Comparison of Rabbit, Kitten and Mammal Milk Replacer Efficiencies in Early Weaning Rabbits
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals, Diets, Milk Replacer Preparation and Experimental Design
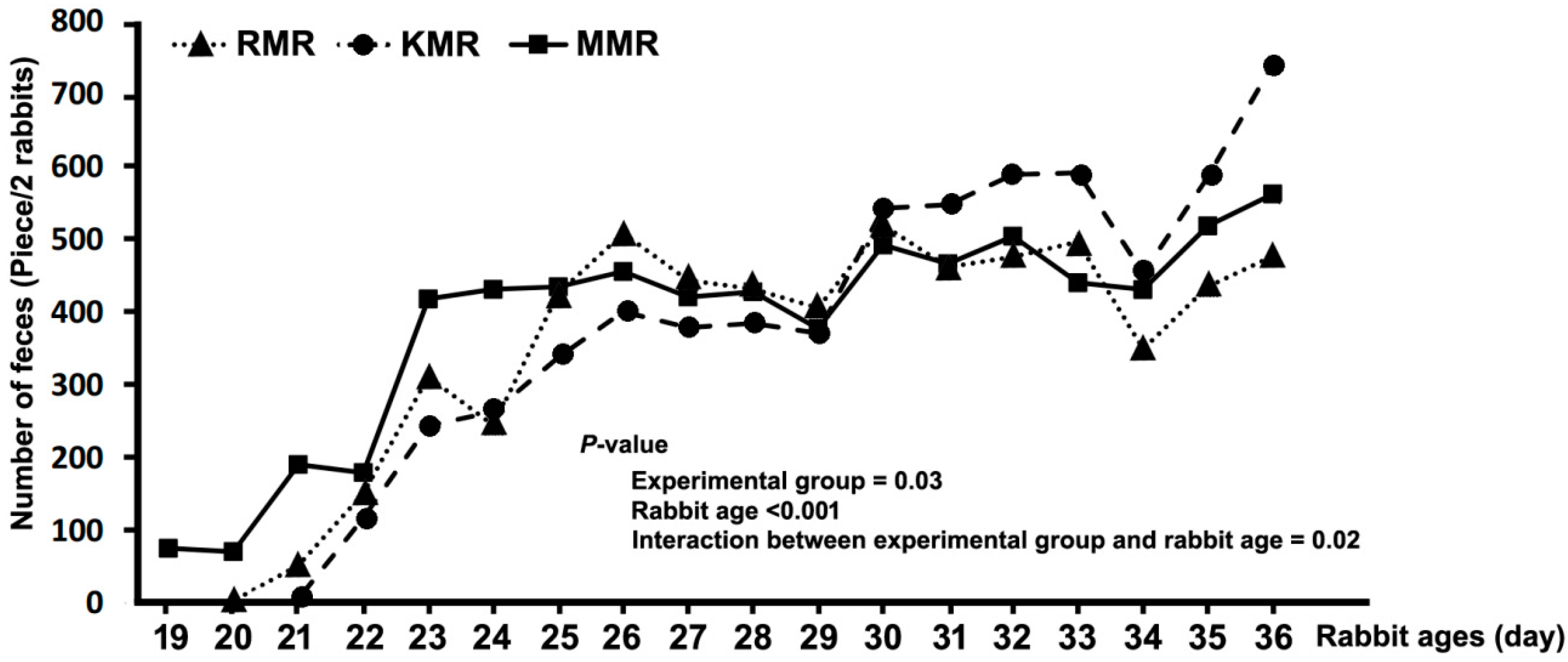
2.3. Perfomance, Digestibility and Faecal Evaluation
2.4. Internal Organs, Gut Histology and Caecal pH
2.5. Caecal Enzyme Activity
2.6. Crude Protein Assessment
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Artificial Milk Replacers | Age (days) | Number of Feces |
|---|---|---|
| RMR A | ||
| 19 | 0 a | |
| 20 | 0 a | |
| 21 | 6 ab | |
| 22 | 117 b | |
| 23 | 244 cd | |
| 24 | 263 c | |
| 25 | 338 def | |
| 26 | 401 efgh | |
| 27 | 379 defg | |
| 28 | 385 defg | |
| 29 | 371 cde | |
| 30 | 543 hi | |
| 31 | 548 fgh | |
| 32 | 590 hi | |
| 33 | 592 ghi | |
| 34 | 454 def | |
| 35 | 587 ghi | |
| 36 | 742 i | |
| KMR AB | ||
| 19 | 75.6 | |
| 20 | 70.8 | |
| 21 | 191 | |
| 22 | 180 | |
| 23 | 418 | |
| 24 | 432 | |
| 25 | 435 | |
| 26 | 456 | |
| 27 | 421 | |
| 28 | 428 | |
| 29 | 378 | |
| 30 | 493 | |
| 31 | 467 | |
| 32 | 505 | |
| 33 | 441 | |
| 34 | 431 | |
| 35 | 519 | |
| 36 | 563 | |
| MMR B | ||
| 19 | 0 | |
| 20 | 3.33 | |
| 21 | 52.3 | |
| 22 | 151 | |
| 23 | 311 | |
| 24 | 245 | |
| 25 | 426 | |
| 26 | 509 | |
| 27 | 444 | |
| 28 | 432 | |
| 29 | 406 | |
| 30 | 520 | |
| 31 | 463 | |
| 32 | 476 | |
| 33 | 497 | |
| 34 | 350 | |
| 35 | 436 | |
| 36 | 477 | |
| SEM | 12.02 | |
| p-Value | ||
| Groups | 0.03 | |
| Age | 0.001 | |
| Groups ∗ Age | 0.02 |
References
- Grand View Research. Pet Care Market Size, Share & Trends Analysis Report by Pet Type (Dog, Cat, Fish, Bird), by Products, Competitive Landscape, and Segment Forecasts, 2018–2025. Available online: https://www.grandviewresearch.com/industry-analysis/pet-care-market (accessed on 20 April 2020).
- Morley, C.; Fook, J. The importance of pet loss and some implications for services. Mortality 2005, 10, 127–143. [Google Scholar] [CrossRef]
- Rooney, N.J.; Blackwell, E.J.; Mullan, S.M.; Saunders, R.; Baker, P.E.; Hill, J.M.; Sealey, C.E.; Turner, M.J.; DE Held, S. The current state of welfare, housing and husbandry of the English pet rabbit population. BMC Res. Notes 2014, 7, 942–954. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, I.; Espinosa, A.; Gracía, J.; Carabano, R.; De Blas, J.C. Effect of levels of starch, fiber, and lactose on digestion and growth performance of early-weaned rabbits. J. Anim. Sci. 2002, 80, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Kovitvadhi, A.; Chandang, P.; Tirawattanawanich, C.; Kasemsuwan, S.; Rukkwamsuk, T. The study on cluster and obstacles of meat rabbit production in Thailand. J. Mahanakorn. Vet. Med. 2019, 14, 115–127. [Google Scholar]
- De Blas, C.; Wiseman, J. Nutrition of the Rabbit, 2nd ed.; CAB International: Oxfordshire, UK, 2010; pp. 1–19, 180–185, 229. [Google Scholar]
- Varga, M. Textbook of Rabbit Medicine, 2nd ed.; Butterworth Heinemann: London, UK, 2014; pp. 84–87. [Google Scholar]
- Paul, G.; Friend, D.G. Comparison of outcomes using two milk replacer formulars based on commercially available products in two species of infant cottontail rabbits. J. Wildl. Rehabil. 2017, 37, 13–19. [Google Scholar]
- Ferguson, F.A.; Lukefahr, S.D.; McNitt, J.L. A technical note on artificial milk feeding of rabbit kits weaned at 14 days. World Rabbit Sci. 1997, 5, 65–70. [Google Scholar] [CrossRef]
- Maertens, L.; Lebas, F.; Szendrö, Z.S. Rabbit milk: A review of quantity, quality and non-dietary affecting factors. World Rabbit Sci. 2006, 14, 205–230. [Google Scholar] [CrossRef]
- AVMA (American Veterinary Medical Association). AVMA Guidelines for the Euthanasia of Animals: 2020 Edition; American Veterinary Medical Association: Schaumburg, IL, USA, 2020; pp. 63–64. [Google Scholar]
- AOAC (Association of Official Analytical Chemists). Official Method of Analysis, 18th ed.; Association of Official Analytical Chemists: Rockville, MD, USA, 2006. [Google Scholar]
- AFFCO (Association of American Feed Control Officials). 2020 Official Publication; Association of American Feed Control Officials Inc.: Champaign, IL, USA, 2020. [Google Scholar]
- Kovitvadhi, A.; Gasco, L.; Ferrocino, I.; Rotolo, L.; Dabbou, S.; Malfatto, V.; Gai, F.; Peiretti, P.G.; Falzone, M.; Vignolini, C.; et al. Effect of purple loosestrife (Lythrum salicaria) diet supplementation in rabbit nutrition on performance, digestibility, health and meat quality. Animal 2016, 10, 10–18. [Google Scholar] [CrossRef]
- Areekijseree, M.; Engkagul, A.; Kovitvadhi, U.; Thongpan, A.; Mingmuang, M.; Pakkong, P.; Rungruangsak-Torrissen, K. Temperature and pH characteristics of amylase and proteinase of adult fresh water pearl mussel, Hyriopsis (Hyriopsis) bialatus, Simpson (1900). Aquaculture 2004, 234, 575–578. [Google Scholar] [CrossRef]
- Vatanparast, M.; Hosseininaveh, V.; Ghadamyari, M.; Minoo-Sajjadian, S. Plant cell wall degrading enzymes, pectinase and cellulase, in the digestive system of the red palm weevil, Rhynchophorus ferrugineus (Coleoptera: Curculionidae). Plant Prot. Sci. 2014, 50, 190–198. [Google Scholar] [CrossRef]
- Folin, O.; Ciocalteu, V. On tyrosine and tryptophan determinations in proteins. J. Biol. Chem. 1927, 73, 627–650. [Google Scholar]
- R: A Language and Environment for Statistical Computing. Available online: https://www.R-project.org (accessed on 19 March 2020).
- Combes, S.; Fortun-Lamothe, L.; Cauquil, L.; Gidenne, T. Engineering the rabbit digestive ecosystem to improve digestive health and efficacy. Animal 2013, 7, 1429–1439. [Google Scholar] [CrossRef] [PubMed]
- Falcão-e-Cunha, L.; Castro-Solla, L.; Maertens, L.; Marounek, M.; Pinheiro, V.; Freire, J.; Mourão, J.L. Alternatives to antibiotic growth promoters in rabbit feeding: A review. World Rabbit Sci. 2007, 15, 127–140. [Google Scholar] [CrossRef]
- Kinoshita, M.; Suzuki, Y.; Saito, Y. Butyrate reduces colonic paracellular permeability by enhancing PPARγ activation. Biochem. Biophys. Res. Commun. 2002, 293, 827–831. [Google Scholar] [CrossRef]
- Makovicky, P.; Tumova, E.; Volek, Z.; Makovicky, P.; Vodicka, P. Histological aspects of the small intestine under variable feed restriction: The effects of short and intense restriction on a growing rabbit model. Exp. Med. 2014, 8, 1623–1627. [Google Scholar] [CrossRef]
| Items | Artificial Milk Replacers | Rabbit Diet a | Rabbit Milk b | ||
|---|---|---|---|---|---|
| RMR | KMR | MMR | |||
| Ingredients (%) | |||||
| Sodium caseinate | 43.8 | - | - | - | - |
| Skimmed milk powder | 7.73 | - | - | - | - |
| Hydrogenated palm fat | 41.5 | - | - | - | - |
| Monodicalcium phosphate | 3.13 | - | - | - | - |
| Limestone | 2.30 | - | - | - | - |
| Salt | 0.94 | - | - | - | - |
| Premix c | 0.50 | - | - | - | - |
| Polyoxyethylene (80) sorbitan monooleate | 0.10 | - | - | - | - |
| Chemical composition | |||||
| Dry matter (%FM) | 95.8 | 96.2 | 95.2 | 90.7 | 29.8 |
| Crude ash (%DM) | 9.77 | 6.66 | 8.27 | 5.69 | 7.38 |
| Crude protein (%DM) | 41.4 | 48.5 | 30.1 | 16.1 | 41.3 |
| Ether extract (%DM) | 43.8 | 22.9 | 52.5 | 2.42 | 43.3 |
| Crude fiber (%DM) | ND | ND | ND | 25.2 | ND |
| Nitrogen free extract (%DM) d | 5.03 | 22.0 | 9.13 | 50.6 | 8.05 |
| Metabolizable energy content (kcal/100g DM) e | 535 | 441 | 584 | 254 | 540 |
| Parameters | Factors | SEM | p-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Artificial Milk Replacers (AMR) | Age (days) | AMR | Age | AMR * Age | |||||||
| RMR | KMR | MMR | 0 | 6 | 12 | 18 | |||||
| BW (g/head) | 210 | 198 | 202 | 134 a | 157 b | 214 c | 308 d | 9.086 | 0.38 | 0.001 | 0.94 |
| 19–24 | 25–30 | 31–36 | - | ||||||||
| ADFI (g/head/day) | 22.1 | 26.7 | 22.1 | 7.47 a | 22.1 b | 39.6 c | - | 2.239 | 0.59 | 0.001 | 0.56 |
| ADG (g/day) | 9.62 | 9.33 | 8.52 | 4.40 a | 10.8 b | 14.6 c | - | 0.808 | 0.84 | 0.001 | 0.77 |
| FCR | 2.39 | 2.62 | 2.74 | 1.91 a | 2.21 a | 2.75 b | - | 0.115 | 0.44 | 0.01 | 0.56 |
| Water intake (g/head/day) | 41.8 | 30.9 | 34.2 | 10.1 a | 31.9 b | 68.0 c | - | 3.902 | 0.1 | 0.001 | 0.38 |
| Faeces excretion (g/head/day) | 15 | 14.3 | 15.4 | 1.29 a | 5.49 b | 8.14 c | - | 0.936 | 0.81 | 0.001 | 0.32 |
| Crude protein intake (g/head/day) 1 | |||||||||||
| Diet | 3.72 | 3.73 | 3.9 | 1.20 a | 3.56 b | 6.37 c | - | 0.361 | 0.59 | 0.001 | 0.56 |
| Diet and milk | 5.95 | 6.34 | 5.52 | 3.39 a | 5.65 b | 8.56 c | - | 0.364 | 0.11 | 0.001 | 0.56 |
| 23–27 | 31–35 | - | - | ||||||||
| Apparent digestibility (%) | |||||||||||
| Dry matter | 59.6 | 63.3 | 63.5 | 60.5 | 63.4 | - | - | 1.126 | 0.29 | 0.22 | 0.96 |
| Organic matter | 60.7 | 65.1 | 65.2 | 62.8 | 64.2 | - | - | 1.123 | 0.18 | 0.51 | 0.85 |
| Ether extract | 68.3 | 73.8 | 70.7 | 71.3 | 70.3 | - | - | 2.418 | 0.75 | 0.88 | 0.97 |
| Crude protein | 76.5 | 80.8 | 81.1 | 81.5 b | 77.1 a | - | - | 1.007 | 0.07 | 0.03 | 0.34 |
| Parameters | Artificial Milk Replacers (AMR) | SEM | p-Value | ||
|---|---|---|---|---|---|
| RMR | KMR | MMR | |||
| Internal organs characteristics (g/live body weight) | |||||
| Liver | 4.46 | 4.00 | 3.81 | 0.162 | 0.25 |
| Spleen | 0.14 | 0.10 | 0.10 | 0.015 | 0.51 |
| Kidney | 1.24 | 1.18 | 1.12 | 0.046 | 0.61 |
| Thoracic organs 1 | 1.07 | 1.09 | 1.15 | 0.063 | 0.38 |
| Pancreas | 0.06 | 0.07 | 0.05 | 0.007 | 0.99 |
| Full stomach | 7.40 | 7.44 | 7.22 | 0.555 | 0.79 |
| Stomach wall | 2.09 | 2.01 | 1.99 | 0.061 | 0.65 |
| Intestinal organs 2 | 7.38 | 6.87 | 7.37 | 0.242 | 0.54 |
| Full caecum | 16.3 | 16.1 | 16.2 | 0.422 | 0.97 |
| Caecal wall | 2.75 | 2.49 | 2.34 | 0.134 | 0.48 |
| Caecal pH | 6.50 | 6.30 | 6.38 | 0.081 | 0.63 |
| Caecal digestive enzyme activities (U) | |||||
| Amylase | 12.2 ab | 18.5 b | 10.4 a | 1.41 | 0.04 |
| Protease (×10−1) | 7.26 b | 6.84 ab | 5.79 a | 0.253 | 0.04 |
| Cellulase | 3.71 | 3.50 | 3.60 | 0.057 | 0.31 |
| Duodenal villi morphology (µm) | |||||
| Villus height | 320 a | 361 ab | 380 b | 8.50 | 0.04 |
| Villus crypt | 66.2 ab | 62.6 a | 68.4 b | 0.737 | 0.007 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chankuang, P.; Linlawan, A.; Junda, K.; Kuditthalerd, C.; Suwanprateep, T.; Kovitvadhi, A.; Chundang, P.; Sanyathitiseree, P.; Yinharnmingmongkol, C. Comparison of Rabbit, Kitten and Mammal Milk Replacer Efficiencies in Early Weaning Rabbits. Animals 2020, 10, 1087. https://doi.org/10.3390/ani10061087
Chankuang P, Linlawan A, Junda K, Kuditthalerd C, Suwanprateep T, Kovitvadhi A, Chundang P, Sanyathitiseree P, Yinharnmingmongkol C. Comparison of Rabbit, Kitten and Mammal Milk Replacer Efficiencies in Early Weaning Rabbits. Animals. 2020; 10(6):1087. https://doi.org/10.3390/ani10061087
Chicago/Turabian StyleChankuang, Panthiphaporn, Achira Linlawan, Kawisara Junda, Chittikan Kuditthalerd, Tuksaorn Suwanprateep, Attawit Kovitvadhi, Pipatpong Chundang, Pornchai Sanyathitiseree, and Chaowaphan Yinharnmingmongkol. 2020. "Comparison of Rabbit, Kitten and Mammal Milk Replacer Efficiencies in Early Weaning Rabbits" Animals 10, no. 6: 1087. https://doi.org/10.3390/ani10061087
APA StyleChankuang, P., Linlawan, A., Junda, K., Kuditthalerd, C., Suwanprateep, T., Kovitvadhi, A., Chundang, P., Sanyathitiseree, P., & Yinharnmingmongkol, C. (2020). Comparison of Rabbit, Kitten and Mammal Milk Replacer Efficiencies in Early Weaning Rabbits. Animals, 10(6), 1087. https://doi.org/10.3390/ani10061087







