Assessing Environmental Control Strategies in Cage-Free Egg Production Systems: Effect on Spatial Occupancy and Natural Behaviors
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
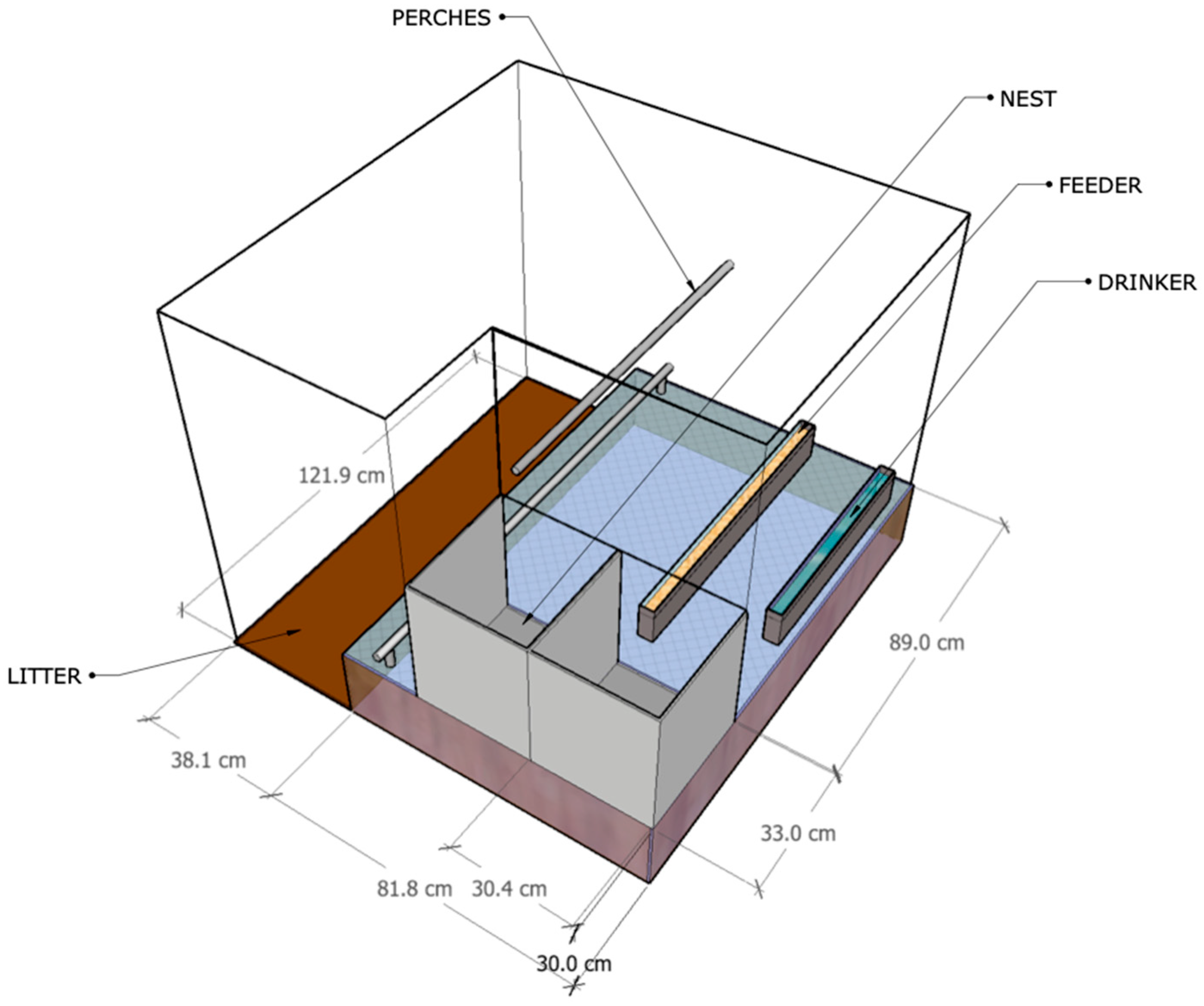
2.1. Experimental Bench-Scale Rooms
2.2. Animal and Housing
2.3. Environmental Control Strategies (ECS)
2.4. Spatial Occupancy and Laying Hen Ethology Analysis
2.5. Video Recordings and Observation Analysis
2.6. Statistical Analysis
3. Results
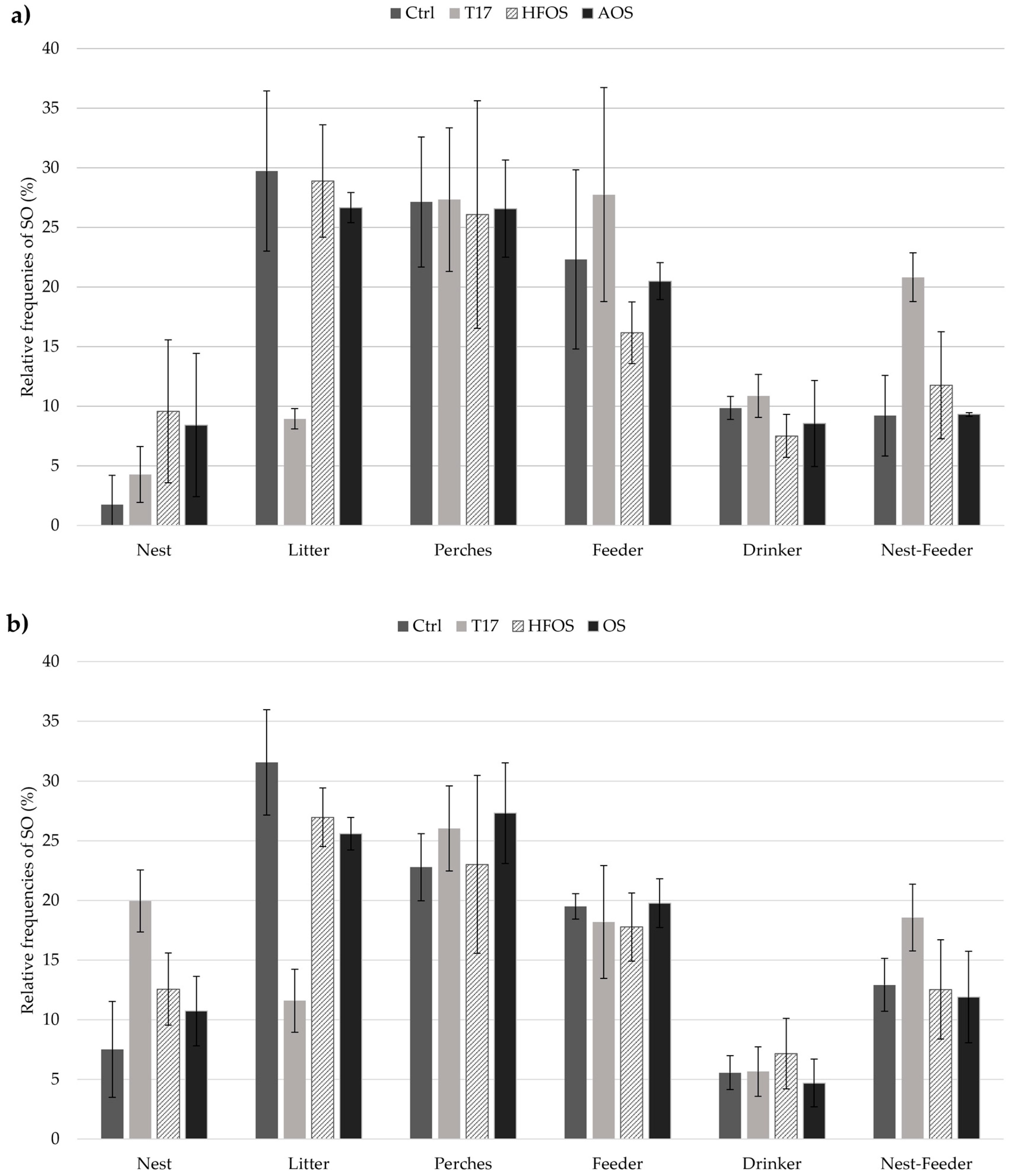
3.1. Spatial Occupancy (SO)
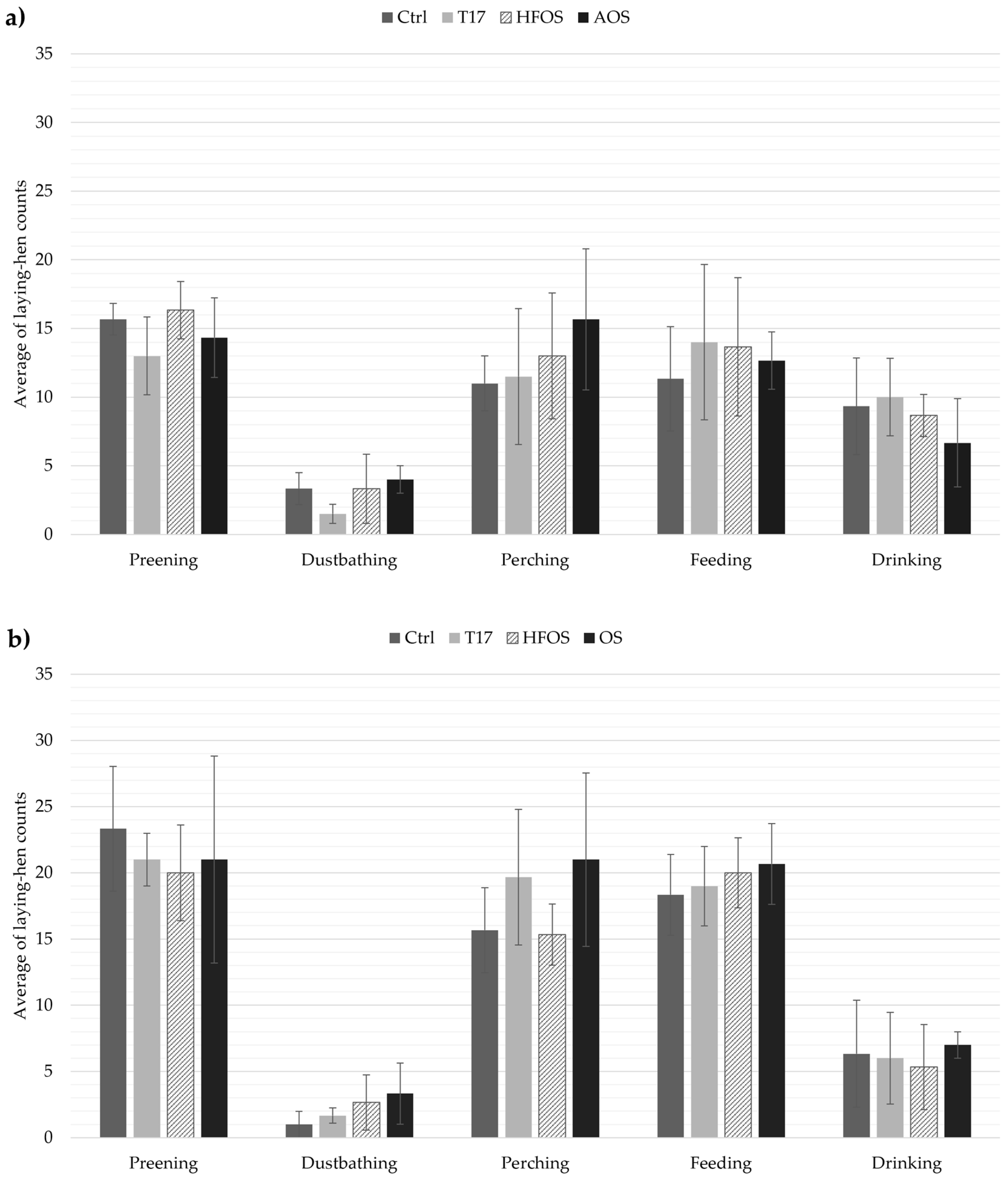
3.2. Laying Hen Behavior (LHB)
4. Discussion
4.1. Space Occupancy (SO) Preference
4.2. Patterns in Animal Behaviors
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Commission. Council Directive 99/74/EC of 19 July 1999 laying down minimum standards for the protection of laying hens. Off. J. Eur. Communities 1999, 203, 53–57. [Google Scholar]
- Appleby, M.C. The European Union ban on conventional cages for laying hens: History and prospects. J. Appl. Anim. Welf. Sci. 2003, 6, 103–121. [Google Scholar] [CrossRef] [PubMed]
- NFACC. Code of Practice for the Care and Handling of Pullets and Laying Hens. Available online: https://www.nfacc.ca/pdfs/pullets_and_laying_hens_code_of_practice.pdf (accessed on 29 November 2019).
- Shields, S.; Shapiro, P.; Rowan, A. A decade of progress toward ending the intensive confinement of farm animals in the United States. Animals 2017, 7, 40. [Google Scholar] [CrossRef] [PubMed]
- Hartcher, K.; Jones, B. The welfare of layer hens in cage and cage-free housing systems. World’s Poult. Sci. J. 2017, 73, 767–782. [Google Scholar] [CrossRef]
- Colson, S. Bien-être de Poules Pondeuses Logées en Volière de Ponte: Comparaison à des Poules Logées en Cage Conventionnelle et Influence des Conditions D’élevage des Poulettes sur leur Adaptation à la Volière de Ponte; Université de Rennes: Rennes, France, 2006. [Google Scholar]
- Oliveira, J.L.; Xin, H.; Chai, L.; Millman, S.T. Effects of litter floor access and inclusion of experienced hens in aviary housing on floor eggs, litter condition, air quality, and hen welfare. Poult. Sci. 2018, 98, 1664–1677. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, F.; Godbout, S. Étude De L’efficacité Environnementale de Différents Systèmes de Production Dans Les Élevages de Poules Pondeuses; Institut de Recherche et de Développement en Agroenvironnement (IRDA): Québec, QC, Canada, 2016; p. 38. [Google Scholar]
- Ritz, C.; Fairchild, B.; Lacy, M. Implications of ammonia production and emissions from commercial poultry facilities: A review. J. Appl. Poult. Res. 2004, 13, 684–692. [Google Scholar] [CrossRef]
- Beker, A.; Vanhooser, S.; Swartzlander, J.; Teeter, R. Atmospheric ammonia concentration effects on broiler growth and performance. J. Appl. Poult. Res. 2004, 13, 5–9. [Google Scholar] [CrossRef]
- Oloyo, A. The use of housing system in the management of heat stress in poultry production in hot and humid climate: A review. Poult. Sci. J. 2018, 6, 1–9. [Google Scholar]
- Olanrewaju, H.; Miller, W.; Maslin, W.; Thaxton, J.; Dozier, W.; Purswell, J.; Branton, S. Interactive effects of ammonia and light intensity on ocular, fear and leg health in broiler chickens. Int. J. Poult. Sci. 2007, 6, 762–769. [Google Scholar] [CrossRef]
- David, B.; Mejdell, C.; Michel, V.; Lund, V.; Moe, R.O. Air quality in alternative housing systems may have an impact on laying hen welfare. Part II—Ammonia. Animals 2015, 5, 886–896. [Google Scholar] [CrossRef]
- Kristensen, H.H.; Burgess, L.R.; Demmers, T.G.; Wathes, C.M. The preferences of laying hens for different concentrations of atmospheric ammonia. Appl. Anim. Behav. Sci. 2000, 68, 307–318. [Google Scholar] [CrossRef]
- Costa, A.; Guarino, M. Particulate matter concentration and emission factor in three different laying hen housing systems. J. Agric. Eng. 2009, 40, 15–24. [Google Scholar] [CrossRef]
- Yang, W.; Guo, M.; Liu, G.; Yu, G.; Wang, P.; Wang, H.; Chai, T. Detection and analysis of fine particulate matter and microbial aerosol in chicken houses in Shandong Province, China. Poult. Sci. 2017, 97, 995–1005. [Google Scholar] [CrossRef] [PubMed]
- Farokhi, A.; Heederik, D.; Smit, L.A. Respiratory health effects of exposure to low levels of airborne endotoxin—A systematic review. Environ. Health 2018, 17, 14. [Google Scholar] [CrossRef]
- WHO. Hazard Prevention and Control in the Work Environment: Airborne Dust; World Health Organization: Geneva, Switzerland, 1999. [Google Scholar]
- Löndahl, J.; Massling, A.; Pagels, J.; Swietlicki, E.; Vaclavik, E.; Loft, S. Size-Resolved Respiratory-Tract Deposition of Fine and Ultrafine Hydrophobic and Hygroscopic Aerosol Particles During Rest and Exercise. Inhal. Toxicol. 2007, 19, 109–116. [Google Scholar] [CrossRef]
- Michel, V.; Huonnic, D. A comparison of welfare, health and production performance of laying hens reared in cages or in aviaries. Br. Poult. Sci. 2003, 44, 775–776. [Google Scholar] [CrossRef]
- David, B.; Moe, R.O.; Michel, V.; Lund, V.; Mejdell, C. Air quality in alternative housing systems may have an impact on laying hen welfare. Part I—Dust. Animals 2015, 5, 495–511. [Google Scholar] [CrossRef]
- Lin, X.; Zhang, R.; Jiang, S.; El-Mashad, H.; Xin, H. Emissions of ammonia, carbon dioxide and particulate matter from cage-free layer houses in California. Atmos. Environ. 2017, 152, 246–255. [Google Scholar] [CrossRef]
- Godbout, S.; Pelletier, F.; Fournel, S. Mesure et Évaluation des Émissions de Gaz et D’odeurs Issues de Différentes Stratégies de Gestion des Déjections en Production D’oeufs de Consommation; IRDA: Québec, QC, Canada, 2011; p. 88. [Google Scholar]
- Romero, C.; Onyango, E.M.; Powers, W.; Angel, R.; Applegate, T.J. Effect of a partial replacement of limestone by a CaSO4-zeolite mixture combined with a slight protein reduction on production indices, egg quality, and excreta pH in laying hens. J. Appl. Poult. Res. 2012, 21, 325–334. [Google Scholar] [CrossRef]
- Prasai, T.P.; Walsh, K.B.; Midmore, D.J.; Jones, B.E.H.; Bhattarai, S.P. Manure from biochar, bentonite and zeolite feed supplemented poultry: Moisture retention and granulation properties. J. Environ. Manag. 2018, 216, 82–88. [Google Scholar] [CrossRef]
- Schneider, A.F.; Almeida, D.S.D.; Yuri, F.M.; Zimmermann, O.F.; Gerber, M.W.; Gewehr, C.E. Natural zeolites in diet or litter of broilers. Br. Poult. Sci. 2016, 57, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Weiss, A.; Lin, C.; Li, H.; Joerger, R.; Chiu, P. Effects of Multiple Litter Amendment Applications in Commercial Broiler Houses on Ammonia Emissions and Litter Microflora. Trans. ASABE 2016, 59, 1393–1401. [Google Scholar] [CrossRef]
- Qasim, W.; Moon, B.E.; Phonsuwan, M.; Jo, J.S.; Lee, M.H.; Nafees, M.; Kim, H.T. Effects of an aluminum sulfate and ferric chloride blend on poultry litter characteristics in vitro. J. Appl. Poult. Res. 2017, 27, 92–102. [Google Scholar] [CrossRef]
- Van Harn, J.; Aarnink, A.J.A.; Mosquera, J.; Van Riel, J.W.; Ogink, N.W.M. Effect of Bedding Material on Dust and Ammonia Emission from Broiler Houses. Trans. ASABE 2012, 55, 219–226. [Google Scholar] [CrossRef]
- Chai, L.; Xin, H.; Zhao, Y.; Wang, T.; Soupir, M.; Liu, K. Mitigating Ammonia and PM Generation of Cage-Free Henhouse Litter with Solid Additive and Liquid Spray. Trans. ASABE 2018, 61, 287–294. [Google Scholar] [CrossRef]
- Griffin, J.; Vardaman, T. Cottonseed oil spray for broiler houses: Effects on dust control, Mycoplasma gallisepticum spread and broiler performances. Poult. Sci. 1970, 49, 1664–1668. [Google Scholar] [CrossRef]
- Aarnink, A.; van Harn, J.; Van Hattum, T.; Zhao, Y.; Ogink, N. Dust reduction in broiler houses by spraying rapeseed oil. Trans. ASABE 2011, 54, 1479–1489. [Google Scholar] [CrossRef]
- Winkel, A.; Mosquera, J.; Aarnink, A.J.A.; Groot Koerkamp, P.W.G.; Ogink, N.W.M. Evaluation of oil spraying systems and air ionisation systems for abatement of particulate matter emission in commercial poultry houses. Biosyst. Eng. 2016, 150, 104–122. [Google Scholar] [CrossRef]
- Fan, Q.; Sun, J.; Chu, L.; Cui, L.; Quan, G.; Yan, J.; Hussain, Q.; Iqbal, M. Effects of chemical oxidation on surface oxygen-containing functional groups and adsorption behavior of biochar. Chemosphere 2018, 207, 33–40. [Google Scholar] [CrossRef]
- Blokhuis, H. Rest in poultry. Appl. Anim. Behav. Sci. 1984, 12, 289–303. [Google Scholar] [CrossRef]
- Weeks, C.; Nicol, C. Behavioural needs, priorities and preferences of laying hens. World’s Poult. Sci. J. 2006, 62, 296–307. [Google Scholar] [CrossRef]
- Bracke, M.B.M.; Hopster, H. Assessing the Importance of Natural Behavior for Animal Welfare. J. Agric. Environ. Ethics 2006, 19, 77–89. [Google Scholar] [CrossRef]
- Shields, S.; Duncan, I.J. A Comparison of the Welfare of Hens in Battery Cages and Alternative Systems; The Humane Society of the United States: Washington, DC, USA, 2009. [Google Scholar]
- Webster, A.; Hurnik, J. An ethogram of White Leghorn-type hens in battery cages. Can. J. Anim. Sci. 1990, 70, 751–760. [Google Scholar] [CrossRef]
- Kozak, M.; Tobalske, B.; Martins, C.; Bowley, S.; Wuerbel, H.; Harlander-Matauschek, A. Use of space by domestic chicks housed in complex aviaries. Appl. Anim. Behav. Sci. 2016, 181, 115–121. [Google Scholar] [CrossRef]
- Garner, J.P. Stereotypies and other abnormal repetitive behaviors: Potential impact on validity, reliability, and replicability of scientific outcomes. Ilar J. 2005, 46, 106–117. [Google Scholar] [CrossRef]
- Taylor, K.D. Displacement Behavior. In The Encyclopedia of Applied Animal Behavior and Welfare; CAB International: Wallingford, UK, 2010; p. 180. [Google Scholar]
- Cordiner, L.; Savory, C. Use of perches and nestboxes by laying hens in relation to social status, based on examination of consistency of ranking orders and frequency of interaction. Appl. Anim. Behav. Sci. 2001, 71, 305–317. [Google Scholar] [CrossRef]
- Engel, J.; Widowski, T.; Tilbrook, A.; Butler, K.; Hemsworth, P. The effects of floor space and nest box access on the physiology and behavior of caged laying hens. Poult. Sci. 2018, 98, 533–547. [Google Scholar] [CrossRef]
- Albentosa, M.; Cooper, J.; Luddem, T.; Redgate, S.; Elson, H.; Walker, A. Evaluation of the effects of cage height and stocking density on the behaviour of laying hens in furnished cages. Br. Poult. Sci. 2007, 48, 1–11. [Google Scholar] [CrossRef]
- Fournel, S.; Rousseau, A.N.; Laberge, B. Rethinking environment control strategy of confined animal housing systems through precision livestock farming. Biosyst. Eng. 2017, 155, 96–123. [Google Scholar] [CrossRef]

| Batch | Abb. 2 | Treatment | Description 1 |
|---|---|---|---|
| 1–2 | T17 | Decrease of litter surface area | 17% of litter area, reduction of litter surface from 33% to 17%. n = 3 (rooms 1, 5 and 11). |
| 1–2 | HFOS | Heated floor + oil sprinkling | 33% of litter area, installation of a heated floor fixed to 27ºC. Spraying an oily emulsion over litter (1.17 L/m2/week). n = 3 (rooms 2, 6 and 10). |
| 1 | AOS | Litter adsorbent + oil sprinkling | 33% of litter area, addition of 10%-litter of acid adsorbent (Active biochar). Spraying an oily emulsion over litter (1.17 L/m2/week). n = 3 (rooms 3, 7 and 9). |
| 2 | OS | Oil sprinkling | 33% of litter area. Spraying an oily emulsion over litter (1.17 L/m2/week). n = 2 (rooms 7 and 9). |
| Behaviors | Description |
|---|---|
| Scratching | Bird scratching itself or scratching the litter with its feet. [37] |
| Kneeling | Includes events when the hen lies down over the litter, wire floor, or even on perches in a relaxing position. |
| Ruffling Feathers | The hen ruffles the feather without shaking its body. This activity could be observed in a standing or sitting position. |
| Body Shaking | Similar to RF but with an instant shaking movement. |
| Preening | The beak is in contact with the feathers. [35] |
| Dustbathing | Sitting position and arbitrary movements where the body, the feathers, the legs, and the beak could be in contact with the litter. [38] |
| Perching | A hen is stand up or sitting on a perch more than 2 s. |
| Feeding | Head in the feeder trough ingesting food. [39] |
| Drinking | Beak within the plane of the drinker. [39] |
| Supplementary behaviors | |
| Other | Other behaviors such as fluttering (wing flapping), sleeping (head through the feathers above the wing base), pecking or foraging, and stretching feathers. |
| Non-reported behavior (nBr) | A hen does not show any of the other behaviors. Stationary or motionless at the time of observation. |
| Batch 1 | Batch 2 | |||
|---|---|---|---|---|
| F-Value | Pr < F | F-Value | Pr < F | |
| Scratching | 0.36 | 0.78 | 1.75 | 0.23 |
| Kneeling | 0.06 | 0.98 | 1.33 | 0.33 |
| Ruffling Feathers | 1.65 | 0.26 | 0.63 | 0.62 |
| Body Shaking | 0.94 | 0.47 | 0.20 | 0.89 |
| Preening 1 | 0.35 | 0.79 | 0.28 | 0.84 |
| Dustbathing 1 | 0.77 | 0.55 | 1.38 | 0.32 |
| Perching 1 | 0.97 | 0.46 | 1.35 | 0.33 |
| Feeding 1 | 0.30 | 0.82 | 0.17 | 0.92 |
| Drinking 1 | 0.65 | 0.61 | 0.23 | 0.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzalez-Mora, A.F.; Larios, A.D.; Rousseau, A.N.; Godbout, S.; Morin, C.; Palacios, J.H.; Grenier, M.; Fournel, S. Assessing Environmental Control Strategies in Cage-Free Egg Production Systems: Effect on Spatial Occupancy and Natural Behaviors. Animals 2021, 11, 17. https://doi.org/10.3390/ani11010017
Gonzalez-Mora AF, Larios AD, Rousseau AN, Godbout S, Morin C, Palacios JH, Grenier M, Fournel S. Assessing Environmental Control Strategies in Cage-Free Egg Production Systems: Effect on Spatial Occupancy and Natural Behaviors. Animals. 2021; 11(1):17. https://doi.org/10.3390/ani11010017
Chicago/Turabian StyleGonzalez-Mora, Andrés F., Araceli D. Larios, Alain N. Rousseau, Stéphane Godbout, Cédric Morin, Joahnn H. Palacios, Michèle Grenier, and Sébastien Fournel. 2021. "Assessing Environmental Control Strategies in Cage-Free Egg Production Systems: Effect on Spatial Occupancy and Natural Behaviors" Animals 11, no. 1: 17. https://doi.org/10.3390/ani11010017
APA StyleGonzalez-Mora, A. F., Larios, A. D., Rousseau, A. N., Godbout, S., Morin, C., Palacios, J. H., Grenier, M., & Fournel, S. (2021). Assessing Environmental Control Strategies in Cage-Free Egg Production Systems: Effect on Spatial Occupancy and Natural Behaviors. Animals, 11(1), 17. https://doi.org/10.3390/ani11010017









