Prevalence of Keel Bone Damage in Red Jungle Fowls (Gallus gallus)—A Pilot Study
Simple Summary
Abstract
1. Introduction
2. Material and Method
2.1. Birds and Management
2.2. Production Data
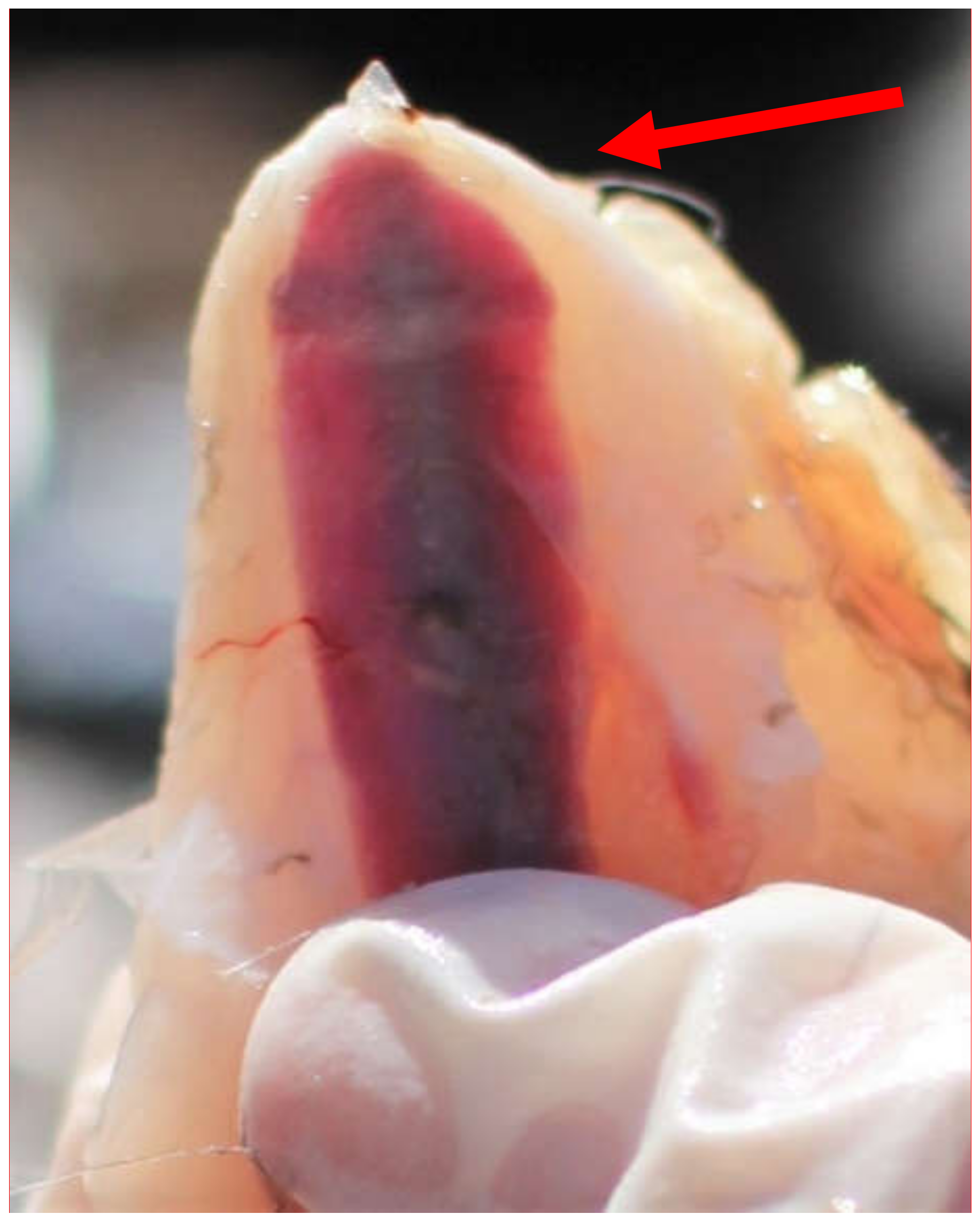
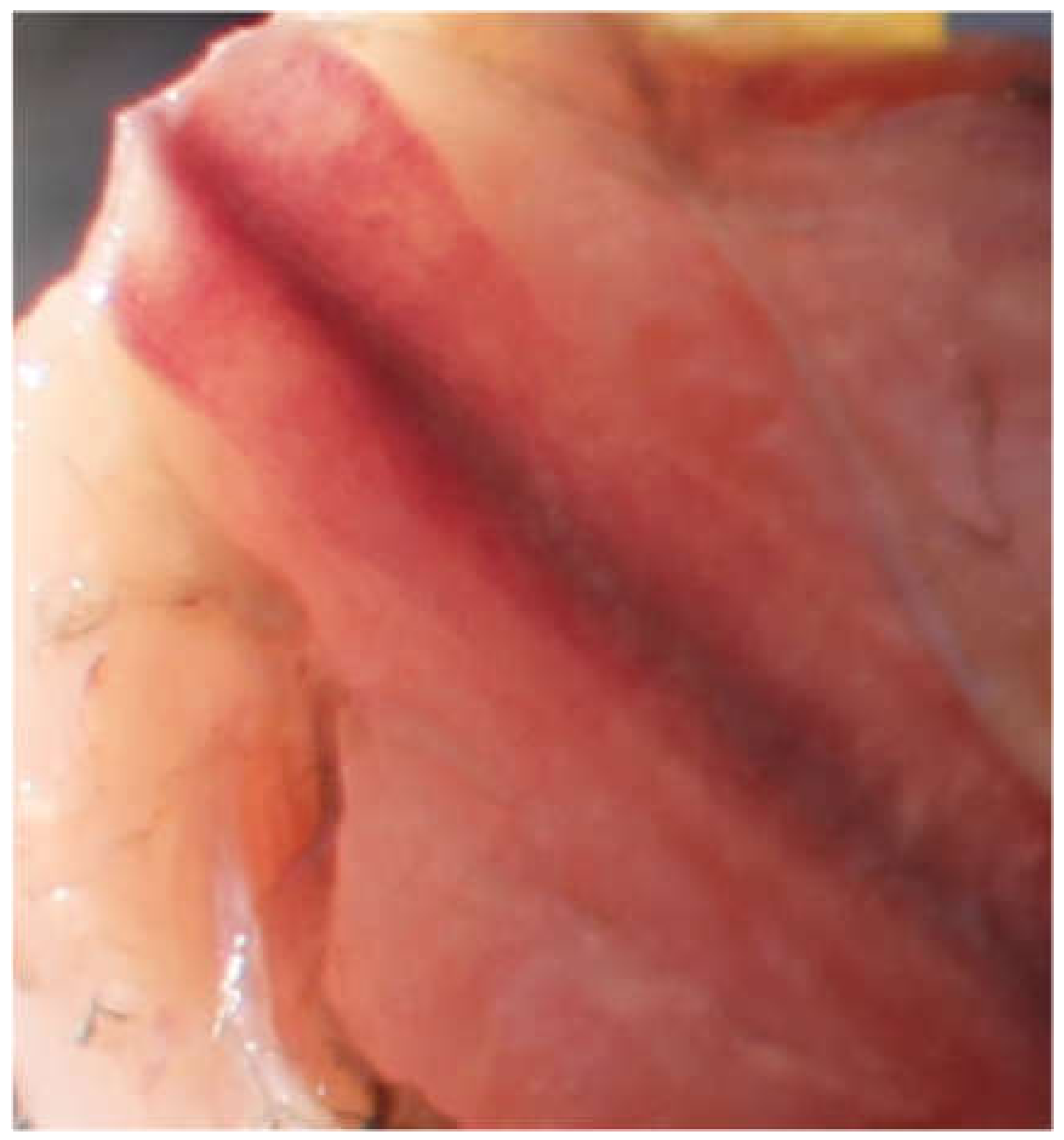
2.3. Keel Bone Examination
2.4. Statistics
3. Results
3.1. Keel Bone Deviations
3.2. Keel Bone Fractures
3.3. Palpation of Fractures versus Autopsy
4. Discussions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Casey-Trott, T.; Heerkens, J.; Petrik, M.; Regmi, P.; Schrader, L.; Toscano, M.J.; Widowski, T. Methods for assessment of keel bone damage in poultry. Poult. Sci. 2015, 94, 2339–2350. [Google Scholar] [CrossRef] [PubMed]
- Heerkens, J.; Delezie, E.; Rodenburg, T.B.; Kempen, I.; Zoons, J.; Ampe, B.; Tuyttens, F. Risk factors associated with keel bone and foot pad disorders in laying hens housed in aviary systems. Poult. Sci. 2016, 95, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Rodenburg, T.; Tuyttens, F.; De Reu, K.; Herman, L.; Zoons, J.; Sonck, B. Welfare assessment of laying hens in furnished cages and non-cage systems: An on-farm comparison. Anim. Welf. 2008, 17, 363–373. [Google Scholar]
- Gebhardt-Henrich, S.G.; Fröhlich, E.K. Early onset of laying and bumblefoot favor keel bone fractures. Animals 2015, 5, 1192–1206. [Google Scholar] [CrossRef]
- Blokhuis, H.; Van Niekerk, T.F.; Bessei, W.; Elson, A.; Guémené, D.; Kjaer, J.; Levrino, G.M.; Nicol, C.; Tauson, R.; Weeks, C. The laywel project: Welfare implications of changes in production systems for laying hens. World Poult. Sci. J. 2007, 63, 101–114. [Google Scholar] [CrossRef]
- Harlander-Matauschek, A.; Rodenburg, T.; Sandilands, V.; Tobalske, B.; Toscano, M.J. Causes of keel bone damage and their solutions in laying hens. World Poult. Sci. J. 2015, 71, 461–472. [Google Scholar] [CrossRef]
- Bestman, M.; Wagenaar, J.-P. Health and welfare in dutch organic laying hens. Animals 2014, 4, 374–390. [Google Scholar] [CrossRef]
- Nasr, M.A.; Nicol, C.J.; Murrell, J.C. Do laying hens with keel bone fractures experience pain? PLoS ONE 2012, 7, e0042420. [Google Scholar] [CrossRef]
- Nasr, M.; Browne, W.; Caplen, G.; Hothersall, B.; Murrell, J.; Nicol, C. Positive affective state induced by opioid analgesia in laying hens with bone fractures. Appl. Anim. Behav. Sci. 2013, 147, 127–131. [Google Scholar] [CrossRef]
- Nasr, M.; Murrell, J.; Wilkins, L.; Nicol, C. The effect of keel fractures on egg-production parameters, mobility and behaviour in individual laying hens. Anim. Welf. Ufaw J. 2012, 21, 127. [Google Scholar] [CrossRef]
- Nasr, M.A.; Nicol, C.J.; Wilkins, L.; Murrell, J.C. The effects of two non-steroidal anti-inflammatory drugs on the mobility of laying hens with keel bone fractures. Vet. Anaesth. Analg. 2015, 42, 197–204. [Google Scholar] [CrossRef]
- Riber, A.B.; Casey-Trott, T.M.; Herskin, M.S. The influence of keel bone damage on welfare of laying hens. Front. Vet. Sci. 2018, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Rentsch, A.K.; Rufener, C.B.; Spadavecchia, C.; Stratmann, A.; Toscano, M.J. Laying hen’s mobility is impaired by keel bone fractures and does not improve with paracetamol treatment. Appl. Anim. Behav. Sci. 2019, 216, 19–25. [Google Scholar] [CrossRef]
- Farm Animal Welfare Council. An Open Letter to Great Britain Governments: Keel Bone Fracture in Laying Hens; Farm Animal Welfare Council: London, UK, 2013. [Google Scholar]
- Buijs, S.; Heerkens, J.; Ampe, B.; Delezie, E.; Rodenburg, T.; Tuyttens, F. Assessing keel bone damage in laying hens by palpation: Effects of assessor experience on accuracy, inter-rater agreement and intra-rater consistency. Poult. Sci. 2019, 98, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Richards, G.; Nasr, M.; Brown, S.; Szamocki, E.; Murrell, J.; Barr, F.; Wilkins, L. Use of radiography to identify keel bone fractures in laying hens and assess healing in live birds. Vet. Rec. 2011, 169, 279. [Google Scholar] [CrossRef]
- Cornell, C.N.; Lane, J.M. Newest factors in fracture healing. Clin. Orthop. Relat. Res. 1992, 277, 297–311. [Google Scholar] [CrossRef]
- Thøfner, I.; Hougen, H.P.; Villa, C.; Lynnerup, N.; Christensen, J.P. Pathological characterization of keel bone fractures in laying hens does not support external trauma as the underlying cause. PLoS ONE 2020, 15, e0229735. [Google Scholar] [CrossRef]
- Tracy, L.M.; Temple, S.M.; Bennett, D.C.; Sprayberry, K.A.; Makagon, M.M.; Blatchford, R.A. The reliability and accuracy of palpation, radiography, and sonography for the detection of keel bone damage. Animals 2019, 9, 894. [Google Scholar] [CrossRef]
- Toscano, M.J.; Dunn, I.C.; Christensen, J.-P.; Petow, S.; Kittelsen, K.; Ulrich, R. Explanations for keel bone fractures in laying hens: Are there explanations in addition to elevated egg production? Poult. Sci. 2020, 99, 4183–4194. [Google Scholar] [CrossRef]
- Wilkins, L.; Brown, S.; Zimmerman, P.; Leeb, C.; Nicol, C. Investigation of palpation as a method for determining the prevalence of keel and furculum damage in laying hens. Vet. Rec. 2004, 155, 547–549. [Google Scholar] [CrossRef]
- Candelotto, L.; Stratmann, A.; Gebhardt-Henrich, S.G.; Rufener, C.; van de Braak, T.; Toscano, M.J. Susceptibility to keel bone fractures in laying hens and the role of genetic variation. Poult. Sci. 2017, 96, 3517–3528. [Google Scholar] [CrossRef]
- Eusemann, B.K.; Baulain, U.; Schrader, L.; Thöne-Reineke, C.; Patt, A.; Petow, S. Radiographic examination of keel bone damage in living laying hens of different strains kept in two housing systems. PLoS ONE 2018, 13, e0194974. [Google Scholar] [CrossRef]
- Whitehead, C.C.; Fleming, R.H.; Julian, R.J.; Sørensen, P. Skeletal problems associated with selection for increased production. Poult. Genet. Breed. Biotechnol. 2003, 29, 52. [Google Scholar]
- Sandilands, V. The laying hen and bone fractures. Vet. Rec. 2011, 169, 411–412. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.L. A chicken for every pot: The economics, evolution and ethics of the modern chicken. Aust. Zool. 2017, 39, 43–51. [Google Scholar] [CrossRef]
- Fleming, R.; McCormack, H.; McTeir, L.; Whitehead, C. Incidence, pathology and prevention of keel bone deformities in the laying hen. Br. Poult. Sci. 2004, 45, 320–330. [Google Scholar] [CrossRef]
- Eusemann, B.K.; Patt, A.; Schrader, L.; Weigend, S.; Thöne-Reineke, C.; Petow, S. The role of egg production in the etiology of keel bone damage in laying hens. Front. Vet. Sci. 2020, 7, 81. [Google Scholar] [CrossRef]
- Håkansson, J.; Bratt, C.; Jensen, P. Behavioural differences between two captive populations of red jungle fowl (gallus gallus) with different genetic background, raised under identical conditions. Appl. Anim. Behav. Sci. 2007, 102, 24–38. [Google Scholar] [CrossRef]
- ECOLEX. Council Directive 1999/74/ec Laying down Minimum Standards for the Protection of Laying Hens; ECOLEX: Gland, Switzerland, 1999. [Google Scholar]
- Statacorp LLC. Stata Statistical Software: Release 15; Statacorp LLC: College Station, TX, USA, 2017. [Google Scholar]
- Riber, A.B.; Hinrichsen, L.K. Keel-bone damage and foot injuries in commercial laying hens in denmark. Anim. Welf. 2016, 25, 179–184. [Google Scholar] [CrossRef]
- Käppeli, S.; Gebhardt-Henrich, S.; Fröhlich, E.; Pfulg, A.; Stoffel, M.H. Prevalence of keel bone deformities in swiss laying hens. Br. Poult. Sci. 2011, 52, 531–536. [Google Scholar] [CrossRef]
- Stratmann, A.; Fröhlich, E.K.; Harlander-Matauschek, A.; Schrader, L.; Toscano, M.J.; Würbel, H.; Gebhardt-Henrich, S.G. Soft perches in an aviary system reduce incidence of keel bone damage in laying hens. PLoS ONE 2015, 10, e0122568. [Google Scholar] [CrossRef]
- Buckner, G.D.; Insko, W., Jr.; Henry, A.H.; Wachs, E.F. Rate of growth and calcification of the sternum of male and female new hampshire chickens having crooked keels. Poult. Sci. 1949, 28, 289–292. [Google Scholar] [CrossRef]
- Pottgüter, R. Feeding laying hens to 100 weeks of age. Lohmann Inf. 2016, 50, 18–21. [Google Scholar]
- Karcher, D.M.; Mench, J.A. Overview of commercial poultry production systems and their main welfare challenges. In Advances in Poultry Welfare; Elsevier: Amsterdam, The Netherlands, 2018; pp. 3–25. [Google Scholar]
- Lohmann Tierzucht. Brown Lite Layers Management Guide; Lohmann Tierzucht: Cuxhaven, Germany, 2017. [Google Scholar]
- Eusemann, B.K.; Sharifi, A.R.; Patt, A.; Reinhard, A.-K.; Schrader, L.; Thöne-Reineke, C.; Petow, S. Influence of a sustained release deslorelin acetate implant on reproductive physiology and associated traits in laying hens. Front. Physiol. 2018, 9, 1846. [Google Scholar] [CrossRef]
- Nasr, M.; Murrell, J.; Nicol, C. The effect of keel fractures on egg production, feed and water consumption in individual laying hens. Br. Poult. Sci. 2013, 54, 165–170. [Google Scholar] [CrossRef]
- Rufener, C.; Baur, S.; Stratmann, A.; Toscano, M.J. Keel bone fractures affect egg laying performance but not egg quality in laying hens housed in a commercial aviary system. Poult. Sci. 2019, 98, 1589–1600. [Google Scholar] [CrossRef]
| Hens 1 | Roosters 2 | |||||
|---|---|---|---|---|---|---|
| n | % | 95% CI 3 | n | % | 95% CI | |
| Fractures | ||||||
| Yes | 1 | 8.3% | 0–48% | 0 | 0% | - |
| No | 11 | 92% | 52–99% | 17 | 100% | - |
| Deviations4 | ||||||
| Yes | 10 3 | 83% | 47–97% | 1 3 | 5.9% | 0.7–36% |
| No | 2 | 17% | 3.5–52% | 16 | 94% | 64–100% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kittelsen, K.E.; Jensen, P.; Christensen, J.P.; Toftaker, I.; Moe, R.O.; Vasdal, G. Prevalence of Keel Bone Damage in Red Jungle Fowls (Gallus gallus)—A Pilot Study. Animals 2020, 10, 1655. https://doi.org/10.3390/ani10091655
Kittelsen KE, Jensen P, Christensen JP, Toftaker I, Moe RO, Vasdal G. Prevalence of Keel Bone Damage in Red Jungle Fowls (Gallus gallus)—A Pilot Study. Animals. 2020; 10(9):1655. https://doi.org/10.3390/ani10091655
Chicago/Turabian StyleKittelsen, Käthe Elise, Per Jensen, Jens Peter Christensen, Ingrid Toftaker, Randi Oppermann Moe, and Guro Vasdal. 2020. "Prevalence of Keel Bone Damage in Red Jungle Fowls (Gallus gallus)—A Pilot Study" Animals 10, no. 9: 1655. https://doi.org/10.3390/ani10091655
APA StyleKittelsen, K. E., Jensen, P., Christensen, J. P., Toftaker, I., Moe, R. O., & Vasdal, G. (2020). Prevalence of Keel Bone Damage in Red Jungle Fowls (Gallus gallus)—A Pilot Study. Animals, 10(9), 1655. https://doi.org/10.3390/ani10091655





