A Preliminary Study Investigating the Impact of Musical Concerts on the Behavior of Captive Fiordland Penguins (Eudyptes pachyrhynchus) and Collared Peccaries (Pecari tajacu)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
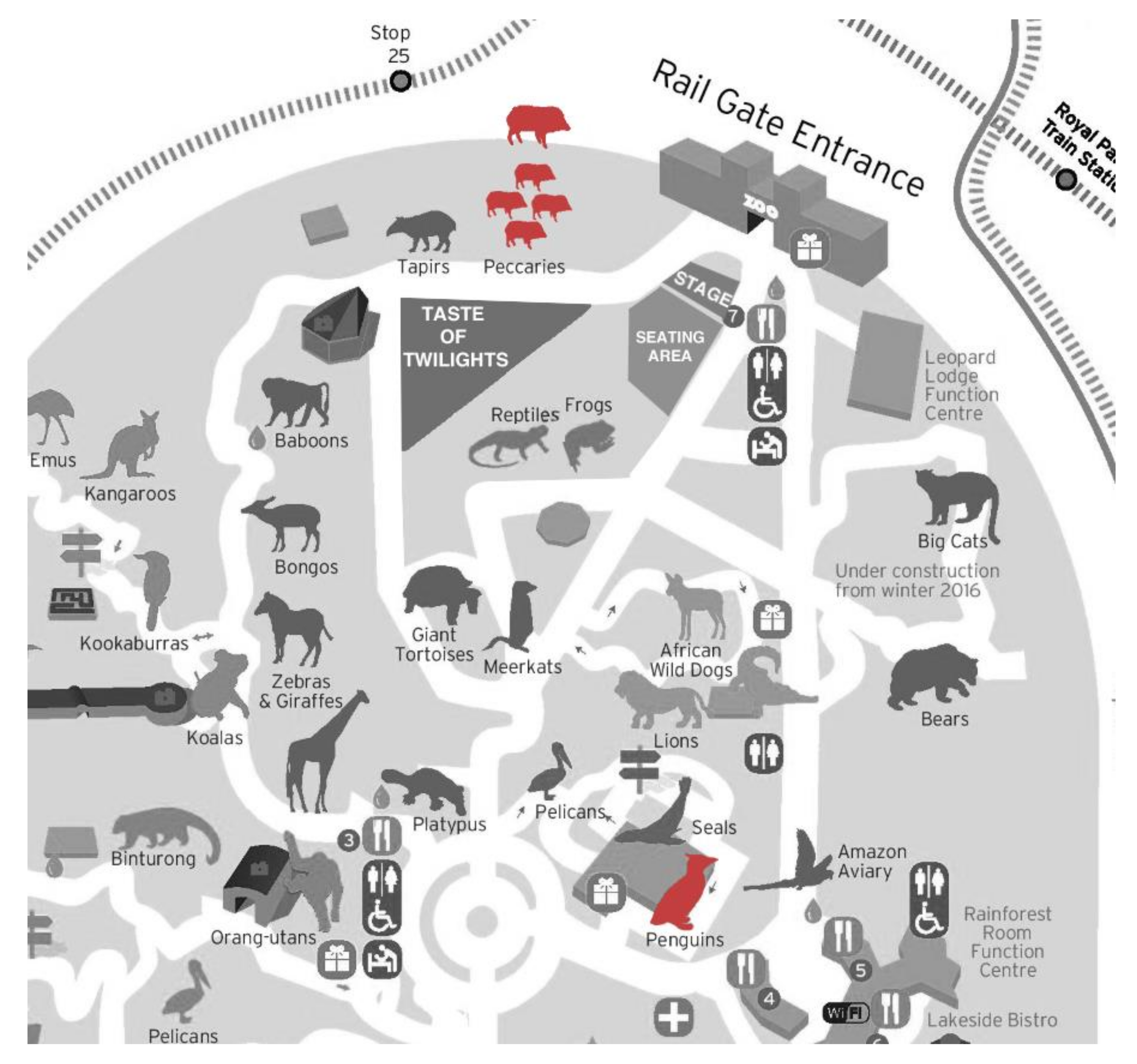
2.1. Study Site
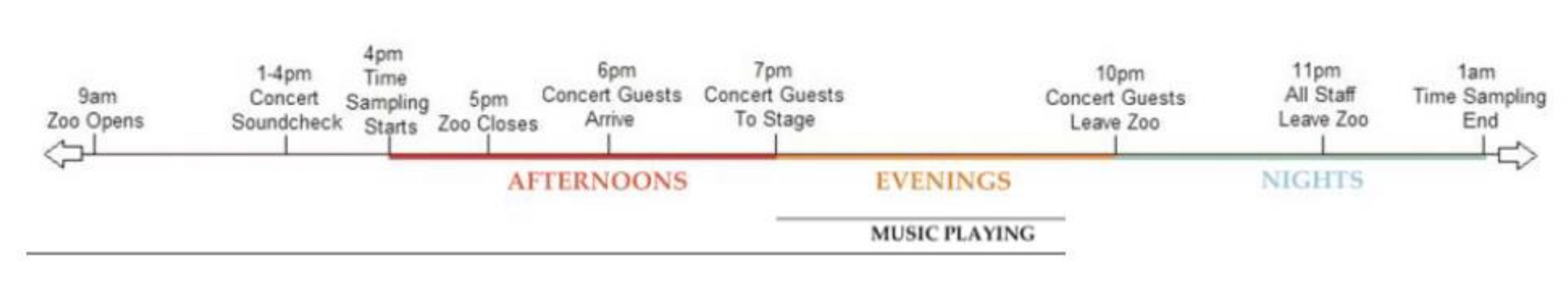
2.2. The Melbourne Zoo Twilights Concert Series
2.3. Study Animals and Exhibits
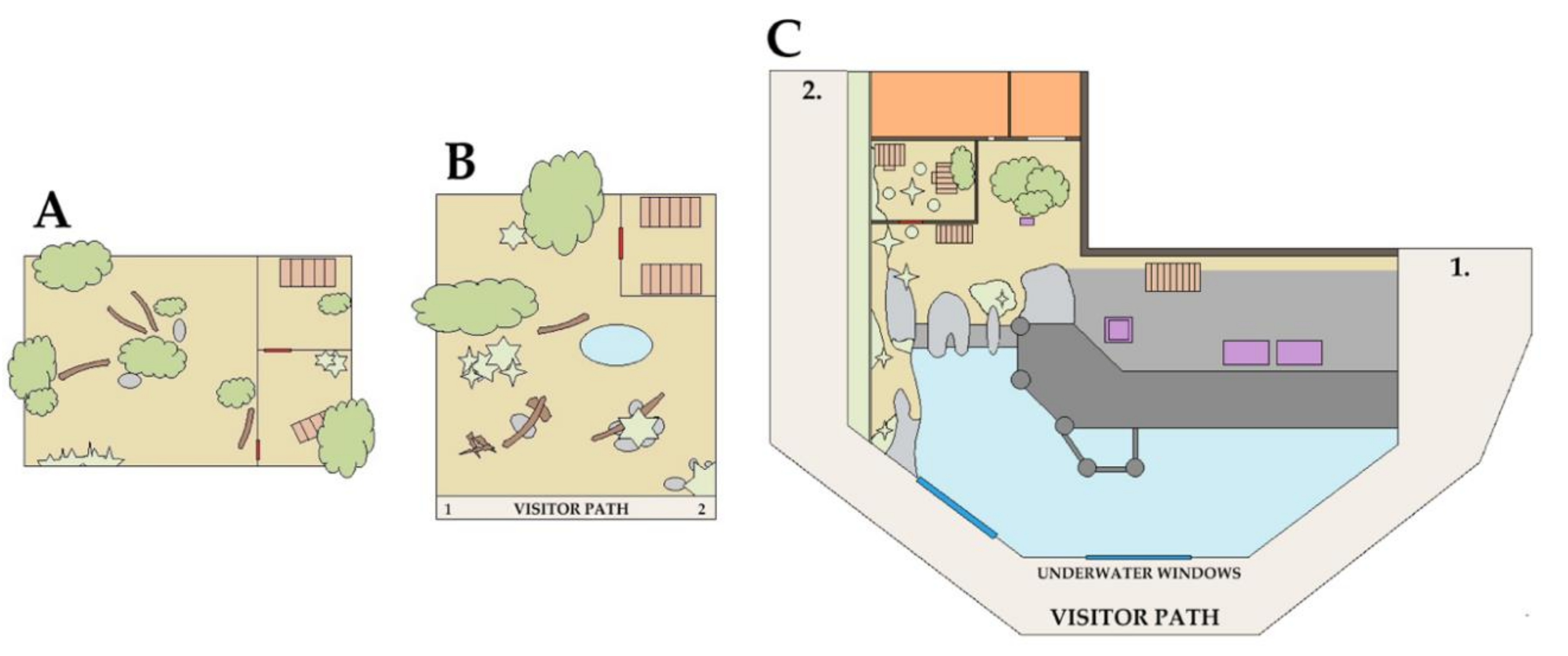
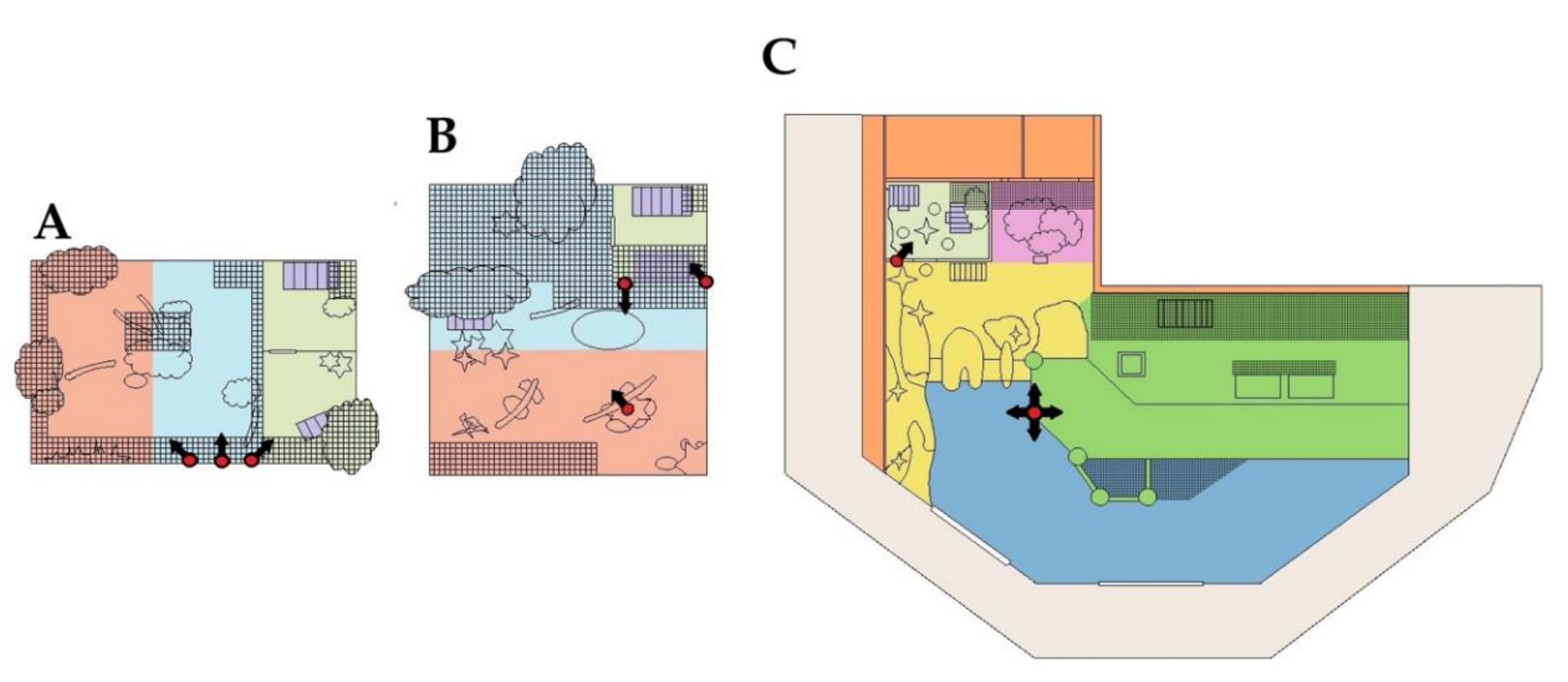
2.3.1. Fiordland Penguins
2.3.2. Peccaries
2.4. Video and Acoustic Monitoring
2.5. Experimental Design
2.6. Behavioural Data Collection
2.7. Visitor Observations
2.8. Statistical Analysis
3. Results
3.1. Fiordland Penguins
3.1.1. Visitor Number at Penguin Exhibit
3.1.2. Impact of Concert on Location and Behaviour
3.1.3. Individual Differences
3.1.4. Unknowns
3.2. Sound Pressure Levels at the Peccary Exhibits
3.3. Solitary Peccary
3.3.1. Impact of Concert on Location and Behaviour
3.3.2. Unknowns
3.4. Peccary Group
3.4.1. Visitor Number at Group-Housed Peccary Exhibit
3.4.2. Impact of Concert on Location and Behaviour
3.4.3. Unknowns
4. Discussion
4.1. Fiordland Penguins
4.2. Peccaries
4.3. Future Evidence-Based Management Considerations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Morgan, K.N.; Tromborg, C.T. Sources of stress in captivity. Appl. Anim. Behav. Sci. 2007, 102, 262–302. [Google Scholar] [CrossRef]
- Mason, G.J. Species differences in responses to captivity: Stress, welfare and the comparative method. Trends Ecol. Evol. 2010, 25, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Mason, G.J.; Mendl, M. Why is there no simple way of measuring animal welfare? Anim. Welf. 1993, 2, 301–319. [Google Scholar]
- Melfi, V. There are big gaps in our knowledge, and thus approach, to zoo animal welfare: A case for evidence-based zoo animal management. Zoo Biol. Publ. Affil. Am. Zoo Aquar. Assoc. 2009, 28, 574–588. [Google Scholar] [CrossRef]
- Broom, D.M.; Johnson, K.G. Stress and Animal Welfare; Chapman and Hall: Melbourne/London, UK, 1993. [Google Scholar]
- Carlstead, K.; Shepherdson, D. Alleviating stress in zoo animals with environmental enrichment. In The Biology Animal Stress: Basic Principles and Implications for Animal Welfare; CAB International: Wallingford, UK, 2000; pp. 337–354. [Google Scholar]
- Quadros, S.; Goulart, V.D.; Passos, L.; Vecci, M.A.; Young, R.J. Zoo visitor effect on mammal behaviour: Does noise matter? Appl. Anim. Behav. Sci. 2014, 156, 78–84. [Google Scholar] [CrossRef]
- Bastian, M.L.; Glendinning, D.R.; Brown, J.L.; Boisseau, N.P.; Edwards, K.L. Effects of a recurring late-night event on the behavior and welfare of a population of zoo-housed gorillas. Zoo Biol. 2020. [Google Scholar] [CrossRef]
- Alworth, L.C.; Buerkle, S.C. The effects of music on animal physiology, behavior and welfare. Lab Anim. 2013, 42, 54. [Google Scholar] [CrossRef]
- Robbins, L.; Margulis, S.W. Music for the birds: Effects of auditory enrichment on captive bird species. Zoo Biol. 2016, 35, 29–34. [Google Scholar] [CrossRef]
- Wallace, E.K.; Altschul, D.; Körfer, K.; Benti, B.; Kaeser, A.; Lambeth, S.; Waller, B.M.; Slocombe, K.E. Is music enriching for group-housed captive chimpanzees (Pan troglodytes)? PLoS ONE 2017, 12, e0172672. [Google Scholar] [CrossRef] [PubMed]
- Davey, G. Visitor behavior in zoos: A review. Anthrozoös 2006, 19, 143–157. [Google Scholar] [CrossRef]
- Wells, D.L. A note on the influence of visitors on the behaviour and welfare of zoo-housed gorillas. Appl. Anim. Behav. Sci. 2005, 93, 13–17. [Google Scholar] [CrossRef]
- Davis, N.; Schaffner, C.M.; Smith, T.E. Evidence that zoo visitors influence HPA activity in spider monkeys (Ateles geoffroyii rufiventris). Appl. Anim. Behav. Sci. 2005, 90, 131–141. [Google Scholar] [CrossRef]
- Rajagopal, T.; Archunan, G.; Sekar, M. Impact of zoo visitors on the fecal cortisol levels and behavior of an endangered species: Indian blackbuck (Antelope cervicapra L.). J. Appl. Anim. Welf. Sci. 2011, 14, 18–32. [Google Scholar] [CrossRef]
- Sherwen, S.L.; Magrath, M.J.; Butler, K.L.; Hemsworth, P.H. Little penguins, Eudyptula minor, show increased avoidance, aggression and vigilance in response to zoo visitors. Appl. Anim. Behav. Sci. 2015, 168, 71–76. [Google Scholar] [CrossRef]
- Chiew, S.J.; Butler, K.L.; Sherwen, S.L.; Coleman, G.J.; Fanson, K.V.; Hemsworth, P.H. Effects of regulating visitor viewing proximity and the intensity of visitor behaviour on little penguin (Eudyptula minor) behaviour and welfare. Animals 2019, 9, 285. [Google Scholar] [CrossRef]
- Ozella, L.; Favaro, L.; Carnovale, I.; Pessani, D. Pond use by captive African penguins (Spheniscus demersus) in an immersive exhibit adjacent to human bathers. J. Appl. Anim. Welf. Sci. 2015, 18, 303–309. [Google Scholar] [CrossRef]
- Bloomfield, R.C.; Gillespie, G.R.; Kerswell, K.J.; Butler, K.L.; Hemsworth, P.H. Effect of partial covering of the visitor viewing area window on positioning and orientation of zoo orangutans: A preference test. Zoo Biol. 2015, 34, 223–229. [Google Scholar] [CrossRef]
- Whitworth, A.W. An investigation into the determining factors of zoo visitor attendances in UK zoos. PLoS ONE 2012, 7, e29839. [Google Scholar] [CrossRef]
- Diebold, E.; Branch, S.; Henry, L. Management of penguin populations in North American zoos and aquariums. Mar. Ornithol. 1999, 27, 171–176. [Google Scholar]
- Ozella, L.; Anfossi, L.; Di Nardo, F.; Pessani, D. Effect of weather conditions and presence of visitors on adrenocortical activity in captive African penguins (Spheniscus demersus). Gen. Comp. Endocrinol. 2017, 242, 49–58. [Google Scholar] [CrossRef]
- Collins, C.K.; Quirke, T.; Overy, L.; Flannery, K.; O’Riordan, R. The effect of the zoo setting on the behavioural diversity of captive gentoo penguins and the implications for their educational potential. J. Zoo Aquar. Res. 2016, 4, 85–90. [Google Scholar]
- Warham, J. The Fiordland crested penguin Eudyptes pachyrhynchus. Ibis 1974, 116, 1–27. [Google Scholar] [CrossRef]
- Fowler, M. Husbandry and diseases of captive wild swine and peccaries. Rev. Sci. Tech. Off. Int. Epizoot. 1996, 15, 141–154. [Google Scholar] [CrossRef]
- Sowls, L.K. The Peccaries; University Arizona Press: Tucson, AZ, USA, 1984. [Google Scholar]
- Biondo, C.; Izar, P.; Miyaki, C.Y.; Bussab, V.S. Social structure of collared peccaries (Pecari tajacu): Does relatedness matter? Behav. Process. 2014, 109, 70–78. [Google Scholar] [CrossRef]
- De Faria, C.M.; de Souza Sá, F.; Costa, D.D.L.; da Silva, M.M.; da Silva, B.C.; Young, R.J.; de Azevedo, C.S. Captive-born collared peccary (Pecari tajacu, Tayassuidae) fails to discriminate between predator and non-predator models. Acta Ethol. 2018, 21, 175–184. [Google Scholar] [CrossRef]
- Orban, D.A.; Soltis, J.; Perkins, L.; Mellen, J.D. Sound at the zoo: Using animal monitoring, sound measurement, and noise reduction in zoo animal management. Zoo Biol. 2017, 36, 231–236. [Google Scholar] [CrossRef]
- Bleicher, S.S.; Rosenzweig, M.L. Too much of a good thing? A landscape of fear analysis for peccaries reveals hikers act as a greater deterrent than thorny or bitter food. Can. J. Zool. 2017, 96, 317–324. [Google Scholar] [CrossRef]
- Mendes Pontes, A.R.; Chivers, D.J. Peccary movements as determinants of the movements of large cats in Brazilian Amazonia. J. Zool. 2007, 273, 257–265. [Google Scholar] [CrossRef]
- Hosey, G. Hediger revisited: How do zoo animals see us? J. Appl. Anim. Welf. Sci. 2013, 16, 338–359. [Google Scholar] [CrossRef]
- Kagan, R.; Carter, S.; Allard, S. A universal animal welfare framework for zoos. J. Appl. Anim. Welf. Sci. 2015, 18, S1–S10. [Google Scholar] [CrossRef]
- Australian Government, Bureau of Meteorology. Monthly Climate Summary for Greater Melbourne; Australian Government, Bureau of Meteorology: Docklands, VIC, Australia, 2018.
- Olson, W.C.; Cunningham, E.M. Time-sampling techniques. Child Dev. 1934, 5, 41–58. [Google Scholar] [CrossRef]
- Tyler, S. Time-sampling: A matter of convention. Anim. Behav. 1979, 27, 801–810. [Google Scholar] [CrossRef]
- Babisch, W. The noise/stress concept, risk assessment and research needs. Noise Health 2002, 4, 1. [Google Scholar]
- Nummela, S.; Thewissen, J. The physics of sound in air and water. In Sensory Evolution on the Threshold: Adaptations in Secondarily Aquatic Vertebrates; University of California Press: Berkeley, CA, USA, 2008; pp. 175–181. [Google Scholar]
- Stevens, J.; Thyssen, A.; Laevens, H.; Vervaecke, H. The influence of zoo visitor numbers on the behaviour of harbour seals (Phoca vitulina). J. Zoo Aquar. Res. 2013, 1, 31–34. [Google Scholar]
- Pelletier, C.; Weladji, R.B.; Lazure, L.; Paré, P. Zoo soundscape: Daily variation of low-to-high-frequency sounds. Zoo Biol. 2020. [Google Scholar] [CrossRef]
- Sumpter, D.J. Collective Animal Behavior; Princeton University Press: Princeton, NJ, USA, 2010. [Google Scholar]
- Meagher, R.K.; Campbell, D.L.; Dallaire, J.A.; Díez-León, M.; Palme, R.; Mason, G.J. Sleeping tight or hiding in fright? The welfare implications of different subtypes of inactivity in mink. Appl. Anim. Behav. Sci. 2013, 144, 138–146. [Google Scholar] [CrossRef]
- Fureix, C.; Meagher, R.K. What can inactivity (in its various forms) reveal about affective states in non-human animals? A review. Appl. Anim. Behav. Sci. 2015, 171, 8–24. [Google Scholar] [CrossRef]
- Beauchamp, G.; Ruxton, G.D. False alarms and the evolution of antipredator vigilance. Anim. Behav. 2007, 74, 1199–1206. [Google Scholar] [CrossRef]
- Dawkins, M.S. From an animal’s point of view: Motivation, fitness, and animal welfare. Behav. Brain Sci. 1990, 13, 1–9. [Google Scholar] [CrossRef]
- Kight, C.R.; Swaddle, J.P. How and why environmental noise impacts animals: An integrative, mechanistic review. Ecol. Lett. 2011, 14, 1052–1061. [Google Scholar] [CrossRef]
| Category | Behaviour | Description |
|---|---|---|
| Locomotion | Walk | Slow movement in any direction while animal stands in an upright position. Head and chest may be hunched. |
| Run | Fast movement in any direction while animal stands in an upright position. Head and chest may be hunched. | |
| Surface swim | Surface swim | Floating or locomotion on surface of water. |
| Dive | Dive | Locomotion whilst completely submerged under water. |
| Vigilance | Vigilance | Stands in erect position with neck stretched above the shoulders and eyes open. Head and eyes may be orientated towards one point or searching for disturbance. |
| Rest | Rest | Lies on belly with head placed against the ground or supported on chests OR stands upright with head resting on neck/shoulders and flippers tucked to body. |
| Stand | Remains stationary with two feet on the ground for at least 5 s, but body, head, and limbs may move. Head held upright (not propped against chest). | |
| Habitat engagement | Exploration | Penguin examines item or environmental feature for at least 3 s, orientating eyes and head towards point of interest and often touching with beak. May be stationary or walking. |
| Forage | Using beak, penguin picks up food item or nesting substrate including sticks, rocks, twigs, and leaves. | |
| Preen | Preen | Uses beak to peck, stroke, or comb feathers in any region of the body. Animal either stationary on land or standing in or floating on water. |
| Human interaction | Keeper interaction | Orientates body and eyes to keeper; moves towards keeper or is in gentle contact with keeper. Excludes antagonistic behaviours towards keeper. |
| Visitor interaction | Tapping visitor window glass with beak when visitor is present, or following visitor in the water or on the land, or orientating head and eyes to watch visitor through glass screen or over enclosure wall. | |
| Cohabitant interaction | Affiliative | Using beak, pecks, strokes, or combs feathers of conspecific; standing beside or before another penguin and bowing the head towards it; touching beaks and/or the head with a conspecific; all mating behaviours. |
| Antagonistic | Uses the beak to peck at another penguin, resulting in other penguin moving away or showing aggression; pursuing another penguin at a run with head lowered below shoulders, and eyes and head orientated towards target being chased; penguin faces another penguin with head held at same level or higher, and stares at other bird for at least two seconds; penguin stands with feathers raised and neck outstretched while conspecific lowers head. |
| Category | Behaviour | Description |
|---|---|---|
| Locomotion | Walk | Locomotion in any direction. At least two hooves remain on ground. |
| Rest | Rest | Lies either with belly or side of body touching the ground. Eyes may be opened or closed; however, peccary remains responsive to small environmental changes. |
| Sleep | Sleep | Lies either with belly or side of body touching the ground. Eyes are closed. Unresponsive to small changes in environment. |
| Stand | Stand | Remains stationary with all four hooves on the ground and legs straight. Head held level with back or below back. |
| Habitat engagement | Forage | Snout disturbs grass, vegetation, substrate, or water in a continuous “searching” motion. Snout may be used to root ground. |
| Eat | Navigation of food item into mouth. | |
| Sniff | Snout touches and moves across a substrate. Head may also be lifted into the air while snout twitches or continues to move. | |
| Wallow | Substrate is manipulated by head, body, or legs so that peccary’s head, body, or legs are partially or entirely coated in substrate. May be performed on land or in water, while standing, sitting, or lying. | |
| Enrichment use | Uses any part of the body to touch enrichment source. | |
| Vigilance | Vigilance | Head held level with or above the shoulders, ears pointed towards one point and eyes focused on same point. Animal may be standing or lying. |
| No Concert | Concert | Trt × Time | Trt × ID | Time × ID | Trt × Time × ID | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Afternoon | Evening | Night | Afternoon | Evening | Night | Trt | Time | ID | |||||
| Location | |||||||||||||
| Vegetated patch | 5.3 ± 2.2 | 8.3 ± 2.8 | 0.0 ± 0.0 | 7.9 ± 3.1 | 6.9 ± 3.9 | 0.0 ± 0.0 | 0.114 | 0.351 | <0.001 | 0.033 | 0.005 | 0.927 | 0.996 |
| Nest | 0.7 ± 0.5 | 10.2 ± 6.1 | 25.5 ± 9.8 | 0.0 ± 0.0 | 13.5 ± 6.2 | 66.7 ± 21.1 | <0.001 | <0.001 | <0.001 | 0.096 | 0.001 | 0.588 | 0.257 |
| Entrance platform | 11.8 ± 1.7 | 1.4 ± 0.5 | 25.0 ± 16.4 | 6.9 ± 2.0 | 5.1 ±1.2 | 5.6 ± 4.1 | 0.056 | <0.001 | 0.939 | <0.001 | 0.423 | 0.818 | 0.769 |
| Shoreline | 8.7 ± 2.2 | 12.7 ± 2.6 | 0.0 ± 0.0 | 4.8 ± 0.8 | 9.3 ± 2.2 | 0.0 ± 0.0 | <0.001 | 1.000 | 0.186 | 1.000 | 0.102 | 0.867 | 0.905 |
| Pool | 15.5 ± 3.4 | 8.3 ± 1.5 | 0.0 ± 0.0 | 29.4 ± 3.2 | 20.7 ± 3.1 | 0.0 ± 0.0 | <0.001 | <0.001 | 0.271 | 0.711 | 0.617 | 0.828 | 0.835 |
| Den | 3.0 ± 1.1 | 7.7 ± 2.5 | 25.5 ± 9.8 | 2.0 ± 1.3 | 5.3 ± 1.4 | 0.0 ± 0.0 | 0.155 | <0.001 | 0.436 | 0.818 | 0.683 | 0.822 | 0.611 |
| Unknown location | 18.9 ± 1.2 | 12.9 ± 1.7 | 3.4 ± 1.7 | 17.6 ± 1.5 | 10.2 ± 2.2 | 3.3 ± 2.1 | 0.048 | <0.001 | 0.139 | 0.037 | 0.008 | 0.492 | 0.125 |
| Behaviour | |||||||||||||
| Locomotion | 4.8 ± 1.2 | 4.3 ± 1.1 | 3.1 ± 3.1 | 4.4 ± 0.9 | 3.9 ±1.5 | 0.0 ± 0.0 | 0.917 | 0.002 | 0.070 | 0.555 | 0.831 | 0.725 | 0.036 |
| Surface swimming | 6.4 ± 1.4 | 2.8 ± 0.9 | 0.0 ± 0.0 | 12.9 ± 1.5 | 8.4 ± 2.4 | 0.0 ± 0.0 | <0.001 | <0.001 | 0.029 | 0.826 | 0.292 | 0.683 | 0.486 |
| Diving | 3.8 ± 1.3 | 3.1 ± 0.7 | 0.0 ± 0.0 | 12.5 ± 1.3 | 4.7 ± 1.4 | 0.0 ± 0.0 | <0.001 | <0.001 | 0.266 | 0.182 | 0.568 | 0.594 | 0.741 |
| Vigilance | 6.3 ± 1.3 | 4.3 ± 1.6 | 1.6 ± 1.1 | 4.2 ± 1.0 | 7.8 ± 1.8 | 1.4 ± 1.4 | 0.986 | 0.001 | 0.763 | 0.198 | 0.222 | 0.632 | 0.097 |
| Resting | 9.8 ± 2.7 | 9.5 ± 2.0 | 18.8 ± 11.2 | 6.0 ± 2.0 | 4.9 ± 2.0 | 4.2 ± 2.8 | <0.001 | 0.001 | <0.001 | 0.024 | 0.561 | 0.003 | 0.690 |
| Habitat engagement | 1.0 ± 0.6 | 4.9 ± 2.5 | 5.2 ± 5.2 | 0.4 ± 0.3 | 0.6 ± 0.4 | 0.0 ± 0.0 | <0.001 | 0.002 | 0.278 | 0.808 | 0.106 | 0.028 | - |
| Preening | 7.2 ± 0.0 | 4.6 ± 1.4 | 4.2 ± 2.7 | 3.8 ± 1.1 | 4.7 ± 1.2 | 0.0 ± 0.0 | 0.019 | 0.001 | 0.275 | 0.303 | 0.874 | 0.433 | 0.837 |
| Human interaction | 0.0 ± 0.0 | 0.0 ± 0.00 | 0.0 ± 0.0 | 2.2 ± 0.8 | 0.0 ± 0.0 | 0.0 ± 0.0 | - | ||||||
| Cohabitant interaction | 1.0 ± 0.3 | 3.0 ± 0.0 | 0.0 ± 0.0 | 1.4 ± 0.5 | 5.0 ± 1.4 | 0.0 ± 0.0 | 0.370 | 0.041 | 0.873 | 0.996 | 0.882 | 0.821 | 0.833 |
| Unknown | |||||||||||||
| Unknown behaviour | 4.7 ± 1.2 | 11.0 ± 16.4 | 46.4 ± 16.4 | 3.9 ± 1.6 | 17.3 ± 6.7 | 66.7 ± 21.1 | 0.001 | <0.001 | 0.660 | 0.002 | <0.001 | <0.001 | 0.521 |
| Footage missing | 2.4 ± 2.4 | 2.6 ± 0.5 | 0.5 ± 0.5 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | - | ||||||
| Animal not visible | 15.5 ± 2.0 | 3.1 ± 0.5 | 0.5 ± 0.5 | 28.0 ± 3.2 | 9.1 ± 1.6 | 1.4 ± 1.4 | <0.001 | <0.001 | 0.587 | 0.418 | 0.868 | 0.475 | 0.854 |
| Camera inactive | 37.1± 4.4 | 41.8 ± 9.7 | 19.8 ± 9.7 | 20.4 ± 3.1 | 26.6 ± 4.8 | 26.4 ± 17.0 | 0.001 | <0.001 | 0.588 | 0.195 | <0.001 | 0.002 | 0.266 |
| No Concert | Concert | |||||
|---|---|---|---|---|---|---|
| Sound Pressure Level | Afternoon | Evening | Night | Afternoon | Evening | Night |
| LAavg | 50.1 ± 2.0 a | 48.0 ± 0.7 a | 48.7 ± 1.8 a | 61.1 ± 1.3 b | 72.8 ± 1.7 c | 48.0 ± 1.6 a |
| LAmin | 44.7 ± 0.3 a | 43.3 ± 0.5 a | 41.8 ± 0.8 a | 48.9 ± 1.2 b | 54.1 ± 1.4 c | 42.4 ± 0.4 a |
| LAmax | 65.0 ± 3.8 a | 57.3 ± 1.4 ad | 61.3 ± 2.2 a d | 82.2 ± 1.67 b | 87.5 ± 1.2 c | 55.9 ± 2.5 d |
| No Concert | Concert | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Afternoon | Evening | Night | Afternoon | Evening | Night | Trt | Time | Trt × Time | |
| Location | |||||||||
| Den | 0.0 ± 0.0 | 2.3 ± 0.9 | 0.0 ± 0.0 | 0.0 ± 0.0 | 4.1 ± 1.7 | 1.4 ± 0.7 | 0.087 | 0.009 | - |
| Exhibit front | 7.9 ± 4.1 | 16.2 ± 6.5 | 12.5 ± 2.9 | 15.5 ± 2.9 | 31.6 ± 7.5 | 32.3 ± 8.7 | <0.001 | <0.001 | 0.498 |
| Exhibit back | 2.8 ± 2.3 | 11.5 ± 6.5 | 30.4 ± 11.2 | 20.4 ± 11.1 | 6.3 ± 5.2 | 13.2 ± 7.1 | 0.011 | <0.001 | 0.715 |
| Behaviour | |||||||||
| Locomotion | 0.0 ± 0.0 | 7.4 ± 2.3 | 3.2 ± 1.8 | 0.3 ± 0.3 | 5.6 ± 2.1 | 2.8 ± 1.0 | 0.481 | 0.002 | 0.831 |
| Habitat engagement | 7.4 ± 3.6 | 22.7 ± 7.6 | 12.5 ± 3.6 | 9.4 ± 4.8 | 24.7 ± 6.3 | 8.3 ± 3.7 | 0.851 | <0.001 | 0.241 |
| Rest | 0.0 ± 0.0 | 0.0 ± 0.0 | 1.9 ± 1.4 | 5.2 ± 4.8 | 11.1 ± 5.8 | 8.3 ± 2.2 | <0.001 | 0.048 | - |
| Sleep | 3.2 ± 0.0 | 0.0 ± 0.0 | 14.7 ± 10.8 | 0.0 ± 0.0 | 0.0 ± 0.0 | 26.7 ± 7.7 | 0.035 | <0.001 | - |
| Vigilance | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.7 ± 0.7 | 0.7 ± 0.5 | - | ||
| Stand | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.5 ± 0.5 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | - | ||
| Unknown | |||||||||
| Footage missing | 60.2 ± 20.0 a | 16.7 ± 8.6 b | 32.7 ± 0.5 c | 54.2 ± 15.1 a | 4.5 ± 3.8 d | 31.9 ± 0.5 c | <0.001 | <0.001 | 0.001 |
| Animal not visible | 9.7 ± 6.6 a | 40.7 ± 6.1 b | 28.5 ± 8.1 b | 17.4 ± 5.7 abc | 27.4 ± 6.7 b | 8.0 ± 2.7 ac | <0.001 | <0.001 | <0.001 |
| Camera inactive | 19.4 ± 12.1 a | 12.5 ± 5.5 bc | 6.1 ± 3.2 c | 13.5 ± 6.8 ab | 26.0 ± 11.5 ad | 13.2 ± 8.6 abc | 0.010 | 0.001 | 0.001 |
| No Concert | Concert | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Afternoon | Evening | Night | Afternoon | Evening | Night | Trt | Time | Trt × Time | |
| Location | |||||||||
| Box | 0.0 ± 0.0 | 7.2 ± 7.2 | 7.8 ± 7.8 | 5.8 ± 5.8 | 8.6 ± 5.3 | 10.4 ± 6.8 | 0.002 | 0.054 | 0.171 |
| Den | 26.4 ± 16.7 a | 85.2 ± 6.4 b | 55.4 ± 18.7 c | 60.8 ± 15.3 c | 61.6 ± 9.8 b | 48.7 ± 12.7 c | <0.001 | <0.001 | 0.007 |
| Exhibit front | 2.7 ± 1.8 | 4.2 ± 1.7 | 20.1 ± 16.1 | 3.9 ± 2.5 | 4.3 ± 1.7 | 11.1 ± 6.1 | 0.032 | 0.162 | 0.334 |
| Exhibit back | 4.2 ± 2.6 a | 3.3 ± 1.9 a | 0.0 ± 0.0 a | 4.5 ± 4.5 a | 12.9 ± 3.9 b | 4.7 ± 3.7 a | 0.039 | 0.012 | 0.012 |
| Behaviour | |||||||||
| Locomotion | 0.6 ± 0.4 | 3.1 ± 0.6 | 0.8 ± 0.2 | 0.9 ± 0.7 | 4.3 ± 1.9 | 1.6 ± 0.7 | 0.062 | <0.001 | 0.812 |
| Habitat engagement | 5.7 ± 3.9 a | 11.3 ± 3.4 b | 2.8 ± 1.4 c | 2.8 ± 1.0 c | 10.8 ± 1.9 b | 4.3 ± 1.7 a | 0.349 | <0.001 | 0.003 |
| Rest | 0.1 ± 0.1 a | 3.8 ± 3.8 b | 1.9 ± 1.9 bcd | 0.6 ± 0.6 ad | 3.3 ± 2.4 b | 4.2 ± 3.0 b e | 0.040 | <0.001 | 0.016 |
| Sleep | 0.0 ± 0.0 a | 0.2 ± 0.2 a | 5.4 ± 5.4 b | 6.3 ± 6.3 b | 4.0 ± 4.0 b | 3.2 ± 3.0 b | <0.001 | <0.001 | <0.001 |
| Vigilance | 0.0 ± 0.0 a | 3.6 ± 2.1 b | 0.1 ± 0.1 a | 0.4 ± 0.3 ac | 3.3 ± 1.5 b | 1.6 ± 1.1 b c | 0.015 | <0.001 | 0.009 |
| Stand | 0.0 ± 0.0 | 0.6 ± 0.2 | 0.1 ± 0.1 | 0.3 ± 0.3 | 0.5 ± 0.2 | 0.2 ± 0.1 | 0.826 | 0.139 | 0.708 |
| Social | 0.0 ± 0.0 | 0.1 ± 0.1 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.5 ± 0.3 | 0.0 ± 0.0 | 0.164 | 0.097 | - |
| Unknown | |||||||||
| Unknown | 12.6 ± 6.1 a | 46.6 ± 10.7 b | 43.9 ± 6.7 bd | 19.2 ± 7.9 c | 39.2 ± 11.1 b d | 37.1 ± 11.6 d | 0.623 | <0.001 | <0.001 |
| Camera inactive | 11.6 ± 6.4 a | 22.2 ± 8.6 b | 12.0 ± 5.2 a | 15.3 ± 8.7 a | 26.0 ± 9.9 b | 23.6 ± 13.2 b | <0.001 | <0.001 | 0.002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fanning, L.; Larsen, H.; Taylor, P.S. A Preliminary Study Investigating the Impact of Musical Concerts on the Behavior of Captive Fiordland Penguins (Eudyptes pachyrhynchus) and Collared Peccaries (Pecari tajacu). Animals 2020, 10, 2035. https://doi.org/10.3390/ani10112035
Fanning L, Larsen H, Taylor PS. A Preliminary Study Investigating the Impact of Musical Concerts on the Behavior of Captive Fiordland Penguins (Eudyptes pachyrhynchus) and Collared Peccaries (Pecari tajacu). Animals. 2020; 10(11):2035. https://doi.org/10.3390/ani10112035
Chicago/Turabian StyleFanning, Lara, Hannah Larsen, and Peta S. Taylor. 2020. "A Preliminary Study Investigating the Impact of Musical Concerts on the Behavior of Captive Fiordland Penguins (Eudyptes pachyrhynchus) and Collared Peccaries (Pecari tajacu)" Animals 10, no. 11: 2035. https://doi.org/10.3390/ani10112035
APA StyleFanning, L., Larsen, H., & Taylor, P. S. (2020). A Preliminary Study Investigating the Impact of Musical Concerts on the Behavior of Captive Fiordland Penguins (Eudyptes pachyrhynchus) and Collared Peccaries (Pecari tajacu). Animals, 10(11), 2035. https://doi.org/10.3390/ani10112035







