Decline in Distribution and Abundance: Urban Hedgehogs under Pressure
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
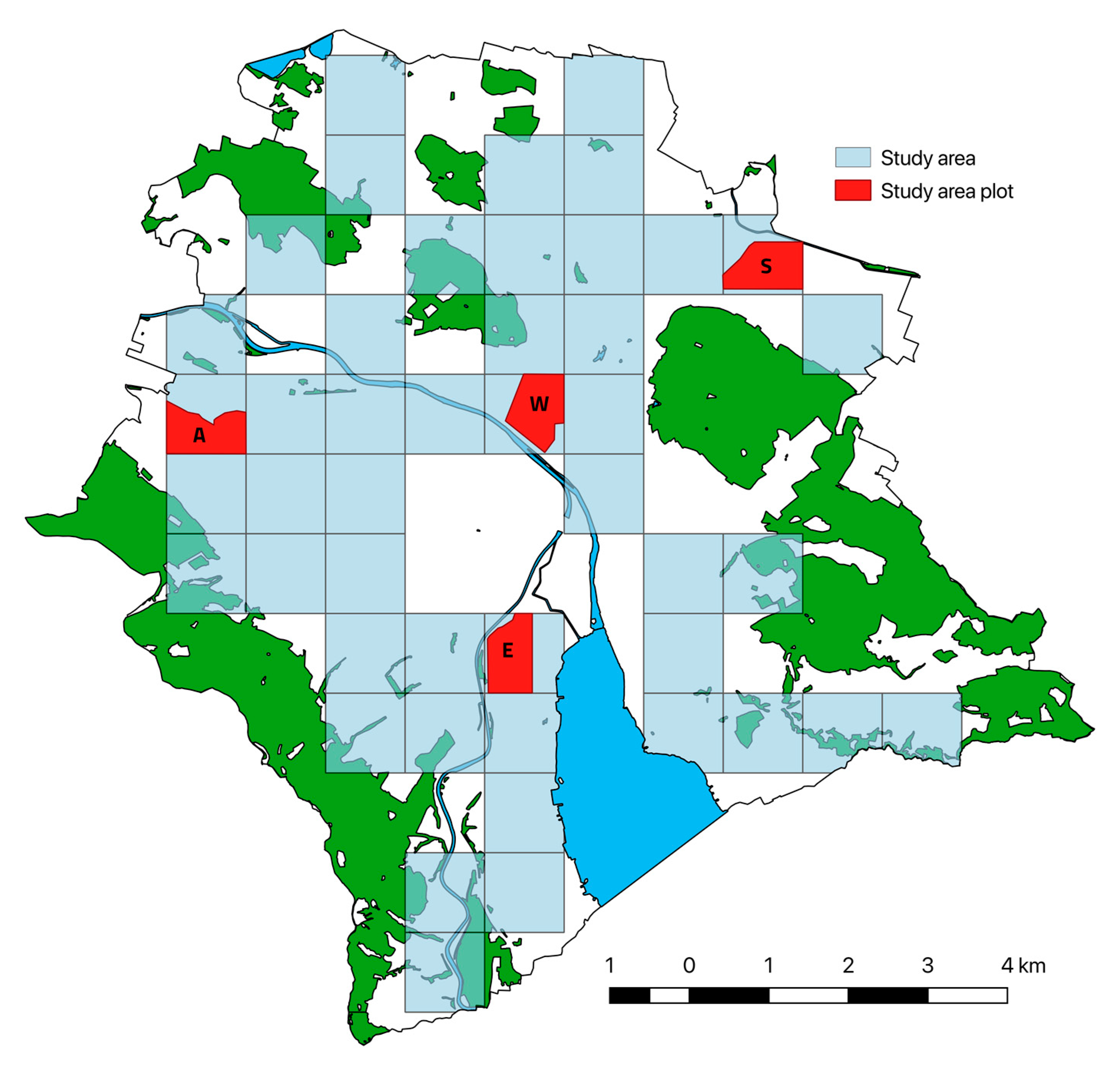
2.1. Study Species and Site
2.2. Past Study (1992)
2.3. Recent Study (2016–2018)
2.3.1. Surveys of Hedgehog Distribution and Abundance Today
2.3.2. Capture Mark Recapture Study
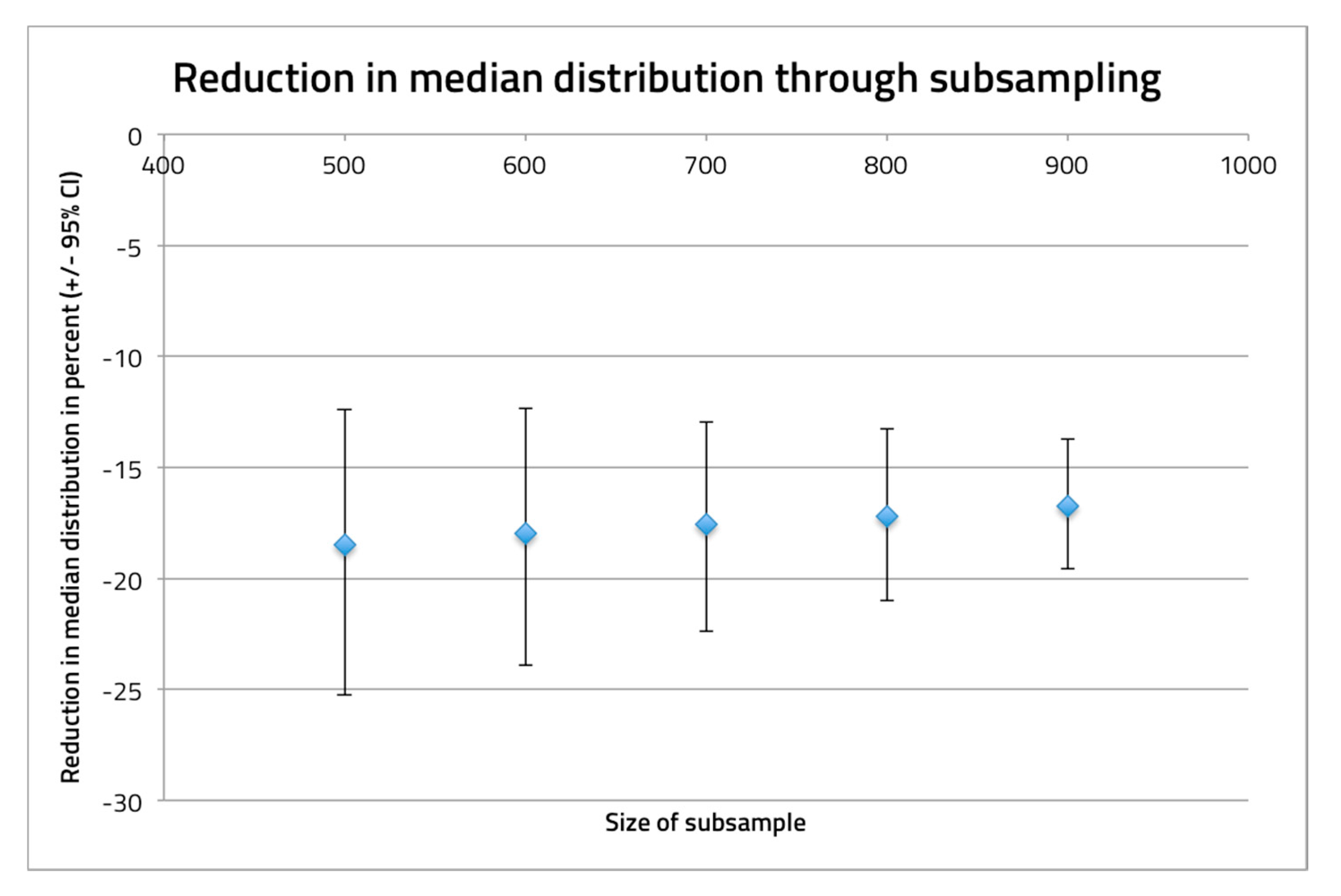
2.4. Analyses
3. Results
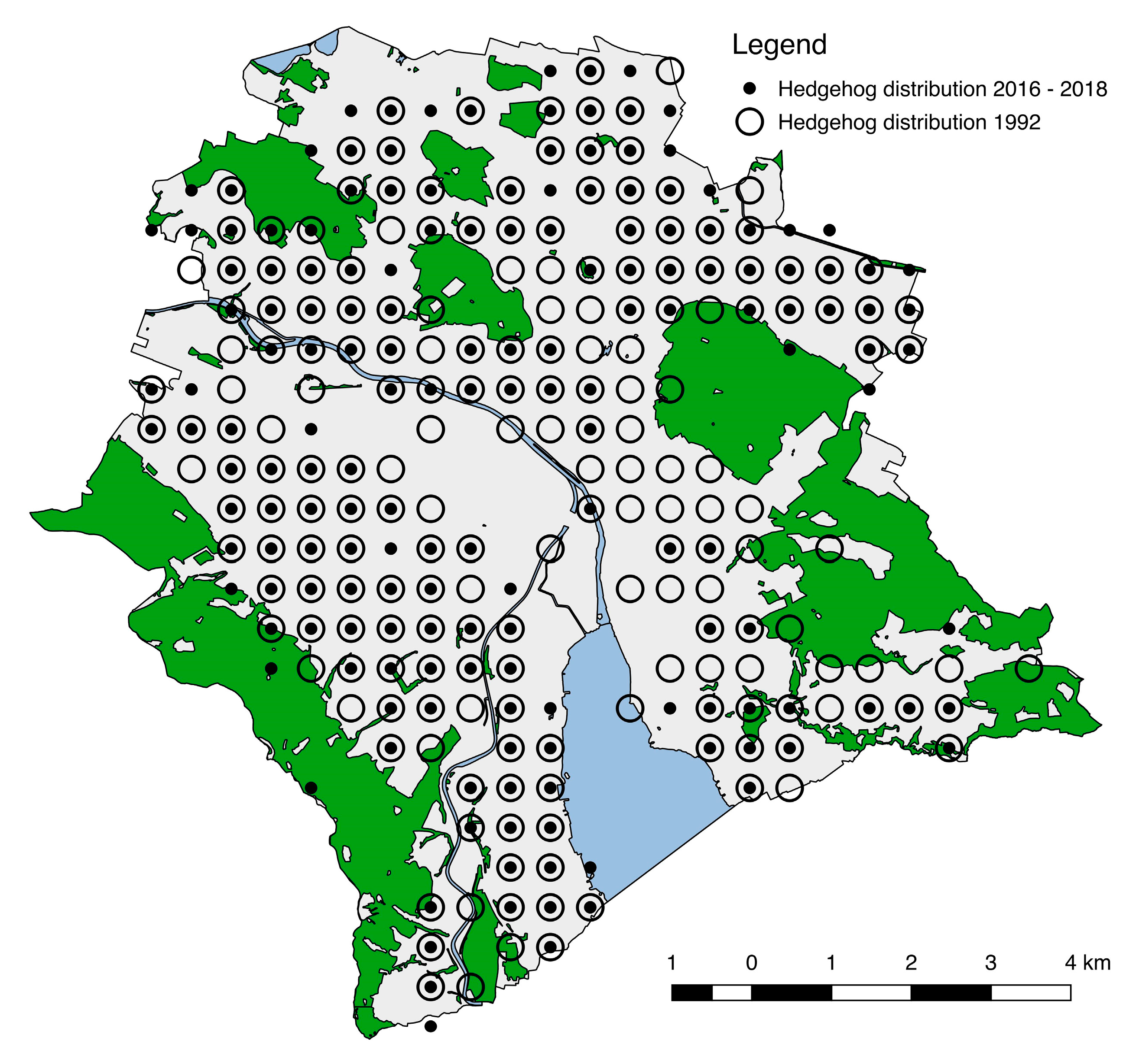
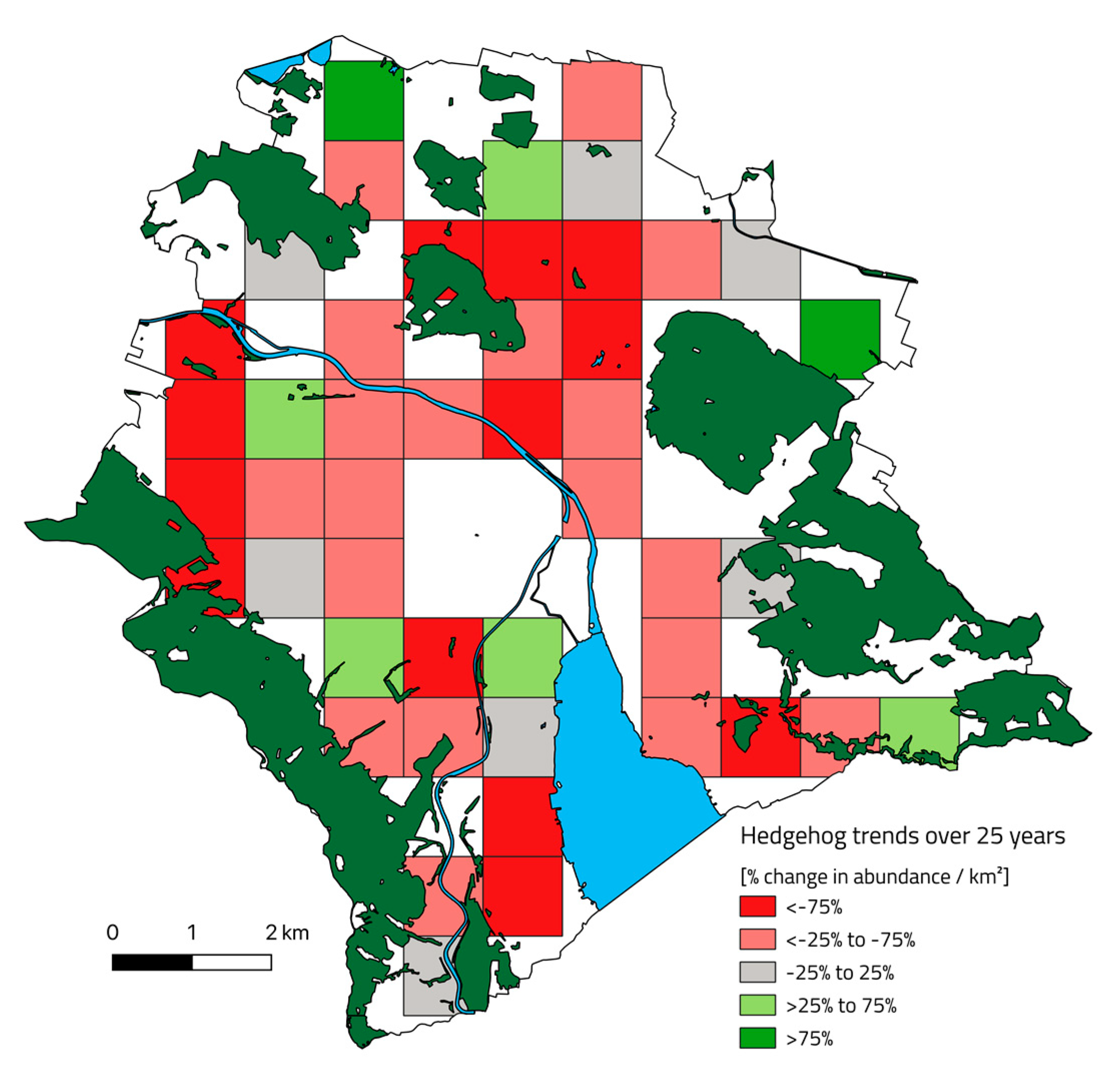
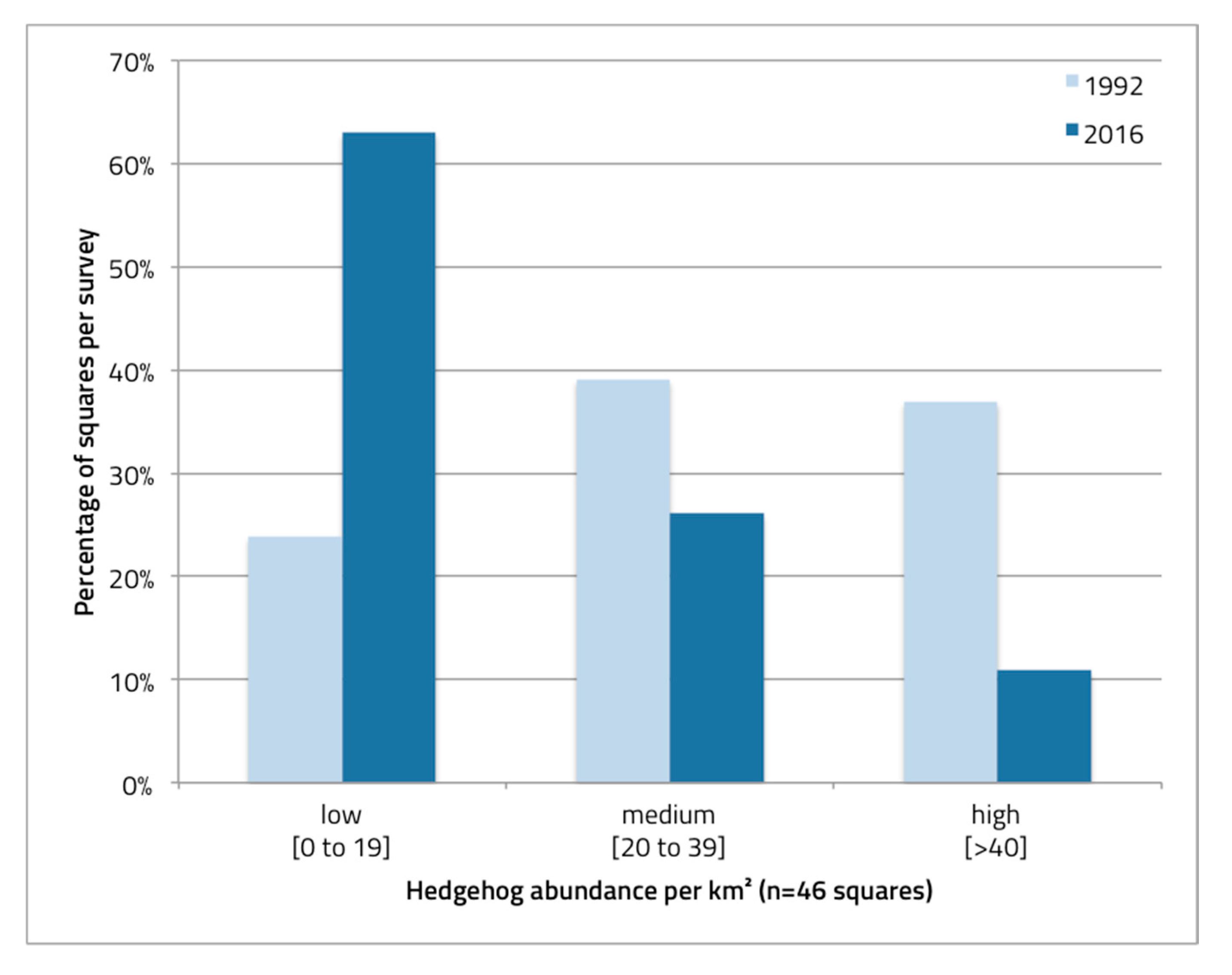
3.1. Change of Hedgehog Distribution in 25 Years
3.2. Change of Hedgehog Abundance and Density in 25 Years
4. Discussion
4.1. Potential Factors Negatively Affecting Hedgehog Populations
4.1.1. Habitat
4.1.2. Food
4.1.3. Poison
4.1.4. Predation
4.1.5. Diseases and Parasites
4.1.6. Extinction Debt
4.2. Comparability of the Studies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A


| Study Area Plot | Individual | Sex | Weight (Range in g) | Health Status | Number of Captures |
|---|---|---|---|---|---|
| Altstetten | A_A | female | 1430 | good | 1 |
| Altstetten | A_B | unknown | 1460 | good | 1 |
| Altstetten | A_C | unknown | 1230 | good | 1 |
| Altstetten | A_D | female | 1000 | good | 1 |
| Schwamendingen | S_A | female | 580–1150 | good | 3 |
| Schwamendingen | S_B | female | 1030–1220 | good | 4 |
| Schwamendingen | S_C | unknown | 860–1010 | good | 2 |
| Schwamendingen | S_D | unknown | 1020 | good | 1 |
| Schwamendingen | S_E | female | 640 | good | 1 |
| Schwamendingen | S_F | female | 710–1120 | good | 4 |
| Schwamendingen | S_G | female | 820–1040 | good | 5 |
| Schwamendingen | S_H | female | 870–950 | good | 2 |
| Schwamendingen | S_I | female | 890–1050 | good | 3 |
| Schwamendingen | S_J | male | 840 | good | 1 |
| Schwamendingen | S_K | male | 790–820 | good | 2 |
| Schwamendingen | S_L | female | 1010–1040 | good | 2 |
| Schwamendingen | S_M | female | 910–980 | good | 4 |
| Schwamendingen | S_N | unknown | 1060 | good | 1 |
| Schwamendingen | S_O | male | 400–830 | good | 3 |
| Schwamendingen | S_P | female | 1130–1260 | good | 2 |
| Schwamendingen | S_Q | female | 1240–1340 | good | 4 |
| Schwamendingen | S_R | unknown | 700–900 | good | 4 |
| Schwamendingen | S_S | female | 1000 | good | 1 |
| Schwamendingen | S_T | male | 1050 | good | 2 |
| Schwamendingen | S_U | male | 920–1080 | good | 3 |
| Schwamendingen | S_V | male | 880–980 | good | 2 |
| Schwamendingen | S_W | female | 1040 | good | 1 |
| Schwamendingen | S_X | female | 470–500 | good | 2 |
| Schwamendingen | S_Y | male | 920 | good | 1 |
| Schwamendingen | S_Z | female | 840 | good | 1 |
| Schwamendingen | S_AA | female | 930 | good | 1 |
| Schwamendingen | S_AB | female | 1210 | good | 2 |
| Schwamendingen | S_AC | male | 1000–1120 | good | 2 |
| Schwamendingen | S_AD | male | 1070 | good | 1 |
| Enge | E_1 | female | 1225–1125 | good | 5 |
| Enge | E_2 | male | 1275–1375 | good | 6 |
| Enge | E_3 | female | 825–875 | good | 4 |
| Enge | E_4 | male | 875–1025 | good | 3 |
| Enge | E_5 | female | 1225 | good | 1 |
| Enge | E_6 | female | 875–925 | good | 3 |
| Enge | E_7 | female | 1125 | good | 1 |
| Enge | E_8 | female | 1275–1325 | good | 3 |
| Enge | E_9 | female | 1275–1325 | good | 2 |
| Enge | E_10 | male | 925–1025 | good | 3 |
| Enge | E_11 | unknown | 825–875 | good | 2 |
| Enge | E_12 | unknown | 975–1075 | good | 2 |
| Enge | E_13 | unknown | 675 | good | 2 |
| Enge | E_14 | unknown | 1025 | good | 1 |
| Enge | E_15 | female | 925–1075 | good, medium | 2 |
| Enge | E_16 | unknown | 1125 | good | 1 |
| Enge | E_17 | female | 1175 | good | 1 |
| Enge | E_18 | male | 1350 | many flees | 1 |
| Enge | E_19 | female | 1125 | good | 1 |
| Wipkingen | W_A | male | 1275 | good | 1 |
| Wipkingen | W_B | female | 750 | good | 2 |
| Wipkingen | W_C | male | 1100 | good | 1 |
| Study Area | Estimate | Standard Error | Unconditional Standard Error |
|---|---|---|---|
| Enge | 22.72 | 3.28 | 3.63 |
| Schwamendingen | 35.19 | 3.78 | 3.87 |
| Wipkingen | 3.52 | 0.79 | NA |
| Altstetten | 4.69 | 0.92 | NA |
References
- World Urbanization Prospects—The 2018 Revision; United Nations: New York, NY, USA, 2018; Volume 12.
- Seto, K.C.; Güneralp, B.; Hutyra, L.R. Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. Proc. Natl. Acad. Sci. USA 2012, 109, 16083–16088. [Google Scholar] [CrossRef]
- Shochat, E.; Warren, P.S.; Faeth, S.H.; McIntyre, N.E.; Hope, D. From patterns to emerging processes in mechanistic urban ecology. Trends Ecol. Evol. 2006, 21, 186–191. [Google Scholar] [CrossRef]
- McKinney, M.L. Urbanization as a major cause of biotic homogenization. Biol. Conserv. 2006, 127, 247–260. [Google Scholar] [CrossRef]
- Lin, B.; Meyers, J.; Barnett, G. Understanding the potential loss and inequities of green space distribution with urban densification. Urban For. Urban Green. 2015, 14, 952–958. [Google Scholar] [CrossRef]
- Haaland, C.; van den Bosch, C.K. Challenges and strategies for urban green-space planning in cities undergoing densification: A review. Urban For. Urban Green. 2015, 14, 760–771. [Google Scholar] [CrossRef]
- Fernandez-Juricic, E.; Telleria, J.L. Effects of human disturbance on spatial and temporal feeding patterns of Blackbird Turdus merula in urban parks in Madrid, Spain. Bird Study 2000, 47, 13–21. [Google Scholar] [CrossRef]
- Reijnen, R.; Foppen, R.; Veenbaas, G. Disturbance by traffic of breeding birds: Evaluation of the effect and considerations in planning and managing road corridors. Biodivers. Conserv. 1997, 581, 567–581. [Google Scholar] [CrossRef]
- Dowding, C.V.; Harris, S.; Poulton, S.; Baker, P.J. Nocturnal ranging behaviour of urban hedgehogs, Erinaceus europaeus, in relation to risk and reward. Anim. Behav. 2010, 80, 13–21. [Google Scholar] [CrossRef]
- Werner, P. The ecology of urban areas and their functions for species diversity. Landsc. Ecol. Eng. 2011, 7, 231–240. [Google Scholar] [CrossRef]
- Lowry, H.; Lill, A.; Wong, B.B.M. Behavioural responses of wildlife to urban environments. Biol. Rev. 2013, 88, 537–549. [Google Scholar] [CrossRef]
- Evans, K.L.; Hatchwell, B.J.; Parnell, M.; Gaston, K.J. A conceptual framework for the colonisation of urban areas: The blackbird Turdus merula as a case study. Biol. Rev. 2010, 85, 643–667. [Google Scholar] [CrossRef]
- Diamond, S.E.; Chick, L.; Perez, A.B.E.; Strickler, S.A.; Martin, R.A. Rapid evolution of ant thermal tolerance across an urban-rural temperature cline. Biol. J. Linn. Soc. 2017, 1–10. [Google Scholar] [CrossRef]
- Cheptou, P.-O.; Carrue, O.; Rouifed, S.; Cantarel, A. Rapid evolution of seed dispersal in an urban environment in the weed Crepis sancta. Proc. Natl. Acad. Sci. USA 2008, 105, 3796–3799. [Google Scholar] [CrossRef]
- Reid, N.M.; Proestou, D.A.; Clark, B.W.; Warren, W.C.; Colbourne, J.K.; Shaw, J.R.; Karchner, S.I.; Hahn, M.E.; Nacci, D.; Oleksiak, M.F.; et al. The genomic landscape of rapid repeated evolutionary adaptation to toxic pollution in wild fish. Science 2016, 354, 1305–1308. [Google Scholar] [CrossRef] [PubMed]
- Hendry, A.P.; Farrugia, T.J.; Kinnison, M.T. Human influences on rates of phenotypic change in wild animal populations. Mol. Ecol. 2008, 17, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Ineichen, S.; Ruckstuhl, M. Stadtfauna—600 Tierarten der Stadt Zürich; Haupt Verlag: Bern, Switzerland, 2010. [Google Scholar]
- Hubert, P.; Julliard, R.; Biagianti, S.; Poulle, M.L. Ecological factors driving the higher hedgehog (Erinaceus europeaus) density in an urban area compared to the adjacent rural area. Landsc. Urban Plan. 2011, 103, 34–43. [Google Scholar] [CrossRef]
- Zingg, R. Aktivität Sowie Habitat-und Raumnutzung von Igeln (Erinaceus europaeus) in Einem Ländlichen Siedlungsgebiet; Zürich Zentralstelle der Studentenschaft: Zurich, Switzerland, 1994. [Google Scholar]
- Pettett, C.E.; Moorhouse, T.P.; Johnson, P.J.; Macdonald, D.W. Factors affecting hedgehog (Erinaceus europaeus) attraction to rural villages in arable landscapes. Eur. J. Wildl. Res. 2017, 63. [Google Scholar] [CrossRef]
- Van de Poel, J.L.; Dekker, J.; van Langevelde, F. Dutch hedgehogs Erinaceus europaeus are nowadays mainly found in urban areas, possibly due to the negative Effects of badgers Meles meles. Wildlife Biol. 2015, 21, 51–55. [Google Scholar] [CrossRef]
- Williams, B.M.; Baker, P.J.; Thomas, E.; Wilson, G.; Judge, J.; Yarnell, R.W. Reduced occupancy of hedgehogs (Erinaceus europaeus) in rural England and Wales: The influence of habitat and an asymmetric intra-guild predator. Sci. Rep. 2018, 8, 17–20. [Google Scholar] [CrossRef]
- Rasmussen, S.L.; Berg, T.B.; Dabelsteen, T.; Jones, O.R. The ecology of suburban juvenile European hedgehogs (Erinaceus europaeus) in Denmark. Ecol. Evol. 2019, 1–14. [Google Scholar] [CrossRef]
- Egli, R.; Bontadina, F.; Deplazes, P.; Arlettaz, R. Comparison of Physical Condition and Parasite Burdens in Rural, Suburban and Urban Hedgehogs Erinaceus europaeus: Implications for Conservation; Universität Bern: Bern, Germany, 2004. [Google Scholar]
- Doncaster, C.P.; Dickman, C.R.; Macdonald, D.W. Feeding ecology of red foxes (Vulpes vulpes) in the city of Oxford, England. J. Mammal. 1990, 71, 188–194. [Google Scholar] [CrossRef]
- Contesse, P.; Hegglin, D.; Gloor, S.; Bontadina, F.; Deplazes, P. The diet of urban foxes (Vulpes vulpes) and the availability of anthropogenic food in the city of Zurich, Switzerland. Mamm. Biol. 2004, 69, 81–95. [Google Scholar] [CrossRef]
- Baker, P.J.; Harris, S. Urban mammals: What does the future hold? An analysis of the factors affecting patterns of use of residential gardens in Great Britain. Mamm. Rev. 2007, 37, 297–315. [Google Scholar] [CrossRef]
- Pickett, S.T.A.; Cadenasso, M.L.; Grove, J.M.; Nilon, C.H.; Pouyat, R.V.; Zipperer, W.C.; Costanza, R. Urban ecological systems: Linking terrestrial ecological, physical, and socioeconomic components of metropolitan areas. Annu. Rev. Ecol. Syst. 2001, 32, 127–157. [Google Scholar] [CrossRef]
- Møller, A.P. Urban areas as refuges from predators and flight distance of prey. Behav. Ecol. 2012, 23, 1030–1035. [Google Scholar] [CrossRef]
- Gering, J.C.; Blair, R.B. Predation on artificial bird nests along an urban gradient: Predatory risk or relaxation in urban environments? Ecography 1999, 22, 532–541. [Google Scholar] [CrossRef]
- Huijser, M.P.; Bergers, P.J.M. The effect of roads and traffic on hedgehog (Erinaceus europaeus) populations. Biol. Conserv. 2000, 95, 6–9. [Google Scholar] [CrossRef]
- Hof, A.R.; Bright, P.W. Quantifying the long-term decline of the West European hedgehog in England by subsampling citizen-science datasets. Eur. J. Wildl. Res. 2016, 62, 407–413. [Google Scholar] [CrossRef]
- Krange, M. Change in the occurrence of the West European Hedgehog (Erinaceus europaeus) in Western Sweden during 1950–2010; Karlstad University: Karlstad, Sweden, 2015. [Google Scholar]
- Harris, S.; Morris, P.; Wray, S.; Yalden, D. A review of British mammals: Population estimates and conservation status of British mammals other than cetaceans; Joint Nature Conservation Committee: Peterborough, UK, 1995; p. 216. [Google Scholar]
- Battersby, J. UK Mammals: Species Status and Population Trends; JNCC: Peterborough, UK, 2005. [Google Scholar]
- Wembridge, D. The State of Britain’s hedgehogs 2011. Available online: https://ptes.org/wp-content/uploads/2015/11/SOBH2011.pdf (accessed on 23 March 2020).
- Roos, S.; Johnston, A.; Noble, D. UK Hedgehog datasets and their potential for long-term monitoring. British Trust for Ornithology Research Report 598. 2012. Available online: https://www.bto.org/our-science/projects/gbw/publications/papers/monitoring/btorr598 (accessed on 15 May 2020).
- Wilson, E. Conservation Strategy for West-European Hedgehog (Erinaceus europaeus) in the United Kingdom (2015–2025); People’s Trust for Endangered Species: London, UK, 2018. [Google Scholar]
- Mathews, F.; Harrower, C. Regional Red List of British Mammals; The Mammal Society: Dorset, UK, 2020. [Google Scholar]
- Yarnell, R.W.; Pacheco, M.; Williams, B.; Neumann, J.L.; Rymer, D.J.; Baker, P.J. Using occupancy analysis to validate the use of footprint tunnels as a method for monitoring the hedgehog Erinaceus europaeus. Mamm. Rev. 2014, 44, 234–238. [Google Scholar] [CrossRef]
- Williams, B.; Mann, N.; Neumann, J.L.; Yarnell, R.W.; Baker, P.J. A prickly problem: Developing a volunteer-friendly tool for monitoring populations of a terrestrial urban mammal, the Western European hedgehog (Erinaceus europaeus). Urban Ecosyst. 2018. [Google Scholar] [CrossRef]
- Bontadina, F.; Gloor, S.; Hotz, T. Igel—Wildtiere in der Stadt; Gartenbau- und Landwirtschaftsamt: Zurich, Switzerland, 1993. [Google Scholar]
- Hof, A.R.; Snellenberg, J.; Bright, P.W. Food or fear? Predation risk mediates edge refuging in an insectivorous mammal. Anim. Behav. 2012, 83, 1099–1106. [Google Scholar] [CrossRef]
- White, G.C.; Burnham, K.P. Program MARK: Survival estimation from populations of marked animals. Bird Study 1999, 46, S120–S139. [Google Scholar] [CrossRef]
- RStan: The R Interface to Stan, R package version 2.21.2; Stan Development Team, 2018. Available online: https://mc-stan.org/ (accessed on 13 March 2020).
- Becker, R.A.; Chambers, J.M.; Wilks, A.R. The New S Language: A Programming Environment for Data Analysis and Graphics; Wadsworth & Brooks/Cole Computer Science Series; Chapman & Hall: London, UK, 1988. [Google Scholar]
- QGIS Geographic Information System, Version 2.18, QGIS Development Team, Open Source Geospatial Foundation Project; 2009. Available online: https://qgis.org/en/site/ (accessed on 28 March 2020).
- R: A Language and Environment for Statistical Computing. Version 3.6.2, R Core Team; R Foundation for Statistical Computing: Vienna, Austria, 2008.
- Parrott, D.; Etherington, T.R.; Dendy, J. A geographically extensive survey of hedgehogs (Erinaceus europaeus) in England. Eur. J. Wildl. Res. 2014, 60, 399–403. [Google Scholar] [CrossRef]
- Davey, P.A.; Aebischer, N.J. Participation of the National Gamebag Census in the Mammal Surveillance Network; JNCC: Peterborough, UK, 2006. [Google Scholar]
- Hof, A.R.; Bright, P.W. The value of green-spaces in built-up areas for western hedgehogs. Lutra 2009, 52, 69–82. [Google Scholar]
- Braaker, S.; Kormann, U.; Bontadina, F.; Obrist, M.K. Prediction of genetic connectivity in urban ecosystems by combining detailed movement data, genetic data and multi-path modelling. Landsc. Urban Plan. 2017, 160, 107–114. [Google Scholar] [CrossRef]
- Pettett, C.E.; Johnson, P.J.; Moorhouse, T.P.; Macdonald, D.W. National predictors of hedgehog Erinaceus europaeus distribution and decline in Britain. Mamm. Rev. 2018, 48, 1–6. [Google Scholar] [CrossRef]
- Rondinini, A.C.; Doncaster, C.P. Roads as barriers to movement for hedgehogs. Funct. Ecol. 2002, 16, 504–509. [Google Scholar] [CrossRef]
- Braaker, S.; Moretti, M.; Boesch, R.; Ghazoul, J.; Obrist, M.K.; Bontadina, F. Assessing habitat connectivity for ground-dwelling animals in an urban environment. Ecol. Appl. 2014, 24, 1583–1595. [Google Scholar] [CrossRef]
- Hof, A.R.; Bright, P.W. The value of agri-environment schemes for macro-invertebrate feeders: Hedgehogs on arable farms in Britain. Anim. Conserv. 2010, 13, 467–473. [Google Scholar] [CrossRef]
- Hof, A.R. A Study of the current status of the hedgehog (Erinaceus europaeus), and its decline in Great Britain since 1960. Ph.D. Thesis, University of London, London, UK, 2009; pp. 15–50. [Google Scholar]
- Zürich. Statistisches Jahrbuch der Stadt Zürich 2017; Stadt Zürich Präsidialdepartement: Zurich, Switzerland, 2017. [Google Scholar]
- Felber, S. Grünflächenveränderungen in Siedlungsgebieten der Schweiz: Mit Fernerkundungsdaten zum Vergleich der Grünflächenveränderungen auf Gemeindeebene; Swiss Federal Institute of Technology: Zurich, Switzerland, 2020. [Google Scholar]
- Wright, P.G.R.; Coomber, F.G.; Bellamy, C.C.; Perkins, S.E.; Mathews, F. Predicting hedgehog mortality risks on British roads using habitat suitability modelling. Biodiver. Conserv. 2019, 2050, 1–22. [Google Scholar] [CrossRef]
- Geiger, M.; Taucher, A.L.; Gloor, S.; Hegglin, D.; Bontadina, F. In the footsteps of city foxes: Evidence for a rise of urban badger populations in Switzerland. Hystrix 2018. [Google Scholar] [CrossRef]
- Wroot, A.J. Feeding Ecology of the European Hedgehog. Ph.D. Thesis, University of London, London, UK, 1984. [Google Scholar]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Müller, A.; Sumser, H.; Hörren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Krebs, J.R.; Wilson, J.D.; Bradbury, R.B.; Gavin, M. The second silent spring? Nature 1999, 400. [Google Scholar] [CrossRef]
- Gazzard, A.; Baker, P.J. Patterns of feeding by householders affect activity of hedgehogs (Erinaceus europaeus) during the hibernation period. Animals 2020, 10, 1344. [Google Scholar] [CrossRef] [PubMed]
- Yalden, D.W. The food of the hedgehog in England. Acta Theriol. 1976, 21, 401–424. [Google Scholar] [CrossRef]
- Dowding, C.V.; Shore, R.F.; Worgan, A.; Baker, P.J.; Harris, S. Accumulation of anticoagulant rodenticides in a non-target insectivore, the European hedgehog (Erinaceus europaeus). Environ. Pollut. 2010, 158, 161–166. [Google Scholar] [CrossRef]
- D’Havé, H.; Mubiana, V.K.; Blust, R.; De Coen, W.; Scheirs, J.; Verhagen, R. Non-destructive pollution exposure assessment in the European hedgehog (Erinaceus europaeus): II. Hair and spines as indicators of endogenous metal and as concentrations. Environ. Pollut. 2006, 142, 438–448. [Google Scholar] [CrossRef]
- Spurr, E.B.; Wright, G.R.G.; Radford, C.D.; Brown, L.E.; Maitland, M.J.; Taylor, G.E. Residues of brodifacoum and other anticoagulant pesticides in target and non-target species, Nelson Lakes National Park, New Zealand. N. Z. J. Zool. 2005, 32, 237–249. [Google Scholar] [CrossRef]
- Ruiz-Suárez, N.; Boada, L.D.; Henríquez-Hernández, L.A.; González-Moreo, F.; Suárez-Pérez, A.; Camacho, M.; Zumbado, M.; Almeida-González, M.; del Mar Travieso-Aja, M.; Luzardo, O.P. Continued implication of the banned pesticides carbofuran and aldicarb in the poisoning of domestic and wild animals of the Canary Islands (Spain). Sci. Total Environ. 2015, 505, 1093–1099. [Google Scholar] [CrossRef]
- Vermeulen, F.; Covaci, A.; D’Havé, H.; Van den Brink, N.W.; Blust, R.; De Coen, W.; Bervoets, L. Accumulation of background levels of persistent organochlorine and organobromine pollutants through the soil-earthworm-hedgehog food chain. Environ. Int. 2010, 36, 721–727. [Google Scholar] [CrossRef]
- Reeve, N. Hedgehogs (Poyser Natural History); Poyser: London, UK, 1994; ISBN 0-05661-081-X. [Google Scholar]
- Rautio, A.; Kunnasranta, M.; Valtonen, A.; Ikonen, M.; Hyvärinen, H.; Holopainen, I.J.; Kukkonen, J.V.K. Sex, age, and tissue specific accumulation of eight metals, arsenic, and selenium in the European Hedgehog (Erinaceus europaeus). Arch. Environ. Contam. Toxicol. 2010, 59, 642–651. [Google Scholar] [CrossRef] [PubMed]
- BLW (Bundesamt für Landwirtschaft). Verkaufsstatistik 2018 von Pflanzenschutzmitteln in der Schweiz. 2019. Available online: https://www.admin.ch/gov/de/start/dokumentation/medienmitteilungen.msg-id-77550.html (accessed on 20 April 2020).
- Meftaul, I.M.; Venkateswarlu, K.; Dharmarajan, R.; Annamalai, P.; Megharaj, M. Pesticides in the urban environment: A potential threat that knocks at the door. Sci. Total Environ. 2020, 711. [Google Scholar] [CrossRef] [PubMed]
- Reeve, N.J.; Huijser, M.P. Mortality factors affecting wild hedgehogs: A study of records from wildlife rescue centres. Lutra 1999, 42, 7–24. [Google Scholar]
- Doncaster, C.P. Testing the role of intraguild predation in regulating hedgehog populations. Proc. R. Soc. B 1992, 249, 113–117. [Google Scholar] [PubMed]
- Judge, J.; Wilson, G.J.; Macarthur, R.; Delahay, R.J.; McDonald, R.A. Density and abundance of badger social groups in England and Wales in 2011–2013. Sci. Rep. 2014, 4, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Young, R.P.; Davison, J.; Trewby, I.D.; Wilson, G.J.; Delahay, R.J.; Doncaster, C.P. Abundance of hedgehogs (Erinaceus europaeus) in relation to the density and distribution of badgers (Meles meles). J. Zool. 2006, 269, 349–356. [Google Scholar] [CrossRef]
- Laundré, J.W.; Hernández, L.; Ripple, W.J. The Landscape of Fear: Ecological Implications of Being Afraid. Open Ecol. J. 2010, 3, 1–7. [Google Scholar] [CrossRef]
- Micol, T.; Doncaster, C.; Mackinlay, L. Correlates of local variation in the abundance of hedgehogs Erinaceus europaeus. J. Anim. Ecol. 1994, 63, 851–860. [Google Scholar] [CrossRef]
- Doncaster, C.P. Factors regulating local variations in abundance: Field tests on hedgehogs, Erinaceus europaeus. Oikos 1994, 69, 182–192. [Google Scholar] [CrossRef]
- Trewby, I.D.; Young, R.; McDonald, R.A.; Wilson, G.J.; Davison, J.; Walker, N.; Robertson, A.; Patrick Doncaster, C.; Delahay, R.J. Impacts of removing badgers on localised counts of hedgehogs. PLoS ONE 2014, 9, 2–5. [Google Scholar] [CrossRef]
- Gulland, F.M.D. The role of nematode parasites in Soay sheep (Ovis aries L.) mortality during a population crash. Parasitology 1992, 105, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Gaglio, G.; Allen, S.; Bowden, L.; Bryant, M.; Morgan, E.R. Parasites of European hedgehogs (Erinaceus europaeus) in Britain: Epidemiological study and coprological test evaluation. Eur. J. Wildl. Res. 2010, 56, 839–844. [Google Scholar] [CrossRef]
- Daniel, M.; Danielová, V.; Kříž, B.; Jirsa, A.; Nožička, J. Shift of the tick Ixodes ricinus and tick-borne encephalitis to higher altitudes in Central Europe. Eur. J. Clin. Microbiol. Infect. Dis. 2003, 22, 327–328. [Google Scholar] [CrossRef] [PubMed]
- Scharlemann, J.P.W.; Johnson, P.J.; Smith, A.A.; Macdonald, D.W.; Randolph, S.E. Trends in ixodid tick abundance and distribution in Great Britain. Med. Vet. Entomol. 2008, 22, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Gillis-Germitsch, N.; Tritten, L.; Hegglin, D.; Deplazes, P.; Schnyder, M. Conquering Switzerland: Emergence of Angiostrongylus vasorum over three decades and rapid regional increase in the fox population contrasts with the stable prevalence of lungworms. Parasitology 2020, 1–28. [Google Scholar] [CrossRef]
- Morgan, E.R.; Tomlinson, A.; Hunter, S.; Nichols, T.; Roberts, E.; Fox, M.T.; Taylor, M.A. Angiostrongylus vasorum and Eucoleus aerophilus in foxes (Vulpes vulpes) in Great Britain. Vet. Parasitol. 2008, 154, 48–57. [Google Scholar] [CrossRef]
- Gloor, S.; Bontadina, F.; Hegglin, D.; Deplazes, P.; Breitenmoser, U. The rise of urban fox populations in Switzerland. Mamm. Biol. Zeitschrift für Säugetierkd. 2001, 66, 155–164. [Google Scholar]
- Deplazes, P.; Hegglin, D.; Gloor, S.; Romig, T. Wilderness in the city: The urbanization of Echinococcus multilocularis. Trends Parasitol. 2004, 20, 77–84. [Google Scholar] [CrossRef]
- Pfäffle, M.; Černábolfíková, B.; Hulva, P.; Petney, T. Different parasite faunas in sympatric populations of sister hedgehog species in a secondary contact zone. PLoS ONE 2014, 9, 1–14. [Google Scholar] [CrossRef]
- Lund, S.; Id, R.; Id, J.L.; Wijk, R.E., Van; Jones, O.R.; Bj, T.; Angen, Ø.; Larsen, A.R. European hedgehogs (Erinaceus europaeus) as a natural reservoir of methicillin-resistant Staphylococcus aureus carrying mecC in Denmark. PLoS ONE 2019, 114, 1–13. [Google Scholar]
- Monecke, S.; Gavier-Widen, D.; Mattsson, R.; Rangstrup-Christensen, L.; Lazaris, A.; Coleman, D.C.; Shore, A.C.; Ehricht, R. Detection of mecC-Positive Staphylococcus aureus (CC130-MRSA-XI) in diseased European hedgehogs (Erinaceus europaeus) in Sweden. PLoS ONE 2013, 8, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, B.; Persson, L.; Ekström, K.; Unnerstad, H.E.; Uhlhorn, H.; Börjesson, S. High occurrence of mecC-MRSA in wild hedgehogs (Erinaceus europaeus) in Sweden. Vet. Microbiol. 2017, 207, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Mayt, R.M.; Lehman, C.L.; Nowakt, M.A. Habitat destruction and the extinction debt. Nature 1994, 371, 65–66. [Google Scholar] [CrossRef]
- Keller, L.F.; Waller, D.M. Inbreeding effects in the wild. Trends Ecol. Evol. 2002, 17, 230–241. [Google Scholar] [CrossRef]
- Hahs, A.K.; McDonnell, M.J.; McCarthy, M.A.; Vesk, P.A.; Corlett, R.T.; Norton, B.A.; Clemants, S.E.; Duncan, R.P.; Thompson, K.; Schwartz, M.W.; et al. A global synthesis of plant extinction rates in urban areas. Ecol. Lett. 2009, 12, 1165–1173. [Google Scholar] [CrossRef]
- Lambert, M.R.; Donihue, C.M. Urban biodiversity management using evolutionary tools. Nat. Ecol. Evol. 2020. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taucher, A.L.; Gloor, S.; Dietrich, A.; Geiger, M.; Hegglin, D.; Bontadina, F. Decline in Distribution and Abundance: Urban Hedgehogs under Pressure. Animals 2020, 10, 1606. https://doi.org/10.3390/ani10091606
Taucher AL, Gloor S, Dietrich A, Geiger M, Hegglin D, Bontadina F. Decline in Distribution and Abundance: Urban Hedgehogs under Pressure. Animals. 2020; 10(9):1606. https://doi.org/10.3390/ani10091606
Chicago/Turabian StyleTaucher, Anouk L., Sandra Gloor, Adrian Dietrich, Madeleine Geiger, Daniel Hegglin, and Fabio Bontadina. 2020. "Decline in Distribution and Abundance: Urban Hedgehogs under Pressure" Animals 10, no. 9: 1606. https://doi.org/10.3390/ani10091606
APA StyleTaucher, A. L., Gloor, S., Dietrich, A., Geiger, M., Hegglin, D., & Bontadina, F. (2020). Decline in Distribution and Abundance: Urban Hedgehogs under Pressure. Animals, 10(9), 1606. https://doi.org/10.3390/ani10091606






