 1992, 17, 91–99. [Google Scholar] [CrossRef]
- Wen, R.; Song, Y.; Cheng, T.; Matthes, M.T.; Yasumura, D.; LaVail, M.M.; Steinberg, R.H. Injury-induced upregulation of bFGF and CNTF mRNAS in the rat retina. J. Neurosci. 1995, 15, 7377–7385. [Google Scholar] [CrossRef] [PubMed]
- Li, G.C.; Hahn, G.M. A proposed operational model of thermotolerance based on effects of nutrients and the initial treatment temperature. Cancer Res. 1980, 40, 4501–4508. [Google Scholar]
- Li, G.C.; Meyer, J.L.; Mak, J.Y.; Hahn, G.M. Heat-induced protection of mice against thermal death. Cancer Res. 1983, 43, 5758–5760. [Google Scholar]
- Shilja, S.; Sejian, V.; Bagath, M.; Mech, A.; David, C.G.; Kurien, E.K.; Varma, G.; Bhatta, R. Adaptive capability as indicated by behavioral and physiological responses, plasma HSP70 level, and PBMC HSP70 mRNA expression in Osmanabadi goats subjected to combined (heat and nutritional) stressors. Int. J. Biometeorol. 2016, 60, 1311–1323. [Google Scholar] [CrossRef]
- Aleena, J.; Sejian, V.; Bagath, M.; Krishnan, G.; Beena, V.; Bhatta, R. Resilience of three indigenous goat breeds to heat stress based on phenotypic traits and PBMC HSP70 expression. Int. J. Biometeorol. 2018, 62, 1995–2005. [Google Scholar] [CrossRef]
- Madhusoodan, A.P.; Bagath, M.; Sejian, V.; Krishnan, G.; Rashamol, V.P.; Savitha, S.T.; Awachat, V.B.; Bhatta, R. Summer season induced changes in quantitative expression patterns of different heat shock response genes in Salem black goats. Trop. Anim. Health Prod. 2020, 52, 2725–2730. [Google Scholar] [CrossRef]
- Aleena, J.; Pragna, P.; Archana, P.R.; Sejian, V.; Bagath, M.; Krishnan, G.; Manimaran, A.; Beena, V.; Kurien, E.K.; Varma, G.; et al. Significance of Metabolic Response in Livestock for Adapting to Heat Stress Challenges. Asian J. Anim. Sci. 2016, 10, 224–234. [Google Scholar] [CrossRef]
- McManus, C.; Louvandini, H.; Gugel, R.; Sasaki, L.C.B.; Bianchini, E.; Bernal, F.E.M.; Paiva, S.R.; Paim, T.P. Skin and coat traits in sheep in Brazil and their relation with heat tolerance. Trop. Anim. Health Prod. 2011, 43, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Mwacharo, J.M.; Kim, E.-S.; Elbeltagy, A.R.; Aboul-Naga, A.M.; Rischkowsky, B.A.; Rothschild, M.F. Genomic footprints of dryland stress adaptation in Egyptian fat-tail sheep and their divergence from East African and western Asia cohorts. Sci. Rep. 2017, 7, 17647. [Google Scholar] [CrossRef] [PubMed]
- Sejian, V.; Bagath, M.; Krishnan, G.; Rashamol, V.P.; Pragna, P.; Devaraj, C.; Bhatta, R. Genes for resilience to heat stress in small ruminants: A review. Small Rumin. Res. 2019, 173, 42–53. [Google Scholar] [CrossRef]
- Wei, S.; Shen, X.; Gong, Z.; Deng, Y.; Lai, L.; Liang, H. FSHR and LHR Expression and Signaling as Well as Maturation and Apoptosis of Cumulus-Oocyte Complexes Following Treatment with FSH Receptor Binding Inhibitor in Sheep. Cell Physiol. Biochem. 2017, 43, 660–669. [Google Scholar] [CrossRef]
- Ozawa, M.; Tabayashi, D.; Latief, T.A.; Shimizu, T.; Oshima, I.; Kanai, Y. Alterations in follicular dynamics and steroidogenic abilities induced by heat stress during follicular recruitment in goats. Reproduction 2005, 129, 621–630. [Google Scholar] [CrossRef]
- Ritossa, F. A new puffing pattern induced by temperature shock and DNP in drosophila. Experientia 1962, 18, 571–573. [Google Scholar] [CrossRef]
- Matz, J.M.; Blake, M.J.; Tatelman, H.M.; Lavoi, K.P.; Holbrook, N.J. Characterization and regulation of cold-induced heat shock protein expression in mouse brown adipose tissue. Am. J. Physiol. Renal Physiol. 1995, 269, R38–R47. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Ohwatari, N.; Matsumoto, T.; Kosaka, M.; Ohtsuru, A.; Yamashita, S. TGF-β1 mediates 70-kDa heat shock protein induction due to ultraviolet irradiation in human skin fibroblasts. Pflugers Arch. Eur. J. Physiol. 1999, 438, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Laplante, A.F.; Moulin, V.; Auger, F.A.; Landry, J.; Li, H.; Morrow, G.; Tanguay, R.M.; Germain, L. Expression of Heat Shock Proteins in Mouse Skin During Wound Healing. J. Histochem. Cytochem. 1998, 46, 1291–1301. [Google Scholar] [CrossRef] [PubMed]
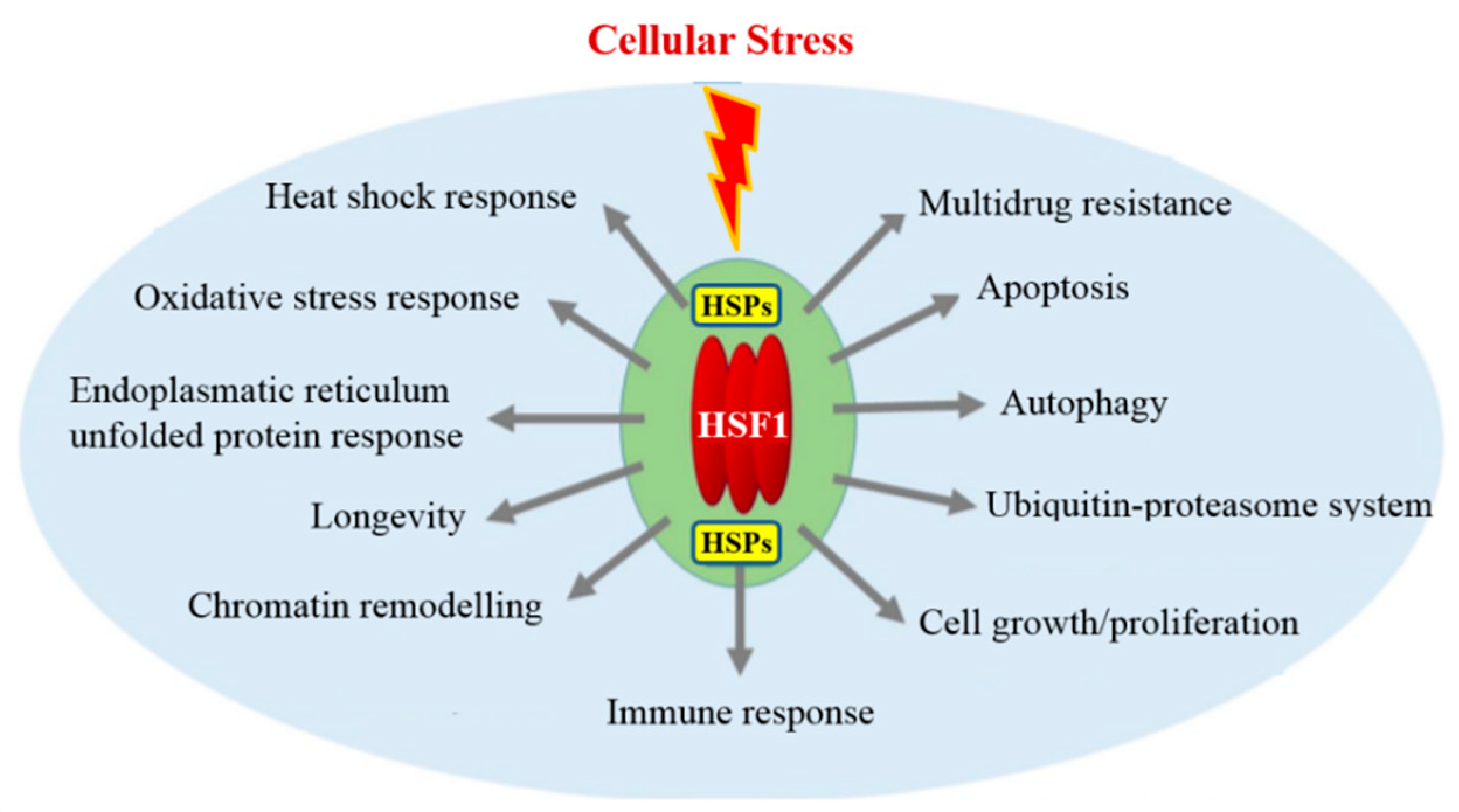
- Barna, J.; Csermely, P.; Vellai, T. Roles of heat shock factor 1 beyond the heat shock response. Cell. Mol. Life Sci. 2018, 75, 2897–2916. [Google Scholar] [CrossRef] [PubMed]
- Moler, F.W.; Silverstein, F.S.; Holubkov, R.; Slomine, B.S.; Christensen, J.R.; Nadkarni, V.M.; Meert, K.L.; Browning, B.; Pemberton, V.L.; Page, K.; et al. Therapeutic Hypothermia after Cardiac Arrest. N. Eng. J. Med. 2017, 376, 318–329. [Google Scholar] [CrossRef] [PubMed]
- Sonna, L.A.; Fujita, J.; Gaffin, S.L.; Lilly, C.M. Invited review: Effects of heat and cold stress on mammalian gene expression. J. Appl. Physiol. 2002, 92, 1725–1742. [Google Scholar] [CrossRef] [PubMed]
- Sahara, T.; Goda, T.; Ohgiya, S. Comprehensive Expression Analysis of Time-dependent Genetic Responses in Yeast Cells to Low Temperature. J. Biol. Chem. 2002, 277, 50015–50021. [Google Scholar] [CrossRef]
- Zhu, X.; Bührer, C.; Wellmann, S. Cold-inducible proteins CIRP and RBM3, a unique couple with activities far beyond the cold. Cell. Mol. Life Sci. 2016, 73, 3839–3859. [Google Scholar] [CrossRef]
- Fujita, J. Cold shock response in mammalian cells. J. Mol. Microbiol. Biotechnol. 1999, 2, 243–255. [Google Scholar]
- Leonart, M.E. A new generation of proto-oncogenes: Cold-inducible R.N.A. binding proteins. Biochimica et Biophysica Acta (BBA)—Rev. Cancer 2010, 1805, 43–52. [Google Scholar] [CrossRef]
- De Leeuw, F.; Zhang, T.; Wauquier, C.; Huez, G.; Kruys, V.; Gueydan, C. The cold-inducible RNA-binding protein migrates from the nucleus to cytoplasmic stress granules by a methylation-dependent mechanism and acts as a translational repressor. Exp. Cell Res. 2007, 313, 4130–4144. [Google Scholar] [CrossRef]
- Anderson, P.; Kedersha, N. RNA granules: Post-transcriptional and epigenetic modulators of gene expression. Nat. Rev. Mol. Cell Biol. 2009, 10, 430–436. [Google Scholar] [CrossRef]
- Nishiyama, H.; Itoh, K.; Kaneko, Y.; Kishishita, M.; Yoshida, O.; Fujita, J. A Glycine-rich RNA-binding Protein Mediating Cold-inducible Suppression of Mammalian Cell Growth. J. Cell Biol. 1997, 137, 899–908. [Google Scholar] [CrossRef]
- Sakurai, T.; Itoh, K.; Higashitsuji, H.; Nonoguchi, K.; Liu, Y.; Watanabe, H.; Nakano, T.; Fukumoto, M.; Chiba, T.; Fujita, J. Cirp protects against tumor necrosis factor-α-induced apoptosis via activation of extracellular signal-regulated kinase. Biochim. Biophys. Mol. Cell Res. 2006, 1763, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Murry, C.E.; Jennings, R.B.; Reimer, K.A. Preconditioning with ischemia: A delay of lethal cell injury in ischemic myocardium. Circulation 1986, 74, 1124–1136. [Google Scholar] [CrossRef]
- Barone, F.C.; White, R.F.; Spera, P.A.; Ellison, J.; Currie, R.W.; Wang, X.; Feuerstein, G.Z. Ischemic Preconditioning and Brain Tolerance: Temporal Histological and Functional Outcomes, Protein Synthesis Requirement, and Interleukin-1 Receptor Antagonist and Early Gene Expression. Stroke 1998, 29, 1937–1951. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, N.; Wang, Y.; Mukasa, A.; Furuya, K.; Shimizu, T.; Hamakubo, T.; Aburatani, H.; Kodama, T.; Kirino, T. Genome-Wide Gene Expression Analysis for Induced Ischemic Tolerance and Delayed Neuronal Death following Transient Global Ischemia in Rats. J. Cereb. Blood Flow Metab. 2004, 24, 212–233. [Google Scholar] [CrossRef] [PubMed]
- Kirino, T. Ischemic Tolerance. J. Cereb. Blood Flow Metab. 2002, 22, 1283–1296. [Google Scholar] [CrossRef] [PubMed]
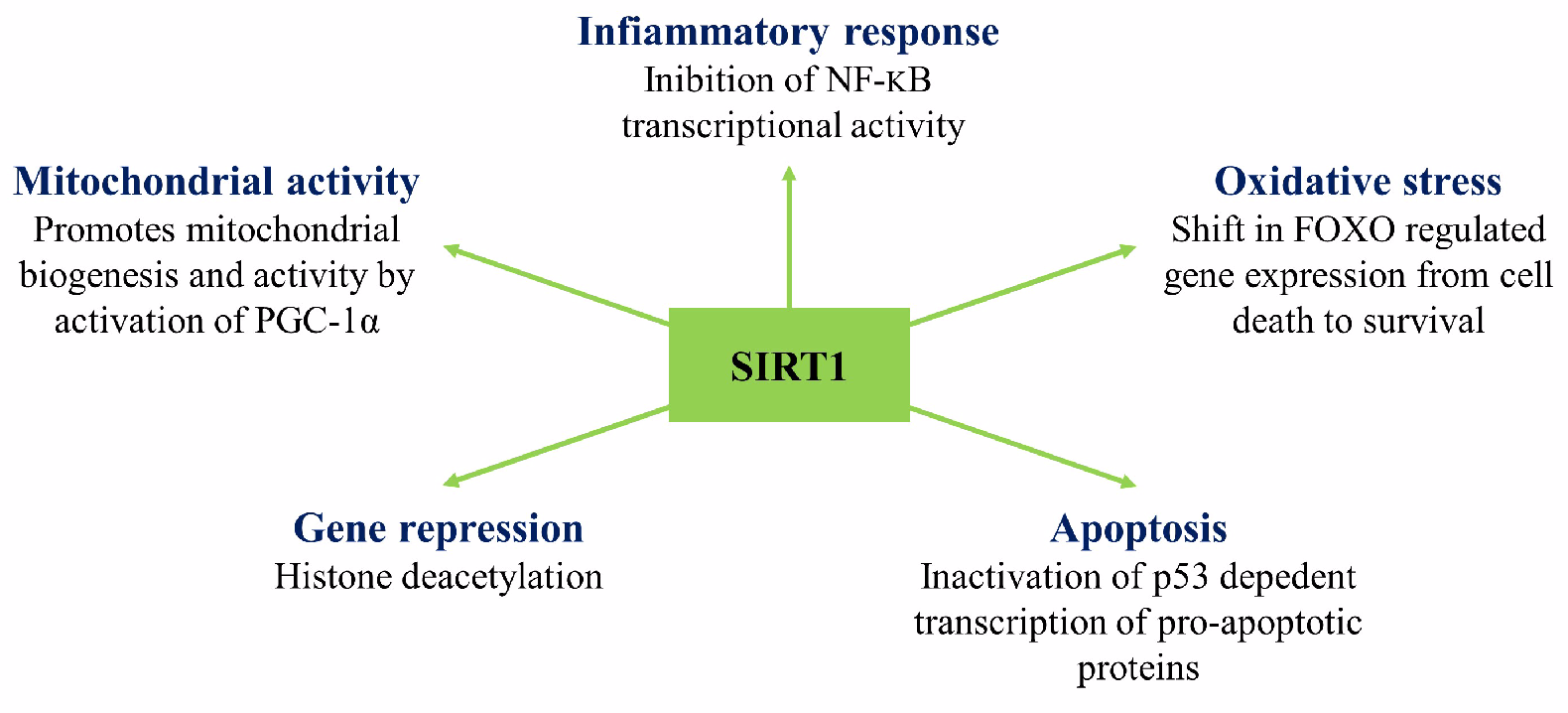
- Morris, K.C.; Lin, H.W.; Thompson, J.W.; Perez-Pinzon, M.A. Pathways for Ischemic Cytoprotection: Role of Sirtuins in Caloric Restriction, Resveratrol, and Ischemic Preconditioning. J. Cereb. Blood Flow Metab. 2011, 31, 1003–1019. [Google Scholar] [CrossRef]
- Brooks, M.J.; Andrews, D.T. Molecular mechanisms of ischemic conditioning: Translation into patient outcomes. Future Cardiol. 2013, 9, 549–568. [Google Scholar] [CrossRef]
- Liu, W.; Shen, S.M.; Zhao, X.Y.; Chen, G.Q. Targeted genes and interacting proteins of hypoxia inducible factor-1. Int. J. Biochem. Mol. Biol. 2012, 3, 165–178. [Google Scholar]
- Lim, J.-H.; Lee, Y.-M.; Chun, Y.-S.; Chen, J.; Kim, J.-E.; Park, J.-W. Sirtuin 1 Modulates Cellular Responses to Hypoxia by Deacetylating Hypoxia-Inducible Factor 1α. Mol. Cell 2010, 38, 864–878. [Google Scholar] [CrossRef]
- Guelfi, G.; Casano, A.B.; Menchetti, L.; Bellicci, M.; Suvieri, C.; Moscati, L.; Carotenuto, P.; Santoro, M.M.; Diverio, S. A cross-talk between blood-cell neuroplasticity-related genes and environmental enrichment in working dogs. Sci. Rep. 2019, 9, 6910. [Google Scholar] [CrossRef]
- Menchetti, L.; Guelfi, G.; Speranza, R.; Carotenuto, P.; Moscati, L.; Diverio, S. Benefits of dietary supplements on the physical fitness of German Shepherd dogs during a drug detection training course. PLoS ONE 2019, 14, e0218275. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-W.; Friso, S. Epigenetics: A New Bridge between Nutrition and Health. Adv. Nutr. 2010, 1, 8–16. [Google Scholar] [CrossRef]
- Camporeale, G.; Giordano, E.; Rendina, R.; Zempleni, J.; Eissenberg, J.C. Drosophila melanogaster Holocarboxylase Synthetase Is a Chromosomal Protein Required for Normal Histone Biotinylation, Gene Transcription Patterns, Lifespan, and Heat Tolerance. J. Nutr. 2006, 136, 2735–2742. [Google Scholar] [CrossRef][Green Version]
- Zempleni, J.; Chew, Y.C.; Bao, B.; Pestinger, V.; Wijeratne, S.S.K. Repression of Transposable Elements by Histone Biotinylation. J. Nutr. 2009, 139, 2389–2392. [Google Scholar] [CrossRef] [PubMed]
- Landecker, H. Food as exposure: Nutritional epigenetics and the new metabolism. BioSocieties 2011, 6, 167–194. [Google Scholar] [CrossRef] [PubMed]
- Bolhuis, J.E.; Schouten, W.G.P.; Schrama, J.W.; Wiegant, V.M. Effects of rearing and housing environment on behaviour and performance of pigs with different coping characteristics. Appl. Anim. Behav. Sci. 2006, 101, 68–85. [Google Scholar] [CrossRef]
- Oostindjer, M.; Kemp, B.; van den Brand, H.; Bolhuis, J.E. Facilitating ‘learning from mom how to eat like a pig’ to improve welfare of piglets around weaning. Appl. Anim. Behav. Sci. 2014, 160, 19–30. [Google Scholar] [CrossRef]
- Chapillon, P.; Manneché, C.; Belzung, C.; Caston, J. Rearing environmental enrichment in two inbred strains of mice: 1. Effects on emotional reactivity. Behav. Genet. 1999, 1, 41–46. [Google Scholar] [CrossRef]
- Dibner, J.J.; Knight, C.D.; Kitchell, M.L.; Atwell, C.A.; Downs, A.C.; Ivey, F.J. Early Feeding and Development of the Immune System in Neonatal Poultry. J. Appl. Poult. Res. 1998, 7, 425–436. [Google Scholar] [CrossRef]
- Bhanja, S.K.; Bhadauria, P. Behaviour and Welfare Concepts in Laying Hens and Their Association with Housing Systems. India. J. Poult. Sci. 2018, 53, 1. [Google Scholar] [CrossRef]
- Ellen, H.H.; Bottcher, R.W.; von Wachenfelt, E.; Takai, H. Dust levels and control methods in poultry houses. J. Agric. Saf. Health 2000, 6, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Just, N.; Kirychuk, S.; Gilbert, Y.; Létourneau, V.; Veillette, M.; Singh, B.; Duchaine, C. Bacterial diversity characterization of bioaerosols from cage-housed and floor-housed poultry operations. Environ. Res. 2011, 111, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Le Bouquin, S.; Huneau-Salaün, A.; Huonnic, D.; Balaine, L.; Martin, S.; Michel, V. Aerial dust concentration in cage-housed, floor-housed, and aviary facilities for laying hens. Poult. Sci. 2013, 92, 2827–2833. [Google Scholar] [CrossRef] [PubMed]
- Moe, R.O.; Guémené, D.; Bakken, M.; Larsen, H.J.S.; Shini, S.; Lervik, S.; Skjerve, E.; Michel, V.; Tauson, R. Effects of housing conditions during the rearing and laying period on adrenal reactivity, immune response and heterophil to lymphocyte (H/L) ratios in laying hens. Animal 2010, 4, 1709–1715. [Google Scholar] [CrossRef]
- Robinson, G.E.; Fernald, R.D.; Clayton, D.F. Genes and social behavior. Science 2008, 322, 896–900. [Google Scholar] [CrossRef]
- Beery, A.K.; Kaufer, D. Stress, social behavior, and resilience: Insights from rodents. Neurobiol. Stress 2015, 1, 116–127. [Google Scholar] [CrossRef]
- Ellen, E.D.; Rodenburg, T.B.; Albers, G.A.A.; Bolhuis, J.E.; Camerlink, I.; Duijvesteijn, N.; Knol, E.F.; Muir, W.M.; Peeters, K.; Reimert, I.; et al. The prospects of selection for social genetic effects to improve welfare and productivity in livestock. Front Genet. 2014, 5. [Google Scholar] [CrossRef]
- Kanitz, E.; Tuchscherer, M.; Puppe, B.; Tuchscherer, A.; Stabenow, B. Consequences of repeated early isolation in domestic piglets (Sus scrofa) on their behavioural, neuroendocrine, and immunological responses. Brain Behav. Immun. 2004, 18, 35–45. [Google Scholar] [CrossRef]
- Ellen, E.D.; Visscher, J.; van Arendonk, J.A.M.; Bijma, P. Survival of Laying Hens: Genetic Parameters for Direct and Associative Effects in Three Purebred Layer Lines. Poult. Sci. 2008, 87, 233–239. [Google Scholar] [CrossRef]
- Felger, J.C.; Haroon, E.; Miller, A.H. Risk and Resilience: Animal Models Shed Light on the Pivotal Role of Inflammation in Individual Differences in Stress-Induced Depression. Biol. Psychiatry 2015, 78, 7–9. [Google Scholar] [CrossRef]
- Shu, W.; Cho, J.Y.; Jiang, Y.; Zhang, M.; Weisz, D.; Elder, G.A.; Schmeidler, J.; De Gasperi, R.; Sosa, M.A.G.; Rabidou, D.; et al. Altered ultrasonic vocalization in mice with a disruption in the Foxp2 gene. Proc. Natl. Acad. Sci. USA 2005, 102, 9643–9648. [Google Scholar] [CrossRef] [PubMed]
- Toth, A.L.; Robinson, G.E. Evo-devo and the evolution of social behavior. Trends Genet. 2007, 23, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.; Calvo-Torrent, A.; Pico-Alfonso, M.A. Social defeat and subordination as models of social stress in laboratory rodents: A review. Aggr. Behav. 1998, 24, 241–256. [Google Scholar] [CrossRef]
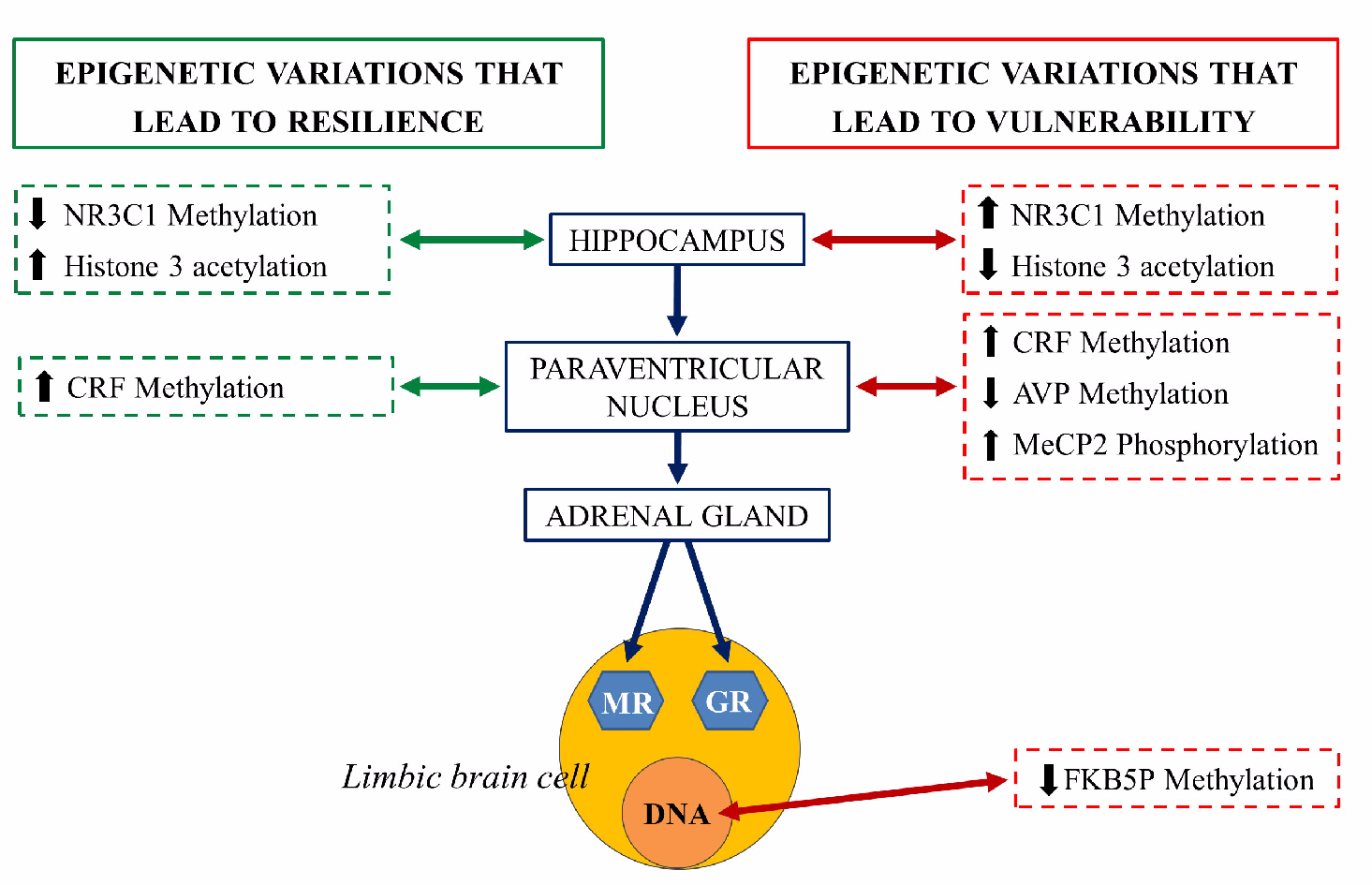
- Zannas, A.S.; West, A.E. Epigenetics and the regulation of stress vulnerability and resilience. Neuroscience 2014, 264, 157–170. [Google Scholar] [CrossRef]
- Dudley, K.J.; Li, X.; Kobor, M.S.; Kippin, T.E.; Bredy, T.W. Epigenetic mechanisms mediating vulnerability and resilience to psychiatric disorders. Neurosci. Biobehav. Rev. 2011, 35, 1544–1551. [Google Scholar] [CrossRef]
- Roth, T.L.; Zoladz, P.R.; Sweatt, J.D.; Diamond, D.M. Epigenetic modification of hippocampal Bdnf D.N.A. in adult rats in an animal model of post-traumatic stress disorder. J. Psychiatr. Res. 2011, 45, 919–926. [Google Scholar] [CrossRef]
- Elliott, E.; Ezra-Nevo, G.; Regev, L.; Neufeld-Cohen, A.; Chen, A. Resilience to social stress coincides with functional DNA methylation of the Crf gene in adult mice. Nat. Neurosci. 2010, 13, 1351–1353. [Google Scholar] [CrossRef]
- Uchida, S.; Hara, K.; Kobayashi, A.; Otsuki, K.; Yamagata, H.; Hobara, T.; Suzuki, T.; Miyata, N.; Watanabe, Y. Epigenetic status of Gdnf in the ventral striatum determines susceptibility and adaptation to daily stressful events. Neuron 2011, 69, 359–372. [Google Scholar] [CrossRef]
- Vitellius, G.; Trabado, S.; Bouligand, J.; Delemer, B.; Lombès, M. Pathophysiology of Glucocorticoid Signaling. Ann. Endocrinol. 2018, 79, 98–106. [Google Scholar] [CrossRef]
- Weaver, I.C.G.; Cervoni, N.; Champagne, F.A.; D’Alessio, A.C.; Sharma, S.; Seckl, J.R.; Dymov, S.; Szyf, M.; Meaney, M.J. Epigenetic programming by maternal behavior. Nat. Neurosci. 2004, 7, 847–854. [Google Scholar] [CrossRef]
- Van Bodegom, M.; Homberg, J.R.; Henckens, M.J.A.G. Modulation of the Hypothalamic-Pituitary-Adrenal Axis by Early Life Stress Exposure. Front. Cell. Neurosci. 2017, 11. [Google Scholar] [CrossRef]
- Zapata-Martín del Campo, C.; Martínez-Rosas, M.; Guarner-Lans, V. Epigenetic programming of synthesis, release, and/or receptor expression of common mediators participating in the risk/resilience for comorbid stress-related disorders and coronary artery disease. Int. J. Mol. Sci. 2018, 19, 1224. [Google Scholar] [CrossRef] [PubMed]
- Murgatroyd, C.; Patchev, A.V.; Wu, Y.; Micale, V.; Bockmühl, Y.; Fischer, D.; Holsboer, F.; Wotjak, C.T.; Almeida, O.F.X.; Spengler, D. Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat. Neurosci. 2009, 12, 1559–1566. [Google Scholar] [CrossRef]
- Schiele, M.A.; Domschke, K. Epigenetics at the crossroads between genes, environment and resilience in anxiety disorders. Genes Brain Behav. 2018, 17, e12423. [Google Scholar] [CrossRef]
- Weiss, I.C.; Franklin, T.B.; Vizi, S.; Mansuy, I.M. Inheritable effect of unpredictable maternal separation on behavioral responses in mice. Front. Behav. Neurosci. 2011, 5. [Google Scholar] [CrossRef] [PubMed]
- Kentner, A.C.; Lambert, K.G.; Hannan, A.J.; Donaldson, S.T. Editorial: Environmental enrichment: Enhancing neural plasticity, resilience, and repair. Front. Behav. Neurosci. 2019, 13, 75. [Google Scholar] [CrossRef] [PubMed]
- Somjen, G.G. The Missing Error Signal—Regulation Beyond Negative Feedback. Physiology 1992, 7, 184–185. [Google Scholar] [CrossRef]
- Carrasco, G.A.; Van de Kar, L.D. Neuroendocrine pharmacology of stress. Eur. J. Pharmacol. 2003, 463, 235–272. [Google Scholar] [CrossRef]
- Ménard, C.; Pfau, M.L.; Hodes, G.E.; Russo, S.J. Immune and Neuroendocrine Mechanisms of Stress Vulnerability and Resilience. Neuropsychopharmacol 2017, 42, 62–80. [Google Scholar] [CrossRef]
- Wang, J.; Hodes, G.E.; Zhang, H.; Zhang, S.; Zhao, W.; Golden, S.A.; Bi, W.; Menard, C.; Kana, V.; Leboeuf, M.; et al. Epigenetic modulation of inflammation and synaptic plasticity promotes resilience against stress in mice. Nat. Commun. 2018, 9, 477. [Google Scholar] [CrossRef]
- Tsigos, C.; Kyrou, I.; Kassi, E.; Chrousos, G.P. Stress: Endocrine Physiology and Pathophysiology. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dungan, K., Eds.; MDText.com Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- McEwen, B.S.; Gray, J.D.; Nasca, C. Recognizing resilience: Learning from the effects of stress on the brain. Neurobiol. Stress 2015, 1, 1–11. [Google Scholar] [CrossRef]
- Mcewen, B.S.; Weiss, J.M.; Schwartz, L.S. Selective retention of corticosterone by limbic structures in rat brain. Nature 1968, 220, 911–912. [Google Scholar] [CrossRef]
- Jankord, R.; Herman, J.P. Limbic regulation of hypothalamo-pituitary-adrenocortical function during acute and chronic stress. Ann. N. Y. Acad. Sci. 2008, 1148, 64–73. [Google Scholar] [CrossRef]
- Lupien, S.; Mcewen, B. The acute effects of corticosteroids on cognition: Integration of animal and human model studies. Brain Res. Rev. 1997, 24, 1–27. [Google Scholar] [CrossRef]
- Derks, N.A.V.; Krugers, H.J.; Hoogenraad, C.C.; Joëls, M.; Sarabdjitsingh, R.A. Effects of early life stress on synaptic plasticity in the developing hippocampus of male and female rats. PLoS ONE 2016, 11, e0164551. [Google Scholar] [CrossRef]
- Kim, J.J.; Song, E.Y.; Kim, J.J.; Song, E.Y.; Kosten, T.A. Stress effects in the hippocampus: Synaptic plasticity and memory. Stress 2006, 9, 1–11. [Google Scholar] [CrossRef]
- Arnsten, A.F.T. Stress signalling pathways that impair prefrontal cortex structure and function. Nat. Rev. Neurosci. 2009, 10, 410–422. [Google Scholar] [CrossRef]
- Liston, C.; Miller, M.M.; Goldwater, D.S.; Radley, J.J.; Rocher, A.B.; Hof, P.R.; Morrison, J.H.; McEwen, B.S. Stress-Induced Alterations in Prefrontal Cortical Dendritic Morphology Predict Selective Impairments in Perceptual Attentional Set-Shifting. J. Neurosci. 2006, 26, 7870–7874. [Google Scholar] [CrossRef]
- Ladd, C.O.; Huot, R.L.; Thrivikraman, K.V.; Nemeroff, C.B.; Plotsky, P.M. Long-term adaptations in glucocorticoid receptor and mineralocorticoid receptor mrna and negative feedback on the hypothalamo-pituitary-adrenal axis following neonatal maternal separation. Biol Psychiatry. 2004, 55, 367–375. [Google Scholar] [CrossRef]
- Covington, H.E.; Lobo, M.K.; Maze, I.; Vialou, V.; Hyman, J.M.; Zaman, S.; LaPlant, Q.; Mouzon, E.; Ghose, S.; Tamminga, C.A.; et al. Antidepressant Effect of Optogenetic Stimulation of the Medial Prefrontal Cortex. J. Neurosci. 2010, 30, 16082–16090. [Google Scholar] [CrossRef]
- Brett, T. Litz, Resilience and Mental Health: Challenges across the Lifespan; Southwick, S.M., Ed.; Cambridge University Press: Cambridge, UK, 2011; ISBN 978-0-521-89839-3. [Google Scholar]
- Peckham, H. Epigenetics: The Dogma-defying Discovery That Genes Learn From Experience. IJNPT 2013, 1, 9–20. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basile, F.; Capaccia, C.; Zampini, D.; Biagetti, T.; Diverio, S.; Guelfi, G. Omics Insights into Animal Resilience and Stress Factors. Animals 2021, 11, 47. https://doi.org/10.3390/ani11010047
Basile F, Capaccia C, Zampini D, Biagetti T, Diverio S, Guelfi G. Omics Insights into Animal Resilience and Stress Factors. Animals. 2021; 11(1):47. https://doi.org/10.3390/ani11010047
Chicago/Turabian StyleBasile, Federica, Camilla Capaccia, Danilo Zampini, Tommaso Biagetti, Silvana Diverio, and Gabriella Guelfi. 2021. "Omics Insights into Animal Resilience and Stress Factors" Animals 11, no. 1: 47. https://doi.org/10.3390/ani11010047
APA StyleBasile, F., Capaccia, C., Zampini, D., Biagetti, T., Diverio, S., & Guelfi, G. (2021). Omics Insights into Animal Resilience and Stress Factors. Animals, 11(1), 47. https://doi.org/10.3390/ani11010047






