B-Mode and Doppler Ultrasonographic Findings of Prostate Gland and Testes in Dogs Receiving Deslorelin Acetate or Osaterone Acetate
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals Selection
2.2. Ultrasonographic Study
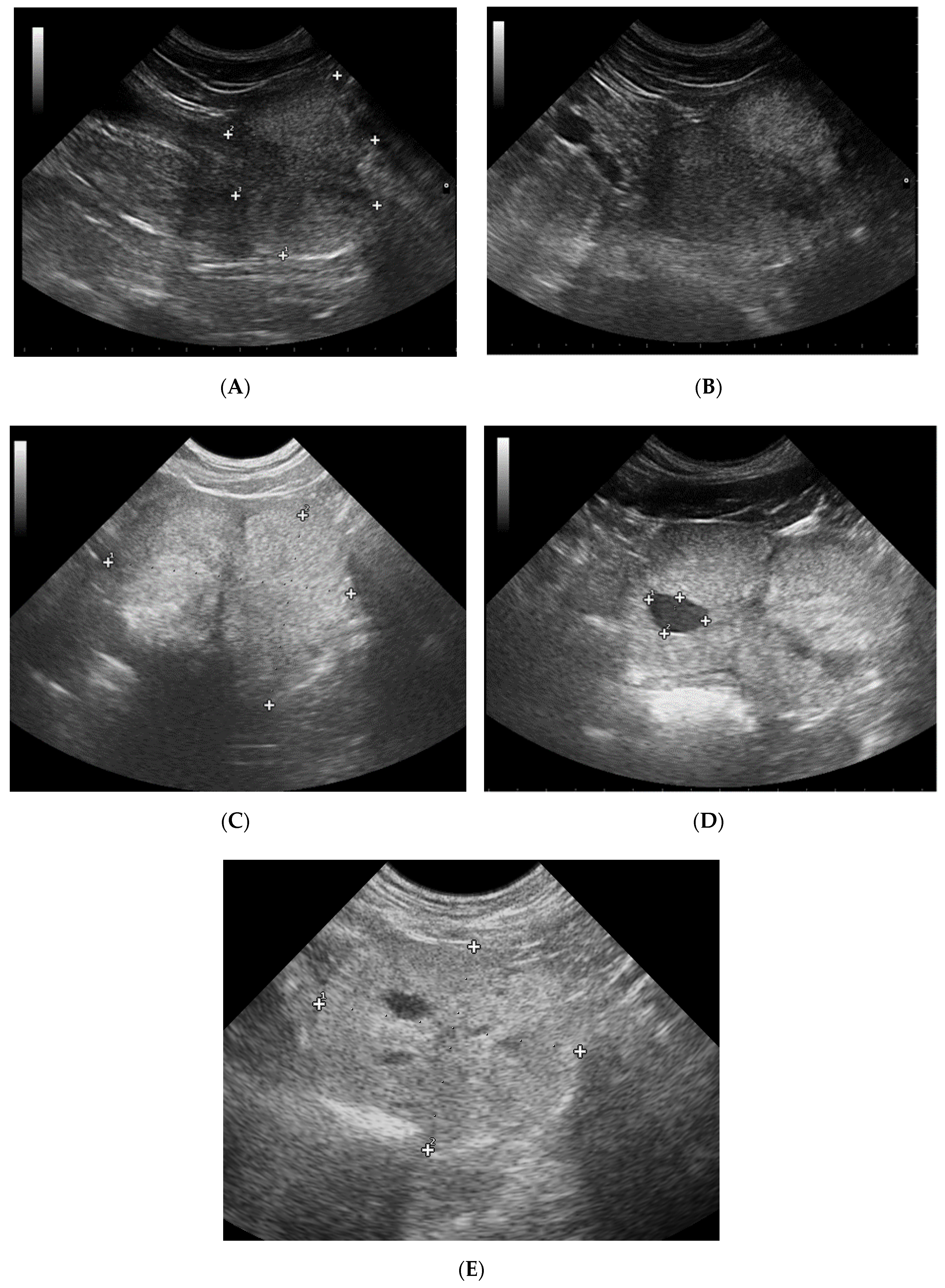
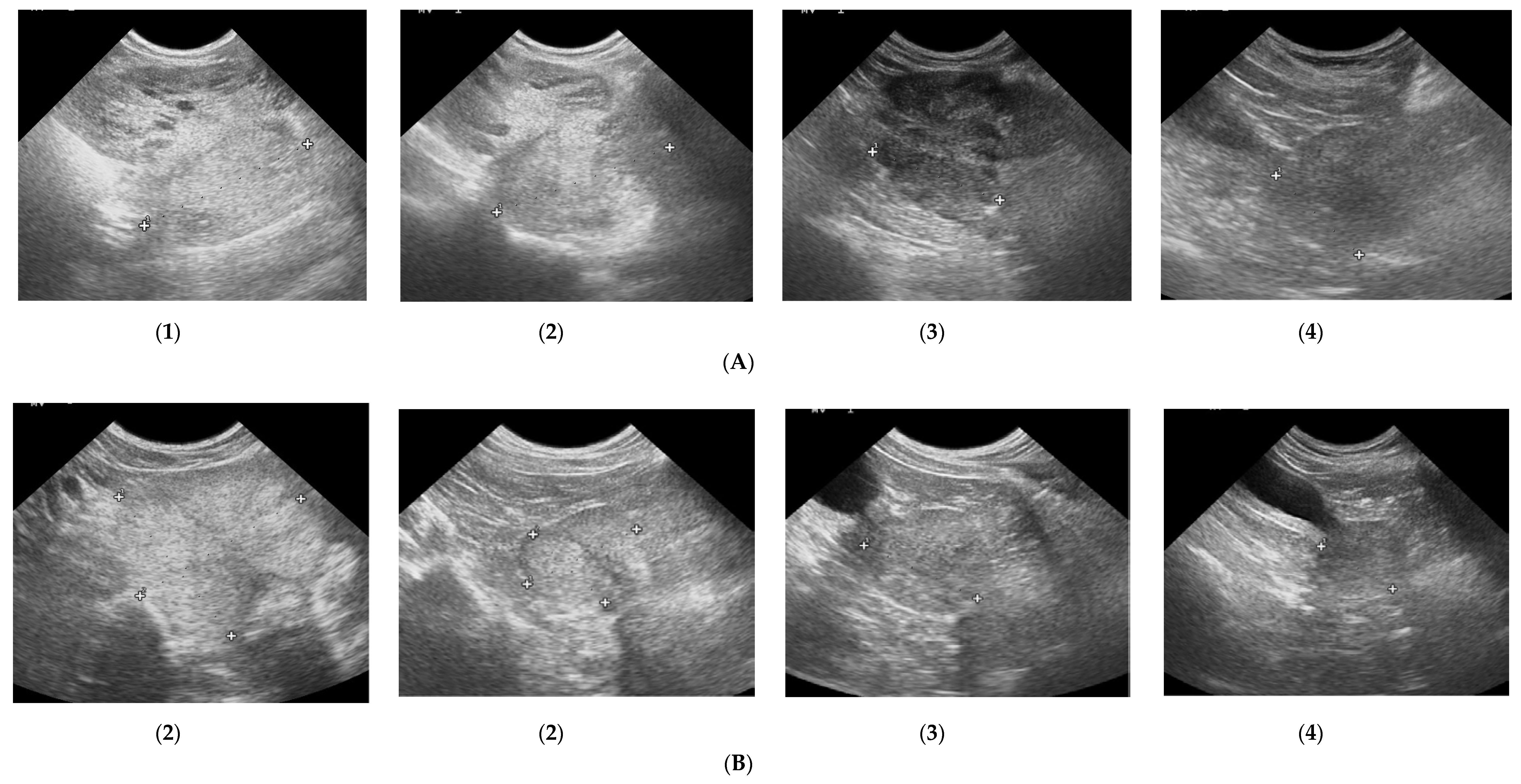
2.3. B-Mode Ultrasonography—Prostate Gland
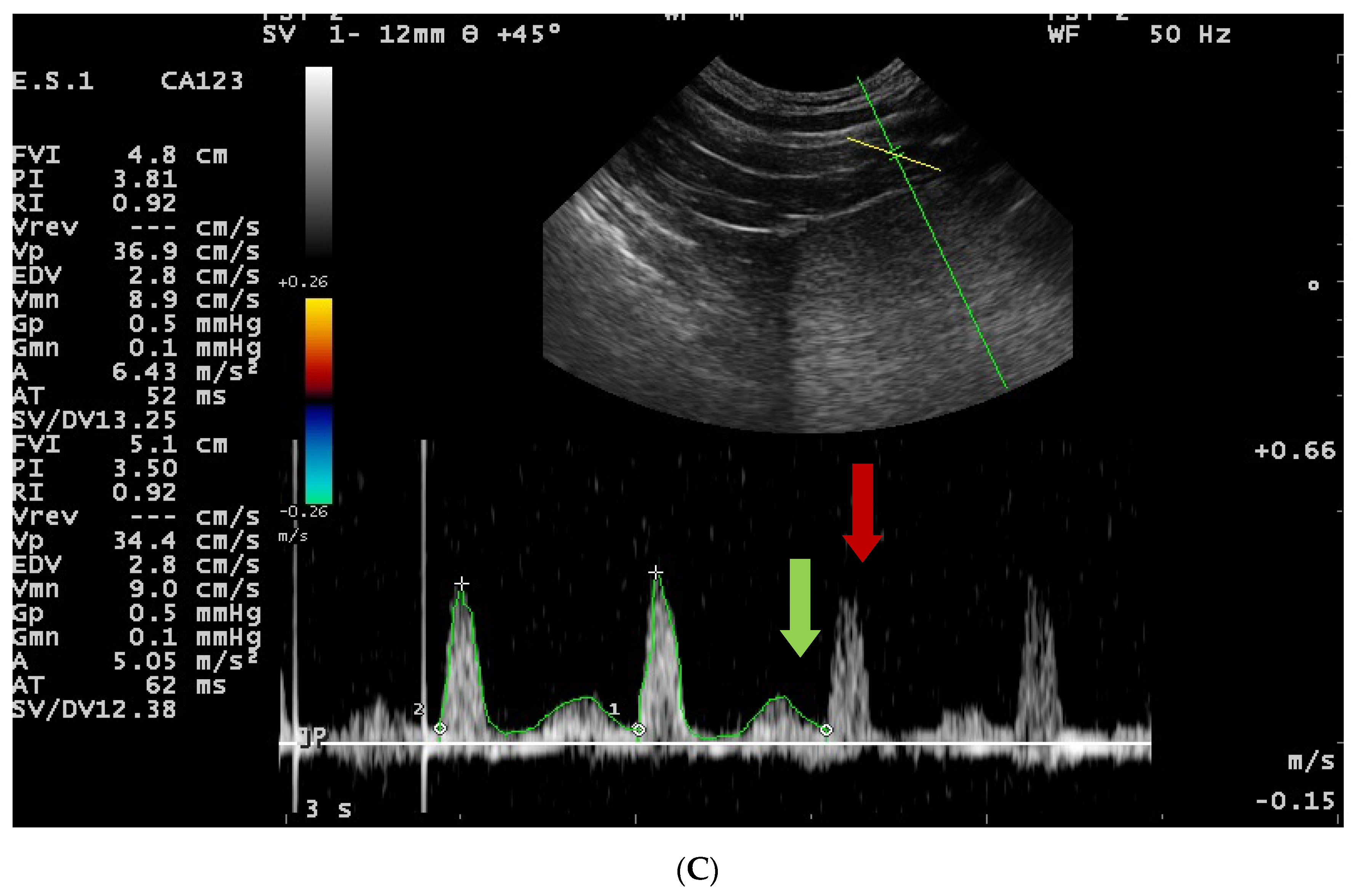
2.4. Doppler Ultrasonography—Prostate Gland
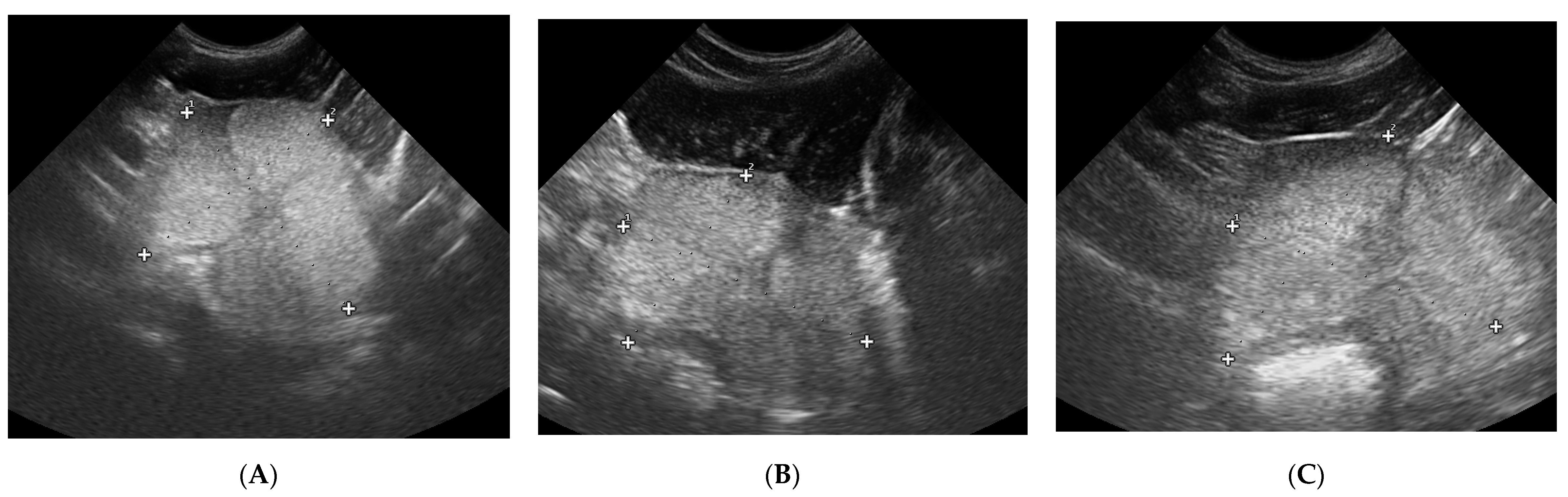
2.5. B-Mode Ultrasonography—Testes
2.6. Doppler Ultrasonography—Testes
2.7. Indications for Inclusion and BPH Confirmation
2.8. Drugs and Schedule
2.9. Study Design
2.10. Statistical Analysis
3. Results
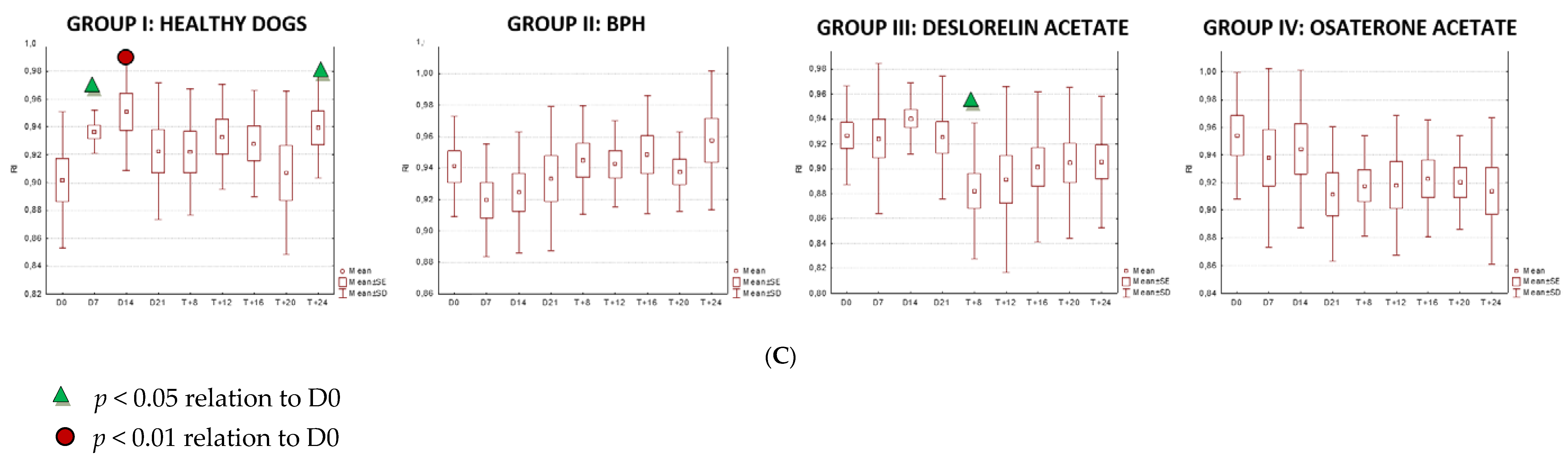
3.1. Prostate B-Mode Appearance
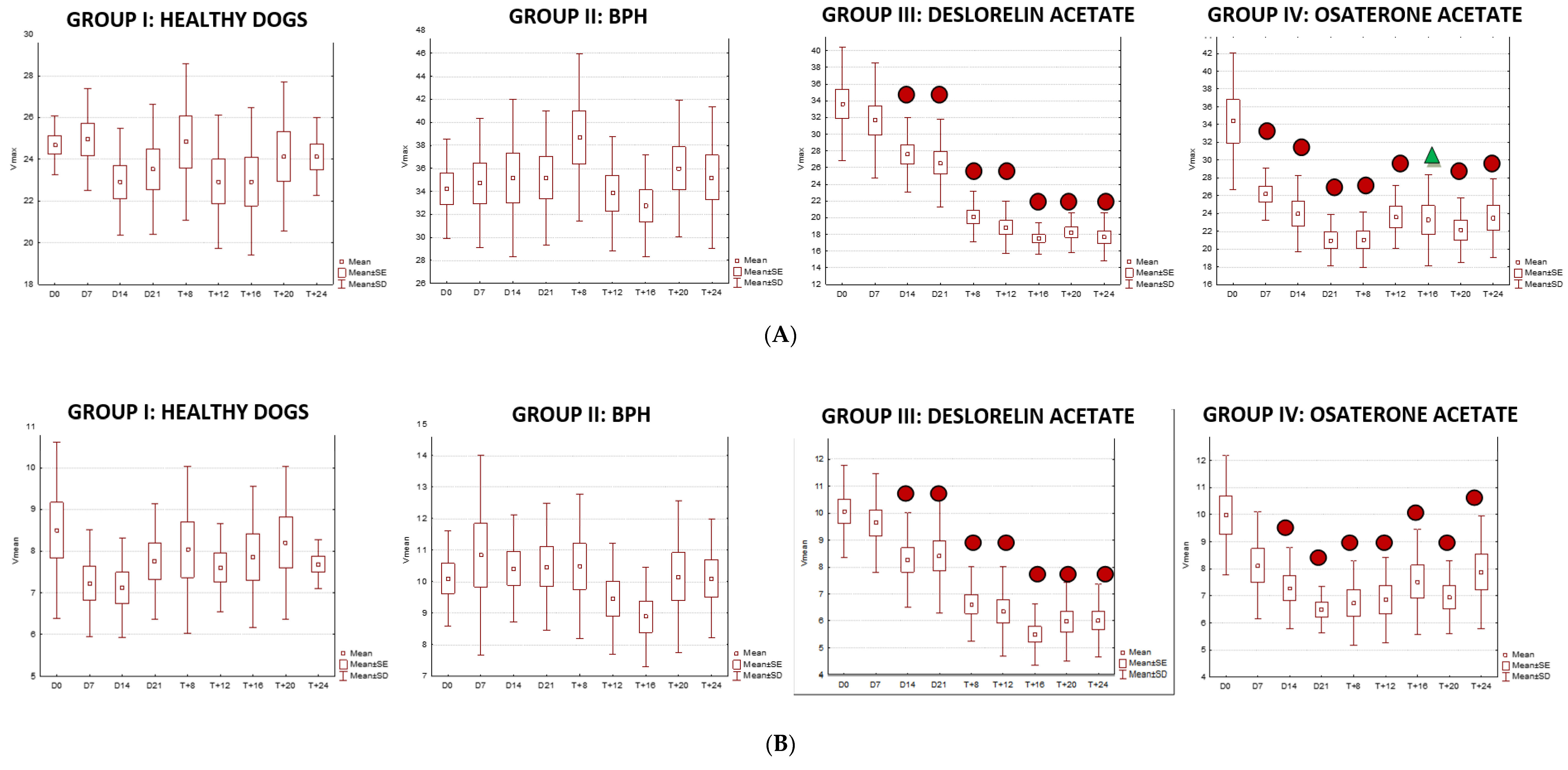
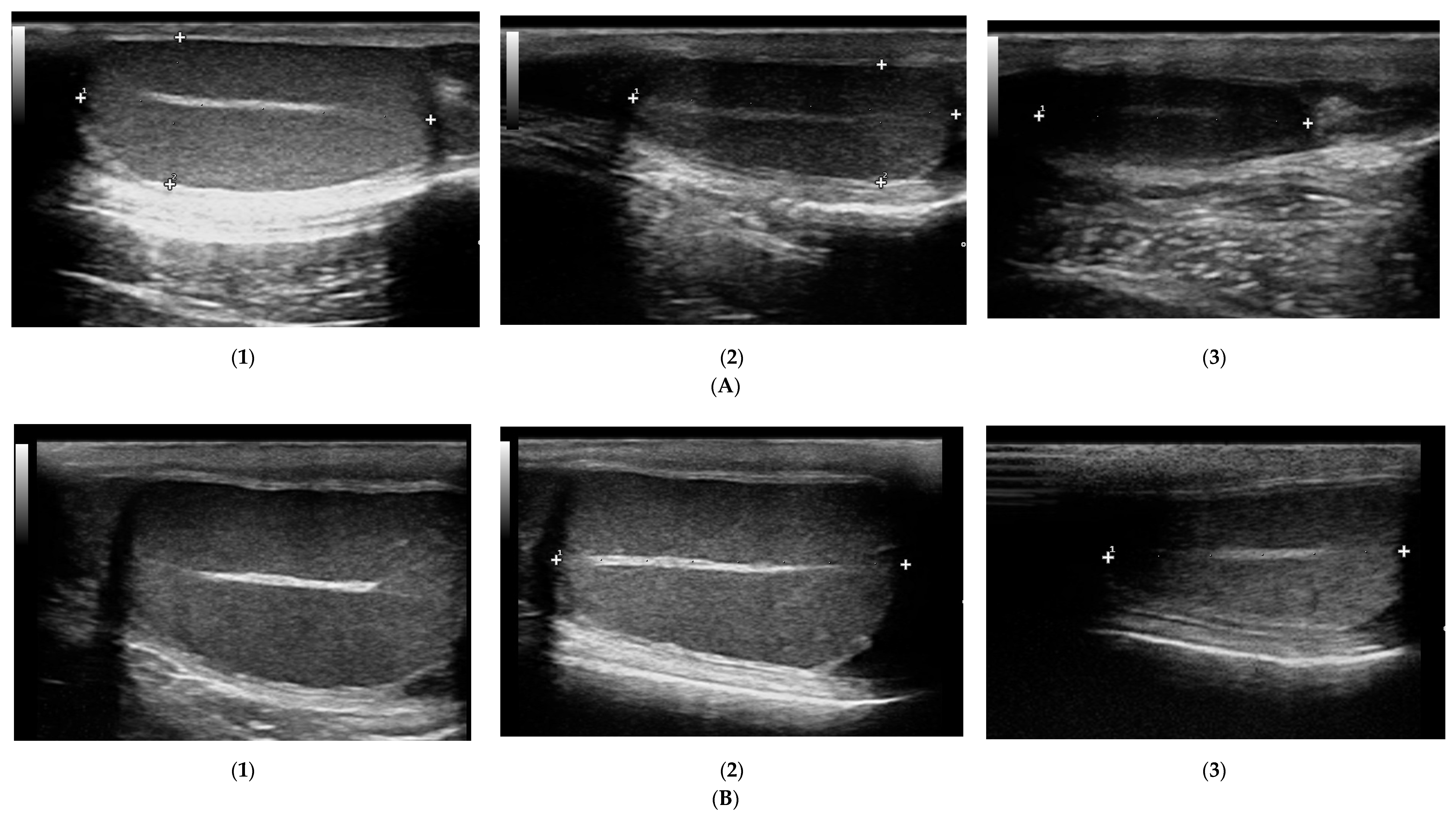
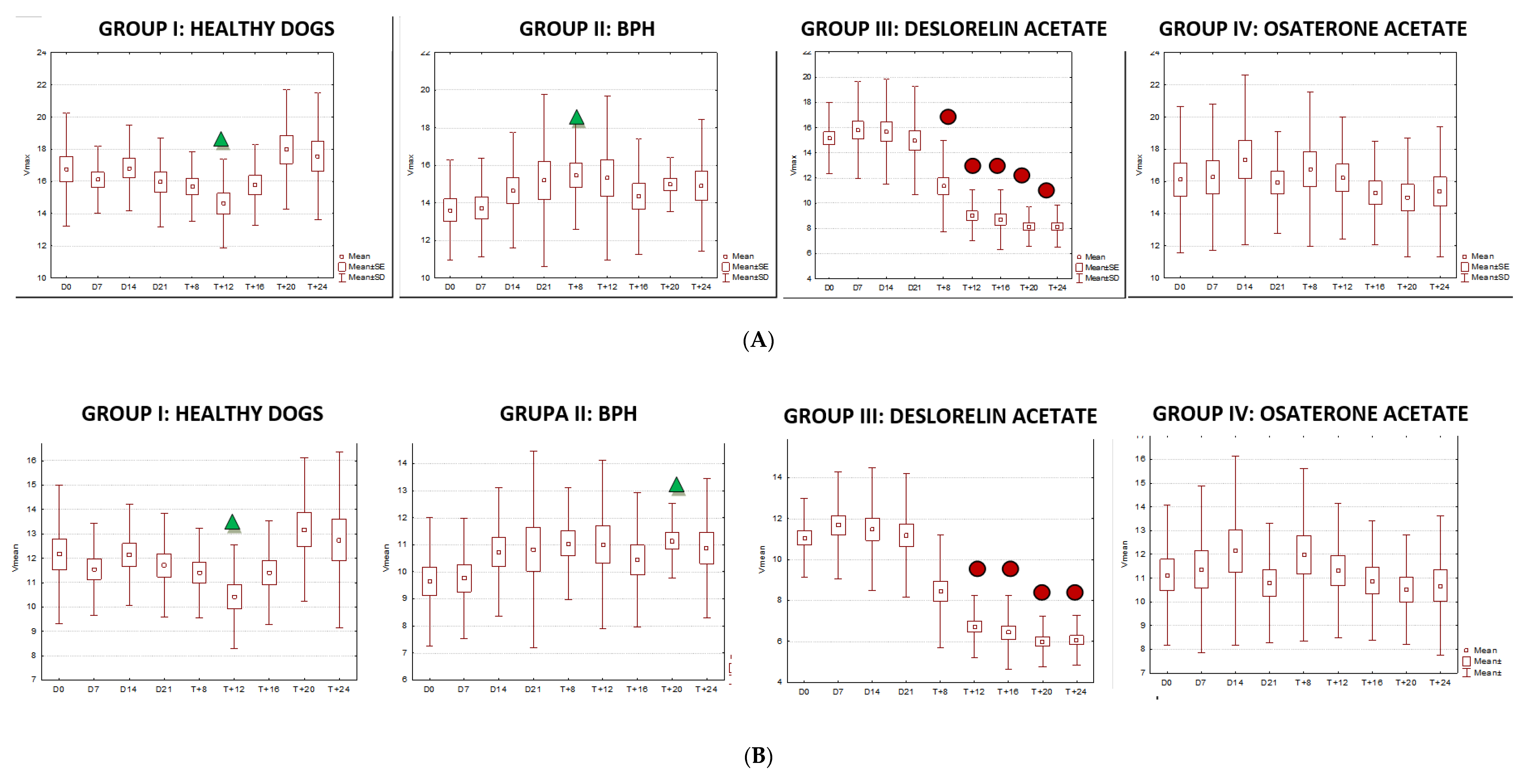
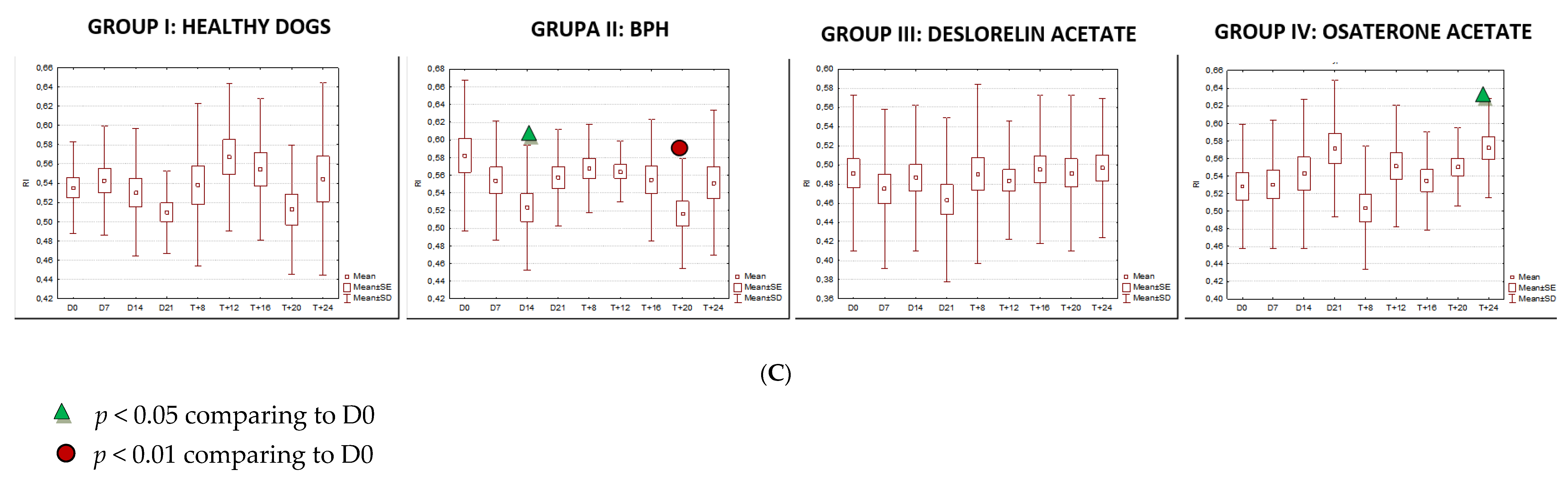
3.2. Doppler Ultrasonography of the Prostatic Artery
3.3. Testicles B-Mode Appearance
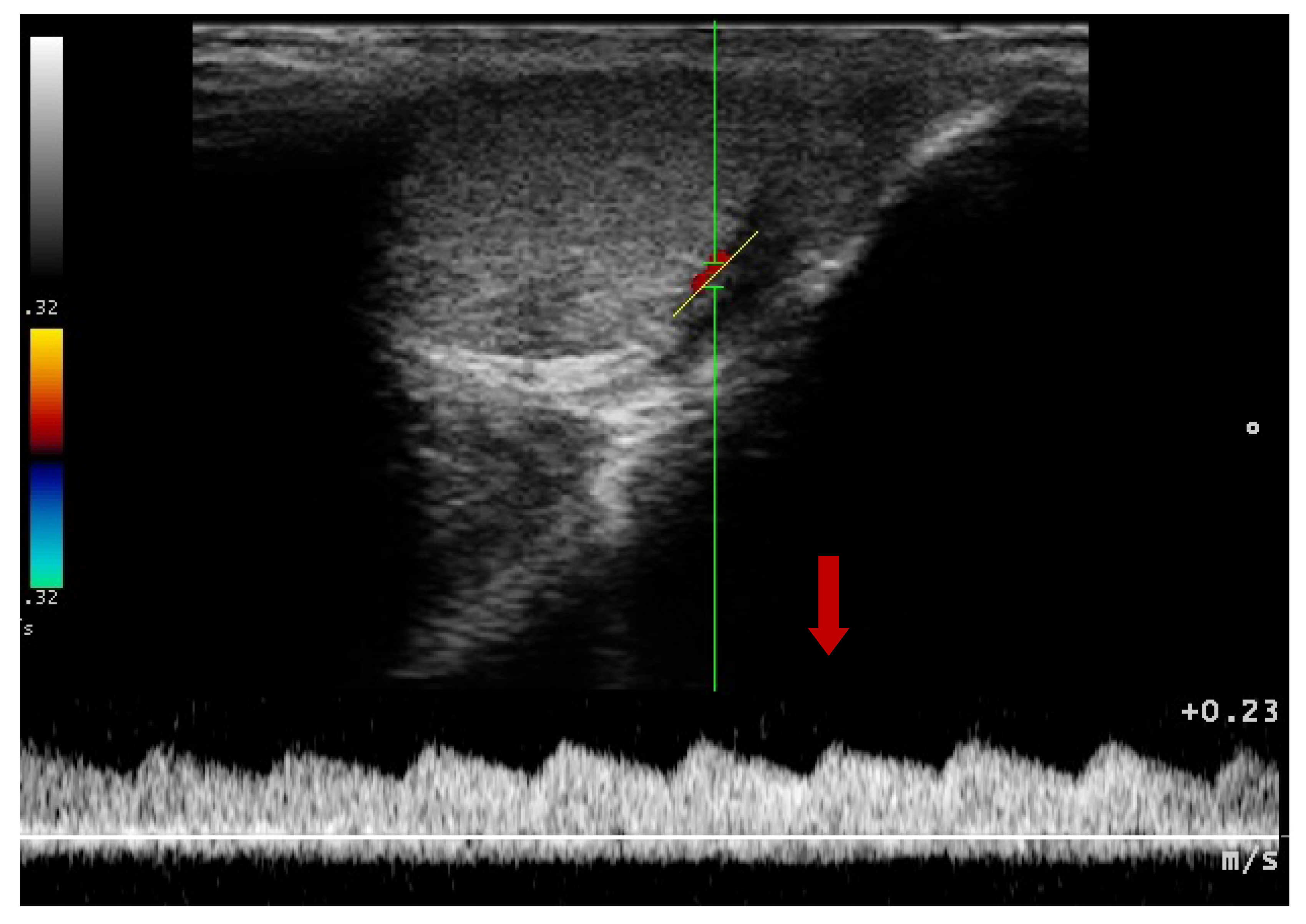
3.4. Doppler Ultrasonography of the Testicular Artery
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mattoon, J.S.; Nyland, T.G. Small Animal Diagnostic Ultrasound. Ch. 17: Prostate and Testes, 3rd ed.; Saunders: Philadephia, PA, USA, 2015; pp. 608–633. [Google Scholar]
- Memon, M.A. Common Causes of Male Dog Infertility. Theriogenology 2007, 68, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Moxon, R.; Bright, L.; Pritchard, B.; Bowen, I.M.; de Souza, M.B.; Da Silva, L.D.; England, G.C. Digital Image Analysis of Testicular and Prostatic Ultrasonographic Echogencity and Heterogeneity in Dogs and the Relation to Semen Quality. Anim. Reprod. Sci. 2015, 160, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Souza, M.B.; Barbosa, C.C.; England, G.C.; Mota Filho, A.C.; Sousa, C.V.; de Carvalho, G.G.; Silva, H.V.R.; Pinto, J.N.; Linhares, C.S.J.; Silva, L.D. Regional Differences of Testicular Artery Blood Flow in Post Pubertal and Pre-Pubertal Dogs. BMC Vet. Res. 2015, 11, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, J.D.; Soler, M.; Lucas, X.; Agut, A. Colour and Pulsed Doppler Ultrasonographic Study of the Canine Testis. Reprod. Domest. Anim. 2012, 47, 655–659. [Google Scholar] [CrossRef]
- Freitas, L.A.; Pinto, J.N.; Silva, H.V.R.; Uchoa, D.C.; Mota Filho, A.C.; Da Silva, L.D.M. Prostatic and Testicular Doppler and Echobiometry in Boxer Dogs. Acta Sci. Vet. 2013, 41, 1121–1129. [Google Scholar]
- Johnston, S.D.; Root-Kustritz, M.V.; Olson, P.N. Disorders of the canine prostate. In Canine and Feline Theriogenology; Johnston, S.D., Root-Kustritz, M.V., Olson, P.N., Eds.; Saunders Co: Philadephia, PA, USA, 2001; pp. 337–355. [Google Scholar]
- Lopate, C. The Problem Stud Dog. Vet. Clin. North Am. Small Anim. Pract. 2012, 42, 469–488. [Google Scholar] [CrossRef]
- Lévy, X.; Niżański, W.; von Heimendahl, A.; Mimouni, P. Diagnosis of Common Prostatic Conditions in Dogs: An Update. Reprod. Domest. Anim. 2014, 49, 50–57. [Google Scholar] [CrossRef]
- Albouy, M.; Sanquer, A.; Maynard, L.; Eun, H.N. Efficacies of Osaterone and Delmadinone in Treatment of Benign Prostatic Hyperplasia in Dogs. Vet. Rec. 2008, 163, 179–183. [Google Scholar] [CrossRef]
- Fontaine, E.; Fontbonne, A. Clinical Use of GnRH Agonists in Canine and Feline Species. Reprod. Domest. Anim. 2011, 46, 344–353. [Google Scholar] [CrossRef]
- Günzel-Apel, A.R.; Mohrke, C.; Polusen Nautrup, C. Colour-Coded and Pulsed Doppler Sonography of the Canine Testis, Epididymis and Prostate Gland: Physiological and Pathological Findings. Reprod. Domest. Anim. 2001, 36, 236–240. [Google Scholar] [CrossRef]
- Gumbsch, P.; Gabler, C.; Holzmann, A. Colour-Coded Duplex Sonography of the Testes of Dogs. Vet. Rec. 2002, 151, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Vignoli, M.; England, G.C.W. B-Mode and Contrast-Enhanced Ultrasonographic Findings in Canine Prostatic Disorders. Reprod. Domest. Anim. 2012, 47, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Polisca, A.; Orlandi, R.; Troisi, A.; Brecchia, G.; Zerani, M.; Boiti, C.; Zelli, R. Clinical Efficacy of the GnRH Agonist (Deslorelin) in Dogs Affected by Benin Prostatic Hyperplasia and Evaluation of Prostatic Blood Flow by Doppler Ultrasound. Reprod. Domest. Anim. 2013, 48, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Atalan, G.; Holt, P.E.; Barr, F.J.; Brown, P.J. Ultrasonographic Estimation of Prostatic Size in Canine Cadavers. Res. Vet. Sci. 1999, 67, 7–15. [Google Scholar] [CrossRef]
- Junaidi, A.; Williamson, P.E.; Martin, G.B.; Blackberry, M.A.; Cummins, J.M.; Trigg, T.E. Dose-Response Studies for Pituitary and Testicular Function in Male Dogs Treated with the GnRH Superagonist, Deslorelin. Reprod. Domest. Anim. 2009, 44, 725–734. [Google Scholar] [CrossRef]
- Niżański, W.; Ochota, M.; Fontaine, C.; Pasikowska, J. Comparison of Clinical Effectiveness of Deslorelin Acetate and Osaterone Acetate in Dogs with Benign Prostatic Hyperplasia. Animals 2020, 10, 1936. [Google Scholar] [CrossRef]
- Root Kustritz, M.V. Collection of tissue and culture samples from the canine reproductive tract. Theriogenology 2006, 66, 567–574. [Google Scholar] [CrossRef]
- Tsutsui, T.; Hori, T.; Shimizu, M.; Orima, H.; Kawakami, E.; Fukuda, S. Regression of Prostatic Hypertrophy by Osaterone Acetate in Dogs. J. Vet. Med. Sci. 2000, 62, 1115–1119. [Google Scholar] [CrossRef]
- Tsuru, N.; Kurita, Y.; Masuda, H.; Suzuki, K.; Fijita, K. Role of Doppler Ultrasound and Resistive Index in Benign Prostatic Hypertrophy. Int. J. Urol. 2002, 9, 427–430. [Google Scholar] [CrossRef]
- Hecht, S. Male Reproductive Tract. In Atlas of Small Animal Ultrasonography, Red; Pennick, D., Andjou, M.A., Eds.; Willey-Blackwell Publishing: Oxford, UK, 2008; pp. 417–445. [Google Scholar]
- Bradley, K. Gruczoł Krokowy. W: Badanie Ultrasonograficzne u Psów I Kotów, Red; I wyd; Barr, F., Gaschen, L., Eds.; Polskie, Galaktyka: Łódź, Poland, 2012; pp. 183–191. [Google Scholar]
- Goericke-Pesch, S.; Wilhelm, E.; Ludwig, C.; Desmoulins, P.O.; Driancourt, M.A.; Hoffmann, B. Evaluation of the Clinical Efficacy of Gonazon Implants in the Treatment of Reproductive Pathologies, Behavioral Problems and Suppression of Reproductive Function in the Male Dog. Theriogenology 2010, 73, 920–926. [Google Scholar] [CrossRef]
- Jurczak, A.; Domoslawska, A.; Janowski, T.; Zdunczyk, S. Treatment of Benign Prostatic Hyperplasia (BPH) in Dogs Using GnRH Agonist Implant Suprelorin®- Preliminary Results. In Proceedings of the 7th EVSSAR Congress, Louvain La Neuve, Belgium, 14–15 May 2010; p. 118. [Google Scholar]
- Ponglowhapan, S.; Lohachit, C. Clinical Use of GnRH Agonist Deslorelin in Benign Prostatic Hyperplasia in Dogs. In Proceedings of the 7th EVSSAR Congress, Louvain La Neuve, Belgium, 14–15 May 2010; p. 140. [Google Scholar]
- Neumaier, C.E.; Martinoli, C.; Derchi, L.E.; Silvestri, E.; Rosenberg, I. Normal Prostate Gland: Examination with Color Doppler US. Radiology 1995, 196, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, T.; Hori, T.; Shimizu, M.; Tatsuzawa, C.; Kawakami, E. Effect of Osaterone Acetate Administration on Prostatic Regression Rate, Peripheral Blood Hormone Levels and Semen Quality in Dogs with Benign Prostatic Hypertrophy. J. Vet. Med. Sci. 2001, 63, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Leaks, E.; Johansson, M.; Widmark, A.; Bergh, A.; Damber, J.E. Decrement of Blood Flow Precedents the Involution of the Ventral Prostate in Rat after Castration. Urol. Res. 1997, 25, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Buttyan, R.; Ghafar, M.A.; Shabsigh, A. The Effects of Androgen Deprivation on the Prostate Gland: Cell Heath Mediatel by Vascular Regression. Curr. Opin. Urol. 2000, 10, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Ochiai, A.; Naya, Y.; Okihara, K.; Ukimura, O.; Miki, T. Doppler Resistive Index in Benign Prostatic Hyperplasia: Correlation with Ultrasonic Appearance of the Prostate and Infravesical Obstruction. Eur. Urol. 2000, 37, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Abdelwahab, O.; El-Barky, E.; Khalil, M.M.; Kamar, A. Evaluation of Resistive Index of Prostatic Blood Flow in Benign Prostatic Hyperplasia. Int. Braz. J. Urol. 2012, 38, 250–257. [Google Scholar] [CrossRef]
- Murakoshi, M.; Inada, R.; Tagawa, M.; Makino, M.; Suzuki, M.; Mieda, M.; Honma, S.; Takezawa, Y.; Yamanaka, H. Inhibitory Influence of a New Steroid al Antiandrogen, TZP-4238, on Prostatic Hyperplasia in the Beagle Dog. Acta Pathol. Jpn. 1992, 42, 151–157. [Google Scholar]
- Pozor, M.A.; McDonnell, S.M. Color Doppler Ultrasound Evaluation of Testicular Blood Flow in Stallions. Theriogenology 2004, 61, 799–810. [Google Scholar] [CrossRef]
- England, G.C.W.; Bright, L.; Pritchard, B.; Bowen, I.M.; De Souza, M.; Silva, L.D.M.; Moxon, R. Canine Reproductive Ultrasound Examination for Predicting Future Sperm Quality. Reprod. Domest. Anim. 2017, 52, 202–207. [Google Scholar] [CrossRef]
- Kutzler, M.; Tyson, R.; Grimes, M.; Timm, K. Determination of Testicular Blood Flow in Camelids Using Vascular Casting and Color Pulsed-Wave Doppler Ultrasonography. Vet. Med. Int. 2011, 2011. [Google Scholar] [CrossRef]
- Biagiotti, G.; Cavallini, G.; Modenini, G.; Vitali, G.; Gianoroli, L. Spermatogenesis and Spectral Echo-Colour Doppler Traces from the Main Testicular Artery. BJU Int. 2002, 90, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Paltiel, H.J.; Rupich, R.C.; Babcock, D. Maturational Changes in Arterial Impendance of the Normal Testis in Boys: Doppler Sonographic Study. AJR 1994, 163, 1189–1193. [Google Scholar] [CrossRef] [PubMed]
- Wielgos, M.; Bablok, L.; Fracki, S.; Marianowski, L. Doppler Flow Measurements in Testicular Artery of Aging Males. Ginekol. Pol. 1998, 69, 537–540. [Google Scholar] [PubMed]
- Jee, W.H.; Choe, J.K.; Byun, J.Y.; Shinn, K.S.; Hwang, T.K. Resistive index of the intrascrotal artery in scrotal inflammatory disease. Acta Radiol. 1997, 38, 1026–1030. [Google Scholar] [CrossRef] [PubMed]
- Lee, F.T.; Winter, D.B.; Madsen, F.A.; Zagzebski, J.A.; Pozniak, M.A.; Chosy, S.G.; Scanlan, K.A. Conventional Color Doppler Velocity Sonography for the Diagnosis of Acute Experimental Torsion of the Spermatic Cord. Am. J. Radiol. 1996, 167, 785–790. [Google Scholar]

| Control Group I—Healthy Dogs | Control Group II—Dogs with BPH | |||||||||||||||||||||
| Background Echotexture | Parenchymal Stippling | General Apperance | Focal Changes | Background Echotexture | Parenchymal Stippling | General Apperance | Focal Changes | |||||||||||||||
| N | HI | HY | R | C | I | HO | HE | CY | M | fHY | N | HI | HY | R | C | I | HO | HE | CY | M | fHY | |
| D0 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| D7 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| D14 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| D21 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| W+8 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| W+12 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| W+16 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| W+20 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| W+24 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 0 | 0 | 0 | 100% | 0 | 30% | 70% | 0 | 0 | 100% | 90% | 10% | 0 |
| Experimental Group III—Deslorelin Acetate | Experimental Group IV—Osaterone Acetate | |||||||||||||||||||||
| Background echotexture | Parenchymal stippling | General apperance | Focal changes | Background echotexture | Parenchymal stippling | General apperance | Focal changes | |||||||||||||||
| N | HI | HY | R | C | I | HO | HE | CY | M | fHY | N | HI | HY | R | C | I | HO | HE | CY | M | fHY | |
| D0 | 0 | 100% | 0 | 7% | 93% | 0 | 0 | 100% | 100% | 0 | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 100% | 0 | 0 |
| D7 | 0 | 100% | 0 | 20% | 80% | 0 | 0 | 100% | 100% | 0 | 0 | 0 | 100% | 0 | 0 | 100% | 0 | 0 | 100% | 100% | 0 | 0 |
| D14 | 0 | 100% | 0 | 20% | 80% | 0 | 0 | 100% | 100% | 0 | 0 | 20% | 80% | 0 | 10% | 90% | 0 | 20% | 80% | 80% | 0 | 0 |
| D21 | 0 | 100% | 0 | 27% | 73% | 0 | 0 | 100% | 100% | 0 | 0 | 40% | 60% | 0 | 50% | 50% | 0 | 50% | 50% | 50% | 0 | 0 |
| W+8 | 40% | 33% | 27% | 87% | 6.5% | 6.5% | 77% | 33% | 33% | 0 | 0 | 40% | 60% | 0 | 50% | 50% | 0 | 60% | 40% | 40% | 0 | 0 |
| W+12 | 40% | 0 | 60% | 87% | 0 | 13% | 93% | 7% | 7% | 0 | 0 | 40% | 60% | 0 | 50% | 50% | 0 | 60% | 40% | 40% | 0 | 0 |
| W+16 | 7% | 0 | 93% | 20% | 0 | 80% | 100% | 0 | 0 | 0 | 0 | 30% | 70% | 0 | 30% | 70% | 0 | 50% | 50% | 50% | 0 | 0 |
| W+20 | 7% | 0 | 93% | 20% | 0 | 80% | 100% | 0 | 0 | 0 | 0 | 20% | 80% | 0 | 30% | 70% | 0 | 50% | 50% | 50% | 0 | 0 |
| W+24 | 7% | 0 | 93% | 20% | 0 | 80% | 100% | 0 | 0 | 0 | 0 | 30% | 70% | 0 | 30% | 70% | 0 | 30% | 70% | 0 | 0 | 0 |
| Group | D0 | D7 | D14 | D21 | W + 8 | W + 12 | W + 16 | W + 20 | W + 24 |
|---|---|---|---|---|---|---|---|---|---|
| I | |||||||||
| Mean | 15.58 | 16.06 | 14.82 | 14.84 | 16.12 | 15.77 | 16.26 | 16.87 | 17.5 |
| SD | 3.48 | 3.53 | 2.97 | 2.65 | 2.46 | 2.8 | 3.62 | 3.42 | 3.63 |
| Range | 11.55–20.8 | 12.46–21.61 | 11.35–20.34 | 11.49–19.35 | 12.61–19.09 | 12.55–19.21 | 12.03–22.11 | 12.41–23.88 | 13.53–24.6 |
| II | |||||||||
| Mean | 18.26 | 19.04 | 19.1 | 19.24 | 19.05 | 20.67 | 21.47 | 21.93 | 20.44 |
| SD | 4.1 | 4.72 | 4.23 | 4.7 | 4 | 5.83 | 6.63 | 7.01 | 6.5 |
| Range | 12.82–23.32 | 12.4–26.7 | 12.64–23.84 | 11.88–26.91 | 13.47–24.63 | 13.93–29.84 | 11.97–36.55 | 12.24–34.5 | 11.75–31.52 |
| III | |||||||||
| Mean | 39.39 | 39.11 | 32.05 | 28.35 | 14.65 * | 11.62 * | 10.7 * | 9.6 * | 9.26 * |
| SD | 31.26 | 26.15 | 19.16 | 20.62 | 7.9 | 5.4 | 3.41 | 1.72 | 1.47 |
| Range | 11.13–115.91 | 16.01–106.49 | 12.88–80.79 | 11.76–92.57 | 8.44–40.94 | 8.5–30.58 | 7.82–21.83 | 7.88–14.52 | 7.7–12.61 |
| IV | |||||||||
| Mean | 24.07 | 20.74 | 14.7 & | 13.21 * | 13.68 * | 13.65 * | 13.79 * | 14.7 & | 18.57 |
| SD | 9.82 | 6.96 | 4.1 | 2.8 | 4.09 | 3.72 | 4.12 | 5.3 | 9.55 |
| Range | 10.29–38.48 | 10.91–32.7 | 8.42–22.5 | 8.3–16.78 | 8.48–19.83 | 8.73–21.53 | 8–19.5 | 8.5–25.11 | 7.61–38.3 |
| Groups Compared | D7 | D14 | D21 | W + 8 | W + 12 | W + 16 | W + 20 | W + 24 |
|---|---|---|---|---|---|---|---|---|
| I C–II C | 0.92020 | 0.04165 & | 0.18131 | 0.62421 | 0.01137 & | 0.08641 | 0.14046 | 0.95508 |
| II C–III T | 0.91166 | 0.03259 & | 0.00279 * | <0.00001 * | 0.00003 * | 0.00003 * | <0.00001 * | <0.00001 * |
| II C–IV T | 0.01195 & | <0.00001 * | <0.00001 * | <0.00001 * | 0.00016 * | 0.00016 * | 0.00016 * | 0.06155 |
| III T–IV T | 0.02650 & | 0.00116 * | 0.01486 & | 0.07162 | 0.00616 * | 0.00141 * | 0.00072 * | 0.00024 * |
| Groups | D7 | D14 | D21 | W + 8 | W + 12 | W + 16 | W + 20 | W + 24 |
|---|---|---|---|---|---|---|---|---|
| III | 0.85193 | 0.41862 | 0.37251 | 0.00264 * | 0.04882 * | 0.46792 | 0.41862 | 0.46792 |
| IV | 0.39270 | 0.02959 * | 0.35476 | 0.76735 | 0.98397 | 0.93458 | 0.67268 | 0.27672 |
| Group | D0 | D7 | D14 | D21 | W+8 | W+12 | W+16 | W+20 | W+24 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SPV | Vmean | SPV | Vmean | SPV | Vmean | SPV | Vmean | SPV | Vmean | SPV | Vmean | SPV | Vmean | SPV | Vmean | SPV | Vmean | |
| I | ||||||||||||||||||
| Mean | 24.68 a | 8.5 | 24.95 a | 7.23 a | 22.91 a | 7.12 a | 23.51 a | 7.75 a | 24.82 a | 8.04 a | 22.93 a | 7.60 a | 22.94 a | 7.86 a | 24.13 a | 8.21 a | 24.82 a | 8.04 a |
| SD | 1.4 | 2.12 | 2.44 | 1.29 | 2.56 | 1.19 | 3.11 | 1.39 | 3.743 | 2.01 | 3.19 | 1.055 | 3.53 | 1.69 | 3.58 | 1.84 | 3.74 | 2.01 |
| SE | 23.1–27.28 | 6.34–13.6 | 21.3–29.53 | 6.02–10.53 | 18.65–27.5 | 5.78–9.24 | 16.18–26.43 | 5.44–10.6 | 30.53–18.03 | 3.7–11.03 | 17.95–25.34 | 6.05–9.74 | 15.9–27.95 | 5.37–11.53 | 19.48–32.27 | 5.48–11.47 | 20.98–26.02 | 6.7–8.48 |
| II | ||||||||||||||||||
| Mean | 34.22 b | 10.11 | 34.71 b | 10.84 b | 35.16 b | 10.43 b | 35.17 b | 10.48 b | 38.69 b | 10.49 b | 33.82 b | 9.46 b | 32.74 b | 8.88 a | 36.00 b | 10.16 b | 35.21 b | 10.10 b |
| SD | 4.34 | 1.51 | 5.60 | 3.18 | 6.85 | 1.7 | 5.81 | 2.014 | 7.29 | 2.3 | 4.97 | 1.756 | 4.41 | 1.57 | 5.920 | 2.41 | 6.14 | 1.87 |
| SE | 24.88–38.88 | 7.13–11.76 | 25.78–44.38 | 5.58–15.23 | 25.54–45.62 | 7.9–12.94 | 27.9–39.6 | 7.1–13.73 | 31.48–52.72 | 7.84–15.84 | 27.9–46.23 | 8.18–13.9 | 29–44.7 | 6.13–11.26 | 25.88–47.47 | 5.85–12.37 | 26.98–45.7 | 7.7–14.35 |
| III | ||||||||||||||||||
| Mean | 33.65 cb | 10.06 | 31.66 cb | 9.64 cb | 27.57 c | 8.27 a | 26.56 ac | 8.42 a | 20.13 c | 6.63 a | 18.85 c | 6.35 c | 17.52 c | 5.50 b | 18.24 c | 5.97 c | 17.7 c | 6.02 c |
| SD | 6.83 | 1.70 | 6.87 | 1.85 | 4.47 | 1.75 | 5.23 | 2.14 | 3.05 | 1.38 | 3.15 | 1.66 | 1.85 | 1.14 | 2.39 | 1.454 | 2.91 | 1.36 |
| SE | 21.22–45.37 | 7–13.37 | 19.98–45.37 | 6.4–12.73 | 20.9–34.03 | 5.78–11.73 | 18.55–33.87 | 5.84–13.76 | 16.3–27.8 | 5.08–9.36 | 14.7–25.83 | 4.52–11.28 | 15.24–19.05 | 3.22–8.22 | 14.62–21.93 | 3.84–9.35 | 14.36–25.08 | 3.73–7.64 |
| IV | ||||||||||||||||||
| Mean | 34.36 dbc | 9.98 | 26.16 a | 8.12 a | 23.98 ac | 7.28 a | 20.99 ad | 6.5 c | 21.05 dc | 6.74 a | 23.59 a | 6.85 ac | 23.25 a | 7.52 a | 22.14 a | 6.95 ac | 23.48 a | 7.87 a |
| SD | 7.66 | 2.21 | 2.91 | 1.98 | 4.32 | 1.5 | 2.91 | 0.86 | 3.13 | 1.57 | 3.55 | 1.58 | 5.13 | 1.94 | 3.61 | 1.34 | 4.43 | 2.09 |
| SE | 17.8–42.87 | 7.5–12.23 | 22.7–31.29 | 6.3–11.57 | 17.27–31.96 | 6.13–10.25 | 16.36–26.8 | 5.36–8.26 | 14.87–25.3 | 3.83–8.92 | 18.96–25.96 | 5.28–9.98 | 17.92–31.13 | 5.34–10.88 | 17.46–29.4 | 5.7–10.27 | 16–29.92 | 3.7–11.22 |
| Group | D0 | D7 | D14 | D21 | W + 8 | W + 12 | W + 16 | W + 20 | W + 24 |
|---|---|---|---|---|---|---|---|---|---|
| I | |||||||||
| Mean | 8.43 | 10.51 | 8.01 | 7.86 | 8.62 | 8.24 | 8.24 | 8.25 | 8.45 |
| SD | 3.49 | 7.59 | 3.02 | 3.03 | 3.23 | 2.49 | 3.43 | 3.2 | 3.34 |
| Range | 5.06–15.07 | 5.33–29.3 | 5.0–13.62 | 5.1–13.68 | 5.85–14.61 | 5.68–13.1 | 4.98–14.12 | 4.92–13.14 | 5.28–13.65 |
| II | |||||||||
| Mean | 4.47 | 4.71 | 4.7 | 4.66 | 4.98 | 4.87 | 4.99 | 4.9 | 5.27 |
| SD | 2.2 | 2.81 | 2.69 | 2.89 | 3.38 | 2.8 | 3.04 | 2.7 | 3.15 |
| Range | 1.56–8.24 | 1.65–10.29 | 1.25–9.51 | 0.85–10.42 | 0.85–12.1 | 1.05–10.41 | 1.4–11.45 | 0.98–9.82 | 1.4–12.01 |
| III | |||||||||
| Mean | 10.22 | 10.75 | 10.12 | 9.23 | 6.97 | 4.92 * | 3.69 * | 3.85 * | 3.68 * |
| SD | 4.93 | 5.67 | 4.72 | 4.18 | 4.3 | 3.85 | 2.18 | 2.5 | 2.26 |
| Range | 3.58–22.8 | 3.6–24.85 | 3.6–20.12 | 3.18–17.79 | 1.41–15.38 | 1.2–17.07 | 0.84–8.9 | 0.88–9.28 | 0.79–8.4 |
| IV | |||||||||
| Mean | 11.61 | 10.43 | 10.53 | 10.68 | 10.47 | 11.25 | 10.6 | 10.23 | 11.28 |
| SD | 7.03 | 5.95 | 6.53 | 6.37 | 6.44 | 6.94 | 6.58 | 6.05 | 7.78 |
| Range | 2.02–24.22 | 2.43–20.42 | 2.4–23.8 | 2.83–21.15 | 2.1–20.23 | 2.7–17.63 | 2.02–23.07 | 2.0–21.04 | 1.93–21.42 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niżański, W.; Ochota, M.; Fontaine, C.; Pasikowska, J. B-Mode and Doppler Ultrasonographic Findings of Prostate Gland and Testes in Dogs Receiving Deslorelin Acetate or Osaterone Acetate. Animals 2020, 10, 2379. https://doi.org/10.3390/ani10122379
Niżański W, Ochota M, Fontaine C, Pasikowska J. B-Mode and Doppler Ultrasonographic Findings of Prostate Gland and Testes in Dogs Receiving Deslorelin Acetate or Osaterone Acetate. Animals. 2020; 10(12):2379. https://doi.org/10.3390/ani10122379
Chicago/Turabian StyleNiżański, Wojciech, Małgorzata Ochota, Christelle Fontaine, and Joanna Pasikowska. 2020. "B-Mode and Doppler Ultrasonographic Findings of Prostate Gland and Testes in Dogs Receiving Deslorelin Acetate or Osaterone Acetate" Animals 10, no. 12: 2379. https://doi.org/10.3390/ani10122379
APA StyleNiżański, W., Ochota, M., Fontaine, C., & Pasikowska, J. (2020). B-Mode and Doppler Ultrasonographic Findings of Prostate Gland and Testes in Dogs Receiving Deslorelin Acetate or Osaterone Acetate. Animals, 10(12), 2379. https://doi.org/10.3390/ani10122379






