Toxic Wasting Disorders in Sheep
Simple Summary
Abstract
1. Introduction
2. Inorganic and Organic Toxic Compounds
2.1. Copper
2.2. Nitrogen-Containing Compounds
2.2.1. Nonprotein Nitrogen Sources
2.2.2. Nitrates and Nitrites
2.3. Fluoride/Fluorine
2.4. Lead
2.5. Selenium
2.6. Zinc
2.7. Arsenic
2.8. Others
3. Toxic Plants
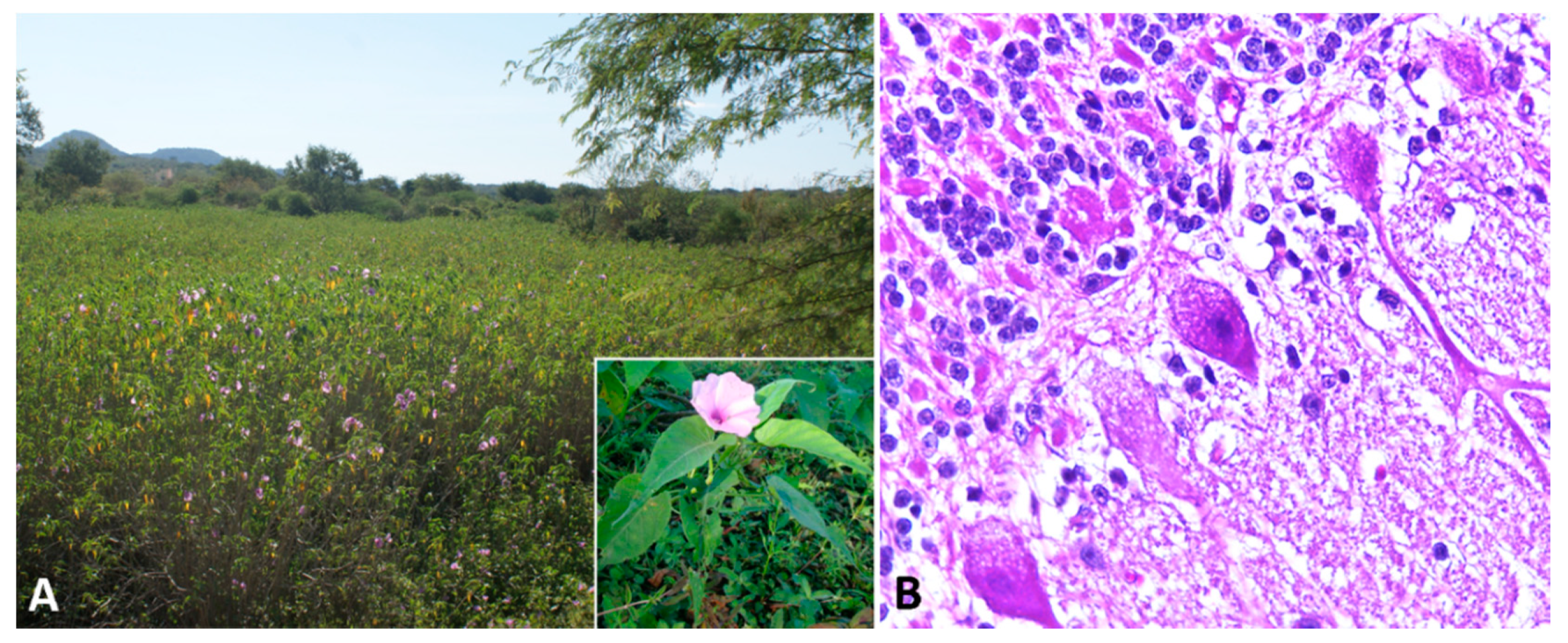
3.1. Pyrrolizidine Alkaloid-Containing Plants
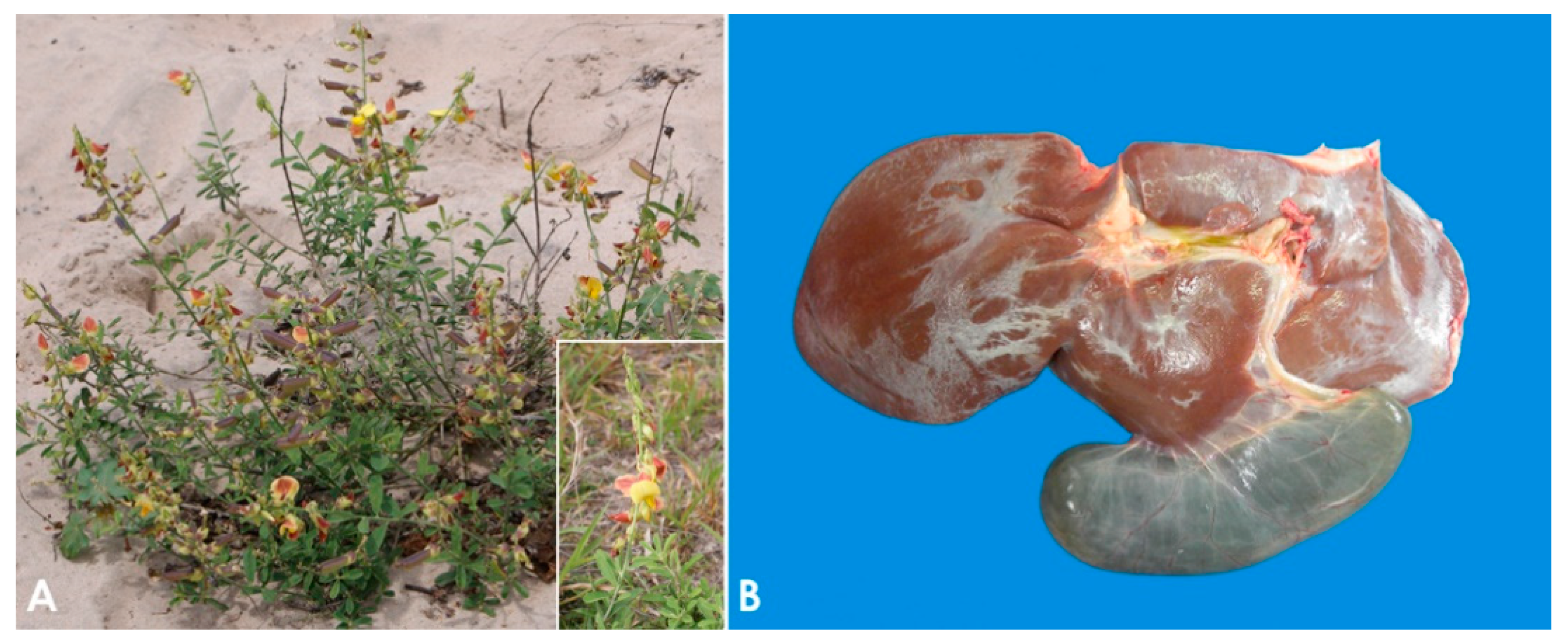
3.2. Indolizidine Alkaloids (Swainsonine-Containing Plants)
3.3. Calcinogenic Glycoside-Containing Plants
3.4. Nitrate-Containing Plants
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zajac, A.M. Gastrointestinal nematodes of small ruminants: Life cycle, anthelmintics, and diagnosis. Vet. Clin. N. Am. Food Anim. Pract. 2006, 22, 529–541. [Google Scholar] [CrossRef]
- Kalogianni, A.I.; Bossis, I.; Ekateriniadou, L.V.; Gelasakis, A.I. Etiology, Epizootiology and Control of Maedi-Visna in Dairy Sheep: A Review. Animals 2020, 10, 616. [Google Scholar] [CrossRef]
- Gascoigne, E.; Ogden, N.; Lovatt, F.; Davies, P. Update on caseous lymphadenitis in sheep. Practice 2020, 42, 105. [Google Scholar] [CrossRef]
- Windsor, P.; Whittington, R. Ovine Paratuberculosis Control in Australia Revisited. Animals 2020, 10, 1623. [Google Scholar] [CrossRef] [PubMed]
- Asín, J.; Ramírez, G.A.; Navarro, M.A.; Nyaoke, A.C.; Henderson, E.H.; Mendonça, F.S.; Molín, J.; Uzal, F.A. Nutritional wasting disorders in sheep. Animals 2021. under review, current issue. [Google Scholar]
- McCaughley, W.J. Inorganic and Organic Poisons. In Diseases of Sheep, 4th ed.; Aitken, I.D., Ed.; Blackwell Publishing: Oxford, UK, 2007; pp. 424–439. [Google Scholar]
- Panter, K.E.; Welch, K.D.; Gardner, D.R.; Lee, S.T.; Green, B.; Pfister, J.; Cook, D.; Davis, T.Z.; Stegelmeier, B. Poisonous plants of the United States. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 825–872. [Google Scholar]
- Stegelmeier, B.L.; Field, R.; Panter, K.E.; Hall, J.O.; Welch, K.D.; Pfister, J.A.; Gardner, D.R.; Lee, S.T.; Colegate, S.; Davis, T.Z.; et al. Selected Poisonous Plants Affecting Animal and Human Health. In Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed.; Haschek, W.M., Rousseaux, C.G., Wallig, M.A., Eds.; Academic Press: Waltham, MA, USA, 2013; pp. 1259–1314. [Google Scholar]
- Cullen, J.M.; Stalker, M.J. Liver and biliary system. In Jubb, Kennedy & Palmer’s Pathology of Domestic Animals: Volume 2, 6th ed.; Maxie, M.G., Ed.; W.B. Saunders: St. Louis, MO, USA, 2016; pp. 258–352. [Google Scholar]
- Thompson, L.J. Copper. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 425–427. [Google Scholar]
- Hamad Minervino, A.H.; López-Alonso, M.; Barrêto Júnior, R.A.; Lopes Rodrigues, F.A.M.; Cabral Araújo, C.A.S.; Sousa, R.S.; Satsuk Mori, C.; Miranda, M.; Costa Oliveira, F.L.; Coutinho Antonelli, A.; et al. Dietary Zinc Supplementation to Prevent Chronic Copper Poisoning in Sheep. Animals 2018, 8, 227. [Google Scholar] [CrossRef]
- Christodoulopoulos, G.; Roubies, N. Diagnosis and treatment of copper poisoning caused by accidental feeding on poultry litter in a sheep flock. Aust Vet. J. 2007, 85, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Cope, R.B. Nonprotein Nitrogen (Urea) and Hyperammonemia. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 1093–1097. [Google Scholar]
- Kromann, R.P.; Joyner, A.E.; Sharp, J.E. Influence of certain nutritional and physiological factors on urea toxicity in sheep. J. Anim. Sci. 1971, 32, 732–739. [Google Scholar] [CrossRef] [PubMed]
- McBarron, E.J.; McInnes, P. Observations on urea toxicity in sheep. Aust. Vet. J. 1968, 44, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Villar, L.; Hegarty, R.; Van Tol, M.; Godwin, I.; Nolan, J. Dietary nitrate metabolism and enteric methane mitigation in sheep consuming a protein-deficient diet. Anim. Prod. Sci. 2020, 60, 232–241. [Google Scholar] [CrossRef]
- Cockrum, R.R.; Austin, K.J.; Ludden, P.A.; Cammack, K.M. Effect of subacute dietary nitrate on production traits and plasma analytes in Suffolk ewes. Animal 2009, 4, 702–708. [Google Scholar] [CrossRef]
- Hall, J.O. Nitrate- and Nitrite-Accumulating Plants. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 941–946. [Google Scholar]
- Thompson, L.J. Fluoride. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 429–431. [Google Scholar]
- Yasar, S.; Yur, F. Antioxidant vitamin and mineral levels in sheep with fluorosis. Biol. Trace Elem. Res. 2008, 123, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Milhaud, G.E.; Borba, M.A.; Krishnaswamy, S. Effect of fluoride ingestion on dental fluorosis in sheep. Am. J. Vet. Res. 1987, 48, 873–879. [Google Scholar] [PubMed]
- Botha, C.J.; Naudé, T.W.; Minnaar, P.P.; Van Amstel, S.R.; Janse van Rensburg, S.D. Two outbreaks of fluorosis in cattle and sheep. J. S. Afr. Vet. Assoc. 1993, 64, 165–168. [Google Scholar] [PubMed]
- Kristinsson, J.; Gunnarsson, E.; Jóhannesson, Þ.; Pálsson, P.; Þormar, H. Experimental fluoride poisoning in Icelandic sheep. Icel. Agric. Sci. 1997, 11, 107–112. [Google Scholar]
- Pareja-Carrera, J.; Mateo, R.; Rodríguez-Estival, J. Lead (Pb) in sheep exposed to mining pollution: Implications for animal and human health. Ecotox. Environ. Saf. 2014, 108, 210–216. [Google Scholar] [CrossRef]
- Clegg, F.G.; Rylands, J.M. Osteoporosis and hydronephrosis of young lambs following the ingestion of lead. J. Comp. Pathol. 1966, 76, 15–22. [Google Scholar] [CrossRef]
- Craig, L.E.; Dittmer, K.E.; Thompson, K.G. Bones and Joints. In Jubb, Kennedy & Palmer’s Pathology of Domestic Animals: Volume 1, 6th ed.; Maxie, M.G., Ed.; W.B. Saunders: St. Louis, MO, USA, 2016; pp. 16–163. [Google Scholar]
- Thompson, L.J. Lead. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 439–443. [Google Scholar]
- Payne, J.; Livesey, C. Lead poisoning in cattle and sheep. Practice 2010, 32, 64. [Google Scholar] [CrossRef]
- Strugnell, B.W.; Payne, J.H.; Matthews, R. Selenium toxicosis in sheep associated with excessive sodium selenite in a commercial supplement. Vet. Rec. 2010, 167, 707–708. [Google Scholar] [CrossRef]
- McKenzie, C.M.; Al-Dissi, A.N. Accidental selenium toxicosis in lambs. Can. Vet. J. 2017, 58, 1110–1112. [Google Scholar]
- Gardiner, M.R. Chronic selenium toxicity studies in sheep. Aust. Vet. J. 1966, 42, 442–448. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, D.; Raisbeck, M.F. Pathology of experimentally induced chronic selenosis (alkali disease) in yearling cattle. J. Vet. Diagn. Investig. 1995, 7, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.O. Selenium. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 469–477. [Google Scholar]
- Glenn, M.W.; Martin, J.L.; Cummins, L.M. Sodium selenate toxicosis: The distribution of selenium within the body after prolongued feeding of toxic quantities of sodium selenate to sheep. Am. J. Vet. Res. 1964, 25, 1495–1499. [Google Scholar] [PubMed]
- Raisbeck, M.F. Selenosis. Vet. Clin. N. Am. Food Anim. Pract. 2000, 16, 465–480. [Google Scholar] [CrossRef]
- Davies, N.T.; Soliman, H.S.; Corrigall, W.; Flett, A. The susceptibility of suckling lambs to zinc toxicity. Br. J. Nutr. 1977, 38, 153–156. [Google Scholar] [CrossRef]
- Allen, J.G.; Masters, H.G.; Peet, R.L.; Mullins, K.R.; Lewis, R.D.; Skirrow, S.Z.; Fry, J. Zinc toxicity in ruminants. J. Comp. Pathol. 1983, 93, 363–377. [Google Scholar] [CrossRef]
- Garland, T. Zinc. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 489–492. [Google Scholar]
- Sattar, A.; Xie, S.; Hafeez, M.A.; Wang, X.; Hussain, H.I.; Iqbal, Z.; Pan, Y.; Iqbal, M.; Shabbir, M.A.; Yuan, Z. Metabolism and toxicity of arsenicals in mammals. Environ. Toxicol. Pharmacol. 2016, 48, 214–224. [Google Scholar] [CrossRef]
- Garland, T. Arsenic. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 411–415. [Google Scholar]
- Jubb, K.V.F.; Kennedy, P.C.; Palmer, N.; Maxie, M.G. Nervous system. In Jubb, Kennedy & Palmer’s Pathology of Domestic Animals: Volumen 1, 6th ed.; Maxie, M.G., Ed.; W.B. Saunders: St. Louis, MO, USA, 2016; pp. 250–406. [Google Scholar]
- Ashrafihelan, J.; Amoli, J.S.; Alamdari, M.; Esfahani, T.A.; Mozafari, M.; Nourian, A.R.; Bahari, A.A. Arsenic toxicosis in sheep: The first report from Iran. Interdiscip. Toxicol. 2013, 6, 93–98. [Google Scholar] [CrossRef][Green Version]
- Keshavarzi, B.; Seradj, A.; Akbari, Z.; Moore, F.; Shahraki, A.R.; Pourjafar, M. Chronic Arsenic Toxicity in Sheep of Kurdistan Province, Western Iran. Arch. Environ. Contam. Toxicol. 2015, 69, 44–53. [Google Scholar] [CrossRef]
- Maji, C.; Sarkar, S.; Biswas, S.; Patra, P.H.; Datta, B.K.; Bandyopadhyay, S.; Biswas, T.K.; Jana, C.; Mandal, T.K. Experimental assessment of arsenic toxicity in garole sheep in India. Emerg. Contam. 2016, 2, 128–134. [Google Scholar] [CrossRef]
- Robinson, M.; Trafford, J. A study of early urinary enzyme changes in mercuric chloride nephropathy in sheep. J. Comp. Pathol. 1977, 87, 275–280. [Google Scholar] [CrossRef]
- Gupta, R.C.; Milatovic, D.; Lall, R.; Srivastava, A. Mercury. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Elsevier: London, UK, 2018; pp. 455–462. [Google Scholar]
- Linklater, K.A.; Smith, M.C. Miscellaneous Toxicities. In Color Atlas of Diseases and Disorders of the Sheep and Goat; Wolfe: Aylesbury, UK, 1993; pp. 227–243. [Google Scholar]
- Nation, P.N.; Crowe, S.P.; Harries, W.N. Clinical signs and pathology of accidental monensin poisoning in sheep. Can. Vet. J. 1982, 23, 323–326. [Google Scholar] [PubMed]
- Stegelmeier, B.L.; Molyneux, R.J.; Asano, N.; Watson, A.A.; Nash, R.J. The comparative pathology of the glycosidase inhibitors swainsonine, castanospermine, and calystegines A3, B2, and C1 in mice. Toxicol. Pathol. 2008, 36, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Seaman, J.T. Hepatogenous chronic copper poisoning in sheep associated with grazing Echium plantagineum. Aust. Vet. J. 1985, 62, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Seaman, J.T. Pyrrolizidine alkaloid poisoning of sheep in New South Wales. Aust. Vet. J. 1987, 64, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Harris, D.J.; Nowara, G. The characteristics and causes of sheep losses in the Victorian Mallee. Aust. Vet. J. 1995, 72, 331–340. [Google Scholar] [CrossRef]
- Ilha, M.R.S.; Loretti, A.P.; Barros, S.S.; Barros, C.S.L. Intoxicação espontânea por Senecio brasiliensis (Asteraceae) em ovinos no Rio Grande do Sul. Pesqui. Vet. Bras. 2001, 21, 123–138. [Google Scholar] [CrossRef]
- Grecco, F.B.; Estima-Silva, P.; Marcolongo-Pereira, C.; Soares, M.P.; Collares, G.; Schild, A.L. Seneciose crônica em ovinos no sul do Rio Grande do Sul. Pesqui. Vet. Bras. 2011, 31, 326–330. [Google Scholar] [CrossRef]
- Giaretta, P.R.; Panziera, W.; Hammerschmitt, M.E.; Bianchi, R.M.; Galiza, G.J.N.; Wiethan, I.S.; Bazzi, T.; Barros, C.S.L. Clinical and pathological aspects of chronic Senecio spp poisoning in sheep. Pesqui. Vet. Bras. 2014, 34, 967–973. [Google Scholar] [CrossRef]
- Panter, K.; Colegate, S.; Davis, T.Z.; Stegelmeier, B.; Welsh, S.L.; Gardner, D.; Lee, S.; Cuneo, P.; Stonecipher, C. Fiddleneck (Amsinckia intermedia Lehmann Boraginaceae): Toxicity in Cattle Potentiated by Burrow Weed (Isocoma acradenia). Int. J. Pharm. Res. 2017, 4, 16–24. [Google Scholar]
- Laws, L. Toxicity of Crotalaria mucronata to sheep. Aust. Vet. J. 1968, 44, 453–455. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, A.A. British Poisonous Plants, 2nd ed.; Bull. 161; Ministry of Agriculture, Fishery and Food: London, UK, 1979; pp. 98–102.
- Rosiles, R.; Paasch Martínez, L. Megalocitosis hepatica en ovinos. Vet. Mex. 1982, 13, 151. [Google Scholar]
- Mortimer, P.H.; White, E.P. Toxicity of some composite (Senecio) weeds. In Proceedings of the 28th N. Z. Weed and Pest Control Conference, Hastings, New Zealand, 5–7 August 1975; Volume 28, pp. 88–91. [Google Scholar]
- Craig, A.M.; Latham, C.J.; Blythe, L.L.; Schmotzer, W.B.; O’Connor, O.A. Metabolism of toxic pyrrolizidine alkaloids from tansy ragwort (Senecio jacobaea) in ovine ruminal fluid under anaerobic conditions. Appl. Environ. Microbiol. 1992, 58, 2730–2736. [Google Scholar] [CrossRef] [PubMed]
- Araya, O.; Hernandez, J.R.; Espinoza, A.; Cubillos, V. Serum changes and histologic liver lesions due to experimental ingestion of ragwort (Senecio erraticus) in sheep. Vet. Hum. Toxicol. 1983, 25, 4–7. [Google Scholar]
- Damir, H.A.; Adam, S.E.I.; Tartour, G. The Effects of Heliotropium Ovalifolium on Goats and Sheep. Br. Vet. J. 1982, 138, 463–472. [Google Scholar] [CrossRef]
- Ketterer, P.J.; Glover, P.E.; Smith, L.W. Blue heliotrope (Heliotropium amplexicaule) poisoning in cattle. Aust. Vet. J. 1987, 64, 115–117. [Google Scholar] [CrossRef]
- Stegelmeier, B.L. Pyrrolizidine alkaloid-containing toxic plants (Senecio, Crotalaria, Cynoglossum, Amsinckia, Heliotropium, and Echium spp.). Vet. Clin. N. Am. Food Anim. Pract. 2011, 27, 419–428. [Google Scholar] [CrossRef]
- Seaman, J.T.; Dixon, R.J. Investigations into the toxicity of Echium plantagineum in sheep. 2. Pen feeding experiments. Aust. Vet. J. 1989, 66, 286–292. [Google Scholar] [CrossRef]
- Seaman, J.T.; Turvey, W.S.; Ottaway, S.J.; Dixon, R.J.; Gilmour, A.R. Investigations into the toxicity of Echium plantagineum in sheep. 1. Field grazing experiments. Aust. Vet. J. 1989, 66, 279–285. [Google Scholar] [CrossRef]
- Wiedenfeld, H.; Edgar, J. Toxicity of pyrrolizidine alkaloids to humans and ruminants. Phytochem. Rev. 2011, 10, 137–151. [Google Scholar] [CrossRef]
- de Barros, C.S.; Driemeier, D.; Pilati, C.; Barros, S.S.; Castilhos, L.M. Senecio spp poisoning in cattle in southern Brazil. Vet. Hum. Toxicol. 1992, 34, 241–246. [Google Scholar] [PubMed]
- Olson, B.; Lacey, J. Sheep: A method for controlling rangeland weeds. Sheep Res. J. Spec. Issue 1994, 105–112. [Google Scholar]
- Anjos, B.L.; Nobre, V.M.; Dantas, A.F.; Medeiros, R.M.; Oliveira Neto, T.S.; Molyneux, R.J.; Riet-Correa, F. Poisoning of sheep by seeds of Crotalaria retusa: Acquired resistance by continuous administration of low doses. Toxicon 2010, 55, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Bandarra, P.M.; Oliveira, L.G.d.; Dalto, A.C.; Boabaid, F.M.; Juffo, G.; Riet-Correa, F.; Driemeier, D.; Farias da Cruz, C.E. Sheep production as a Senecio spp. control tool. Pesqui. Vet. Bras. 2012, 32, 1017–1022. [Google Scholar] [CrossRef]
- Howell, J.; Deol, H.; Dorling, P. Experimental copper and Heliotropium europaeum intoxication in sheep: Clinical syndromes and trace element concentrations. Aust. J. Agric. Res. 1991, 42, 979–992. [Google Scholar] [CrossRef]
- Hooper, P. Pyrrolizidine alkaloid poisoning-pathology with particular reference to differences in animal and plant species. In Effects of Poisonous Plants on Livestock; Academic Press: New York, NY, USA, 1978; pp. 161–176. [Google Scholar]
- Nobre, V.; Dantas, A.; Riet-Correa, F.; Barbosa Filho, J.; Tabosa, I.; Vasconcelos, J. Acute intoxication by Crotalaria retusa in sheep. Toxicon 2005, 45, 347–352. [Google Scholar] [CrossRef]
- Cheeke, P.R. Natural Toxicants in Feeds, Forages, and Poisonous Plants; Interstate Publishers, Inc.: Danville, IL, USA, 1998. [Google Scholar]
- Bull, L.B.; Culvenor, C.C.T.; Dick, A.T. The Pyrrolizidine Alkaloids: Their Chemistry, Pathogenicity and Other Biological Properties; North Holland Publiching Co.: Amsterdam, The Netherlands, 1968. [Google Scholar]
- Smith, G.; Allred, K.; Kiehl, D. Swainsonine content of New Mexican locoweeds. Proc. West. Sect. Am. Soc. Anim. Sci. 1992, 43, 405–407. [Google Scholar]
- Burrows, G.E.; Tyrl, R.J. Toxic Plants of North America, 2nd ed.; John Wiley & Sons: Ames, IA, USA, 2013. [Google Scholar]
- Cook, D.; Gardner, D.R.; Pfister, J.A. Swainsonine-containing plants and their relationship to endophytic fungi. J. Agric. Food Chem. 2014, 62, 7326–7334. [Google Scholar] [CrossRef]
- Mendonça, F.S.; Silva Filho, G.B.; Chaves, H.A.; Aires, L.D.; Braga, T.C.; Gardner, D.R.; Cook, D.; Buril, M.T. Detection of swainsonine and calystegines in Convolvulaceae species from the semiarid region of Pernambuco. Pesqui. Vet. Bras. 2018, 38, 2044–2051. [Google Scholar] [CrossRef]
- Tulsiani, D.; Broquist, H.; James, L.; Touster, O. The similar effects of swainsonine and locoweed on tissue glycosidases and oligosaccharides of the pig indicate that the alkaloid is the principal toxin responsible for the induction of locoism. Arch. Biochem. Biophys. 1984, 232, 76–85. [Google Scholar] [CrossRef]
- Dorling, P.R.; Huxtable, C.R.; Colegate, S. Inhibition of lysosomal α-mannosidase by swainsonine, an indolizidine alkaloid isolated from Swainsona canescens. Biochem. J. 1980, 191, 649–651. [Google Scholar] [CrossRef] [PubMed]
- Molyneux, R.J.; McKenzie, R.A.; O’Sullivan, B.M.; Elbein, A.D. Identification of the glycosidase inhibitors swainsonine and calystegine B2 in weir vine (Ipomoea sp. Q6 [aff. calobra]) and correlation with toxicity. J. Nat. Prod. 1995, 58, 878–886. [Google Scholar] [CrossRef]
- Pan, Y.; Ghidoni, J.; Elbein, A.D. The effects of castanospermine and swainsonine on the activity and synthesis of intestinal sucrase. Arch. Biochem. Biophys. 1993, 303, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Van Kampen, K.R.; James, L.F. Pathology of Locoweed (Astragalus lentiginosus) Poisoning in Sheep; Sequential Development of Cyloplasmie Vacuolation in Tissues. Pathol. Vet. 1970, 7, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Stegelmeier, B.; James, L.; Panter, K.; Molyneux, R. Serum swainsonine concentration and alpha-mannosidase activity in cattle and sheep ingesting Oxytropis sericea and Astragalus lentiginosus (locoweeds). Am. J. Vet. Res. 1995, 56, 149–154. [Google Scholar] [PubMed]
- James, L.F. Effect of Astragalus tenellus on sheep. Rangel. Ecol. Manag. 1971, 24, 161. [Google Scholar] [CrossRef]
- Stegelmeier, B.; Molyneux, R.; Elbein, A.; James, L. The lesions of locoweed (Astragalus mollissimus), swainsonine, and castanospermine in rats. Vet. Pathol. 1995, 32, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Stegelmeier, B.L.; James, L.F.; Panter, K.E.; Gardner, D.R.; Ralphs, M.H.; Pfister, J.A. Tissue swainsonine clearance in sheep chronically poisoned with locoweed (Oxytropis sericea). J. Anim. Sci. 1998, 76, 1140–1144. [Google Scholar] [CrossRef]
- Driemeier, D.; Colodel, E.; Gimeno, E.J.; Barros, S. Lysosomal storage disease caused by Sida carpinifolia poisoning in goats. Vet. Pathol. 2000, 37, 153–159. [Google Scholar] [CrossRef]
- Mendonça, F.S.; Albuquerque, R.F.; Evêncio-Neto, J.; Freitas, S.H.; Dória, R.G.; Boabaid, F.M.; Driemeier, D.; Gardner, D.R.; Riet-Correa, F.; Colodel, E.M. Alpha-mannosidosis in goats caused by the swainsonine-containing plant Ipomoea verbascoidea. J. Vet. Diagn. Investig. 2012, 24, 90–95. [Google Scholar] [CrossRef]
- Cook, D.; Ralphs, M.; Welch, K.; Stegelmeier, B. Locoweed poisoning in livestock. Rangelands 2009, 31, 16–21. [Google Scholar] [CrossRef]
- Stegelmeier, B.L.; Ralphs, M.H.; Gardner, D.R.; Molyneux, R.J.; Jame, L.F. Serum α-mannosidase activity and the clinicopathologic alterations of locoweed (Astragalus mollissimus) intoxication in range cattle. J. Vet. Diagn. Investig. 1994, 6, 473–479. [Google Scholar] [CrossRef]
- Rocha, B.P.; Reis, M.O.; Driemeier, D.; Cook, D.; Camargo, L.M.; Riet-Correa, F.; Evêncio-Neto, J.; Mendonça, F.S. Liver biopsy as diagnostic method for poisoning by swainsonina-containing plants. Pesqui. Vet. Bras. 2016, 36, 373–377. [Google Scholar] [CrossRef]
- Braun, U.; Mitchell, K.J.; Schramm, S.; Nogler, S.; Hatt, J.-M.; Malbon, A. Ultrasonographic findings in two sheep with enzootic calcinosis. Schweiz. Arch. Tierheilkd. 2016, 158, 827–832. [Google Scholar] [CrossRef]
- Robinson, W.F.; Robinson, N.A. Cardiovascular System. In Jubb, Kennedy & Palmer’s Pathology of Domestic Animals: Volume 3, 6th ed.; Maxie, M.G., Ed.; W.B. Saunders: St. Louis, MO, USA, 2016; pp. 1–101. [Google Scholar]
- Mello, J.R. Calcinosis—Calcinogenic plants. Toxicon 2003, 41, 1–12. [Google Scholar] [CrossRef]
- O’sullivan, B. Humpy back of sheep. Clinical and pathological observations. Aust. Vet. J. 1976, 52, 414. [Google Scholar] [CrossRef] [PubMed]
- Tustin, R.; Pienaar, C.; Schmidt, J.; Faul, A.; Van der Walt, K.; Boyazoglu, P.; De Boom, H. Enzootic calcinosis of sheep in South Africa. J. S. Afr. Vet. Assoc. 1973, 44, 383–395. [Google Scholar]
- Rietcorrea, F.; Schild, A.; Méndez, M.; Wasserman, R.; Krook, L. Enzootic calcinosis in sheep caused by the ingestion of Nierembergia veitchii (Solanaceae). Pesqui. Vet. Bras. 1987, 7, 85–95. [Google Scholar]
- Rissi, D.R.; Rech, R.R.; Pierezan, F.; Kommers, G.D.; de Barros, C.S.L. Poisoning in sheep by Nierembergia veitchii: Observations in four outbreaks. Cienc. Rural 2007, 37, 1393–1398. [Google Scholar] [CrossRef]
- García y Santos, C.; Pereira, R.; Etcheberry, G.; Goyen, J.M.; Pérez, W.; Capelli, A.; Alonso, E.; Ruiz-Díaz, A.; Riet-Correa, F. Enzootic calcinosis caused by Nierembergia rivularis in sheep. J. Vet. Diagn. Investig. 2012, 24, 423–426. [Google Scholar] [CrossRef] [PubMed]
- Guedes, K.M.; Colodel, E.M.; Castro, M.B.; Souza, M.A.; Arruda, L.P.; Dianezi, D.M.; Borges, J.R.J.; Riet-Correa, F. Calcinose enzoótica em ruminantes no Brasil Central. Pesqui. Vet. Bras. 2011, 31, 643–648. [Google Scholar] [CrossRef]
- Gorniak, S.; Dagli, M.; Maiorka, P.; Raspantini, P.; Jericó, M. Evaluation in rabbits of the fetal effects of maternal ingestion of Solanum malacoxylon. Vet. Res. Commun. 1999, 23, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Angus, K.W. Plant Poisoing in Britain and Ireland. In Diseases of Sheep, 4th ed.; Aitken, I.D., Ed.; Blackwell Publishing: Oxford, UK, 2007; pp. 405–423. [Google Scholar]
- Arnold, M.; Gaskill, C.; Lehmkuhler, J.; Smith, S. Nitrate Poisoning. University of Kentuchy. Agric. Naut. Resour. Publ. 2014, 165. Available online: https://uknowledge.uky.edu/cgi/viewcontent.cgi?article=1165&context=anr_reports (accessed on 29 November 2020).
- Parton, K.; Bruere, A. Plant poisoning of livestock in New Zealand. N. Z. Vet. J. 2002, 50, 22–27. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, R.; Rayner, A.; Thompson, G.; Pidgeon, G.; Burren, B. Nitrate-nitrite toxicity in cattle and sheep grazing Dactyloctenium radulans (button grass) in stockyards. Aust. Vet. J. 2004, 82, 630–634. [Google Scholar] [CrossRef]
- Simões, J.G.; Medeiros, R.M.T.; Medeiros, M.A.; Olinda, R.G.; Dantas, A.F.M.; Riet-Correa, F. Nitrate and nitrite poisoning in sheep and goats caused by ingestion of Portulaca oleracea. Pesqui. Vet. Bras. 2018, 38, 1549–1553. [Google Scholar] [CrossRef]
- Burrows, G. Nitrate intoxication. J. Am. Vet. Med. Assoc. 1980, 177, 82–83. [Google Scholar]

| Toxicant | Treatment | Dose and Mode of Administration |
|---|---|---|
| Copper | Ammonium tetrathiomolybdate | 1.68 mg/kg BW IV (3 times in 5 d) 3.4 mg/kg BW SC (3 alternate d) |
| Nitrates/nitrites | Methylene blue 4% | 10 mg/kg BW, IV infusion |
| Fluoride/Fluorine | Monoacetin (acute cases) | 0.55 mL/kg BW IM every 1/2 h, several hours |
| Ca and Al compounds added to the ration | ||
| Lead | CaEDTA | 75 mg/kg IV, slow infusion, daily during the first 48 h |
| Selenium | No specific chelation therapy available | - |
| Zinc | CaEDTA | 100 mg/kg IV or SQ, into four doses/day for 3 d |
| Arsenic | 2,3-dimercaptopropanol | 23 mg/kg every 4 h for 2 d, repeat every 6 h for 2 d, repeat every 12 h for 2 d |
| Sodium thiosulfate | 30–40 mg/kg 20% solution IV, repeated every 12 h until clinical improvement |
| Toxicant | Gross Lesions * | Histological Lesions | Ancillary Tests |
|---|---|---|---|
| Pyrrolizidin alkaloid | Atrophic and firm liver with/without nodular regeneration | Fibrosis Bile duct hyperplasia Megalocytosis Nuclear invaginations Hepatocyte atrophy | - |
| Indolizidine alkaloid (swainsonine) | - | Cytoplasmic vacuolization in epithelial and neural cells | Lectin histochemistry in liver Increased serum swansonine Decreased serum alpha-mannosidase activity |
| Calcinogenic glycoside | Gritty mineral deposits in tissues | Basophilic mineral deposits | Elevated serum 1,25-dihydroxycholecalciferol |
| Nitrates/nitrites | Brownish discoloration of carcass and organs Brown and dense blood | - | Nitrates/nitrites concentration in ocular fluid |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molín, J.; Mendonça, F.S.; Henderson, E.E.; Nyaoke, A.C.; Ramírez, G.A.; Navarro, M.A.; Uzal, F.A.; Asín, J. Toxic Wasting Disorders in Sheep. Animals 2021, 11, 229. https://doi.org/10.3390/ani11010229
Molín J, Mendonça FS, Henderson EE, Nyaoke AC, Ramírez GA, Navarro MA, Uzal FA, Asín J. Toxic Wasting Disorders in Sheep. Animals. 2021; 11(1):229. https://doi.org/10.3390/ani11010229
Chicago/Turabian StyleMolín, Jéssica, Fábio S. Mendonça, Eileen E. Henderson, Akinyi C. Nyaoke, Gustavo A. Ramírez, Mauricio A. Navarro, Francisco A. Uzal, and Javier Asín. 2021. "Toxic Wasting Disorders in Sheep" Animals 11, no. 1: 229. https://doi.org/10.3390/ani11010229
APA StyleMolín, J., Mendonça, F. S., Henderson, E. E., Nyaoke, A. C., Ramírez, G. A., Navarro, M. A., Uzal, F. A., & Asín, J. (2021). Toxic Wasting Disorders in Sheep. Animals, 11(1), 229. https://doi.org/10.3390/ani11010229







