Utilization of Methionine Sources for Growth and Met+Cys Deposition in Broilers
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
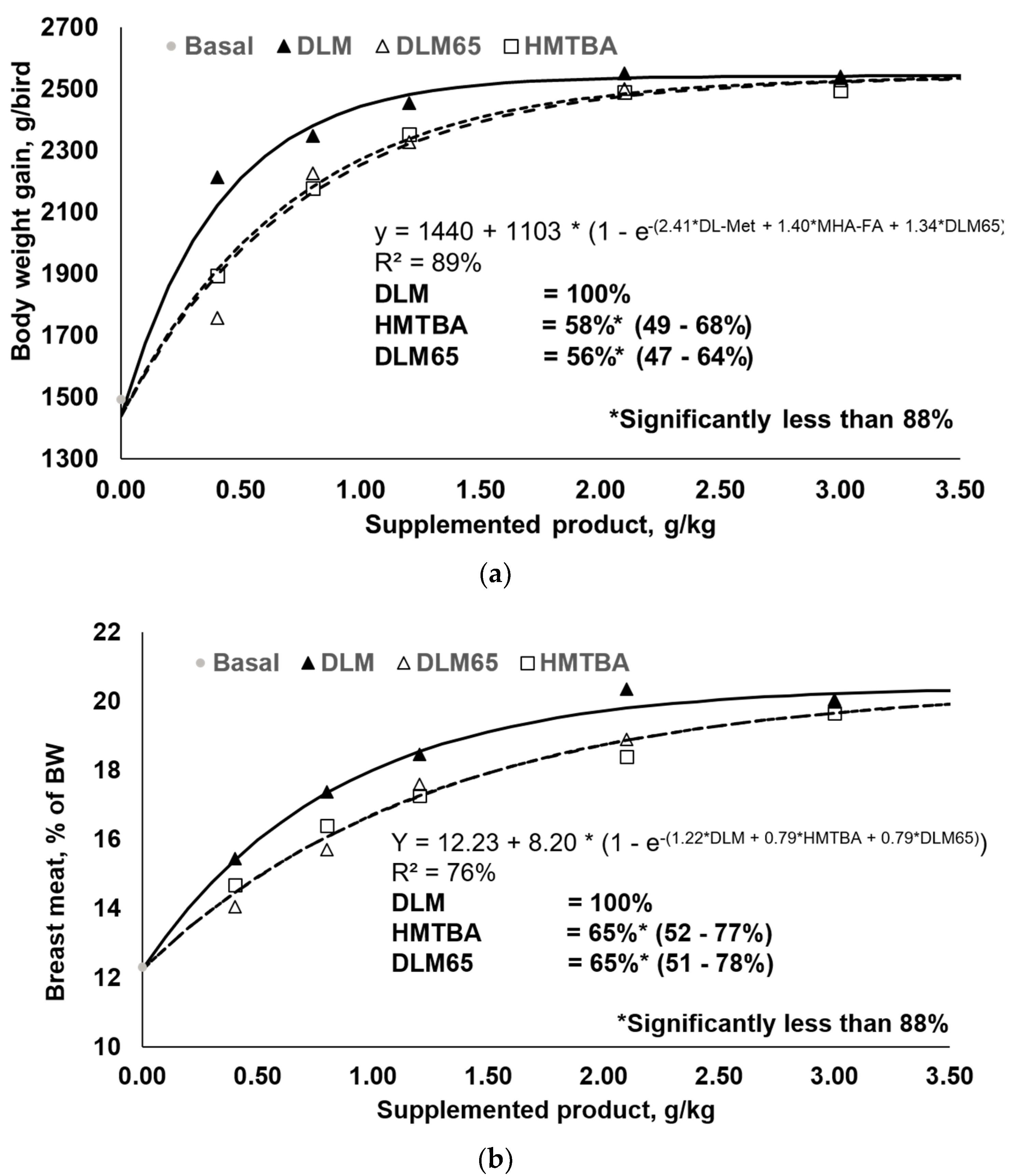
3.1. Performance and Relative Bioavailability Determination
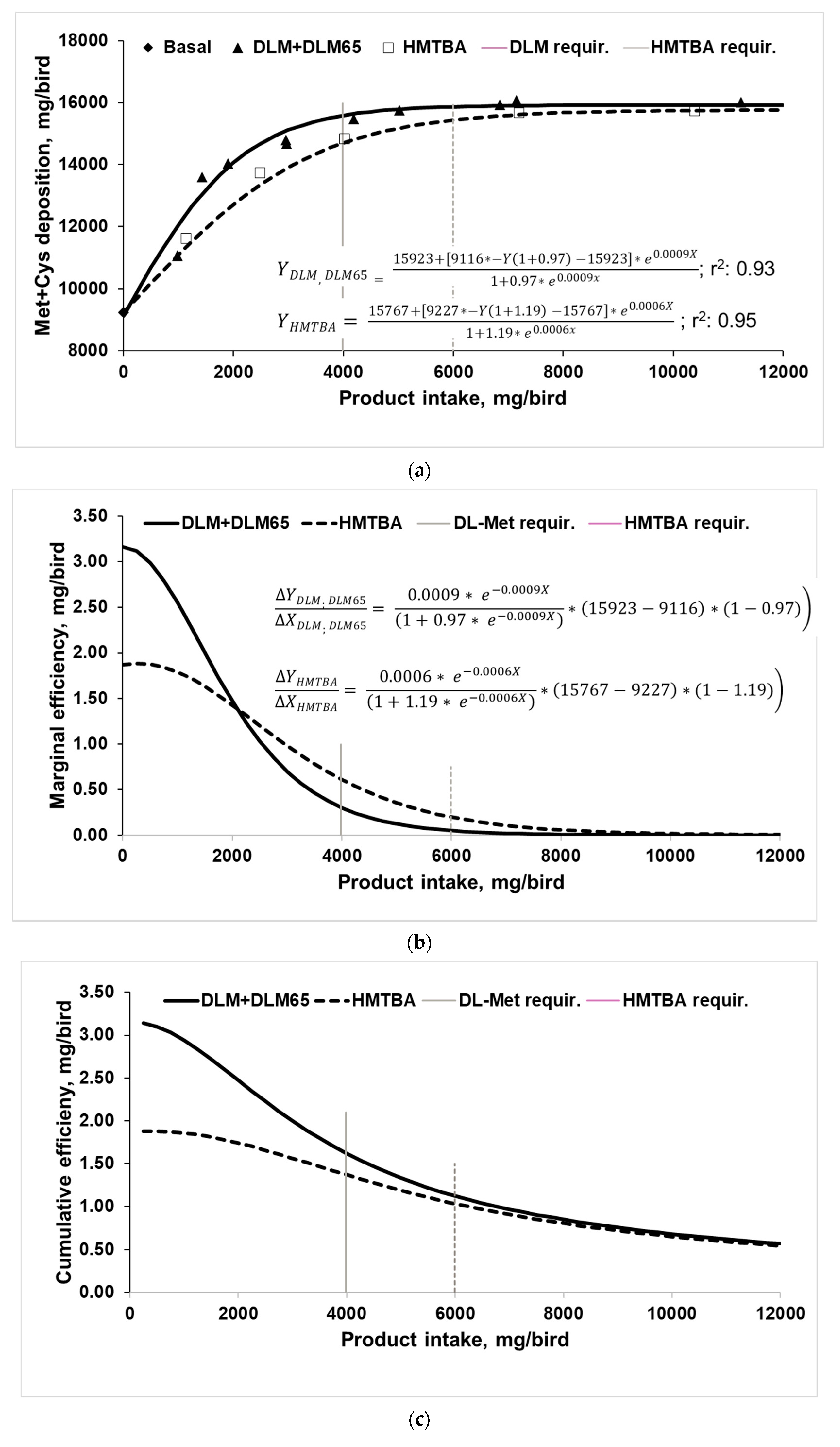
3.2. Utilization of Methionine Sources for Met+Cys Deposition in Body Protein
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Willke, T. Methionine production—A critical review. Appl. Microbiol. Biotechnol. 2014, 98, 9893–9914. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Htoo, J.K.; Liao, S.F. Methionine nutrition in swine and related monogastric animals: Beyond protein biosynthesis. Anim. Feed Sci. Technol. 2020, 268, 114608. [Google Scholar] [CrossRef]
- Sauer, N.; Emrich, K.; Piepho, H.-P.; Lemme, A.; Redshaw, M.S.; Mosenthin, R. Meta-analysis of the relative efficiency of methionine-hydroxy-analogue-free-acid compared with DL-methionine in broilers using nonlinear mixed models. Poult. Sci. 2008, 87, 2023–2031. [Google Scholar] [CrossRef] [PubMed]
- Jansman, A.J.M.; Kan, C.A.; Wiebenga, J. Comparison of the Biological Efficacy of DL-Methionine and Hydroxy-4-Methylthiobutanoic Acid (HMB) in Pigs and Poultry; Centraal Veefoederbureau: Lelystad, The Netherlands, 2003. [Google Scholar]
- Rychen, G.; Aquilina, G.; Azimonti, G.; Bampidis, V.; de Lourdes Bastos, M.; Bories, G.; Chesson, A.; Cocconcelli, P.S.; Flachowsky, G.; Gropp, J.; et al. Safety and efficacy of hydroxy analogue of methionine and its calcium salt (ADRY+®) for all animal species. Efsa J. 2018, 16, e05198. [Google Scholar] [CrossRef] [PubMed]
- Lemme, A. Letter to the editor. World’s Poult. Sci. J. 2020, 76, 168–170. [Google Scholar] [CrossRef]
- Vázquez-Añón, M.; Kratzer, D.; González-Esquerra, R.; Yi, I.G.; Knight, C.D. A multiple regression model approach to contrast the performance of 2-hydroxy-4-methylthio butanoic acid and DL-methionine supplementation tested in broiler experiments and reported in the literature. Poult. Sci. 2006, 85, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Añón, M.; González-Esquerra, R.; Saleh, E.; Hampton, T.; Ritcher, S.; Firman, J.; Knight, C.D. Evidence for 2-hydroxy-4(methylthio) butanoic acid and DL-methionine having different dose responses in growing broilers. Poult. Sci. 2006, 85, 1409–1420. [Google Scholar] [CrossRef]
- Kratzer, D.D.; Littell, R.C. Appropriate Statistical Methods to Compare Dose Responses of Methionine Sources. Poult. Sci. 2006, 85, 947–954. [Google Scholar] [CrossRef]
- Piepho, H.P. A cautionary note on appropriate statistical methods to compare dose responses of methionine sources. Poult. Sci. 2006, 85, 1511–1512. [Google Scholar] [CrossRef]
- Lemme, A.; Hoehler, D.; Brennan, J.J.; Mannion, P.F. Relative effectiveness of methionine hydroxy analog compared to DL-methionine in broiler chickens. Poult. Sci. 2002, 81, 838–845. [Google Scholar] [CrossRef]
- Hoehler, D.; Lemme, A.; Jensen, S.K.; Vieira, S.L. Relative Effectiveness of Methionine Sources in Diets for Broiler Chickens. J. Appl. Poult. Res. 2005, 14, 679–693. [Google Scholar] [CrossRef]
- Hoehler, D.; Mack, S.; Jansman, A.J.M.; de Jong, J. Regression analysis to assess the bioefficacy of different methionine sources in broiler chickens. In Proceedings of the 26th Poultry Science Symposium, Peebles, UK, June 1999. [Google Scholar]
- Elwert, C.; Fernandes, E.d.A.; Lemme, A. Biological Effectiveness of Methionine Hydroxy-analogue Calcium Salt in Relation to DL-Methionine in Broiler Chickens. Asian Australas. J. Anim. Sci. 2008, 21, 1506–1515. [Google Scholar] [CrossRef]
- Littell, R.C.; Henry, P.R.; Lewis, A.J.; Ammerman, C.B. Estimation of relative bioavailability of nutrients using SAS procedures. J. Anim. Sci. 1997, 75, 2672–2683. [Google Scholar] [CrossRef] [PubMed]
- Potter, L.M. Bioavailability of phosphorus from various phosphates based on body weight and toe ash measurements. Poult. Sci. 1988, 67, 96–102. [Google Scholar] [CrossRef]
- Potter, L.M.; Potchanakorn, M.; Ravindran, V.; Kornegay, E.T. Bioavailability of phosphorus in various phosphate sources using body weight and toe ash as response criteria. Poult. Sci. 1995, 74, 813–820. [Google Scholar] [CrossRef]
- Fernandes, J.I.; Lima, F.R.; Mendonça, C.X.; Mabe, I.; Albuquerque, R.; Leal, P.M. Relative bioavailability of phosphorus in feed and agricultural phosphates for poultry. Poult. Sci. 1999, 78, 1729–1736. [Google Scholar] [CrossRef]
- Boling, S.D.; Edwards, H.M.; Emmert, J.L.; Biehl, R.R.; Baker, D.H. Bioavailability of iron in cottonseed meal, ferric sulfate, and two ferrous sulfate by-products of the galvanizing industry. Poult. Sci. 1998, 77, 1388–1392. [Google Scholar] [CrossRef]
- Richards, J.D. Measuring trace mineral bioavailability key. Feedstuffs 2010, 18, 14–15. [Google Scholar]
- Schutte, J.B.; Pack, M. Biological efficacy of L-lysine preparations containing biomass compared to L-lysine-HCl. Arch. Fur Tierernährung 1994, 46, 261–268. [Google Scholar] [CrossRef]
- Payne, R.L.; Lemme, A.; Seko, H.; Hashimoto, Y.; Fujisaki, H.; Koreleski, J.; Swiatkewicz, S.; Szczurek, W.; Rostagno, H.S. Bioavailability of methionine hydroxy analog-free acid relative to DL-methionine in broilers. Anim. Sci. J. 2006, 77, 427–439. [Google Scholar] [CrossRef]
- Goes, E.; Iuspa, M.A.; Naranjo, V.; Almeida, L.; Oliveira, A.; Maiorka, A. The effects of replacing methionine hydroxy-analogue calcium salt with 65% Dl-methionine on growth performance and carcass quality of broilers from 21 to 42 days of age. Poult. Sci. 2017, 96, 102. [Google Scholar]
- Murakami, A.E.; de Oliveira Neto, A.R.; Ospina-Rojas, I.C.; Naranjo, V.; Arruda, N.A. Bioavailability of methionine hydroxy analog relative to DL-methionine in broilers during the starter and grower phases. Poult. Sci. 2017, 96, 102. [Google Scholar]
- Liu, Y.L.; Song, G.L.; Yi, G.F.; Hou, Y.Q.; Huang, J.W.; Vazquez-Anon, M.; Knight, C.D. Effect of supplementing 2-Hydroxy-4-(Methylthio) butanoic acid and DL-methionine in corn-soy-cotton seed meal diets on growth performance and carcass quality of broilers. Asian Australas. J. Anim. Sci. 2006, 19, 1197–1205. [Google Scholar] [CrossRef]
- Agostini, P.S.; Dalibard, P.; Mercier, Y.; van der Aar, P.; van der Klis, J.D. Comparison of methionine sources around requirement levels using a methionine efficacy method in 0 to 28 day old broilers. Poult. Sci. 2016, 95, 560–569. [Google Scholar] [CrossRef]
- Ullrich, C.; Langeheine, M.; Brehm, R.; Taube, V.; Rosillo Galera, M.; Rohn, K.; Popp, J.; Visscher, C. Influence of Different Methionine Sources on Performance and Slaughter Characteristics of Broilers. Animals 2019, 9, 984. [Google Scholar] [CrossRef]
- Ribeiro, A.M.L.; Dahlke, F.; Kessler, A.M. Methionine sources do not affect performance and carcass yield of broilers fed vegetable diets and submitted to cyclic heat stress. Braz. J. Poult. Sci. 2005, 7, 159–164. [Google Scholar] [CrossRef]
- European Union Council. Council directive 2007/43/EC of 28 June 2007 laying down minimum rules for the protection of chickens kept for meat production. Off. J. Europ. Union 2007, 182, 19–28. [Google Scholar]
- Lemme, A. AMINOChick® 2.0; Evonik Operations GmbH: Hanau, Germany, 2014. [Google Scholar]
- Fontaine, J.; Schirmer, B.; Hoerr, J. Near-infrared reflectance spectroscopy (NIRS) enables the fast and accurate prediction of the essential amino acid contents. 2. Results for wheat, barley, corn, triticale, wheat bran/middlings, rice bran, and sorghum. J. Agric. Food Chem. 2002, 50, 3902–3911. [Google Scholar] [CrossRef]
- Fontaine, J.; Hörr, J.; Schirmer, B. Near-infrared reflectance spectroscopy enables the fast and accurate prediction of the essential amino acid contents in soy, rapeseed meal, sunflower meal, peas, fishmeal, meat meal products, and poultry meal. J. Agric. Food Chem. 2001, 49, 57–66. [Google Scholar] [CrossRef]
- Khan, D.R. Studies on Dietary Methionine Efficiency and Requirement in Naked Neck and Normally Feathered Growing Chickens; Niedersächsische Staats- und Universitätsbibliothek Göttingen: Göttingen, Germany, 2015; Available online: https://ediss.uni-goettingen.de/handle/11858/00-1735-0000-0022-5FB9-7 (accessed on 27 November 2020).
- Fatufe, A.A.; Rodehutscord, M. Growth, body composition, and marginal efficiency of methionine utilization are affected by nonessential amino acid nitrogen supplementation in male broiler chicken. Poult. Sci. 2005, 84, 1584–1592. [Google Scholar] [CrossRef]
- Fatufe, A.A.; Timmler, R.; Rodehutscord, M. Response to lysine intake in composition of body weight gain and efficiency of lysine utilization of growing male chickens from two genotypes. Poult. Sci. 2004, 83, 1314–1324. [Google Scholar] [CrossRef] [PubMed]
- Aviagen Ltd. Ross 308 Broiler Performance Objectives; Aviagen Ltd.: Beith, UK, 2019. [Google Scholar]
- Maenz, D.D.; Engele-Schaan, C.M. Methionine and 2-Hydroxy-4-Methylthiobutanoic Acid Are Partially Converted to Nonabsorbed Compounds during Passage through the Small Intestine and Heat Exposure Does Not Affect Small Intestinal Absorption of Methionine Sources in Broiler Chicks. J. Nutr. 1996, 126, 1438–1444. [Google Scholar] [CrossRef] [PubMed]
- Malik, G.; Hoehler, D.; Rademacher, M.; Drew, M.D.; van Kessel, A.G. Apparent absorption of methionine and 2-hydroxy-4-methylthiobutanoic acid from gastrointestinal tract of conventional and gnotobiotic pigs. Animal 2009, 3, 1378–1386. [Google Scholar] [CrossRef] [PubMed]
- Drew, M.D.; van Kessel, A.G.; Maenz, D.D. Absorption of methionine and 2-hydroxy-4-methylthiobutoanic acid in conventional and germ-free chickens. Poult. Sci. 2003, 82, 1149–1153. [Google Scholar] [CrossRef]
- Lemme, A.; Mitchell, M.A. Examination of the Composition of the Luminal Fluid in the Small Intestine of Broilers and Absorption of Amino Acids under Various Ambient Temperatures Measured In vivo. Int. J. Poult. Sci. 2008, 7, 223–233. [Google Scholar] [CrossRef]
- Maenz, D.D.; Engele-Schaan, C.M. Methionine and 2-hydroxy-4-methylthiobutanoic acid are transported by distinct Na+-dependent and H+-dependent systems in the brush boarder membrane of chick intestinal epithelium. J. Nutr. 1996, 126, 529–536. [Google Scholar] [CrossRef]
- Lingens, G.; Molnar, S. Studies on metabolism of broilers by using 14C-labelled DL-methionine and DL-methionine hydroxy analogue calcium salt. Arch. Anim. Nutr. 1996, 49, 113–124. [Google Scholar]
- Zhang, S.; Gilbert, E.R.; Noonan, K.J.T.; Saremi, B.; Wong, E.A. Gene expression and activity of methionine converting enzymes in broiler chickens fed methionine isomers or precursors. Poult. Sci. 2018, 97, 2053–2063. [Google Scholar] [CrossRef]
- van Weerden, E.J.; Schutte, J.B.; Bertram, H.L. Utilisation of the polymers of methionine hydroxy analog free acid (MHA-FA) in broiler chicks. Arch. Geflügelk. 1992, 56, 63–68. [Google Scholar]
- Martín-Venegas, R.; Soriano-García, J.F.; Vinardell, M.P.; Geraert, P.A.; Ferrer, R. Oligomers Are Not the Limiting Factor in the Absorption of DL-2-Hydroxy-4-(methylthio)butanoic Acid in the Chicken Small Intestine. Poult. Sci. 2006, 85, 56–63. [Google Scholar] [CrossRef]
- Richards, J.D.; Atwell, C.A.; Vázquez-Añón, M.; Dibner, J.J. Comparative in vitro and in vivo absorption of 2-hydroxy-4(methylthio) butanoic acid and methionine in the broiler chicken. Poult. Sci. 2005, 84, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.M.; Pacheco, L.G.; Dorigam, J.C.P.; Denadai, J.C.; Viana, G.S.; Varella, H.R.; Nascimento, C.C.N.; van Milgen, J.; Sakomura, N.K. Stable isotopes to study sulfur amino acid utilization in broilers. Animal 2020, 14, s286–s293. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, L.G.; Sakomura, N.K.; Suzuki, R.M.; Dorigam, J.C.P.; Viana, G.S.; van Milgen, J.; Denadai, J.C. Methionine to cystine ratio in the total sulfur amino acid requirements and sulfur amino acid metabolism using labelled amino acid approach for broilers. BMC Vet. Res. 2018, 14, 703. [Google Scholar] [CrossRef] [PubMed]
- D’Mello, J.P.F. Adverse effects of amino acids. In Amino Acids in Animal Nutrition; D’Mello, J.P.F., Ed.; CABI: Wallingford, CT, USA, 2003; pp. 125–142. ISBN 9780851996547. [Google Scholar]
- D’Mello, J.P.F. An outline of pathways in amino acid metabolism. In Amino Acids in Animal Nutrition; D’Mello, J.P.F., Ed.; CABI: Wallingford, CT, USA, 2003; pp. 71–86. ISBN 9780851996547. [Google Scholar]
- Verstegen, M.W.A.; Jongbloed, A.W. Crystalline amino acids and nitrogen emission. In Amino Acids in Animal Nutrition; D’Mello, J.P.F., Ed.; CABI: Wallingford, CT, USA, 2003; pp. 449–458. ISBN 9780851996547. [Google Scholar]
| Ingredients | Starter (day 0 to 11) | Grower (day 11 to 28) | Finisher (day 28 to 35) |
|---|---|---|---|
| Corn | 250.0 | 208.8 | 200.0 |
| Soybean meal | 323.7 | 255.8 | 218.6 |
| Wheat | 286.1 | 364.6 | 365.9 |
| Peas | 50.0 | 100.0 | 139.4 |
| Corn gluten meal | 18.2 | 0.0 | 0.0 |
| Soybean oil | 14.4 | 11.3 | 17.3 |
| Lard | 19.4 | 28.6 | 34.1 |
| Limestone | 14.5 | 11.6 | 9.0 |
| Monocalcium phosphate | 7.6 | 4.3 | 1.3 |
| Salt | 3.3 | 2.5 | 2.6 |
| Sodium bicarbonate | 0.0 | 1.0 | 1.0 |
| L-Lysine-HCl | 2.7 | 1.8 | 1.4 |
| L-Threonine | 1.1 | 0.9 | 0.7 |
| L-Valine | 0.7 | 0.5 | 0.3 |
| Phytase | 3.3 | 3.3 | 3.3 |
| Premix 1 + coccidiostat 2 + NSP degrading enzyme | 5.0 | 5.0 | 5.0 |
| Nutrient composition | |||
| AMEn (kcal/kg) | 2900 | 2975 | 3075 |
| Ash | 5.55 | 4.62 | 3.94 |
| Crude protein | 233.5 | 205.2 | 194.2 |
| Crude fibre | 23.4 | 24.4 | 25.1 |
| Starch (AM) | 349.4 | 385.8 | 396.5 |
| Crude fat (AH) | 60.0 | 64.5 | 75.6 |
| Calcium | 7.86 | 6.11 | 4.61 |
| Phosphorus | 5.39 | 4.47 | 3.72 |
| Sodium | 1.45 | 1.40 | 1.40 |
| Chloride | 3.15 | 2.49 | 2.42 |
| Potassium | 9.85 | 8.99 | 8.53 |
| DEB (mEq/kg) | 226 | 221 | 211 |
| Amino acids | |||
| Total Lysine-calculated | 14.1 | 12.0 | 11.2 |
| Total Lysine-analyzed 3 | 14.5 (103%) | 12.5 (104%) | 11.7 (104%) |
| Total Met + Cys–calculated | 7.1 | 6.2 | 5.8 |
| Total Met + Cys–analyzed 4 | 7.4 (101%) | 6.2 (100%) | 6.3 (108%) |
| Dig. Lysine 5 | 12.6 | 10.7 | 9.90 |
| Dig. Methionine+Cysteine | 6.2 | 5.4 | 5.1 |
| Dig. AA: Dig Lysine | |||
| Methionine | 24 | 24 | 24 |
| Methionine+Cysteine | 49 | 50 | 51 |
| Threonine | 65 | 65 | 65 |
| Tryptophan | 20 | 21 | 21 |
| Arginine | 106 | 111 | 114 |
| Isoleucine | 68 | 69 | 70 |
| Valine | 79 | 80 | 80 |
| Leucine | 132 | 127 | 129 |
| Glycine+Serine | 138 | 143 | 145 |
| Glycineequivalents 6 | 115 | 120 | 122 |
| Treatment | Methionine Sources | Addition of Methionine Sources (g/kg) 1 | Replicates per Treatment | Birds per Treatment | ||
|---|---|---|---|---|---|---|
| 0–11 day | 11–28 day | 28–35 day | ||||
| 1 | Negative control (NC) | 0.00 | 0.00 | 0.00 | 6 | 120 |
| 2 | DLM | 0.40 | 0.40 | 0.40 | 6 | 120 |
| 3 | DLM | 0.80 | 0.80 | 0.80 | 6 | 120 |
| 4 | DLM | 1.20 | 1.20 | 1.20 | 6 | 120 |
| 5 | DLM | 2.10 | 2.10 | 2.10 | 6 | 120 |
| 6 | DLM | 3.00 | 3.00 | 3.00 | 6 | 120 |
| 7 | HMTBA | 0.40 | 0.40 | 0.40 | 6 | 120 |
| 8 | HMTBA | 0.80 | 0.80 | 0.80 | 6 | 120 |
| 9 | HMTBA | 1.20 | 1.20 | 1.20 | 6 | 120 |
| 10 | HMTBA | 2.10 | 2.10 | 2.10 | 6 | 120 |
| 11 | HMTBA | 3.00 | 3.00 | 3.00 | 6 | 120 |
| 12 | DLM65 | 0.40 | 0.40 | 0.40 | 6 | 120 |
| 13 | DLM65 | 0.80 | 0.80 | 0.80 | 6 | 120 |
| 14 | DLM65 | 1.20 | 1.20 | 1.20 | 6 | 120 |
| 15 | DLM65 | 2.10 | 2.10 | 2.10 | 6 | 120 |
| 16 | DLM65 | 3.00 | 3.00 | 3.00 | 6 | 120 |
| Treatment | Met Source | Added Product | Body Weight Gain | Feed Intake | Feed Conversion Ratio | |||
|---|---|---|---|---|---|---|---|---|
| g/kg | g/Bird 1 | Rel. 2 | g/Bird 1 | Rel. 2 | g/g 1 | Rel. 2 | ||
| 1 | Basal | 0.00 | 1492 a | 100 | 2809 a | 100 | 1.885 h | 100 |
| 2 | DLM | 0.40 | 2212 d | 148 | 3550 de | 126 | 1.605 e | 85 |
| 3 | DLM | 0.80 | 2347 e | 157 | 3649 efg | 130 | 1.555 cd | 83 |
| 4 | DLM | 1.20 | 2455 f | 165 | 3750 g | 134 | 1.528 bc | 81 |
| 5 | DLM | 2.10 | 2551 f | 171 | 3716 fg | 132 | 1.457 a | 77 |
| 6 | DLM | 3.00 | 2542 f | 170 | 3726 fg | 133 | 1.467 a | 78 |
| 7 | HMTBA | 0.40 | 1896 c | 127 | 3316 bc | 118 | 1.750 f | 93 |
| 8 | HMTBA | 0.80 | 2179 d | 146 | 3438 cd | 122 | 1.578 de | 84 |
| 9 | HMTBA | 1.20 | 2354 e | 158 | 3658 efg | 130 | 1.554 cd | 82 |
| 10 | HMTBA | 2.10 | 2489 f | 167 | 3709 fg | 132 | 1.490 ab | 79 |
| 11 | HMTBA | 3.00 | 2496 f | 167 | 3673 efg | 131 | 1.472 a | 78 |
| 12 | DLM65 | 0.40 | 1756 b | 118 | 3181 b | 113 | 1.813 g | 96 |
| 13 | DLM65 | 0.80 | 2226 d | 149 | 3592 ef | 128 | 1.614 e | 86 |
| 14 | DLM65 | 1.20 | 2328 e | 156 | 3708 fg | 132 | 1.593 de | 85 |
| 15 | DLM65 | 2.10 | 2501 f | 168 | 3726 fg | 133 | 1.491 ab | 79 |
| 16 | DLM65 | 3.00 | 2529 f | 170 | 3688 fg | 131 | 1.459 a | 77 |
| p-value | <0.001 | <0.001 | <0.001 | |||||
| Treatment | Met Source | Added Product | Carcass Yield | Breast Meat Yield | ||
|---|---|---|---|---|---|---|
| g/kg | % of BW 1 | Rel. 2 | % of BW 1 | Rel. 2 | ||
| 1 | Basal | 0.00 | 58.1 a | 100 | 12.3 a | 100 |
| 2 | DLM | 0.40 | 62.1 c | 107 | 15.4 cd | 125 |
| 3 | DLM | 0.80 | 63.8 def | 110 | 17.4 efg | 141 |
| 4 | DLM | 1.20 | 66.1 i | 114 | 18.5 hi | 150 |
| 5 | DLM | 2.10 | 65.4 ghi | 113 | 20.4 k | 165 |
| 6 | DLM | 3.00 | 65.8 hi | 113 | 20.0 k | 162 |
| 7 | HMTBA | 0.40 | 61.6 c | 106 | 14.7 bc | 119 |
| 8 | HMTBA | 0.80 | 62.9 cde | 108 | 16.4 de | 133 |
| 9 | HMTBA | 1.20 | 64.2 efgh | 111 | 17.3 ef | 140 |
| 10 | HMTBA | 2.10 | 64.1 efg | 110 | 18.4 ghi | 150 |
| 11 | HMTBA | 3.00 | 65.0 fghi | 111 | 19.7 jk | 160 |
| 12 | DLM65 | 0.40 | 60.0 b | 103 | 14.0 b | 114 |
| 13 | DLM65 | 0.80 | 62.4 cd | 107 | 15.7 cd | 128 |
| 14 | DLM65 | 1.20 | 64.7 fghi | 111 | 17.6 fgh | 143 |
| 15 | DLM65 | 2.10 | 65.4 fghi | 112 | 18.9 ij | 154 |
| 16 | DLM65 | 3.00 | 65.5 ghi | 113 | 20.0 k | 163 |
| p-value | <0.001 | <0.001 | ||||
| Parameters | Model | Relative Bioavailability Value 1 | H0:δβ 3 | |
|---|---|---|---|---|
| HMTBA | DLM65 | |||
| Body weight gain | y = 1440 + 1103 × (1 − e-(2.41*DLM + 1.40*HMTBA + 1.34*DLM65)) | 58% (48.9–67.7) 2 | 56% (47.2–64.0) | 0.608 |
| Feed conversion ratio | y = 1.892 − 0.432 × (1 − e-(2.00 × DLM + 1.33 × HMTBA + 1.07 × DLM65) | 66% (53.7–78.8) 2 | 54% (42.8–64.2) | 0.204 |
| Carcass yield | y = 57.892 + 7.74 × (1 − e-(2.09 × DLM + 1.32 × HMTBA + 1.22 × DLM65)) | 63% (38.5–87.2) 2 | 58% (33.8–82.7) | 0.083 |
| Breast meat yield | y = 12.234 + 8.196 × (1 − e-(1.22 × DLM + 0.79 × HMTBA + 0.79 × DLM65)) | 65% (52.1–77.3) 2 | 65% (51.3–77.8) | 0.255 |
| Average relative bioavailability | 63% | 58% | ||
| Weight Gain | Feed Conversion Ratio | Breast Meat Yield | |||||
|---|---|---|---|---|---|---|---|
| Study | Year | DLM65 | HMTBA | DLM65 | HMTBA | DLM65 | HMTBA |
| 1 1 | 1999 | 59% | 57% | 66% | 58% | ||
| 2 2 | 2002 | 60% | 68% | 57% | 67% | 69% | 64% |
| 3 3 | 2005 | 67% | 64% | 59% | 67% | ||
| 4 3 | 2005 | 69% | 63% | 79% | 73% | ||
| 5 3 | 2005 | 59% | 65% | 47% | 49% | ||
| Current | 2020 | 56% | 58% | 54% | 66% | 65% | 65% |
| Average | 62% | 63% | 60% | 63% | 67% | 65% | |
| Overall weighted average across all criteria | DLM65 | HMTBA | |||||
| 62% | 63% | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lemme, A.; Naranjo, V.; de Paula Dorigam, J.C. Utilization of Methionine Sources for Growth and Met+Cys Deposition in Broilers. Animals 2020, 10, 2240. https://doi.org/10.3390/ani10122240
Lemme A, Naranjo V, de Paula Dorigam JC. Utilization of Methionine Sources for Growth and Met+Cys Deposition in Broilers. Animals. 2020; 10(12):2240. https://doi.org/10.3390/ani10122240
Chicago/Turabian StyleLemme, Andreas, Victor Naranjo, and Juliano Cesar de Paula Dorigam. 2020. "Utilization of Methionine Sources for Growth and Met+Cys Deposition in Broilers" Animals 10, no. 12: 2240. https://doi.org/10.3390/ani10122240
APA StyleLemme, A., Naranjo, V., & de Paula Dorigam, J. C. (2020). Utilization of Methionine Sources for Growth and Met+Cys Deposition in Broilers. Animals, 10(12), 2240. https://doi.org/10.3390/ani10122240





