Mouth Pain in Horses: Physiological Foundations, Behavioural Indices, Welfare Implications, and a Suggested Solution
Simple Summary
Abstract
1. Introduction
2. General Features of Pain
2.1. Major Features of Pain Generation and Experience
2.2. Cerebrocortical Involvement in the Conscious Experience of Pain in Mammals
2.3. Terrestrial Mammals Can Have Numerous Other Subjective Experiences in Addition to Pain
2.4. Adjunct Physiological Responses to Pain-Inducing Stimuli
2.5. General Behavioural Responses to Pain
“There is good evidence that facial expression can be a useful, valid and reliable tool for recognising and evaluating pain in humans and other animals. Both the sensory and emotional components of pain have been demonstrated to affect facial expression, which thus gives a true representation of the affective state of the animal. Many of the mammalian species studied to date have similar facial expression responses to pain.”[15]
2.6. Summing up
3. Sensations Generated within the Mouth
3.1. Tissues Supplied by the Trigeminal Nerve
3.2. Tissues Supplied by the Facial Nerve
4. Mouth Pain in Horses
4.1. Bit-Induced Nociceptor Stimulation and Pain
4.1.1. The Interdental Space
- (1).
- The established relationship between tension (T, units N), mass (m, units kg) and gravitational acceleration (g = 9.8 metres/sec2), which is “T = mg” or “T = 9.8 m” [109].
- (2).
- (3).
- An estimated area of bit–gum contact on the interdental space (CAbg) of 0.387 cm2 [2], which is equivalent to a 6.22 × 6.22 mm square.
4.1.2. The Tongue
4.1.3. The Commissures of the Lips and the Buccal Mucosa
4.2. General Comments and Summing up
5. Behavioural Indices of Mouth Pain in Horses
6. Welfare Implications of Bit-Induced Mouth Pain in Horses
6.1. Respiratory Consequences of an Open Mouth and Relocation of the Tongue above or behind the Bit
6.2. Respiratory Impacts of Low Jowl Angles Maintained by the Firm Application of Rein Tension
6.3. Pain-Related Conflict Behaviours and Summing up
7. A Suggested Solution
- What specific competitive events have been tested for control and safety using bit-free bridles?It appears that no data for comparing the responses of horses ridden bitted or bit-free have been collected, although opportunities for doing so are available with dressage. Bit-free dressage, excluding Grand Prix, is supported in the Netherlands [184], and two online American Western dressage associations give riders the choice of competing using bitted or bit-free bridles [185,186]. In addition, it would be worthwhile to set up other studies that compare the safety and control of horses ridden bitted and bit-free in other competitive events, including, but not limited to, flatraces, steeplechases, and harness races. However, the requirement that bits must be worn during such racing events means that off-track trials would probably be needed.
- Have horses trained from the outset to be ridden bit-free been tested in competition for their ease of control and whether or not they can be ridden safely?Not yet, but sufficient time has passed since bit-free riding became more popular for there to be many young horses that would be eligible for such testing.
- Can horses trained from the outset to be ridden with bitted bridles be successfully transitioned to being ridden safely bit-free?Yes, many years of successful transitioning of horses for recreational riding attest to this, as do a number of published studies (Table 2). However, additional studies would be beneficial.
- If so, what proportion of horses are not able to be transitioned to bit-free riding?Evidence is lacking on the proportion of horses that cannot be so transitioned, but anecdotal reports from horse owners or riders familiar with bitted bridle use suggest that a large majority of horses do respond well when ridden bit-free [11]
- In the horses that can be transitioned, how long does it take?
- Are there particular bit-free bridles that improve the success rate of making this transition?No data are yet available. It would be anticipated that there would be greater benefits with bit-free bridles that do not cause pain.
- What are the detailed specifications of the most effective bit-free bridles?Design provisions should not be aimed specifically at causing pain, nor should equipment permit pain to be inflicted, even when misused.
- Are there particular trainers who are more successful with transitioning horses to bit-free riding?This would almost certainly be the case, but none have yet been compared so none have been documented.
- If so, what training of trainers is necessary to increase the number who are successful?The content of training programmes would at least partly depend on the outcomes of the foregoing trials. Presumably such training would involve adequate study of available literature combined with knowledgeable hands-on supervision and involvement. Equestrian organisations could consider sponsoring and encouraging the conduct of training workshops and other such activities in teaching establishments.
- Can riders be trained so that they can successfully transition their own horses to bit-free riding?The answer is yes, but only if they are well informed, willing to take advice, and are well motivated.
- What proportion of horses can never successfully make this transition?As yet, there are apparently no anecdotal or other reports of complete failure to make the transition. The foregoing trials would be expected to provide specific information about success or failure rates, and possible reasons for any failures.
- What is the relative performance of horses ridden bitted and bit-free in different competitive events?Although not yet determined rigorously, anecdotal reports suggest that improvements in performance can be expected. This needs to be investigated in carefully designed studies in which the athletic performance of bitted and bit-free horses is compared during time-based competitions, such as in the cross-country and show jumping elements of three-day-events. This also applies to flatraces, steeplechases, and harness races, but, as noted above, the requirement that bits be worn in such events will probably mean that off-track trials will need to be arranged.
Funding
Acknowledgments
Conflicts of Interest
References
- Cook, W.R. Bit-induced pain: A cause of fear, flight, and facial neuralgia in the horse. Pferdeheilkunde 2003, 19, 75–82. [Google Scholar] [CrossRef]
- Cook, W.R.; Strasser, H. Harmful effects of the bit. In Metal in the Mouth: The Abusive Effects of Bitted Bridles; Kells, S., Ed.; Sabine Kells: Qualicum Beach, BC, Canada, 2003; pp. 3–13. [Google Scholar]
- McLean, A.N.; McGreevy, P.D. Behavioral problems with the ridden horse. In The Domestic Horse: The Origins, Development, and Management of Its Behaviour; Mills, D.S., McDonnell, S.M., Eds.; Cambridge University Press: Cambridge, UK, 2005; pp. 196–211. [Google Scholar]
- Quick, J.S.; Warren-Smith, A.K. Preliminary investigation of horses’ (Equus caballus) responses to different bridles during foundation training. J. Vet. Behav. 2009, 4, 169–176. [Google Scholar] [CrossRef]
- Hannah, C. The Truth about Bits: Facial [Trigeminal] Neuralgia. Horse and Human 2009. Available online: http://www.horseandhuman.co.nz/articles/html/the_truth_about_bits_part4.html (accessed on 18 February 2020).
- Williams, L.R.; Warren-Smith, A.K. Conflict responses exhibited by dressage horses during competition. J. Vet. Behav. Clin. Appl. Res. 2010, 5, 215. [Google Scholar] [CrossRef]
- Cook, W.R. Damage by the bit to the equine interdental space and second lower premolar. Equine Vet. Educ. 2011, 23, 355–360. [Google Scholar] [CrossRef]
- McGreevy, P.D. The fine line between pressure and pain: Ask the horse. Vet. J. 2011, 188, 250–251. [Google Scholar] [CrossRef]
- Jahiel, J. Increase Comfort, Reduce Risk: The Bitless Bridle. Equestrian Medical Safety Association 2014, Fall Newsletter. pp. 5–12. Available online: http://emsaonline.net/wp-content/uploads/gravity_forms/1-5f7def (accessed on 17 February 2020).
- Mellor, D.J.; Beausoleil, N.J. Equine welfare during exercise: An evaluation of breathing, breathlessness and bridles. Animals 2017, 7, 41. [Google Scholar] [CrossRef]
- Cook, W.R.; Kibler, M. Behavioural assessment of pain in 66 horses, with and without a bit. Equine Vet. Educ. 2019, 31, 551–560. [Google Scholar] [CrossRef]
- Merskey, H. Pain terms: A list of definitions and notes on usage. Pain 1979, 6, 249–252. [Google Scholar]
- Mellor, D.J.; Cook, C.J.; Stafford, K.J. Chapter 9: Quantifying some responses to pain as a stressor. In The Biology of Animal Stress: Basic Principles and Implications for Welfare; Moberg, G.P., Mench, J.A., Eds.; CAB International: Wallingford, UK, 2000; pp. 171–198. [Google Scholar]
- Sneddon, L.U.; Elwood, R.W.; Adamo, S.A.; Leach, M.C. Defining and assessing animal pain. Anim. Behav. 2014, 97, 201–212. [Google Scholar] [CrossRef]
- McLennan, K.M.; Miller, A.L.; Dalla Costa, E.; Stucke, D.; Corke, M.J.; Broom, D.M.; Leach, M.C. Conceptual and methodological issues relating to pain assessment in mammals: The development and utilisation of pain facial expression scales. Appl. Anim. Behav. Sci. 2019, 217, 1–15. [Google Scholar] [CrossRef]
- Molony, V. Comments on anand and craig, pain 67 (1996). Pain 1997, 70, 293. [Google Scholar] [CrossRef]
- Mellor, D.J.; Stafford, K.J. Acute castration and/or tailing distress and its alleviation in lambs. N. Z. Vet. J. 2000, 48, 33–43. [Google Scholar] [CrossRef]
- Stafford, K.J.; Mellor, D.J. Dehorning and disbudding distress and its alleviation in calves. Vet. J. 2005, 169, 337–349. [Google Scholar] [CrossRef]
- Stafford, K.J.; Mellor, D.J. The welfare significance of the castration of cattle: A review. N. Z. Vet. J. 2005, 53, 271–278. [Google Scholar] [CrossRef]
- Weary, D.M.; Niel, L.; Flower, F.C.; Fraser, D. Identifying and preventing pain in animals. Appl. Anim. Behav. Sci. 2006, 100, 64–76. [Google Scholar] [CrossRef]
- Prunier, A.; Mounier, L.; Le Neindre, P.; Leterrier, C.; Mormede, P.; Paulmier, V.; Prunet, P.; Terlouw, C.; Guatteo, R. Identifying and monitoring pain in farm animals: A review. Animal 2013, 6, 998–1010. [Google Scholar] [CrossRef]
- Gregory, N.G. Physiology and Behaviour of Animal Suffering; Blackwell Science: Oxford, UK, 2004. [Google Scholar]
- Wynne, C.D.L. What are animals? Why anthropomorphism is still not a scientific approach to behavior. Comp. Cogn. Behav. Rev. 2007, 2, 125–135. [Google Scholar] [CrossRef]
- Serpell, J.A. How happy is your pet? The problem of subjectivity in the assessment of companion animal welfare. Anim. Welf. 2019, 28, 57–66. [Google Scholar] [CrossRef]
- Bermond, D.G. A neuropsychological and evolutionary approach to animal consciousness and animal suffering. Anim. Welf. 2001, 10, S47–S62. [Google Scholar]
- Key, B. Why fish do not feel pain. Anim. Sentience 2016, 3, 1–33. [Google Scholar]
- Dawkins, M.S. Animal welfare with and without consciousness. J. Zool. 2017, 301, 1–10. [Google Scholar] [CrossRef]
- Waran, N.; Randle, H. What we can measure, we can manage: The importance of using robust welfare indicators in equitation science. Appl. Anim. Behav. Sci. 2017, 190, 74–81. [Google Scholar] [CrossRef]
- Lesimple, C. Indicators of horse welfare: State-of-the-art. Animals 2020, 10, 294. [Google Scholar] [CrossRef]
- Mellor, D.J. Welfare-aligned sentience: Enhanced capacities to experience, inte ract, anticipate, choose and survive. Animals 2019, 9, 440. [Google Scholar] [CrossRef]
- Critical Anthropomorphism. Wikipedia. 2019. Available online: https://en.wikipedia.org/wiki/Critical_anthropomorphism (accessed on 15 December 2019).
- Talbot, J.D.; Marrett, S.; Evans, A.C.; Meyer, E.; Bushnell, M.C.; Duncan, G.H. Multiple representations of pain in human cerebral cortex. Science 1991, 251, 1355–1358. [Google Scholar] [CrossRef]
- Jones, A.K.P.; Friston, K.; Frackowiak, R.S.J. Localization of responses to pain in human cerebral cortex. Science 1992, 255, 215–216. [Google Scholar] [CrossRef]
- Treede, R.D.; Kenshalo, D.R.; Gracely, R.H.; Jones, A.K.P. The cortical representation of pain. Pain 1999, 79, 105–111. [Google Scholar] [CrossRef]
- Adrian, E.D.; Matthews, B.H.C. The interpretation of potential waves in the cortex. J. Physiol. 1934, 81, 440–471. [Google Scholar] [CrossRef]
- Coole, J.W.; Tukey, J.W. An algorithm for the machine calculation of complex fourier series. Math. Comput. 1965, 19, 297–301. [Google Scholar] [CrossRef]
- Chen, A.C.N.; Dworkin, S.F.; Haug, J.; Gehrig, J. Topographic brain measures of human pain and pain responsivity. Pain 1989, 37, 129–141. [Google Scholar] [CrossRef]
- Bromm, B.; Lorenz, J. Neurophysiological evaluation of pain. Electroencephalogr. Clin. Neurophysiol. 1998, 107, 227–253. [Google Scholar] [CrossRef]
- Chang, P.F.; Arendt-Nielsen, L.; Graven-Nielsen, T.; Svensson, P.; Chen, A.C.N. Topographic effects of tonic cutaneous nociceptive stimulation on human electroencephalograph. Neurosci. Lett. 2001, 305, 49–52. [Google Scholar] [CrossRef]
- Chang, P.F.; Arendt-Nielsen, L.; Graven-Nielsen, T.; Svensson, P.; Chen, A.C.N. Different EEG topographic effects of painful and non-painful intramuscular stimulation in man. Exp. Brain Res. 2001, 141, 195–203. [Google Scholar] [CrossRef]
- Chang, P.F.; Arendt-Nielsen, L.; Chen, A.C.N. Differential cerebral responses to aversive auditory arousal versus muscle pain: Specific EEG patterns are associated with human pain processing. Exp. Brain Res. 2002, 147, 387–393. [Google Scholar] [CrossRef]
- Ong, R.M.; Morris, J.P.; O’Dwyer, J.K.; Barnett, J.L.; Hemsworth, P.H.; Clarke, I.J. Behavioural and EEG changes in sheep in response to painful acute electrical stimuli. Aust. Vet. J. 1997, 75, 189–193. [Google Scholar] [CrossRef]
- Jongman, E.C.; Morris, J.P.; Barnett, J.L.; Hemsworth, P.H. EEG changes in 4-week- old lambs in response to castration, tail docking and mulesing. Aust. Vet. J. 2000, 78, 339–343. [Google Scholar] [CrossRef]
- Murrell, J.C.; Johnson, C.B.; White, K.L.; Taylor, P.M.; Haberham, Z.L.; Waterman-Pearson, A.E. Changes in the EEG during castration in horses and ponies anaesthetized with halothane. Vet. Anaesth. Analg. 2003, 30, 138–146. [Google Scholar] [CrossRef]
- Murrell, J.C.; White, K.L.; Johnson, C.B.; Taylor, P.M.; Doherty, T.J.; Waterman- Pearson, A.E. Investigation of the EEG effects of intravenous lidocaine during halothane anaesthesia in ponies. Vet. Anaesth. Analg. 2005, 32, 212–221. [Google Scholar] [CrossRef]
- Haga, H.A.; Ranheim, B. Castration of piglets: The analgesic effects of intratesticular and intrafunicular lidocaine injection. Vet. Anaesth. Analg. 2005, 32, 1–9. [Google Scholar] [CrossRef]
- Johnson, C.B.; Stafford, K.J.; Sylvester, S.P.; Ward, R.N.; Mitchinson, S.; Mellor, D.J. Effects of age on the electroencephalographic response to castration in lambs anaesthetised using halothane in oxygen. N. Z. Vet. J. 2005, 53, 433–437. [Google Scholar] [CrossRef]
- Johnson, C.B.; Wilson, P.R.; Woodbury, M.R.; Caulkett, N.A. Comparison of analgesic techniques for antler removal in halothane-anaesthetized red deer (Cervus elaphus): Electroencephalographic responses. Vet. Anaesth. Analg. 2005, 32, 61–71. [Google Scholar] [CrossRef]
- Johnson, C.B.; Gibson, T.J.; Stafford, K.J.; Mellor, D.J. Pain perception at slaughter. Anim. Welf. 2012, 21, 113–122. [Google Scholar] [CrossRef]
- Murrell, J.C.; Johnson, C.B. 2006 Neurophysiological techniques to assess pain in animals (Review). J. Vet. Pharmacol. Ther. 2006, 29, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Gibson, T.J.; Johnson, C.B.; Stafford, K.J.; Mitchinson, S.L.; Mellor, D.J. Validation of the acute electroencephalographic responses of calves to noxious stimulus with scoop dehorning. N. Z. Vet. J. 2007, 55, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Gibson, T.J.; Johnson, C.B.; Murrell, J.C.; Hulls, C.M.; Mitchinson, S.L.; Stafford, K.J.; Johnstone, A.C.; Mellor, D.J. Electroencephalographic responses of calves to slaughter by ventral neck incision without prior stunning. N. Z. Vet. J. 2009, 57, 77–83. [Google Scholar] [CrossRef]
- Gibson, T.J.; Johnson, C.B.; Murrell, J.C.; Chambers, P.J.; Stafford, K.J.; Mellor, D.J. Components of EEG responses to slaughter: Effects of cutting neck tissues compared to major blood vessels. N. Z. Vet. J. 2009, 57, 84–89. [Google Scholar] [CrossRef]
- Gibson, T.J.; Johnson, C.B.; Murrell, J.C.; Mitchinson, S.L.; Stafford, K.J.; Mellor, D.J. Electroencephalographic response to concussive non-penetrating captive bolt stunning in halothane anaesthetised calves. N. Z. Vet. J. 2009, 57, 90–95. [Google Scholar] [CrossRef]
- Gibson, T.J.; Johnson, C.B.; Murrell, J.C.; Mitchinson, S.L.; Stafford, K.J.; Mellor, D.J. Amelioration of electroencephalographic responses to slaughter by non-penetrating captive bolt stunning after ventral neck incision in halothane anaesthetised calves. N. Z. Vet. J. 2009, 57, 96–101. [Google Scholar] [CrossRef]
- Johnson, C.B.; Sylvester, S.; Stafford, K.J.; Mitchinson, S.; Ward, R.N.; Mellor, D.J. Effects of age on the electroencephalographic response to castration in lambs anaesthetised using halothane in oxygen from birth to six weeks old. Vet. Anaesth. Analg. 2009, 36, 273–279. [Google Scholar] [CrossRef]
- Kells, N.J.; Beausoleil, N.J.; Chambers, P.; Sutherland, M.A.; Morrison, R.S.; Johnson, C.B. Electroencephalographic responses of anaesthetized pigs (Sus scrofa) to tail docking using clippers or cautery iron performed at 2 or 20 days of age. Vet. Anaesth. Analg. 2017, 44, 1156–1165. [Google Scholar] [CrossRef]
- Kells, N.J.; Beausoleil, N.J.; Sutherland, M.A.; Johnson, C.B. Electroencephalographic responses of anaesthetised pigs to intraperitoneal injection of sodium pentobarbital. Anim. Welf. 2018, 27, 205–214. [Google Scholar] [CrossRef]
- Kells, N.J.; Beausoleil, N.J.; Sutherland, M.A.; Johnson, C.B. Postnatal development of EEG responses to noxious stimulation in pigs (Sus scrofa) aged 1–15 days. Anim. Welf. 2019, 28, 317–329. [Google Scholar] [CrossRef]
- Diesch, T.J.; Mellor, D.J.; Johnson, C.B.; Lentle, R.G. Developmental changes in the electroencephalogram and responses to a noxious stimulus in anaesthetised tammar wallaby joeys (Macropus eugenii eugenii). Lab. Anim. 2010, 44, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Diesch, T.J.; Mellor, D.J.; Johnson, C.B.; Lentle, R.G. Electroencephalographic responses to tail clamping in anaesthetised rat pups. Lab. Anim. 2009, 43, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Murrell, J.C.; Waters, D.; Mitchinson, S.L.; Johnson, C.B. Comparative effect of thermal, mechanical and electrical noxious stimuli on the electroencephalogram of the rat. Br. J. Anaesth. 2007, 98, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Kongara, K.; McIlhone, A.E.; Kells, N.J.; Johnson, C.B. Electroencephalographic evaluation of decapitation of the anaesthetized rat. Lab. Anim. 2014, 48, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Kongara, K.; Harding, D.; Ward, N.; Dukkipati, V.S.R.; Johnson, C.; Chamber, P. Comparison of electroencephalographic changes in response to acute electrical and thermal stimuli with the tail flick and hot plate test in rats administered with opiorphon. BMC Neurol. 2018, 18, 43. [Google Scholar] [CrossRef]
- Kongara, K.; Chambers, J.P.; Johnson, C.B. 2010 Electroencephalographic responses of tramadol, parecoxib and morphine to acute noxious electrical stimulation in anaesthetised dogs. Res. Vet. Sci. 2010, 88, 127–133. [Google Scholar] [CrossRef]
- Fraser, D. Understanding Animal Welfare: The Science in its Cultural Context; Wiley-Blackwell: Oxford, UK, 2008. [Google Scholar]
- Mellor, D.J. Enhancing animal welfare by creating opportunities for ‘positive affective engagement’. N. Z. Vet. J. 2015, 63, 3–8. [Google Scholar] [CrossRef]
- Mellor, D.J. Positive welfare states and promoting environment-focused and animal-to-animal interactive behaviours. N. Z. Vet. J. 2015, 63, 9–16. [Google Scholar] [CrossRef]
- Mellor, D.J. Positive animal welfare states and reference standards for welfare assessment. N. Z. Vet. J. 2015, 63, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Mellor, D.J. Updating animal welfare thinking: Moving beyond the ‘five freedoms’ towards ‘a life worth living’. Animals 2016, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Mellor, D.J. Operational details of the Five Domains Model and its key applications to the assessment and management of animal welfare. Animals 2017, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Mellor, D.J.; Beausoleil, N.J. Extending the ‘Five Domains’ model for animal welfare assessment to incorporate positive welfare states. Anim. Welf. 2015, 24, 241–253. [Google Scholar] [CrossRef]
- Mellor, D.J.; Beausoleil, N.J. Moving beyond a problem-based focus on poor welfare towards creating opportunities to have positive welfare experiences. In Mental Health and Well-Being in Animals, 2nd ed.; McMillan, F.D., Ed.; CAB International: Wallingford, UK, 2019; pp. 50–66. [Google Scholar]
- Molony, V.; Kent, J.E. Assessment of acute pain in farm animals using behavioural and physiological measurements. J. Anim. Sci. 1997, 75, 266–272. [Google Scholar] [CrossRef]
- Taylor, P.M.; Pascoe, P.J.; Mama, K.R. Diagnosing and treating pain in the horse: Where are we today? Vet. Clin. Equine 2002, 18, 1–19. [Google Scholar] [CrossRef]
- Mellor, D.J.; Stafford, K.J.; Todd, S.E.; Lowe, T.E.; Ward, R.N.; Gregory, N.G.; Bruce, R.A. Comparison of cortisol and catecholamine responses to ring castration and tailing of lambs and amputation dehorning of calves. Aust. Vet. J. 2002, 80, 228–233. [Google Scholar] [CrossRef]
- Molony, V.; Kent, J.E.; McKendrick, I.J. Validation of a method for assessment of an acute pain in lambs. Appl. Anim. Behav. Sci. 2002, 76, 215–238. [Google Scholar] [CrossRef]
- Peers, A.; Mellor, D.J.; Wintour, E.M.; Dodic, M. Blood pressure, heart rate, hormonal and other acute responses to rubber ring castration plus tailing of lambs. N. Z. Vet. J. 2002, 50, 56–62. [Google Scholar] [CrossRef]
- Rutherford, K.M.D. Assessing pain in animals. Anim. Welf. 2002, 11, 31–53. [Google Scholar]
- Stafford, K.J.; Mellor, D.J. Painful husbandry procedures in livestock and poultry. In Improving Animal Welfare, 2nd ed.; Grandin, T., Ed.; CABI: Wallingford, UK, 2015; pp. 96–124. [Google Scholar]
- Sutherland, M.A. Welfare implications of invasive piglet husbandry procedures, methods of alleviation and alternatives: A review. N. Z. Vet. J. 2015, 63, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Sapolsky, R.M.; Romero, L.M.; Munck, A.U. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocrinol. Rev. 2000, 21, 55–89. [Google Scholar] [CrossRef]
- Ashley, F.H.; Waterman-Pearson, A.E.; Whay, H.R. Behavioural assessment of pain in horses and donkeys: Application to clinical practice and future studies. Equine Vet. J. 2005, 37, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Petrie, N.; Stafford, K.J.; Mellor, D.J.; Bruce, R.A.; Ward, R.N. The behaviour of calves tail docked with a rubber ring used with or without local anaesthetic. Proc. N. Z. Soc. Anim. Prod. 1995, 55, 58–60. [Google Scholar]
- Dinniss, A.S.; Stafford, K.J.; Mellor, D.J.; Bruce, R.A.; Ward, R.N. The behaviour pattern of lambs after castration using a rubber ring and/or castrating clamp with or without local anaesthetic. N. Z. Vet. J. 1999, 47, 198–203. [Google Scholar] [PubMed]
- Mellor, D.J.; Stafford, K.J. Physiological and behavioural assessment of pain in ruminants: Principles and caveats. Altern. Lab. Anim. 2004, 32 (Suppl. 1), 267–271. [Google Scholar] [CrossRef]
- Stafford, K.J.; Mellor, D.J. Monitoring pain in animals using behaviour. Proc. N. Z. Soc. Anim. Prod. 2002, 62, 355–358. [Google Scholar]
- Sylvester, S.P.; Stafford, K.J.; Mellor, D.J.; Bruce, R.A.; Ward, R.N. Behavioural responses of calves to amputation dehorning with and without local anaesthesia. Aust. Vet. J. 2004, 82, 697–700. [Google Scholar] [CrossRef]
- Taffarel, M.O.; Luna, S.P.L.; de Oliveira, F.A.; Cardoso, G.S.; de Moura Alonso, J.; Pantoja, J.C.; Brondani, J.T.; Love, E.; Taylor, P.; White, K.; et al. Refinement and partial validation of the UNESP-Botucatu multidimensional composite pain scale for assessing postoperative pain in horses. BMC Vet. Res. 2015, 11, 83. [Google Scholar] [CrossRef]
- Dalla Costa, E.; Minero, M.; Lebelt, D.; Stucke, D.; Canali, E.; Leach, M.C. Development of the Horse Grimace Scale (HGS) as a pain assessment tool in horses undergoing routine castration. PLoS ONE 2014, 9, e92281. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3960217/ (accessed on 21 January 2020). [CrossRef]
- Dalla Costa, E.; Stucke, D.; Dai, F.; Minero, M.; Leach, M.C.; Lebelt, D. Using the horse grimace scale (HGS) to assess pain associated with acute laminitis in horses (Equus caballus). Animals 2016, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Wathan, J.; Burrows, A.M.; Waller, B.M.; McComb, K. EquiFACS: The equine facial action coding system. PLoS ONE 2015, 10, e0131738. [Google Scholar] [CrossRef]
- Gleerup, K.B.; Lindegaard, C. Recognition and quantification of pain in horses: A tutorial review. Equine Vet. Educ. 2016, 28, 47–57. [Google Scholar] [CrossRef]
- Gleerup, K.B.; Forkman, B.; Lindegaard, C.; Andersen, P.H. An equine pain face. Vet. Anaesth. Analg. 2015, 42, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Machteld, C.; van Dierendonck, M.C.; van Loon, J.P.A.M. Monitoring acute equine visceral pain with the equine utrecht university scale for composite pain assessment (EQUUS-COMPASS) and the equine utrecht university scale for facial assessment of pain (EQUUS-FAP): A validation study. Vet. J. 2016, 216, 175–177. [Google Scholar]
- Dyson, S.; Berger, J.M.; Ellis, A.D.; Mullard, J. Can the presence of musculoskeletal pain be determined from the facial expressions of ridden horses (FEReq)? J. Vet. Behav. 2017, 19, 78–89. [Google Scholar] [CrossRef]
- Mullard, J.; Berger, J.M.; Ellis, A.D.; Dyson, S. Development of an ethogram to describe facial expressions in ridden horses (FEReq). J. Vet. Behav. 2017, 18, 7–12. [Google Scholar] [CrossRef]
- Torcivia, C.; McDonell, S. In-person caretaker visits disrupt ongoing discomfort behavior in hospitalized equine orthopedic surgical patients. Animals 2020, 10, 210. [Google Scholar] [CrossRef]
- Haggard, P.; de Boer, L. Oral somatosensory awareness. Neurosci. Biobehav. Rev. 2014, 47, 469–484. [Google Scholar] [CrossRef]
- Aspinall, V.; Cappello, M. Introduction to Veterinary Anatomy and Physiology Textbook, 3rd ed.; Elsevier Health Sciences: Amsterdam, The Netherlands; Butterworth-Heinemann: Oxford, UK, 2015. [Google Scholar]
- Equine Cranial Nerves. Vet Physio Phyle, WordPress. 2019, pp. 1–14. Available online: https://vetphysiophyle.wordpress.com/2019/04/04/equine-cranial-nerves/ (accessed on 31 January 2020).
- Trigeminal Nerve, Wikipedia. 2019, pp. 1–14. Available online: https://en.wikipedia.org/wiki/Trigeminal_nerve (accessed on 31 January 2020).
- Facial Nerve. Wikipedia. 2020, pp. 1–8. Available online: https://en.wikipedia.org/wiki/Facial_nerve (accessed on 31 January 2020).
- Bennett, D.G. Bits and bitting: Form and function. In Proceedings of the 47th Annual Convention of the American Association of Equine Practitioners, San Diego, CA, USA, 24–28 November 2001; pp. 130–141. Available online: https://pdfs.semanticscholar.org/7a26/a4617e7587d72ffcc2435dd7071d3ad3b6b1.pdf (accessed on 17 February 2020).
- Manfredi, J.; Clayton, H.M.; Rosenstein, D. Radiographic study of bit position within the horse’s oral cavity. Equine Comp. Exerc. Physiol. 2005, 2, 195–201. [Google Scholar] [CrossRef]
- Benoist, C.C.; Cross, G.H. A photographic methodology for analyzing bit position under rein tension. J. Equine Vet. Sci. 2018, 67, 102–111. [Google Scholar] [CrossRef]
- Mantyh, P.W. The neurobiology of skeletal pain. Eur. J. Neurosci. 2014, 39, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Van Lancker, S.; van den Broeck, W.; Simoens, P. Incidence and morphology of bone irregularities of the equine interdental space (bars of the mouth). Equine Vet. Educ. 2007, 19, 103–106. [Google Scholar] [CrossRef]
- College Physics. Tension. In Dynamics: Force and Newton’s Laws of Motion; OpenStax College, Rice University: Huston, Tx, USA; Available online: https://opentextbc.ca/physicstestbook2/chapter/normal-tension-and-other-examples-of-forces/ (accessed on 18 February 2020).
- Clayton, H.M.; Singleton, W.H.; Lanovaz, J.; Cloud, G.L. Measurement of rein tension during horseback riding using strain gage transducers. Exp. Tech. 2003, 27, 34–36. [Google Scholar] [CrossRef]
- Clayton, H.M.; Larson, B.; Kaiser, L.A.J.; Lavagnino, M. Length and elasticity of side reins affect rein tension at trot. Vet. J. 2011, 188, 291–294. [Google Scholar] [CrossRef] [PubMed]
- Egenvall, A.; Eisersiö, M.; Rhodin, M.; van Weeren, R.; Roepstorff, L. Rein tension during canter. Comp. Exerc. Physiol. 2015, 11, 107–117. [Google Scholar] [CrossRef]
- Egenvall, A.; Roepstorff, L.; Eisersiö, M.; Rhodin, M.; van Weeren, R. Stride-related rein tension patterns in walk and trot in the ridden horse. Acta Vet. Scand. 2015, 57, 89. [Google Scholar] [CrossRef]
- Egenvall, A.; Clayton, H.M.; Eisersiö, M.; Roepstorff, L.; Byström, A. Rein tensions in transitions and halts during equestrian dressage training. Animals 2019, 9, 712. [Google Scholar] [CrossRef]
- Piccolo, L.; Kienapfel, K. Voluntary rein tension in horses when moving unridden in s dressage frame compared with ridden tests in the same horses—A pilot study. Animals 2019, 9, 321. [Google Scholar] [CrossRef]
- Heleski, C.R.; McGreevy, P.D.; Kaiser, L.J.; Lavagnino, M.; Tans, E.; Bello, N.; Clayton, H.M. Effects on behaviour and rein tension on horses ridden with or without martingales and rein inserts. Vet. J. 2009, 181, 56–62. [Google Scholar] [CrossRef]
- Christensen, J.W.; Zharkikh, T.L.; Antoine, A.; Malmkvist, J. Rein tension acceptance in young horses in a voluntary test situation. Equine Vet. J. 2011, 43, 223–228. [Google Scholar] [CrossRef]
- Mellor, D.J. Equine Welfare during Exercise: Do We Have a ‘Bit’ of a Problem. PowerPoint Slides Presented at a Professional Development Event, Entitled Sport Horse Welfare and Social Licence to Operate, Mounted by Horse South Australia on 13 and 14 February 2018 at Hahndorf, South Australia. Available online: https://www.slideshare.net/SAHorse/equine-welfare-during-exercise-do-we-have-a-bit-of-a-problem (accessed on 9 February 2020).
- Bendrey, R. New methods for the identification of evidence for bitting on horse remains from archaeological sites. J. Archaeol. Sci. 2007, 34, 1036–1050. [Google Scholar] [CrossRef]
- Mata, F.; Johnson, C.; Bishop, C. A cross-sectional epidemiological study of prevalence and severity of bit-induced oral trauma in polo ponies and race horses. J. Appl. Anim. Welf. Sci. 2015, 18, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Tremaine, W.H. Management of equine mandibular injuries. Equine Vet. Educ. 1998, 10, 146–154. [Google Scholar] [CrossRef]
- Johnson, T.J. Surgical removal of mandibular periostitis bone spurs caused by bit damage. Proc. Am. Assoc. Equine Pract. 2002, 48, 458–462. [Google Scholar]
- Johnson, J.; Porter, M. Dental conditions affecting the mature performance horse (5–15 years). Proc. Am. Assoc. Equine Pract. 2006, 50, 31–36. [Google Scholar]
- Tuomola, K.; Maki-Kinnia, N.; Kujala-Wirth, M.; Mykkänen, A.; Valros, A. Oral lesions in the bit area in finnish trotters after a race: Lesion evaluation, scoring and occurrence. Front. Vet. Sci. 2019, 6, 206. Available online: https://doi.org/10.3389/fvets.2019.00206 (accessed on 9 February 2020). [CrossRef]
- Björnsdóttir, S.; Frey, R.; Kristjansson, T.; Lundström, T. Bit-related lesions in Icelandic competition horses. Acta Vet. Scand. 2014, 56, 40. Available online: http://www.actavetscand.com/content/56/1/40 (accessed on 9 February 2020).
- Byers, M.R.; Närhi, M.V. Dental injury models: Experimental tools for understanding neuroinflammatory interactions and polymodal nociceptor functions. Crit. Rev. Oral Biol. Med. 1999, 10, 4–39. [Google Scholar] [CrossRef]
- Equus 2019. Tongue Injuries: Wounds to Your Horse’s Tongue Can Easily Go Unnoticed—But That Doesn’t Mean They Can be Ignored. Available online: https://equusmagazine.com/horse-care/tongue-injuries-12258 (accessed on 9 February 2020).
- Pigg, M.; Svensson, P.; List, T. Orofacial thermal thresholds: Time-dependent variability and influence of spatial summation and test site. J. Orofac. Pain 2011, 25, 39–48. [Google Scholar]
- Tell, A.; Egenvall, A.; Lundstrom, T.; Wattle, O. The prevalence of oral ulceration in Swedish horses when ridden with bit and bridle and when unridden. Vet. J. 2008, 178, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Uldahl, M.; Clayton, H. Lesions associated with the use of bits, nosebands, spurs and whips in Danish competition horses. Equine Vet. Educ. 2018, 51, 154–162. [Google Scholar] [CrossRef]
- Rezaian, M. Absence of hyaline cartilage in the tongue of ‘Caspian miniature horse’. Anat. Histol. Embryol. 2006, 35, 241–246. [Google Scholar] [CrossRef]
- Engelke, E.; Gasse, H. An anatomical study of the rostral part of the equine oral cavity with respect to position and size of a snaffle bit. Equine Vet. Educ. 2003, 15, 158–163. [Google Scholar] [CrossRef]
- Manfredi, J.M.; Rosenstein, D.; Lanovaz, J.L.; Nauwelaerts, S.; Clayton, H.M. Fluoroscopic study of oral behaviours in response to the presence of a bit and the effects of rein tension. Comp. Exerc. Physiol. 2010, 6, 143–148. [Google Scholar] [CrossRef]
- Findley, J.A.; Sealy, H.; Franklin, S.H. Factors associated with tongue tie use in Australian Standardbred racehorses. Equine Vet. J. 2016, 48 (Suppl. 50), 18–19. [Google Scholar]
- Barakzai, S.Z.; Finnegan, C.; Dixon, P.M.; Hillyer, M.H.; Boden, L.A. Use of tongue ties in thoroughbred racehorses in the United Kingdom, and its association with surgery for dorsal displacement of the soft palate. Vet. Rec. 2009, 165, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Porter, D.; Caraguel, C.; Noschka, E.; Samantha Franklin, S. Tongue-tie use in Australian Thoroughbred horses over a 5-year period (2009–2013). In Proceedings of the World Equine Airway Symposium, Copenhagen, Denmark, 13–15 July 2017; p. 155. [Google Scholar]
- Franklin, S.H.; Naylor, J.R.; Lane, J.G. The effect of a tongue-tie in horses with dorsal displacement of the soft palate. Equine Vet. J. 2002, 34, 430–433. [Google Scholar] [CrossRef]
- Vandermark, S.; Wilkins, C. Tongue Ties: Trying to See the Whole Picture. Horses and People. August 2019. Available online: https://horsesandpeople.com.au/tongue-ties-trying-to-see-the-whole-picture/ (accessed on 6 March 2020).
- Franklin, S.; McGreevy, P. Over 20% of Australian Horses Race with Their Tongues Tied to Their Lower Jaw. The Conversation. July 2018. Available online: https://theconversation.com/over-20-of-australian-horses-race-with-their-tongues-tied-to-their-lower-jaw-99584 (accessed on 12 February 2020).
- Marsh, L.; McGreevy, P.; Hazel, S.; Santos, L.; Herbart, M.; Franklin, S. The effect of tongue-tie application on stress responses in resting horses. BioRxiv 2019, 634717. Available online: https://www.biorxiv.org/content/10.1101/634717v1.full (accessed on 24 February 2020).
- Dyson, S.; Berger, J.M.; Ellis, A.D.; Mullard, J. Development of an ethogram for a pain scoring system in ridden horses and its application to determine the presence of musculoskeletal pain. J. Vet. Behav. 2018, 23, 47–57. [Google Scholar] [CrossRef]
- Muir, W. Recognizing and Treating Pain in Horses, Veterian Key 2016. Available online: https://veteriankey.com/recognizing-and-treating-pain-in-horses/ (accessed on 17 February 2020).
- Mair, T.; Lane, G. Head shaking in horses. In Practice 1990, 12, 183–186. [Google Scholar] [CrossRef]
- Newton, S.A.; Knottenbelt, D.C.; Eldridge, P.R. Headshaking in horses: Possible aetiopathogenesis suggested by the results of diagnostic tests and several treatment regimes used in 20 cases. Equine Vet. J. 2000, 32, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Roberts, V. Trigeminal-mediated headshaking in horses: Prevalence, impact, and management strategies. Vet. Med. Res. Rep. 2019, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cook, W.R. Pathophysiology of bit control in the horse. J. Equine Vet. Sci. 1999, 19, 196–204. [Google Scholar] [CrossRef]
- Cook, W.R.; Mills, D.S. Preliminary study of jointed snaffle vs. crossunder bitless bridles: Quantified comparison of behaviour in four horses. Equine Vet. J. 2009, 41, 827–830. [Google Scholar] [CrossRef]
- Hanson, F.; Cook, R. The Bedouin bridle rediscovered: A welfare, safety and performance enhancer. Horse’s Hoof 2015, 60, 1–8. Available online: http://www.bitlessbridle.com/THEBEDOUBRIDLE.pdf (accessed on 24 February 2020).
- Carey, C. The impact of bitless vs. bitted bridles on the therapeutic riding horse research project. Presented at the Horses in Education and Therapy International Conference on Therapeutic Riding, New Taipei City, Taiwan, 22–25 June 2015; Available online: https://festinalente.ie/equine-assisted-programmes/research/impact-bitless-vs-bitted-bridles-therapeutic-riding-horse/ (accessed on 23 February 2020).
- Carey, C.; Hayes-Moriarty, S.; Brennan, R. The impact of bitted and bitless bridles on the therapeutic riding horse. In Proceedings of the 12th International Equitation Science Conference on Understanding Horses to Improve Training and Performance, Wagga Wagga, Australia, 22–25 November 2016; p. 99. Available online: https://festinalente.ie/wp-content/uploads/2017/04/Impact-of-Bitted-vs-Bitless-Bridles-for-Therapeutic-Riding-Equines.pdf (accessed on 23 February 2020).
- Carey, C.; Brennan, R.; Hayes-Moriarty, S. Further Study on the Impact of Bitted vs. Bitless Bridles for Therapeutic Riding Equines. Festiina Lente Newsletter. January 2020. Available online: https://festinalente.ie/equine-assisted-programmes/research/study-impact-bitted-vs-bitless-bridles-therapeutic-riding-equines/ (accessed on 23 February 2020).
- Polito, R.; Minero, M.; Canali, E.; Verga, M. A pilot study on Yearlings’ reactions to handling in relation to the training method. Anthrozöos 2007, 20, 295–303. [Google Scholar] [CrossRef]
- Visser, E.K.; Van Dierendonck, M.; Ellis, A.D.; Rijksen, C.; Van Reenen, C.G. A comparison of sympathetic and conventional training methods on responses to initial horse training. Vet. J. 2009, 181, 48–52. [Google Scholar] [CrossRef]
- Von Borstel, U.U.; Duncan, I.J.H.; Shoveller, A.K.; Merkies, K.; Linda Jane Keeling, L.J.; Millman, S.T. Impact of riding in a coercively obtained Rollkur posture on welfare and fear of performance horses. Appl. Anim. Behav. Sci. 2009, 228–236. [Google Scholar] [CrossRef]
- McLean, A.N.; McGreevy, P.D. Horse-training techniques that may defy the principles of learning theory and compromise welfare. J. Vet. Behav. 2010, 5, 187–195. [Google Scholar] [CrossRef]
- Hall, C.; Kay, R.; Yarnell, K. Assessing ridden horse behavior: Professional judgment and physiological measures. J. Vet. Behav. 2014, 9, 22–29. [Google Scholar] [CrossRef]
- Górecka-Bruzda, A.; Kosinska, I.; Jaworski, Z.; Tadeusz Jezierski, T.; Murphy, J. Conflict behavior in elite show jumping and dressage horses. J. Vet. Behav. 2015, 10, 137–146. [Google Scholar] [CrossRef]
- Clayton, H. Are Horses Stressed When Bitted for the First Time? Eurodressage. 2019. Available online: http://www.eurodressage.com/2019/01/03/are-horses-stressed-when-bitted-first-time (accessed on 24 February 2020).
- Fraser, A.F. The Behaviour of the Horse; CAB International: Wallingford, UK, 1992. [Google Scholar]
- Ransom, J.I.; Cade, B.S. Quantifying equid behavior: A research ethogram for free-roaming feral horses. In U.S. Geological Survey Techniques and Methods Report 2-A9; USGS: Reston, VA, USA, 2009. [Google Scholar]
- King, M. Bitless: A New Breed of Bridle. The Horse, August 2007. Available online: https://thehorse.com/124806/bitless-a-new-breed-of-bridle/ (accessed on 24 February 2020).
- Hanson, F. A positive reinforcement rein: Rule-changer and game-changer for horsemanship? Horse’s Hoof 2019, 76, 1–14. Available online: https://www.horsetalk.co.nz/2019/10/22/positive-reinforcement-rein-game-changer-horsemanship/ (accessed on 29 February 2020).
- Ambrosiano, N. All about Bitless Bridles for Your Horse: Bit-Free Headgear Is Sometimes the Answer for Sensitive Horses or Tough Training Problems. Equus. 2017. Available online: https://equusmagazine.com/riding/bitless-bridles-092206-10523 (accessed on 24 February 2020).
- Bitless Bridle. Wikipedia. 2019. Available online: https://en.wikipedia.org/wiki/Bitless_bridle (accessed on 24 February 2020).
- Bosal, Wikipedia 2018. Available online: https://en.wikipedia.org/wiki/Bosal (accessed on 24 February 2020).
- Hackamore, Wikipedia 2020. Available online: https://en.wikipedia.org/wiki/Hackamore (accessed on 24 February 2020).
- Ramey, D.W. A historical survey of human-equine interactions. In Equine Welfare; McIlwraith, C.W., Rollin, B.E., Eds.; Wiley-Blackwell: Chichester, UK, 2011; pp. 22–58. [Google Scholar]
- Whay, H.R.; Main, D.C.J.; Green, L.E.; Webster, A.J.F. Farmer perception of lameness prevalence. In Proceedings of the 12th International Symposium on Lameness in Ruminants, Orlando, FL, USA, 9–13 January 2002; pp. 355–358. [Google Scholar]
- Barker, Z.E.; Leach, K.A.; Whay, H.R.; Bell, N.J.; Main, D.C.J. Assessment of lameness prevalence and associated risk factors in dairy herds in England and Wales. J. Dairy Sci. 2010, 93, 932–941. [Google Scholar] [CrossRef] [PubMed]
- Horseman, S.V.; Roe, E.J.; Huxley, J.N.; Bell, N.J.; Mason, C.S.; Whay, H.R. The use of in-depth interviews to understand the process of treating lame dairy cows from the farmers’ perspective. Anim. Welf. 2014, 23, 157–165. [Google Scholar] [CrossRef]
- Beausoleil, N.J.; Mellor, D.J. Introducing breathlessness as an animal welfare issue. N. Z. Vet. J. 2015, 63, 44–51. [Google Scholar] [CrossRef]
- Poiseuille’s Law: IV Fluids, Open Anaesthesia. 2017. Available online: https://www.openanesthesia.org/poiseuilles_law_iv_fluids/ (accessed on 7 March 2020).
- Cook, W.R. A hypothetical, aetiological relationship between the horse’s bit, nasopharyngeal oedema and negative pressure pulmonary oedema. Equine Vet. Educ. 2014, 26, 381–389. [Google Scholar] [CrossRef]
- Cook, W.R. Hypothesis article: Bit-induced asphyxia in the racehorse as a cause of sudden death. Equine Vet. J. 2016, 28, 405–409. [Google Scholar] [CrossRef]
- Dixon, P.M.; Railton, D.I.; McGorum, B.C. Temporary bilateral laryngeal paralysis in a horse associated with general anaesthesia and post anaesthetic myositis. Vet. Rec. 1993, 132, 29–32. [Google Scholar] [CrossRef]
- Kollias-Baker, C.A.; Pipers, F.S.; Heard, D.; Seeherman, H. Pulmonary edema associated with transient airway obstruction in three horses. J. Am. Vet. Med Assoc. 1993, 202, 1116–1118. [Google Scholar]
- Tute, A.S.; Wilkins, P.A.; Gleed, R.D.; Credille, K.M.; Murphy, D.J.; Ducharme, N.G. Negative pressure pulmonary edema as a post-anesthetic complication associated with upper airway obstruction in a horse. Vet. Surg. 1996, 25, 519–523. [Google Scholar]
- McGreevy, P.D.; Harman, A.; McLean, A.; Hawson, L. Over-flexing the horse’s neck: A modern equestrian obsession? J. Vet. Behav. 2010, 5, 180–186. [Google Scholar] [CrossRef]
- McLean, A.N.; McGreevy, P.D. Ethical equitation: Capping the price horses pay for human glory. J. Vet. Behav. 2010, 5, 203–209. [Google Scholar] [CrossRef]
- McGreevy, P.D.; McLean, A.N.; Warren-Smith, A.K.; Waran, N.; Goodwin, D. Defining the terms and processes associated with equitation. In Proceedings of the 1st International Equitation Science Symposium 2005, Melbourne, Australia, 26–27 July 2005; Sydney University Press: Sydney, NSW, Australia, 2005; pp. 10–43. [Google Scholar]
- McGreevy, P.; Warren-Smith, A.; Guisard, Y. The effect of double bridles and jaw-clamping crank nosebands on facial cutaneous and ocular temperature in horses. J. Vet. Behav. Clin. Appl. Res. 2012, 7, 142–148. [Google Scholar] [CrossRef]
- Fenner, K.; Yoon, S.; White, P.; Starling, M.; McGreevy, P. The effect of noseband tightening on horses’ behavior, eye temperature, and cardiac responses. PLoS ONE 2016, 11, e0154179. [Google Scholar] [CrossRef]
- Doherty, O.; Conway, T.; Conway, R.; Murray, G.; Casey, V. An objective measure of noseband tightness and its measurement using a novel digital tightness gauge. PLoS ONE 2017, 12, e0168996. [Google Scholar] [CrossRef]
- Global Dressage Forum. Bitless or Not: It about Having the Choice. Eurodressage. March 2011. Available online: http://www.eurodressage.com/2011/11/03/bitless-or-not-its-about-having-choice (accessed on 8 March 2020).
- North American Dressage Association 2020. Available online: https://www.northamericanwesterndressage.com (accessed on 8 March 2020).
- Western Dressage Association of America 2020. Available online: https://westerndressageassociation.org (accessed on 8 March 2020).
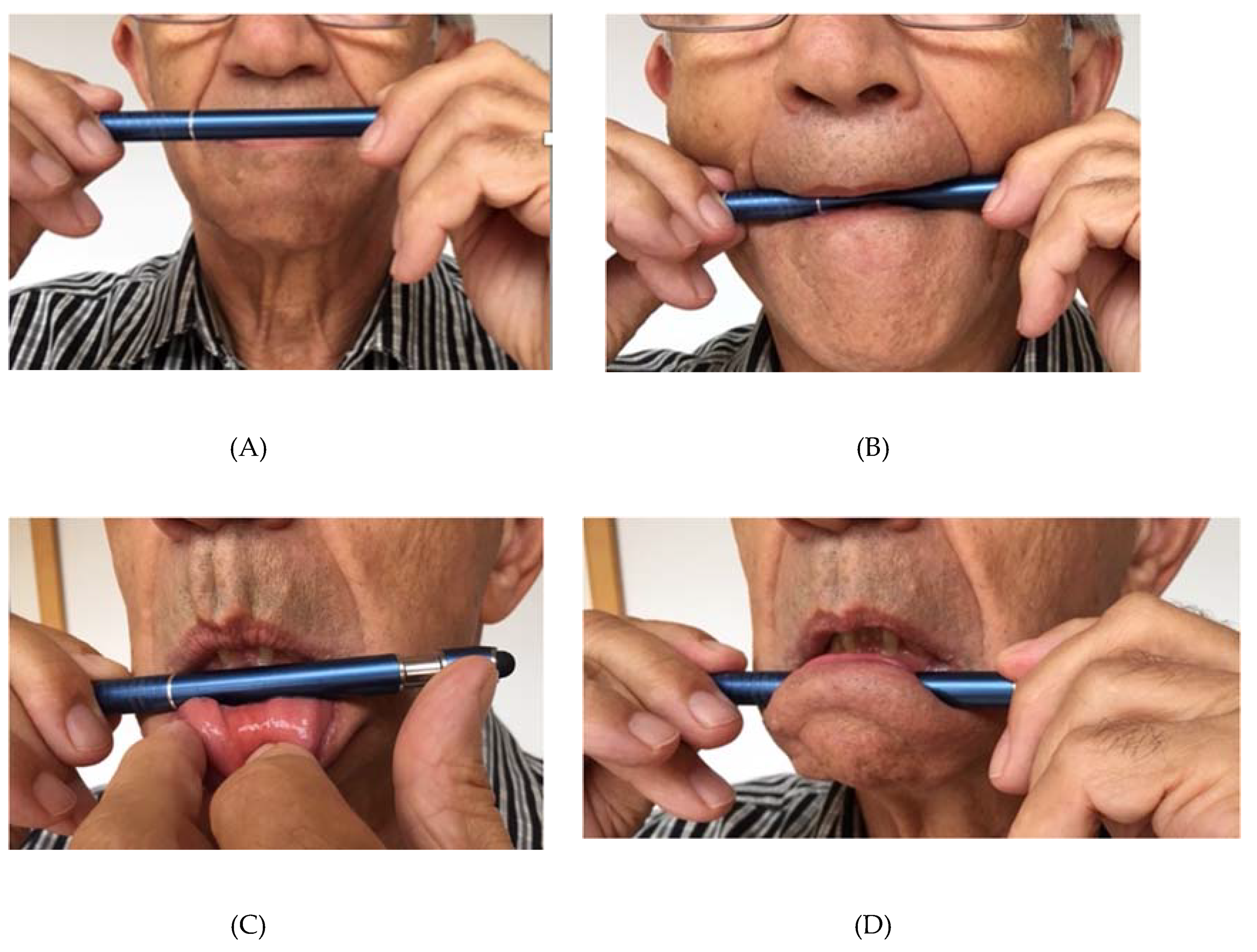
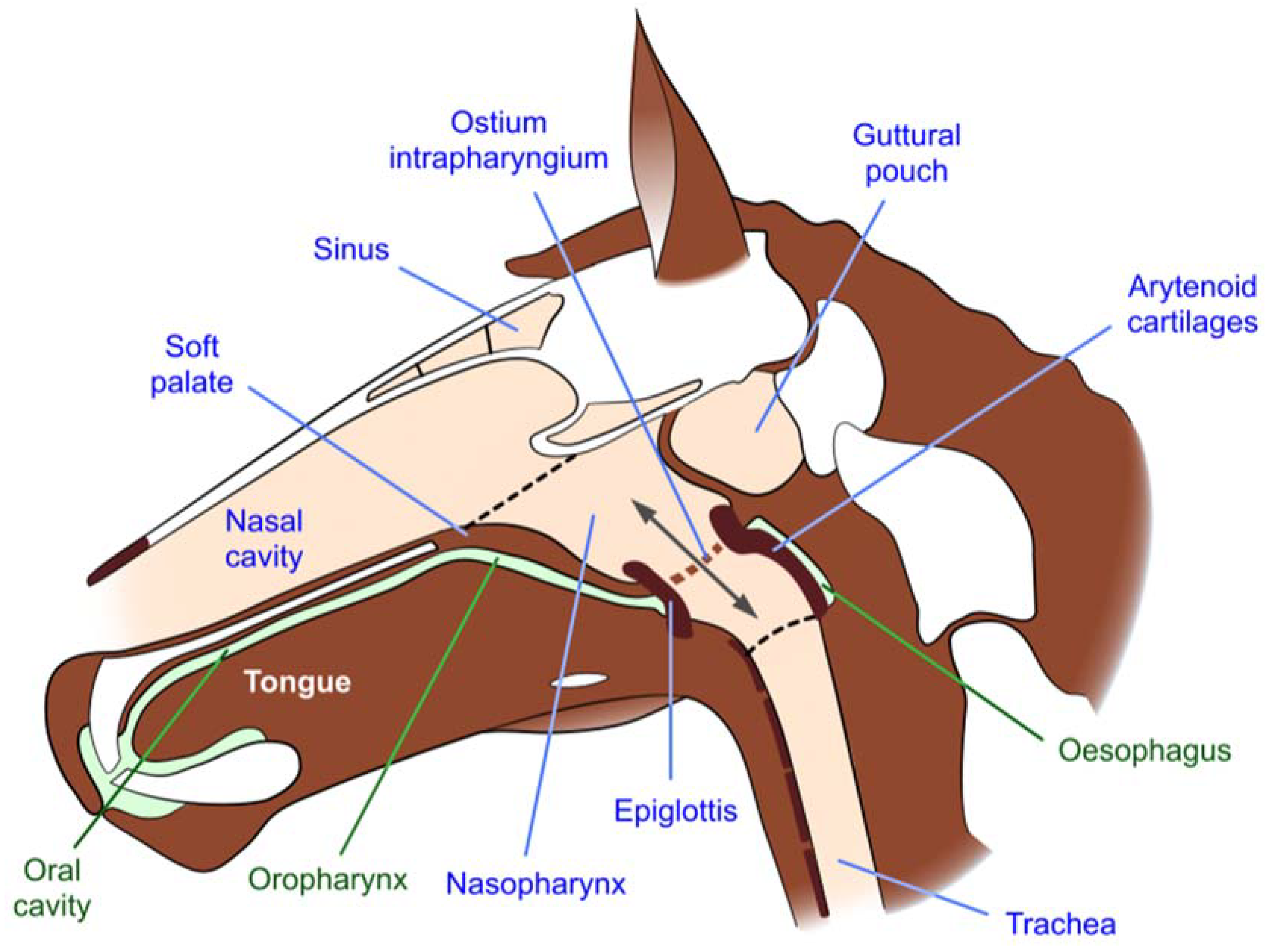
| Attribute | Description |
|---|---|
| Purpose | Pain is understood to have evolutionary survival value. |
| Detection | Pain sensations depend on activation of a discrete set of receptors (nociceptors) by noxious stimuli. |
| Perception | Further processing via nerve pathways to the brain and within the brain enables the noxious stimuli to be consciously perceived as pain. |
| Character | Pain perception varies according to site, duration and intensity of stimulation and can be modified by previous experience, emotional state and innate individual differences. |
| Definition | Pain is defined as an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or is describable in terms of such damage. |
| Variation | The pain-detection threshold is fairly uniform in mammals, whereas pain tolerance may be more species-specific and subject to modification. |
| Indicative Pain-Related Behaviours in Ridden Bitted Horses |
| Mouth: resists bridling; fussing with the bit, persistent jaw movements, chewing; crossing the jaw; slightly open or gaping mouth; teeth grinding, holding the bit between the teeth; tongue persistently moving or protruding from the mouth, tongue placed above the bit or retracted behind it; excessive salivation or drooling. Head-neck: sudden evasive movements due to abrupt increases in rein tension; side-to-side or up-down head shaking, jawline above horizontal; head tilted, stiff necked; rein-induced low jowl-angle, neck arched, nasal plane at or behind the vertical; reaches forward so rider uses longer rein. Pain face: identifiable nostril flare, lip positions, ear positions, eye white visibility and facial muscle tension. Body movement/gait: stiff or choppy stride, hair trigger responses, crabbing; difficult to control, hesitant to move forward, difficult to stop, side-stepping from straight-line motion; bucking; rearing; tail swishing. Refs: [6,28,83,90,96,97,115,133,140,141,146,147,148,149,150,151,152,153,154,155,156,157,158]; plus YouTube archive videos a |
| Bitted to Bit-Free Changes in Ridden Horse Behaviour |
| Mouth: all bit-related mouth behaviours absent; quiet, closed mouth, tongue inside mouth and appropriately placed; little or no teeth grinding; no drooling. Head-neck: head shaking absent; lower head-neck position and wider jowl angle; head, neck and spinal column properly aligned longitudinally. Pain face: no indications of mouth-related pain in healthy animals. Body movement/gait: calm, relaxed and cooperative demeanour; engaged, lively, energised and exhibits vitality of fitness; head freedom supports balanced, aligned and smooth rhythm of motion; tail movement in synchrony with spinal movement. Refs: [1,4,9,11,147,148,149,150,151]; plus YouTube archive videos a |
| Behaviours of Bit-Free Horses at Rest or When Running Free |
| As expected, domesticated horses wearing loosely-but-snugly fitted bit-free bridles do not display any of the bit-related behaviours noted above while standing at rest or engaging in exercise ranging from walking to galloping; nor do horses wearing halters while standing in stalls or moving freely in turnout paddocks. Likewise, neither do wild, free-roaming horses when standing alert or when walking, trotting, cantering and galloping during roundups. Refs: [159,160]; YouTube archive videos of bit-free domesticated horses, and of ~150 free-roaming, wild Brumbies (Australia), Camargue horses (France), Kaimanawa horses (New Zealand) and Mustangs (USA) a |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mellor, D.J. Mouth Pain in Horses: Physiological Foundations, Behavioural Indices, Welfare Implications, and a Suggested Solution. Animals 2020, 10, 572. https://doi.org/10.3390/ani10040572
Mellor DJ. Mouth Pain in Horses: Physiological Foundations, Behavioural Indices, Welfare Implications, and a Suggested Solution. Animals. 2020; 10(4):572. https://doi.org/10.3390/ani10040572
Chicago/Turabian StyleMellor, David J. 2020. "Mouth Pain in Horses: Physiological Foundations, Behavioural Indices, Welfare Implications, and a Suggested Solution" Animals 10, no. 4: 572. https://doi.org/10.3390/ani10040572
APA StyleMellor, D. J. (2020). Mouth Pain in Horses: Physiological Foundations, Behavioural Indices, Welfare Implications, and a Suggested Solution. Animals, 10(4), 572. https://doi.org/10.3390/ani10040572




