Genomewide Mapping of Selection Signatures and Genes for Extreme Feather Pecking in Two Divergently Selected Laying Hen Lines
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design, Data Collection and Editing
2.2. Statistical Analysis
2.2.1. Multidimensional Scaling
2.2.2. Mapping Selection Signatures
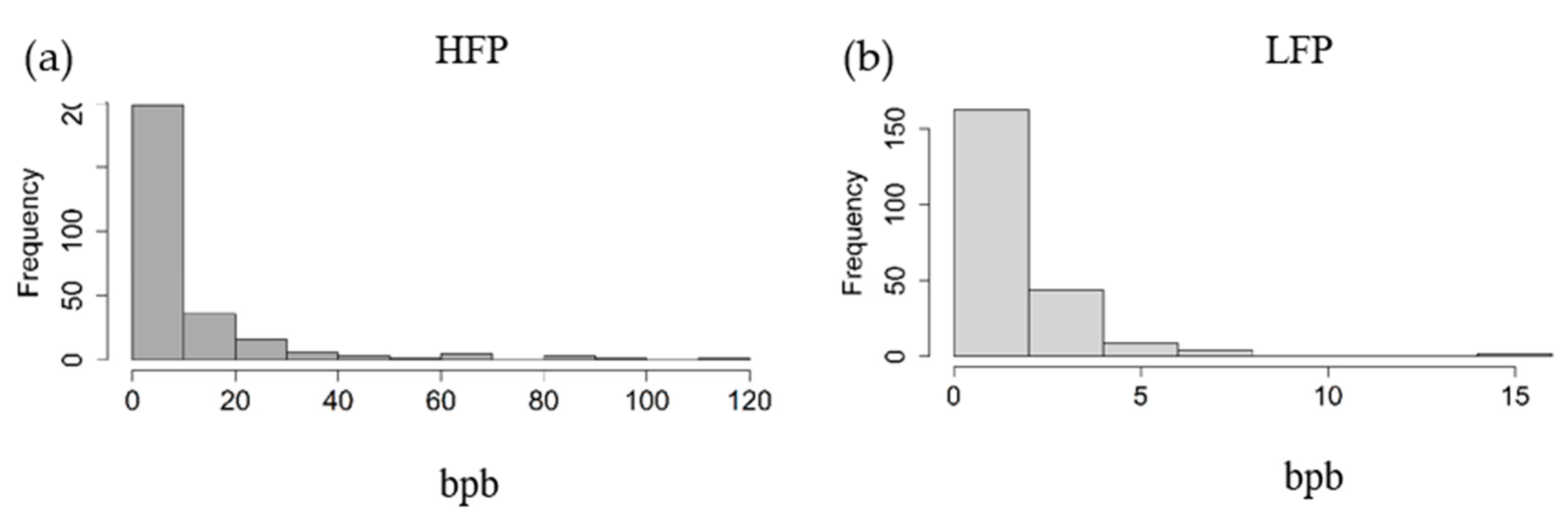
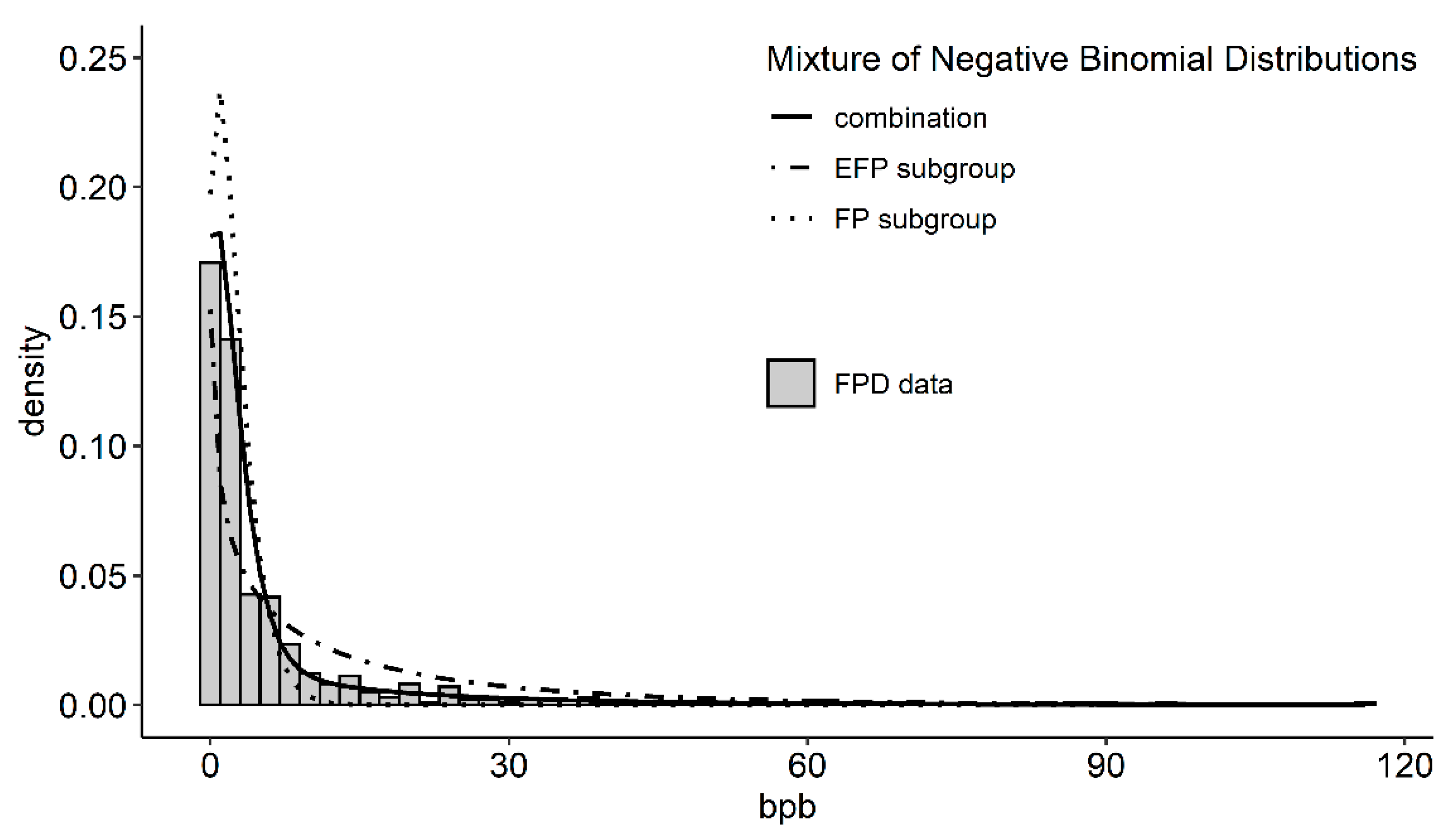
2.2.3. Detecting Extreme Feather Peckers
2.2.4. GWAS and Estimations of Genetic Parameters
2.2.5. Post GWAS Analysis
3. Results
3.1. Multidimensional Scaling
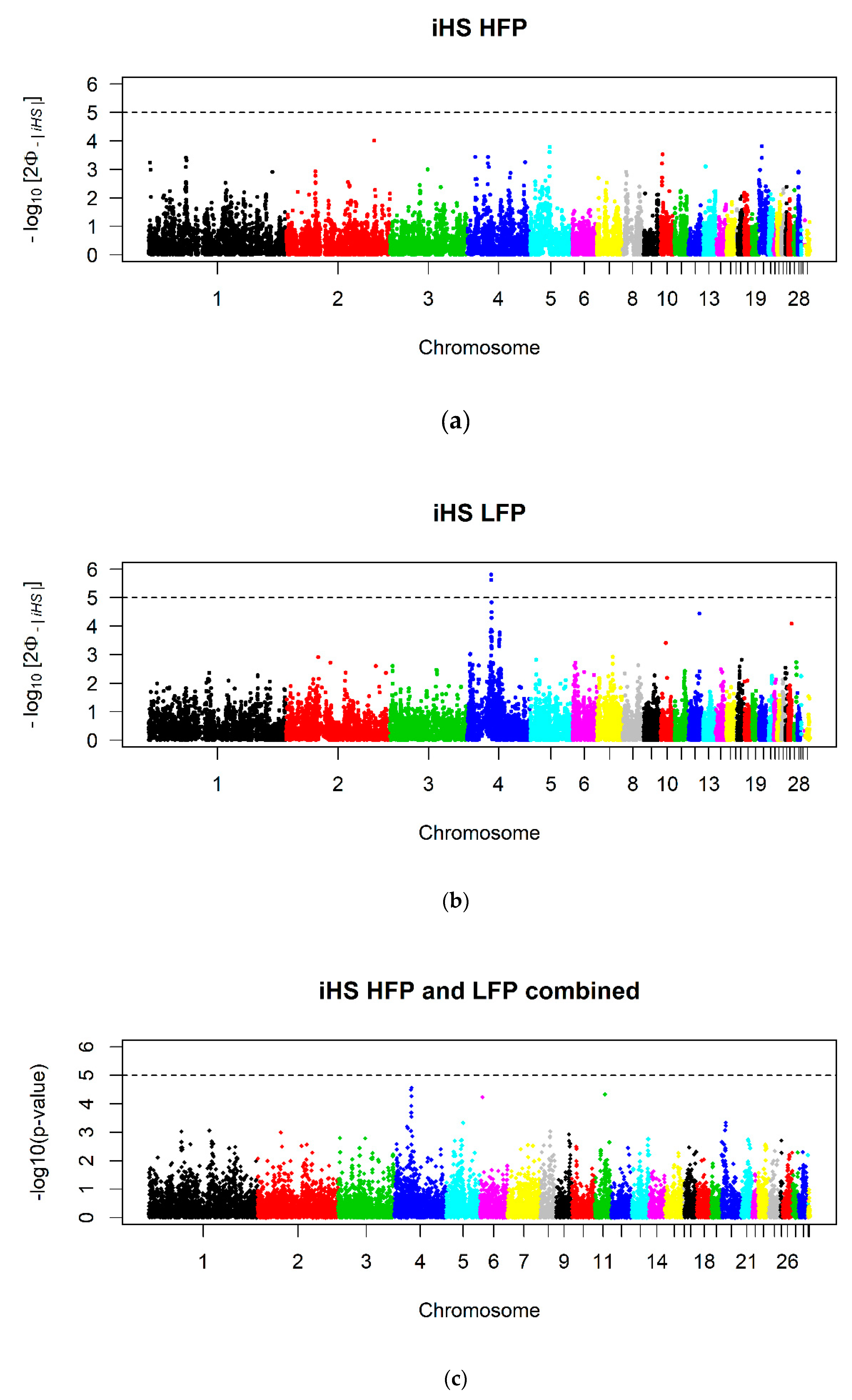
3.2. Selection Signatures
3.3. Extreme Feather Peckers
3.4. GWAS and Estimations of Genetic Parameters
3.5. Post GWAS Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rodenburg, T.B.; van Krimpen, M.M.; de Jong, I.C.; de Haas, E.N.; KOPS, M.S.; Riedstra, B.J.; Nordquist, R.E.; Wagenaar, J.P.; Bestman, M.; Nicol, C.J. The prevention and control of feather pecking in laying hens: Identifying the underlying principles. Worlds Poult. Sci. J. 2013, 69, 361–374. [Google Scholar] [CrossRef]
- Wysocki, M.; Bessei, W.; Kjaer, J.B.; Bennewitz, J. Genetic and physiological factors influencing feather pecking in chickens. Worlds Poult. Sci. J. 2010, 66, 659–672. [Google Scholar] [CrossRef]
- Bennewitz, J.; Bögelein, S.; Stratz, P.; Rodehutscord, M.; Piepho, H.P.; Kjaer, J.B.; Bessei, W. Genetic parameters for feather pecking and aggressive behavior in a large F2-cross of laying hens using generalized linear mixed models. Poult. Sci. 2014, 93, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Buitenhuis, A.J.; Kjaer, J.B. Long term selection for reduced or increased pecking behaviour in laying hens. Worlds Poult. Sci. J. 2008, 64, 477–487. [Google Scholar] [CrossRef]
- Buitenhuis, A.J.; Rodenburg, T.B.; van Hierden, Y.M.; Siwek, M.; Cornelissen, S.J.B.; Nieuwland, M.G.B.; Crooijmans, R.P.M.A.; Groenen, M.A.M.; Koene, P.; Korte, S.M.; et al. Mapping Quantitative Trait Loci Affecting Feather Pecking Behavior and Stress Response in Laying Hens. Poult. Sci. 2003, 82, 1215–1222. [Google Scholar] [CrossRef] [PubMed]
- Lutz, V.; Stratz, P.; Preuß, S.; Tetens, J.; Grashorn, M.A.; Bessei, W.; Bennewitz, J. A genome-wide association study in a large F2-cross of laying hens reveals novel genomic regions associated with feather pecking and aggressive pecking behavior. Genet. Sel. Evol. 2017, 49, 18. [Google Scholar] [CrossRef]
- Qanbari, S.; Simianer, H. Mapping signatures of positive selection in the genome of livestock. Livest. Sci. 2014, 166, 133–143. [Google Scholar] [CrossRef]
- Qanbari, S.; Tapio, M. Genomic Management of Animal Genetic Diversity; Wageningen Academic Publishers: Wageningen, The Netherlands, 2017; pp. 107–137. [Google Scholar]
- Voight, B.F.; Kudaravalli, S.; Wen, X.; Pritchard, J.K. A map of recent positive selection in the human genome. PLoS Biol. 2006, 4, 446–458. [Google Scholar] [CrossRef]
- Holsinger, K.E.; Weir, B.S. Genetics in geographically structured populations: Defining, estimating and interpreting F(ST). Nat. Rev. Genet. 2009, 10, 639–650. [Google Scholar] [CrossRef]
- Weir, B.S.; Cockerham, C.C. Estimating F-Statistics for the Analysis of Population Structure. Evolution 1984, 38, 1358–1370. [Google Scholar]
- Wechsler, B.; Huber-Eicher, B.; Nash, D.R. Feather pecking in growers: A study with individually marked birds. Br. Poult. Sci. 1998, 39, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Labouriau, R.; Kjaer, J.B.; Abreu, G.C.G.; Hedegaard, J.; Buitenhuis, A.J. Analysis of severe feather pecking behavior in a high feather pecking selection line. Poult. Sci. 2009, 88, 2052–2062. [Google Scholar] [CrossRef] [PubMed]
- Piepho, H.P.; Lutz, V.; Kjaer, J.B.; Grashorn, M.A.; Bennewitz, J.; Bessei, W. The presence of extreme feather peckers in groups of laying hens. Animal 2017, 11, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Iffland, H.; Wellmann, R.; Preuß, S.; Tetens, J.; Bessei, W.; Piepho, H.P.; Bennewitz, J. A Novel Model to Explain Extreme Feather Pecking Behavior in Laying Hens. Behav. Genet. 2020, 50, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Liljedahl, L.-E.; Kolstad, N.; Sorensen, P.; Maijala, K. Scandinavian Selection and Crossbreeding Experiment with Laying Hens. Acta Agric. Scand. 1979, 29, 273–286. [Google Scholar] [CrossRef]
- Kjaer, J.B.; Sorensen, P.; Su, G. Divergent selection on feather pecking behavior in laying hens (Gallus gallus domesticus). Appl. Anim. Behav. Sci. 2001, 71, 229–239. [Google Scholar] [CrossRef]
- Grams, V.; Bögelein, S.; Grashorn, M.A.; Bessei, W.; Bennewitz, J. Quantitative genetic analysis of traits related to fear and feather pecking in laying hens. Behav. Genet. 2015, 45, 228–235. [Google Scholar] [CrossRef]
- Bessei, W.; Bauhaus, H.; Bögelein, S. The effect of selection for high and low feather pecking on aggression-related behaviours of laying hens. Arch. Geflügelk. 2013, 77, 10–14. [Google Scholar]
- Lutz, V.; Kjaer, J.B.; Iffland, H.; Rodehutscord, M.; Bessei, W.; Bennewitz, J. Quantitative genetic analysis of causal relationships among feather pecking, feather eating, and general locomotor activity in laying hens using structural equation models. Poult. Sci. 2016, 95, 1757–1763. [Google Scholar] [CrossRef]
- Su, G.; Kjaer, J.B.; Sorensen, P. Variance Components and Selection Response for Feather-Pecking Behavior in Laying Hens. Poult. Sci. 2005, 84, 14–21. [Google Scholar] [CrossRef]
- Browning, S.R.; Browning, B.L. Rapid and accurate haplotype phasing and missing-data inference for whole-genome association studies by use of localized haplotype clustering. Am. J. Hum. Genet. 2007, 81, 1084–1097. [Google Scholar] [CrossRef] [PubMed]
- Wellmann, R. Optimum contribution selection for animal breeding and conservation: The R package optiSel. BMC Bioinform. 2019, 20, 25. [Google Scholar] [CrossRef] [PubMed]
- Peripolli, E.; Munari, D.P.; Silva, M.V.G.B.; Lima, A.L.F.; Irgang, R.; Baldi, F. Runs of homozygosity: Current knowledge and applications in livestock. Anim. Genet. 2016, 48, 255–271. [Google Scholar] [CrossRef] [PubMed]
- Browning, S.R. Estimation of pairwise identity by descent from dense genetic marker data in a population sample of haplotypes. Genetics 2008, 178, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Wellmann, R.; Bennewitz, J. Key Genetic Parameters for Population Management. Front. Genet. 2019, 10, 667. [Google Scholar] [CrossRef]
- De Leeuw, J.; Mair, P. Multidimensional Scaling Using Majorization: SMACOF in R. J. Stat. Softw. 2009, 31, 1–30. [Google Scholar] [CrossRef]
- Gautier, M.; Klassmann, A.; Vitalis, R. Rehh 2.0: A reimplementation of the R package rehh to detect positive selection from haplotype structure. Mol. Ecol. Resour. 2017, 17, 78–90. [Google Scholar] [CrossRef]
- Gautier, M.; Vitalis, R. Rehh: An R package to detect footprints of selection in genome-wide SNP data from haplotype structure. Bioinformatics 2012, 28, 1176–1177. [Google Scholar] [CrossRef]
- Grams, V.; Wellmann, R.; Preuß, S.; Grashorn, M.A.; Kjaer, J.B.; Bessei, W.; Bennewitz, J. Genetic parameters and signatures of selection in two divergent laying hen lines selected for feather pecking behaviour. Genet. Sel. Evol. 2015, 47, 77. [Google Scholar] [CrossRef]
- Storey, J.D.; Bass, A.J.; Dabney, A.; Robinson, D. qvalue: Q-value Estimation for False Discovery Rate Control. R package Version 2.15.0. 2017. Available online: http://github.com/StoreyLab/qvalue (accessed on 15 October 2019).
- Fisher, R.A. Statistical Methods for Research Workers, 4th ed.; Oliver&Boyd: London, UK, 1932. [Google Scholar]
- Macdonald, P.; Du, J. mixdist: Finite Mixture Distribution Models. R Package Version 0.5-5. Available online: https://CRAN.R-project.org/package=mixdist, 2018. (accessed on 15 October 2019).
- Yang, J.; Lee, S.H.; Goddard, M.E.; Visscher, P.M. GCTA: A tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 2011, 88, 76–82. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Bioinformatics enrichment tools: Paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009, 37, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Akey, J.M.; Zhang, G.; Zhang, K.; Jin, L.; Shriver, M.D. Interrogating a High-Density SNP Map for Signatures of Natural Selection. Genome Res. 2002, 12, 1805–1814. [Google Scholar] [CrossRef] [PubMed]
- Sigel, E.; Steinmann, M.E. Structure, Function, and Modulation of GABA(A) Receptors. J. Biol. Chem. 2012, 287, 40224–40231. [Google Scholar] [CrossRef]
- Michels, G.; Moss, S.J. GABA(A) Receptors: Properties and Trafficking. Crit. Rev. Biochem. Mol. Biol. 2007, 42, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Poshivalov, V.P. GABA-ergic correlates between aggressiveness and sociability in isolated mice. Bull. Exp. Biol. Med. 1981, 91, 648. [Google Scholar] [CrossRef]
- Brinker, T.; Bijma, P.; Vereijken, A.; Ellen, E.D. The genetic architecture of socially-affected traits: A GWAS for direct and indirect genetic effects on survival time in laying hens showing cannibalism. Genet. Sel. Evol. 2018, 50, 38. [Google Scholar] [CrossRef]
- Feng, J.; Cai, X.; Zhao, J.; Yan, Z. Serotonin Receptors Modulate GABAA Receptor Channels through Activation of Anchored Protein Kinase C in Prefrontal Cortical Neurons. J. Neurosci. 2001, 21, 6502–6511. [Google Scholar] [CrossRef]
- De Haas, E.N.; van der Eijk, J.A.J. Where in the serotonergic system does it go wrong? Unravelling the route by which the serotonergic system affects feather pecking in chickens. Neurosci. Biobehav. Rev. 2018, 95, 170–188. [Google Scholar] [CrossRef]
- Flisikowski, K.; Schwarzenbacher, H.; Wysocki, M.; Weigend, S.; Preisinger, R.; Kjaer, J.B.; Fries, R. Variation in neighbouring genes of the dopaminergic and serotonergic systems affects feather pecking behaviour of laying hens. Anim. Genet. 2009, 40, 192–199. [Google Scholar] [CrossRef]
- Ellen, E.D.; van der Sluis, M.; Siegford, J.; Guzhva, O.; Toscano, M.J.; Bennewitz, J.; van der Zande, L.E.; van der Eijk, J.A.J.; de Haas, E.N.; Norton, T.; et al. Review of Sensor Technologies in Animal Breeding: Phenotyping Behaviors of Laying Hens to Select Against Feather Pecking. Animals 2019, 9, 108. [Google Scholar] [CrossRef] [PubMed]
- Rodenburg, T.B.; Bennewitz, J.; de Haas, E.N.; Kost’ál, L.; Pichová, K.; Piette, D.; Tetens, J.; van der Eijk, J.A.J.; Visser, B.; Ellen, E.D. The use of sensor technology and genomics to breed for laying hens that show less damaging behaviour. In Proceedings of the 8th European Conference on Precision Livestock Farming, Nantes, France, 12–14 September 2017. [Google Scholar]


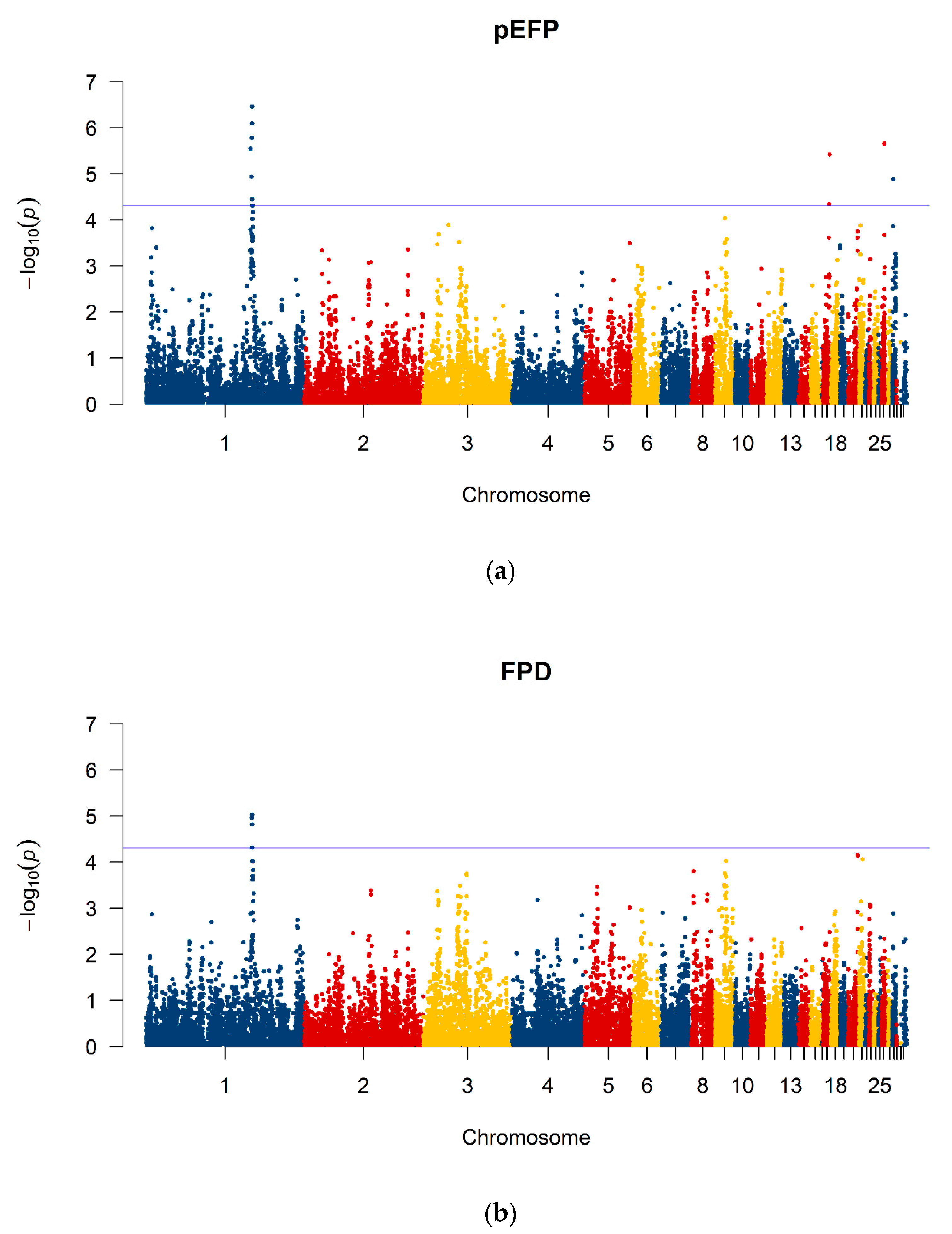
| 1 Trait | 2 Chr | SNP | Position (bp) | −Log10 (p) |
|---|---|---|---|---|
| FPD | 1 | GGaluGA044500 | 132,686,520 | 4.96 |
| 1 | Gga_rs14888608 | 132,789,468 | 5.03 | |
| 1 | GGaluGA044531 | 132,792,863 | 4.82 | |
| 1 | Gga_rs13940234 | 132,960,547 | 4.32 | |
| pEFP | 1 | Gga_rs13938103 | 131,055,669 | 5.55 |
| 1 | Gga_rs14887858 | 132,015,352 | 4.94 | |
| 1 | GGaluGA044500 | 132,686,520 | 5.78 | |
| 1 | Gga_rs14888608 | 132,789,468 | 6.46 | |
| 1 | GGaluGA044531 | 132,792,863 | 6.09 | |
| 1 | Gga_rs13940234 | 132,960,547 | 4.45 | |
| 1 | Gga_rs13624646 | 133,345,452 | 4.31 | |
| 17 | Gga_rs15792349 | 8,366,984 | 4.34 | |
| 17 | Gga_rs14098115 | 8,458,039 | 4.34 | |
| 17 | Gga_rs14097650 | 8,891,679 | 5.42 | |
| 26 | Gga_rs16203090 | 3,684,301 | 5.66 | |
| 28 | Gga_rs15249217 | 1,623,905 | 4.89 |
| 1 Trait | Prevalence | 2 VP (3SE) | 4 VA (SE) | 5 H2obs. (SE) | 6 H2liab. (SE) |
|---|---|---|---|---|---|
| FPD | − | 0.41 (0.03) | 0.08 (0.04) | 0.20 (0.08) | − |
| pEFP | 0.24 | 0.15 (0.01) | 0.02 (0.01) | 0.14 (0.07) | 0.26 (0.14) |
| Category | Term | 2 p | Genes | 3 B |
|---|---|---|---|---|
| Annotation Cluster 1 Enrichment Score 2.22 | ||||
| REACTOME_PATHWAY | R-GGA-977441 | 4.8 × 10−4 | GABRA5, GABRB3, GABRG3 | 1.3 × 10−2 |
| GOTERM_CC_DIRECT | GABA-A receptor complex (GO:1902711) | 8.5 × 10−4 | GABRA5, GABRB3, GABRG3 | 4.1 × 10−2 |
| GOTERM_MF_DIRECT | GABA-A receptor activity (GO:0004890) | 9.2 × 10−4 | GABRA5, GABRB3, GABRG3 | 3.7 × 10−2 |
| INTERPRO | Gamma-aminobutyric acid A receptor (IPR006028) | 1.6 × 10−3 | GABRA5, GABRB3, GABRG3 | 1.3 × 10−1 |
| REACTOME_PATHWAY | R-GGA-975298 | 1.6 × 10−3 | GABRA5, GABRB3, GABRG3 | 2.2 × 10−2 |
| INTERPRO | Neurotransmitter-gated ion-channel transmembrane domain (IPR006029) | 5.1 × 10−3 | GABRA5, GABRB3, GABRG3 | 2.0 × 10−1 |
| INTERPRO | Neurotransmitter-gated ion-channel, conserved site (IPR018000) | 5.1 × 10−3 | GABRA5, GABRB3, GABRG3 | 2.0 × 10−1 |
| INTERPRO | Neurotransmitter-gated ion-channel (IPR006201) | 5.4 × 10−3 | GABRA5, GABRB3, GABRG3 | 1.5 × 10−1 |
| INTERPRO | Neurotransmitter-gated ion-channel ligand-binding (IPR006202) | 5.4 × 10−3 | GABRA5, GABRB3, GABRG3 | 1.5 × 10−1 |
| UP_KEYWORDS | Ion channel | 6.7 × 10−3 | CNGA3, GABRA5, GABRB3, GABRG3 | 3.1 × 10−1 |
| UP_KEYWORDS | Synapse | 1.9 × 10−2 | GABRA5, GABRB3, GABRG3 | 4.1 × 10−1 |
| UP_KEYWORDS | Ion transport | 2.1 × 10−2 | CNGA3, GABRA5, GABRB3, GABRG3 | 3.2 × 10−1 |
| GOTERM_CC_DIRECT | cell junction (GO:0030054) | 4.5 × 10−2 | GABRA5, GABRB3, GABRG3 | 6.7 × 10−1 |
| UP_KEYWORDS | Cell junction | 5.5 × 10−2 | GABRA5, GABRB3, GABRG3 | 5.3 × 10−1 |
| UP_KEYWORDS | Cell membrane | 9.7 × 10−2 | GABRA5, GABRB3, GABRG3, RP2 | 6.7 × 10−1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iffland, H.; Wellmann, R.; Schmid, M.; Preuß, S.; Tetens, J.; Bessei, W.; Bennewitz, J. Genomewide Mapping of Selection Signatures and Genes for Extreme Feather Pecking in Two Divergently Selected Laying Hen Lines. Animals 2020, 10, 262. https://doi.org/10.3390/ani10020262
Iffland H, Wellmann R, Schmid M, Preuß S, Tetens J, Bessei W, Bennewitz J. Genomewide Mapping of Selection Signatures and Genes for Extreme Feather Pecking in Two Divergently Selected Laying Hen Lines. Animals. 2020; 10(2):262. https://doi.org/10.3390/ani10020262
Chicago/Turabian StyleIffland, Hanna, Robin Wellmann, Markus Schmid, Siegfried Preuß, Jens Tetens, Werner Bessei, and Jörn Bennewitz. 2020. "Genomewide Mapping of Selection Signatures and Genes for Extreme Feather Pecking in Two Divergently Selected Laying Hen Lines" Animals 10, no. 2: 262. https://doi.org/10.3390/ani10020262
APA StyleIffland, H., Wellmann, R., Schmid, M., Preuß, S., Tetens, J., Bessei, W., & Bennewitz, J. (2020). Genomewide Mapping of Selection Signatures and Genes for Extreme Feather Pecking in Two Divergently Selected Laying Hen Lines. Animals, 10(2), 262. https://doi.org/10.3390/ani10020262




