The Effect of a Moderate Exercise Program on Serum Markers of Bone Metabolism in Dogs
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Exercise protocol
2.3. Sample collection
2.4. Assays for the bone markers
2.5. Statistical analyses
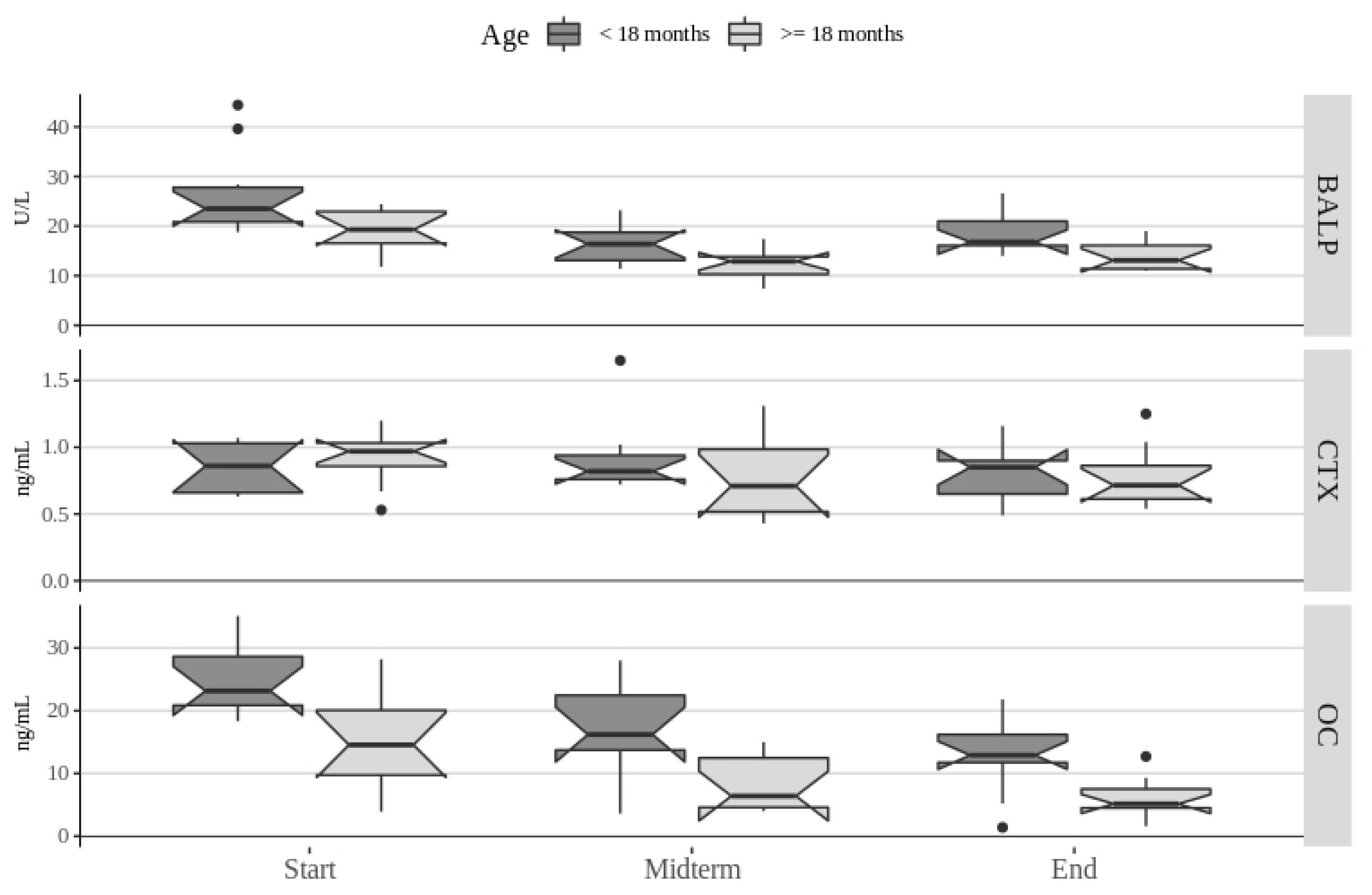
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Arens, D.; Sigrist, I.; Alini, M.; Schawalder, P.; Schneider, E.; Egermann, M. Seasonal changes in bone metabolism in sheep. Vet. J. 2007, 174, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Banfi, G.; Lombardi, G.; Colombini, A.; Lippi, G.; Banfi, P.G. Bone metabolism markers in sports medicine. Sports Med. 2010, 40, 697–714. [Google Scholar] [CrossRef] [PubMed]
- Liesegang, A.; Reutter, R.; Sassi, M.L.; Risteli, J.; Kraenzlin, M.; Riond, J.L.; Wanner, M. Diurnal variation in concentrations of various markers of bone metabolism in dogs. Am. J. Vet. Res. 1999, 60, 949–953. [Google Scholar] [PubMed]
- Allen, M.J.; Hoffmann, W.E.; Richardson, D.C.; Breur, G.J. Serum markers of bone metabolism in dogs. Am. J. Vet. Res. 1998, 59, 250–254. [Google Scholar] [PubMed]
- Allen, L.; Allen, M.; Breur, G.; Hoffmann, W.; Richardson, D. A comparison of two techniques for the determination of serum bone-specific alkaline phosphatase activity in dogs. Res. Vet. Sci. 2000, 68, 231–235. [Google Scholar] [CrossRef]
- Allen, M.J. Biochemical markers of bone metabolism in animals: Uses and limitations. Vet. Clin. Pathol. 2003, 32, 101–113. [Google Scholar] [CrossRef]
- Belić, M.; Svetina, A.; Kušec, V.; Rakočević, S.; Grizelj, J.; Robić, M.; Turk, R. Bone alkaline phosphatase, osteocalcin and C-terminal telopeptide as bone turnover in canine bitches. Vet. Arhiv 2010, 80, 705–713. [Google Scholar]
- Hatayama, K.; Nishihara, Y.; Kimura, S.; Goto, K.; Nakamura, D.; Wakita, A.; Urasoko, Y. Serum alkaline phosphatase isoenzymes in laboratory beagle dogs detected by polyacrylamide-gel disk electrophoresis. J. Toxicol. Sci. 2011, 36, 653–660. [Google Scholar] [CrossRef]
- Belic, M.; Kušec, V.; Svetina, A.; Grizelj, J.; Robić, M.; Vrbanac, Z.; Benić, M.; Turk, R. The influence of sex on biochemical markers of bone turnover in dogs. Res. Vet. Sci. 2012, 93, 918–920. [Google Scholar] [CrossRef]
- Maïmoun, L.; Manetta, J.; Couret, I.; Dupuy, A.-M.; Mariano-Goulart, D.; Micallef, J.P.; Peruchon, E.; Rossi, M. The intensity level of physical exercise and the bone metabolism response. Int. J. Sports Med. 2006, 27, 105–111. [Google Scholar] [CrossRef]
- Vainionpää, A.; Korpelainen, R.; Vaananen, H.K.; Haapalahti, J.; Jämsä, T.; Leppäluoto, J. Effect of impact exercise on bone metabolism. Osteoporos. Int. 2009, 20, 1725–1733. [Google Scholar] [CrossRef] [PubMed]
- Eliakim, A.; Raisz, L.G.; Brasel, J.A.; Cooper, D.M. Evidence for increased bone formation following a brief endurance-type training intervention in adolescent males. J. Bone Miner. Res. 1997, 12, 1708–1713. [Google Scholar] [CrossRef] [PubMed]
- Woitge, H.W.; Friedmann, B.; Suttner, S.; Farahmand, I.; Müller, M.; Schmidt-Gayk, H.; Baertsch, P.; Ziegler, R.; Seibel, M.J. Changes in bone turnover induced by aerobic and anaerobic exercise in young males. J. Bone Miner. Res. 1998, 13, 1797–1804. [Google Scholar] [CrossRef]
- Shibata, Y.; Ohsawa, I.; Watanabe, T.; Miura, T.; Sato, Y. Effects of physical training on bone mineral density and bone metabolism. J. Physiol. Anthr. Appl. Hum. Sci. 2003, 22, 203–208. [Google Scholar] [CrossRef]
- Karabulut, M.; Bemben, D.A.; Sherk, V.D.; Anderson, M.A.; Abe, T.; Bemben, M.G. Effects of high-intensity resistance training and low-intensity resistance training with vascular restriction on bone markers in older men. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 111, 1659–1667. [Google Scholar] [CrossRef]
- Kemmler, W.; Lauber, D.; Weineck, J.; Hensen, J.; Kalender, W.; Engelke, K. Benefits of 2 years of intense exercise on bone density, physical fitness, and blood lipids in early postmenopausal osteopenic women. Arch. Intern. Med. 2004, 164, 1084–1091. [Google Scholar] [CrossRef]
- Kato, T.; Terashima, T.; Yamashita, T.; Hatanaka, Y.; Honda, A.; Umemura, Y. Effect of low-repetition jump training on bone mineral density in young women. J. Appl. Physiol. 2006, 100, 839–843. [Google Scholar] [CrossRef]
- Puustjärvi, K.; Lappalainen, R.; Niemitukia, L.; Arnala, I.; Nieminen, J.; Tammi, M.; Helminen, H.J. Long-distance running alters bone anthropometry, elemental composition and mineral density of young dogs. Scand. J. Med. Sci. Sports 2007, 5, 17–23. [Google Scholar] [CrossRef]
- Hiney, K.M.; Potter, G.D.; Gibbs, P.G.; Bloomfield, S.M. Response of serum biochemical markers of bone metabolism to training in the juvenile racehorse. J. Equine Vet. Sci. 2000, 20, 851–857. [Google Scholar] [CrossRef]
- Jackson, B.F.; Goodship, A.E.; Eastell, R.; Price, J.S. Evaluation of serum concentrations of biochemical markers of bone metabolism and insulin-like growth factor I associated with treadmill exercise in young horses. Am. J. Vet. Res. 2003, 64, 1549–1556. [Google Scholar] [CrossRef]
- Fletcher, K.; Topliff, D.; Cooper, S.; Freeman, D.; Geisert, R. Influence of age and sex on serum osteocalcin concentrations in horses at weaning and during physical conditioning. J. Equine Vet. Sci. 2000, 20, 124–126. [Google Scholar] [CrossRef]
- Tharwat, M.; Al-Sobayil, F.; Buczinski, S. Influence of racing on the serum concentrations of acute-phase proteins and bone metabolism biomarkers in racing greyhounds. Vet. J. 2014, 202, 372–377. [Google Scholar] [CrossRef] [PubMed]
- IGDF. IGDF Standards. Available online: https://www.igdf.org.uk/about-us/igdf-standards/ (accessed on 15 July 2020).
- National Research Council. Nutrient Requirements of Dogs and Cats; The National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Vrbanac, Z.; Belić, M.; Brkljača Bottegaro, N.; Blažević, I.; Kolarić, D.; Vojvodić-Schuster, S.; Benić, M.; Kušec, V.; Stanin, D. The effect of long term moderate intensity exercise on heart rate and metabolic status in sedentary Labrador Retrievers. Vet. Arhiv 2016, 86, 553–564. [Google Scholar]
- Ladlow, J.; Hoffmann, W.; Breur, G.; Richardson, D.; Allen, M. Biological variability in serum and urinary indices of bone formation and resorption in dogs. Calcif. Tissue Int. 2002, 70, 186–193. [Google Scholar] [CrossRef]
- Schoenherr, W.D.; MacLeay, J.M.; Yamka, R.M. Evaluation of body composition and cartilage biomarkers in large-breed dogs fed two foods designed for growth. Am. J. Vet. Res. 2010, 71, 934–939. [Google Scholar] [CrossRef]
- Black, A.; Schoknecht, P.A.; Ralston, S.L.; Shapses, S.A. Diurnal variation and age differences in the biochemical markers of bone turnover in horses. J. Anim. Sci. 1999, 77, 75–83. [Google Scholar] [CrossRef]
- Calvo, M.S.; Eyre, D.R.; Gundberg, C.M. Molecular basis and clinical application of biological markers of bone turnover. Endocr. Rev. 1996, 17, 333–368. [Google Scholar]
- Sanecki, R.K.; Hoffmann, W.E.; Hansen, R.; Schaeffer, D.J. Quantification of bone alkaline phosphatase in canine serum. Vet. Clin. Pathol. 1993, 22, 17–23. [Google Scholar] [CrossRef]
- Nielsen, B.; Potter, G.; Greene, L.; Morris, E.; Murray-Gerzik, M.; Smith, W.; Martin, M. Characterization of changes related to mineral balance and bone metabolism in the young racing Quarter Horse. J. Equine Vet. Sci. 1998, 18, 190–200. [Google Scholar] [CrossRef]
- Franck, H.; Beuker, F.; Gurk, S. The effect of physical activity on bone turnover in young adults. Exp. Clin. Endocrinol. Diabetes 2009, 98, 42–46. [Google Scholar] [CrossRef]
- McCarthy, R.; Jeffcott, L.B. Effects of treadmill exercise on cortical bone in the third metacarpus of young horses. Res. Vet. Sci. 1992, 52, 28–37. [Google Scholar] [CrossRef]
- Roghani, T.; Torkaman, G.; Movasseghe, S.; Hedayati, M.; Goosheh, B.; Bayat, N. Effects of short-term aerobic exercise with and without external loading on bone metabolism and balance in postmenopausal women with osteoporosis. Rheumatol. Int. 2012, 33, 291–298. [Google Scholar] [CrossRef]
- Delmas, P.D. Markers of bone turnover for monitoring treatment of osteoporosis with antiresorptive drugs. Osteoporos. Int. 2000, 11, S66–S76. [Google Scholar] [CrossRef]
- Jackson, B.F.; Blumsohn, A.; Goodship, A.E.; Wilson, A.M.; Price, J. Circadian variation in biochemical markers of bone cell activity and insulin-like growth factor-I in two-year-old horses. J. Anim. Sci. 2003, 81, 2804–2810. [Google Scholar] [CrossRef]
- Stein, G.S.; Lian, J.B.; Owen, T.A. Relationship of cell growth to the regulation of tissue-specific gene expression during osteoblast differentiation. FASEB J. 1990, 4, 3111–3123. [Google Scholar] [CrossRef]
- Chubb, S.P. Measurement of C-terminal telopeptide of type I collagen (CTX) in serum. Clin. Biochem. 2012, 45, 928–935. [Google Scholar] [CrossRef]
- Rechkalov, A.V.; Gorshkova, N.E. Blood biochemical parameters in athletes after combined muscular exercise and food loading. Hum. Physiol. 2011, 37, 449–454. [Google Scholar] [CrossRef]
- Zhang, Y.; Xun, P.; Wang, R.; Mao, L.; He, K. Can magnesium enhance exercise performance? Nutrients 2017, 9, 946. [Google Scholar] [CrossRef]
- Price, J.; Jackson, B.; Eastell, R.; Wilson, A.; Russell, R.G.; Lanyon, L.; Goodship, A. The response of the skeleton to physical training: A biochemical study in horses. Bone 1995, 17, 221–227. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vrbanac, Z.; Brkljaca Bottegaro, N.; Skrlin, B.; Bojanic, K.; Kusec, V.; Stanin, D.; Belic, M. The Effect of a Moderate Exercise Program on Serum Markers of Bone Metabolism in Dogs. Animals 2020, 10, 1481. https://doi.org/10.3390/ani10091481
Vrbanac Z, Brkljaca Bottegaro N, Skrlin B, Bojanic K, Kusec V, Stanin D, Belic M. The Effect of a Moderate Exercise Program on Serum Markers of Bone Metabolism in Dogs. Animals. 2020; 10(9):1481. https://doi.org/10.3390/ani10091481
Chicago/Turabian StyleVrbanac, Zoran, Nika Brkljaca Bottegaro, Branimir Skrlin, Krunoslav Bojanic, Vesna Kusec, Damir Stanin, and Maja Belic. 2020. "The Effect of a Moderate Exercise Program on Serum Markers of Bone Metabolism in Dogs" Animals 10, no. 9: 1481. https://doi.org/10.3390/ani10091481
APA StyleVrbanac, Z., Brkljaca Bottegaro, N., Skrlin, B., Bojanic, K., Kusec, V., Stanin, D., & Belic, M. (2020). The Effect of a Moderate Exercise Program on Serum Markers of Bone Metabolism in Dogs. Animals, 10(9), 1481. https://doi.org/10.3390/ani10091481





