The Effect of Behind-The-Scenes Encounters and Interactive Presentations on the Welfare of Captive Servals (Leptailurus serval)
Simple Summary
Abstract
1. Introduction
- Encounter frequency,
- Type of encounter (behind-the-scenes encounter involving a small number of visitors and close visitor–animal proximity, versus an interactive presentation with higher numbers of visitors but lower visitor–animal proximity).
2. Materials and Methods
2.1. Study Animals—Housing and Husbandry Routine
2.2. Visitor Interaction Program
2.3. Experimental Design
2.4. Behavioural Observations
2.5. Adrenocortical Activity
2.6. Statistical Analysis
3. Results
3.1. Passive, Active, and Maintenance Behaviours
3.2. Abnormal Repetitive Behaviours
3.3. Behavioural Diversity
3.4. Effect of Individual and Time of Day on Behaviour
3.5. Adrenocortical Activity
4. Discussion
4.1. Behavioural Measures
4.2. Adrenocortical Activity
4.3. Other Influences
5. Conclusions
- The current study found no evidence of a negative welfare impact in relation to participation in interactive presentations or behind-the-scenes encounters, at either a behavioural or physiological level. In fact, participation in these activities was shown to significantly reduce the incidence of pacing, suggesting that visitor interaction may contribute to positive welfare for the servals.
- A significant reduction in behavioural diversity was observed on weeks when cats participated in both presentations and BTS. Although not considered a negative welfare impact in the short term, one cannot disregard potential long-term negative effects of a more intense activity schedule, hence careful monitoring would be advised if the level of interaction was to be increased on a more permanent basis.
- It could not be determined whether visitor interaction per se or some other factors (i.e., food rewards, cognitive stimulation, etc.) was responsible for the effects observed, but for management purposes, the current findings are sufficient to lend support to the continued involvement of the servals in the encounter program, at the level of interaction that the cats normally experience.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Herrmann, E.; Kamler, J.F.; Avenant, N.L. New records of servals Leptailurus serval in central South Africa. S. Afr. J. Wildl. Res. 2008, 38, 185–188. [Google Scholar] [CrossRef]
- Perrin, M.R. Space use by a reintroduced serval in Mount Currie Nature Reserve. S. Afr. J. Wildl. Res. 2002, 32, 79–86. [Google Scholar]
- Ramesh, T.; Downs, C.T. Impact of land use on occupancy and abundance of terrestrial mammals in the Drakensberg Midlands, South Africa. J. Nat. Conserv. 2015, 23, 9–18. [Google Scholar] [CrossRef]
- Smithers, R.H.N. The serval Felis serval Schreber, 1776. S. Afr. J. Wildl. Res. 1978, 8, 29–37. [Google Scholar]
- IUCN 2018. IUCN Red List of Threatened Species, Version 2018.1. Available online: www.iucnredlist.org (accessed on 13 February 2019).
- Foley, C.; Foley, L.; Lobora, A.; De Luca, D.; Msuha, M.; Davenport, T.R.B.; Durant, S. A Field Guide to the Larger Mammals of Tanzania, Carnivores: Carnivora; Princeton University Press: Princeton, NJ, USA, 2008. [Google Scholar]
- Sunquist, M.; Sunquist, F. Wild Cats of the World; The University of Chicago Press: Chicago, IL, USA, 2001. [Google Scholar]
- Mellen, J.D. Factors influencing reproductive success in small captive exotic felids (Felis spp.): A multiple regression analysis. Zoo Biol. 1991, 10, 95–110. [Google Scholar] [CrossRef]
- Brodie, J.F. Is research effort allocated efficiently for conservation? Felidae as a global case study. Biodivers. Conserv. 2009, 18, 2927–2939. [Google Scholar] [CrossRef]
- Ripple, W.J.; Estes, J.A.; Beschta, R.L.; Wilmers, C.C.; Ritchie, E.G.; Hebblewhite, M.; Berger, J.; Elmhagen, B.; Letnic, M.; Nelson, M.P.; et al. Status and ecological effects of the world’s largest carnivores. Science 2014, 343, 1241484. [Google Scholar] [CrossRef]
- Bashaw, M.J.; Bloomsmith, M.A.; Marr, M.J.; Maple, T.L. To hunt or not to hunt? A feeding enrichment experiment with captive large felids. Zoo Biol. 2003, 22, 189–198. [Google Scholar] [CrossRef]
- McPhee, M.E. Intact carcasses as enrichment for large felids: Effects on on- and off-exhibit behaviours. Zoo Biol. 2002, 21, 37–47. [Google Scholar] [CrossRef]
- Ruskell, A.D.; Meiers, S.T.; Jenkins, S.E.; Santymire, R.M. Effect of bungee-carcass enrichment on behaviour and faecal glucocorticoid metabolites in two species of zoo-housed felids. Zoo Biol. 2015, 34, 170–177. [Google Scholar] [CrossRef]
- Skibiel, A.L.; Trevino, H.S.; Naugher, K. Comparison of several types of enrichment for captive felids. Zoo Biol. 2007, 26, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Breton, G.; Barrot, S. Influence of enclosure size on the distances covered and paced by captive tigers (Panthera tigris). Appl. Anim. Behav. Sci. 2014, 154, 66–75. [Google Scholar] [CrossRef]
- Lyons, J.; Young, R.J.; Deag, J.M. The effects of physical characteristics of the environment and feeding regime on the behaviour of captive felids. Zoo Biol. 1997, 16, 71–83. [Google Scholar] [CrossRef]
- De Rouck, M.; Kitchener, A.C.; Law, G.; Nelissen, M. A comparative study of the influence of social housing conditions on the behaviour of captive tigers (Panthera tigris). Anim. Welf. 2005, 14, 229–238. [Google Scholar]
- Miller, L.J.; Bettinger, T.; Mellen, J. The reduction of stereotypic pacing in tigers (Panthera tigris) by obstructing the view of neighbouring individuals. Anim. Welf. 2008, 17, 255–258. [Google Scholar]
- Resende, L.S.; Remy, G.L.; de Almeida Ramos, V., Jr.; Andriolo, A. The influence of feeding enrichment on the behaviour of small felids (Carnivora: Felidae) in captivity. Zoologia 2009, 26, 601–605. [Google Scholar] [CrossRef]
- Moreira, N.; Brown, J.L.; Moraes, W.; Swanson, W.F.; Monteiro-Filho, E.L.A. Effect of housing and environmental enrichment on adrenocortical activity, behaviour and reproductive cyclicity in the female tigrina (Leopardus tigrinus) and margay (Leopardus wiedii). Zoo Biol. 2007, 26, 441–460. [Google Scholar] [CrossRef]
- Wells, D.L.; Egli, J.M. The influence of olfactory enrichment on the behaviour of captive black-footed cats, Felis nigripes. Appl. Anim. Behav. Sci. 2004, 85, 107–119. [Google Scholar] [CrossRef]
- Hosey, G.R. Zoo animals and their human audiences: What is the visitor effect? Anim. Welf. 2000, 9, 343–357. [Google Scholar]
- Szokalski, M.S.; Foster, W.K.; Litchfield, C.A. Behavioural monitoring of big cats involved in ‘behind-the-scenes’ zoo visitor tours. Int. J. Comp. Psychol. 2013, 26, 83–104. [Google Scholar]
- Acaralp-Rehnberg, L.K. Human-Animal Interaction in the Modern Zoo: Live Animal Encounter Programs and Associated Effects on Animal Welfare. Ph.D. Thesis, The University of Melbourne, Melbourne, Australia, 2019. [Google Scholar]
- Narayan, E.J.; Parnell, T.; Clark, G.; Martin-Vegue, P.; Mucci, A.; Hero, J.-M. Faecal cortisol metabolites in Bengal (Panthera tigris tigris) and Sumatran tigers (Panthera tigris sumatrae). Gen. Comp. Endocr. 2013, 194, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Wolfensohn, S.; Shotton, J.; Bowley, H.; Davies, S.; Thompson, S.; Justice, W.S.M. Assessment of welfare in zoo animals: Towards optimum quality of life. Animals 2008, 8, 110. [Google Scholar] [CrossRef] [PubMed]
- Wielebnowski, N.; Watters, J. Applying faecal endocrine monitoring to conservation and behaviour studies of wild mammals: Important considerations and preliminary tests. Isr. J. Ecol. Evol. 2007, 53, 439–460. [Google Scholar] [CrossRef]
- Mellor, D.J.; Beausoleil, N.J. Extending the ‘Five Domains’ model for animal welfare assessment to incorporate positive welfare states. Anim. Welf. 2015, 24, 241–253. [Google Scholar] [CrossRef]
- Boissy, A.; Manteuffel, G.; Bak Jensen, M.; Oppermann Moe, R.; Spruijt, B.; Keeling, L.J.; Winckler, C.; Forkman, B.; Dimitrov, I.; Langbein, J.; et al. Assessment of positive emotions in animals to improve their welfare. Physiol. Behav. 2007, 92, 375–397. [Google Scholar] [CrossRef] [PubMed]
- Altmann, J. Observational study of behaviour: Sampling methods. Behaviour 1974, 49, 227–267. [Google Scholar] [CrossRef]
- Stanton, L.A.; Sullivan, M.S.; Fazio, J.M. A standardized ethogram for the Felidae: A tool for behavioural researchers. Appl. Anim. Behav. Sci. 2015, 173, 3–16. [Google Scholar] [CrossRef]
- Keay, J.M.; Singh, J.; Gaunt, M.C.; Kaur, T. Faecal glucocorticoids and their metabolites as indicators of stress in various mammalian species: A literature review. J. Zoo Wildl. Med. 2006, 37, 234–244. [Google Scholar] [CrossRef]
- Graham, L.H.; Brown, J.L. Cortisol metabolism in the domestic cat and implications for non-invasive monitoring of adrenocortical function in endangered felids. Zoo Biol. 1996, 15, 71–82. [Google Scholar] [CrossRef]
- Wasser, S.K.; Hunt, K.E.; Brown, J.L.; Cooper, K.; Crockett, C.M.; Bechert, U.; Millspaugh, J.J.; Larson, S.; Monfort, S.L. A generalized faecal glucocorticoid assay for use in a diverse array of nondomestic mammalian and avian species. Gen. Comp. Endocr. 2000, 120, 260–275. [Google Scholar] [CrossRef]
- Bashaw, M.J.; Sicks, F.; Palme, R.; Schwarzenberger, F.; Tordiffe, A.S.W.; Ganswindt, A. Non-invasive assessment of adrenocortical activity as a measure of stress in giraffe (Giraffa camelopardalis). BMC Vet. Res. 2016, 12. [Google Scholar] [CrossRef] [PubMed]
- Fanson, K.V.; Wielebnowski, N.C.; Shenk, T.M.; Lucas, J.R. Comparative patterns of adrenal activity in captive and wild Canada lynx (Lynx canadensis). J. Comp. Physiol. Behav. 2012, 182, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.; Munro, C.; Edwards, K.L.; Norton, V.; Brown, J.L.; Walker, S.L. Development of a versatile enzyme immunoassay for non-invasive assessment of glucocorticoid metabolites in a diversity of taxonomic species. Gen. Comp. Endocr. 2013, 186, 16–24. [Google Scholar] [CrossRef]
- Hobbs, R.; (Taronga Western Plains Zoo, Dubbo, New South Wales, Australia). Personal communication, 2017.
- Markowitz, H.; LaForse, S. Artificial prey as behavioural enrichment devices for felines. Appl. Anim. Behav. Sci. 1987, 18, 31–43. [Google Scholar] [CrossRef]
- De Souza Resende, L.; Lima e Neto, G.; Gonçalves Duarte Carvalho, P.; Landau-Remy, G.; de Almeida Ramos-Júnior, V.; Andriolo, A.; Genaro, G. Time budget and activity patterns of oncilla cats (Leopardus tigrinus) in captivity. JAAWS 2014, 17, 73–81. [Google Scholar]
- Shepherdson, D.J.; Carlstead, K.; Mellen, J.D.; Seidensticker, J. The influence of food presentation on the behaviour of small cats in confined environments. Zoo Biol. 1993, 12, 203–216. [Google Scholar] [CrossRef]
- Weller, S.H.; Bennett, C.L. Twenty-four hour activity budgets and patterns of behaviour in captive ocelots (Leopardus pardalis). Appl. Anim. Behav. Sci. 2001, 71, 67–79. [Google Scholar] [CrossRef]
- Bashaw, M.J.; Kelling, A.S.; Bloomsmith, M.A.; Maple, T.L. Environmental effects on the behaviour of zoo-housed lions and tigers, with a case study of the effects of a visual barrier on pacing. JAAWS 2007, 10, 95–109. [Google Scholar]
- Macri, A.M.; Patterson-Kane, E. Behavioural analysis of solitary versus socially housed snow leopards (Panthera uncia), with the provision of simulated social contact. Appl. Anim. Behav. Sci. 2011, 130, 115–123. [Google Scholar] [CrossRef]
- Parker, M.; Lamoureux, S.; Allouche, B.; Brossier, J.-A.; Weber, M.; Feugier, A.; Moniot, D.; Deputte, B.; Biourge, V.; Serra, J. Accuracy assessment of spatial organization and activity of indoor cats using a system based on ultrawide band technology. J. Vet. Behav. 2017, 21, 13–19. [Google Scholar] [CrossRef]
- Carlstead, K.; Brown, J.L.; Seidensticker, J. Behavioural and adrenocortical responses to environmental changes in leopard cats (Felis bengalensis). Zoo Biol. 1993, 12, 321–331. [Google Scholar] [CrossRef]
- Rehnberg, L.K.; Robert, K.A.; Watson, S.; Peters, R.A. The effects of social interaction and environmental enrichment on the space use, behaviour and stress of owned housecats facing a novel environment. Appl. Anim. Behav. Sci. 2015, 169, 51–61. [Google Scholar] [CrossRef]
- Kry, K.; Casey, R. The effect of hiding enrichment on stress levels and behaviour of domestic cats (Felis sylvestris catus) in a shelter setting and the implications for adoption potential. Anim. Welf. 2007, 16, 375–383. [Google Scholar]
- Rochlitz, I. Recommendations for the housing and care of domestic cats in laboratories. Lab. Anim. 2000, 34, 1–9. [Google Scholar] [CrossRef]
- Clubb, R.; Mason, G.J. Natural behavioural biology as a risk factor in carnivore welfare: How analysing species differences could help zoos improve enclosures. Appl. Anim. Behav. Sci. 2007, 102, 303–328. [Google Scholar] [CrossRef]
- Clubb, R.; Vickery, S.S. Locomotory stereotypies in carnivores: Does pacing stem from hunting, ranging or frustrated escape? In Stereotypies in Captive Animals: Fundamentals and Applications for Welfare, 2nd ed.; Mason, G., Rushen, J., Eds.; CABI Publishing: Wallingford, UK, 2006. [Google Scholar]
- Livingston, S.E. The nutrition and natural history of the serval (Felis serval) and caracal (Caracal caracal). Vet. Clin. N. Am. Exot. Anim. Pract. 2009, 12, 327–334. [Google Scholar] [CrossRef]
- Mason, G.; Clubb, R.; Latham, N.; Vickery, S. Why and how should we use environmental enrichment to tackle stereotypic behaviour? Appl. Anim. Behav. Sci. 2007, 102, 163–188. [Google Scholar] [CrossRef]
- Spruijt, B.M.; Van Den Bos, R.; Pijlman, F.T. A concept of welfare based on reward evaluating mechanisms in the brain: Anticipatory behaviour as an indicator for the state of reward systems. Appl. Anim. Behav. Sci. 2001, 72, 145–171. [Google Scholar] [CrossRef]
- Watters, J.V. Searching for behavioural indicators of welfare in zoos: Uncovering anticipatory behaviour. Zoo Biol. 2014, 33, 251–256. [Google Scholar] [CrossRef]
- Broom, D.M. Sentience and Animal Welfare; CABI Publishing: Wallingford, UK, 2014. [Google Scholar]
- Broom, D.M. The scientific assessment of Animal Welfare. Appl. Anim. Behav. Sci. 1988, 20, 5–19. [Google Scholar] [CrossRef]
- Quirke, T.; O’Riordan, R.M. The effect of a randomised enrichment treatment schedule on the behaviour of cheetahs (Acinonyx jubatus). Appl. Anim. Behav. Sci. 2011, 135, 103–109. [Google Scholar] [CrossRef]
- Wielebnowski, N.C.; Fletchall, N.; Carlstead, K.; Busso, J.M.; Brown, J.L. Non-invasive assessment of adrenal activity associated with husbandry and behavioural factors in the North American clouded leopard population. Zoo Biol. 2002, 21, 77–98. [Google Scholar] [CrossRef]
- Wielebnowski, N.C. Stress and distress: Evaluating their impact for the well-being of zoo animals. JAVMA 2003, 223, 973–977. [Google Scholar] [CrossRef] [PubMed]
- Whitham, J.C.; Wielebnowski, N.C. New directions for zoo Anim. Welf. science. Appl. Anim. Behav. Sci. 2013, 147, 247–260. [Google Scholar] [CrossRef]
- Benn, A.L.; McLelland, D.J.; Whittaker, A.L. A review of welfare assessment methods in reptiles, and preliminary application of the welfare quality protocol to the pygmy blue-tongue skink, Tiliqua adelaidensis, using animal-based measures. Animals 2019, 9, 27. [Google Scholar] [CrossRef]
- Carlstead, K. A comparative approach to the study of keeper-animal relationships in the zoo. Zoo Biol. 2009, 28, 589–608. [Google Scholar] [CrossRef]
- Woolway, E.E.; Goodenough, A.E. Effects of visitor numbers on captive European red squirrels (Sciurus vulgaris) and impacts on visitor experience. Zoo Biol. 2017, 36, 112–119. [Google Scholar] [CrossRef]
- Forkman, B.A. Some problems with the current patch choice theory: A study on the Mongolian gerbil. Behaviour 1991, 117, 243–254. [Google Scholar] [CrossRef]
- Forkman, B.A. Self-reinforced behaviour does not explain contra-freeloading in the Mongolian gerbil. Ethology 1993, 94, 109–112. [Google Scholar] [CrossRef]
- Duncan, I.J.H.; Hughes, B.O. Free and operant feeding in domestic fowls. Anim. Behav. 1972, 20, 775–777. [Google Scholar] [CrossRef]
- Neuringer, A.J. Animals respond for food in the presence of free food. Science 1969, 166, 399–401. [Google Scholar] [CrossRef] [PubMed]
- Meehan, C.L.; Mench, J.A. The challenge of challenge: Can problem-solving opportunities enhance Anim. Welfare? Appl. Anim. Behav. Sci. 2007, 102, 246–261. [Google Scholar] [CrossRef]
- Edwards, C.; Heiblum, M.; Tejeda, A.; Galindo, F. Experimental evaluation of attachment behaviours in owned cats. J. Vet. Behav. 2007, 2, 119–125. [Google Scholar] [CrossRef]
- Bonato, M.; Malecki, I.A.; Wang, M.D.; Cloete, S.W.P. Extensive human presence at an early age of ostriches improves the docility of birds at a later stage of life. Appl. Anim. Behav. Sci. 2013, 148, 232–239. [Google Scholar] [CrossRef]
- Claxton, A.M. The potential of the human-animal relationship as an environmental enrichment for the welfare of zoo-housed animals. Appl. Anim. Behav. Sci. 2011, 133, 1–10. [Google Scholar] [CrossRef]
- Hosey, G.R. A preliminary model of human-animal relationships in the zoo. Appl. Anim. Behav. Sci. 2008, 109, 105–127. [Google Scholar] [CrossRef]

| Treatment | Presentations | Behind-the-Scenes (BTS) |
|---|---|---|
| 1—Presentation. | Yes | No |
| 2—BTS | No | Yes |
| 3—Presentation. + BTS | Yes | Yes |
| 4—No interaction | No | No |
| Session | Block | Time of Day | Duration | Total Observation Time Per Animal |
|---|---|---|---|---|
| Morning | 1 | 9:00–9:30 | 30 min | 30 min for both cats |
| Presentation | 1 | 10:45–11:30 * | 45 min | 45 min for the non-participating cat, 30 min for the participating cat |
| BTS | 2 | 13:15–14:15 ** | 60 min | 45 min for the non-participating cat, 30 min for the participating cat |
| Afternoon | 2 | 16:00–16:30 | 30 min | 30 min for both cats |
| Passive Behaviours | Active Behaviours | Maintenance Behaviours | Abnormal Repetitive Behaviours |
|---|---|---|---|
| Sitting: Cat is in an upright position, with the hind legs flexed and resting on the ground, while front legs are extended and straight, or crouching on top of all fours. Resting awake: Cat is lying down with its head raised and eyes open. Sleeping: Cat is lying down with its head down and eyes closed, performing minimal head or leg movement, and is not easily disturbed. Standing: Cat is in an upright position and immobile, with all four paws on the ground and legs extended, supporting the body. Hidden: Cat is fully concealed behind dense vegetation or burlap drape, to the point where it cannot be determined confidently which other behaviour(s) it is currently engaged in. | Walking: Forward locomotion at a slow gait. Running: Forward locomotion in a rapid gait, which is faster than walking or trotting. Investigate: Cat moves around attentively while sniffing the ground and/or objects or shows attention towards a specific stimulus by sniffing and/or pawing at it. Climbing: Cat ascends and/or descends an object or structure. Jumping: Cat leaps from one point to another, either vertically or horizontally. Playing: Cat interacts with and manipulates an object in a “playful” manner. | Eat: Cat ingests food (or other edible substances) by means of chewing with the teeth and swallowing. Groom: Cat cleans itself by licking, scratching, biting or chewing the fur on its body. May also include the licking of a front paw and wiping it over one’s head. Defecate: Cat releases faeces on the ground while in a squatting position. Urinate: Cat releases urine on the ground while in a squatting position. Clawing: Cat drags front claws along an object or surface. Scratching: Cat scratches its body using the claws of its hind feet. Stretching: Cat extends its forelegs while curving its back inwards. Scent mark: While standing with tail raised vertically, cat releases a jet of urine backwards against a vertical surface or object. The tail may quiver as urine is discharged. Yawn: Cat opens its mouth widely while inhaling, then closes mouth while exhaling deeply. | Pacing: Cat walks or runs back and forth in a repetitive manner along a designated path, without obvious purpose or intention. The cat had to traverse the same path at least twice to be considered pacing. |
| Presentations | BTS | Presentations + BTS | No Inter-Action | Standard Error of Difference | p-Value | |
|---|---|---|---|---|---|---|
| Passive behaviours * | ||||||
| Total inactivity | 10.58 | 9.88 | 10.17 | 11.38 | 0.75 | ns |
| Sitting | 5.20 bc | 3.74 ab | 4.58 b | 5.02 bc | 0.53 | 0.05 |
| Standing | 1.75 | 2.22 | 1.55 | 1.99 | 0.25 | 0.06 |
| Resting awake | 1.97 | 2.78 | 2.46 | 2.65 | 0.60 | ns |
| Sleeping | 0.40 | 0.02 | 0.15 | 0.39 | 0.19 | ns |
| Hidden | 1.26 | 1.11 | 1.43 | 1.33 | 0.59 | ns |
| Active behaviours * | ||||||
| Total activity | 1.80 b | 2.22 ab | 1.45 bc | 1.84 b | 0.25 | 0.04 |
| Walking | 1.28 b | 1.51 ab | 0.96 bc | 1.40 ab | 0.18 | 0.03 |
| Investigating | 0.31 | 0.33 | 0.21 | 0.31 | 0.08 | ns |
| Playing | 0.06 | 0.11 | 0.12 | 0.02 | 0.07 | ns |
| Other active (running, climbing, jumping) | 0.16 a | 0.26 b | 0.16 a | 0.11 a | 0.04 | 0.01 |
| Maintenance Behaviours * | ||||||
| Total maintenance | 1.19 | 1.28 | 0.78 | 1.18 | 0.33 | ns |
| Eating | 0.74 | 0.85 | 0.57 | 0.72 | 0.29 | ns |
| Grooming | 0.25 | 0.26 | 0.14 | 0.33 | 0.07 | 0.06 |
| Scent marking | 0.13 ab | 0.06 b | 0.03 bc | 0.03 bc | 0.04 | 0.04 |
| Other maintenance (urinating, defecating, clawing, scratching, stretching, yawning) | 0.07 | 0.11 | 0.04 | 0.10 | 0.03 | ns |
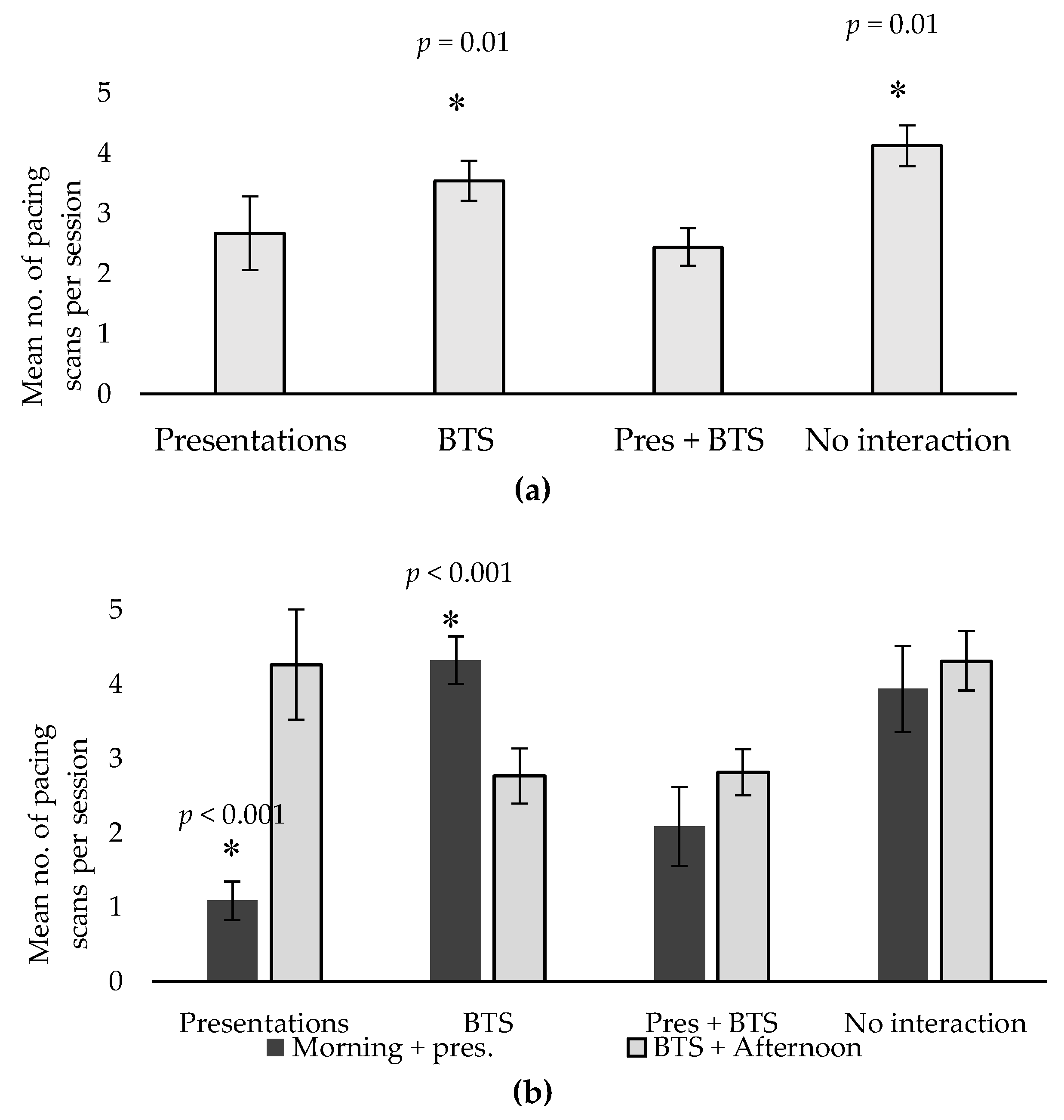
| Pacing * | 2.67 a | 3.54 b | 2.44 a | 4.12 b | 0.45 | 0.01 |
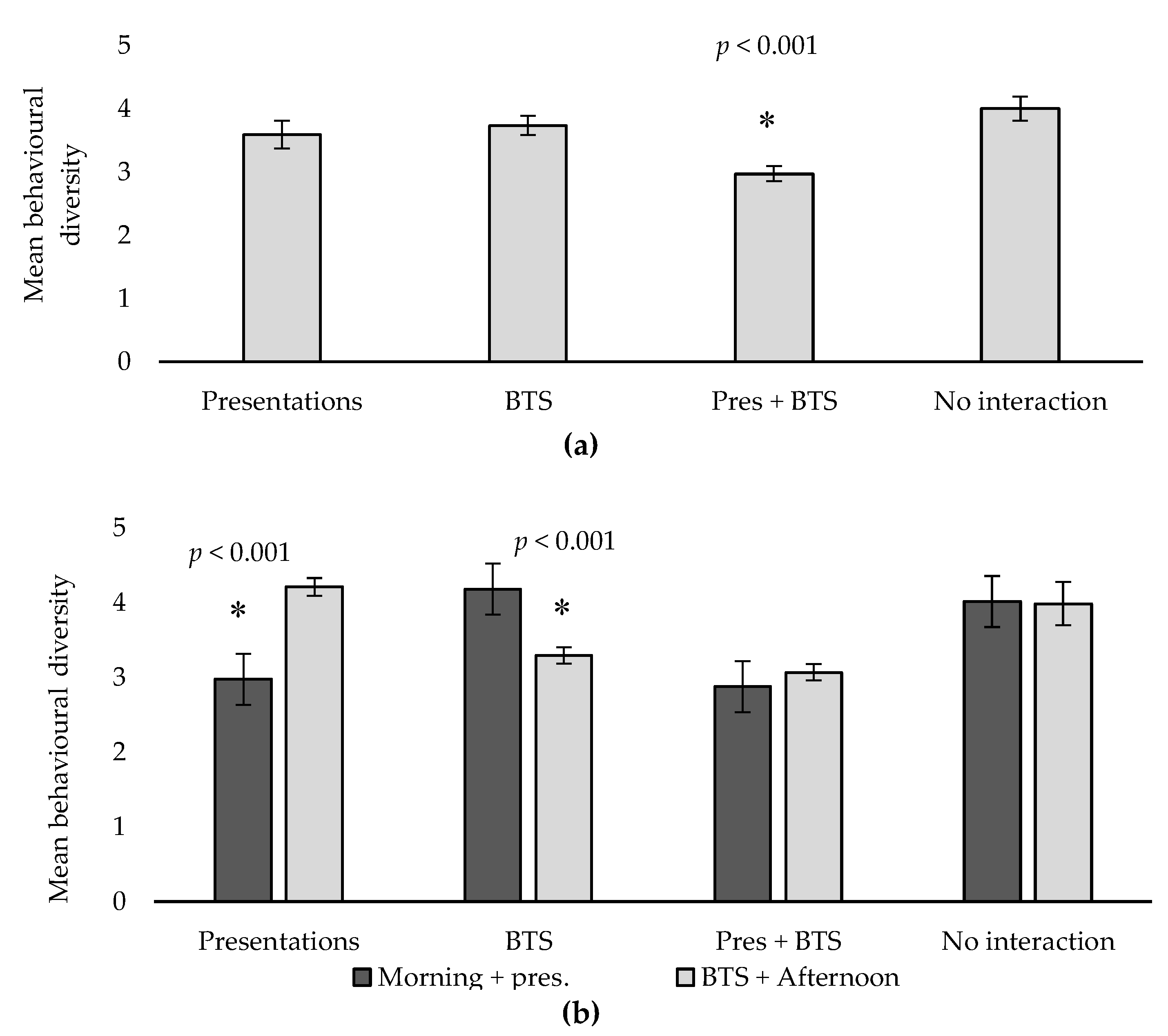
| Behavioural diversity ** | 3.59 a | 3.74 a | 2.97 b | 4.00 a | 0.20 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acaralp-Rehnberg, L.K.; Coleman, G.J.; Magrath, M.J.L.; Melfi, V.; Fanson, K.V.; Bland, I.M. The Effect of Behind-The-Scenes Encounters and Interactive Presentations on the Welfare of Captive Servals (Leptailurus serval). Animals 2020, 10, 743. https://doi.org/10.3390/ani10040743
Acaralp-Rehnberg LK, Coleman GJ, Magrath MJL, Melfi V, Fanson KV, Bland IM. The Effect of Behind-The-Scenes Encounters and Interactive Presentations on the Welfare of Captive Servals (Leptailurus serval). Animals. 2020; 10(4):743. https://doi.org/10.3390/ani10040743
Chicago/Turabian StyleAcaralp-Rehnberg, Lydia K., Grahame J. Coleman, Michael J. L. Magrath, Vicky Melfi, Kerry V. Fanson, and Ian M. Bland. 2020. "The Effect of Behind-The-Scenes Encounters and Interactive Presentations on the Welfare of Captive Servals (Leptailurus serval)" Animals 10, no. 4: 743. https://doi.org/10.3390/ani10040743
APA StyleAcaralp-Rehnberg, L. K., Coleman, G. J., Magrath, M. J. L., Melfi, V., Fanson, K. V., & Bland, I. M. (2020). The Effect of Behind-The-Scenes Encounters and Interactive Presentations on the Welfare of Captive Servals (Leptailurus serval). Animals, 10(4), 743. https://doi.org/10.3390/ani10040743





