Vaginal Microbiota Is Stable throughout the Estrous Cycle in Arabian Mares
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Design
2.2. Progesterone Testing
2.3. Vaginal Sampling Procedures
2.4. Cytological Analysis
2.5. Culture-Dependent Processing
2.6. Culture-Independent Processing
2.6.1. Library Preparation and Sequencing
2.6.2. Amplicon Sequences Processing and Analysis
2.7. Statistical Analysis
2.7.1. Cytological and Culture-Dependent Data
2.7.2. Amplicon Sequencing Data
2.8. Core Microbiome Calculation
2.9. Nucleotide Sequences
3. Results
3.1. Vaginal Microbiome Characterization by Culture-Processing
3.2. Vaginal Microbiome Characterization by Non-Culture Processing
3.2.1. Alpha Diversity
3.2.2. Beta Diversity
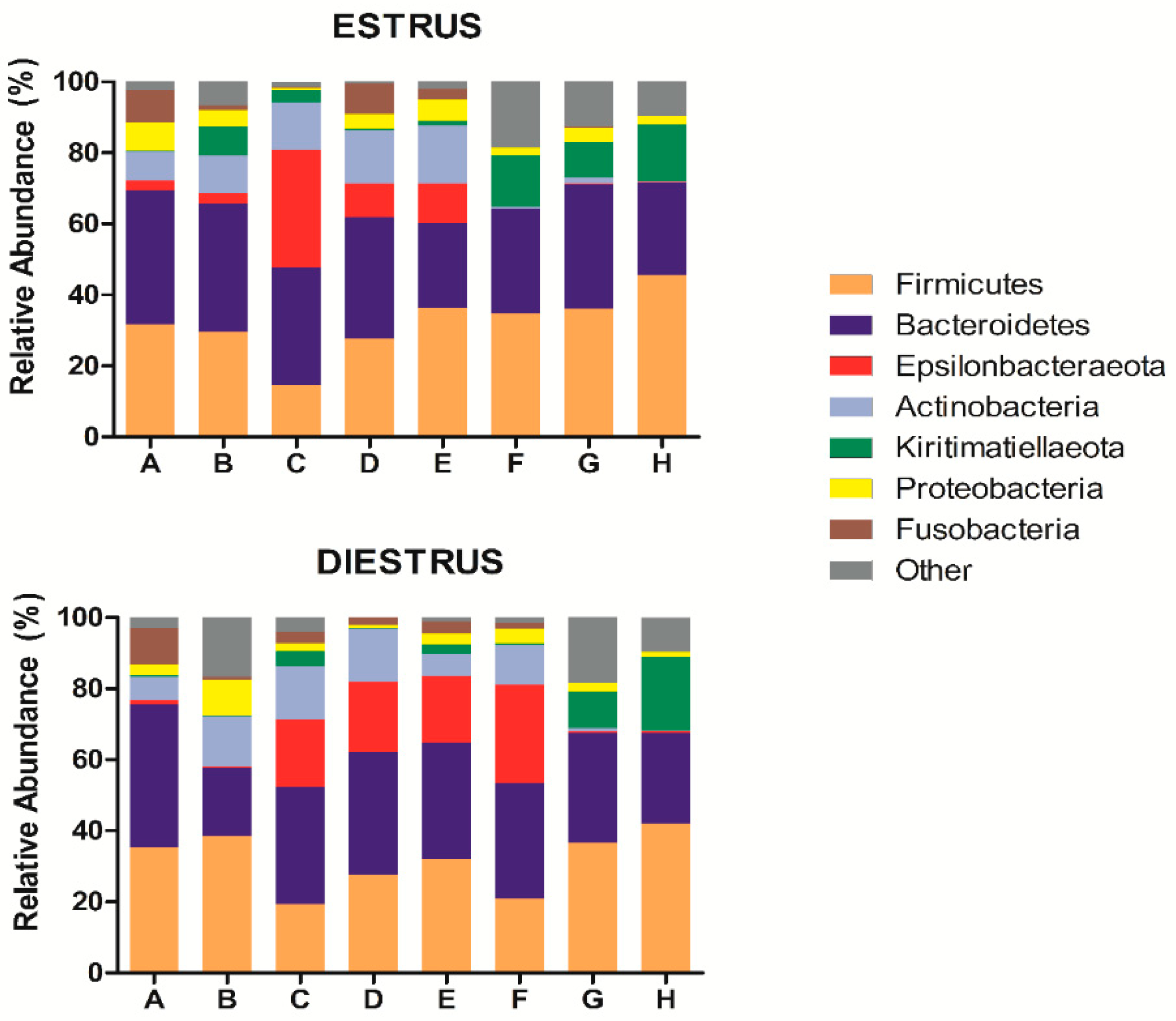
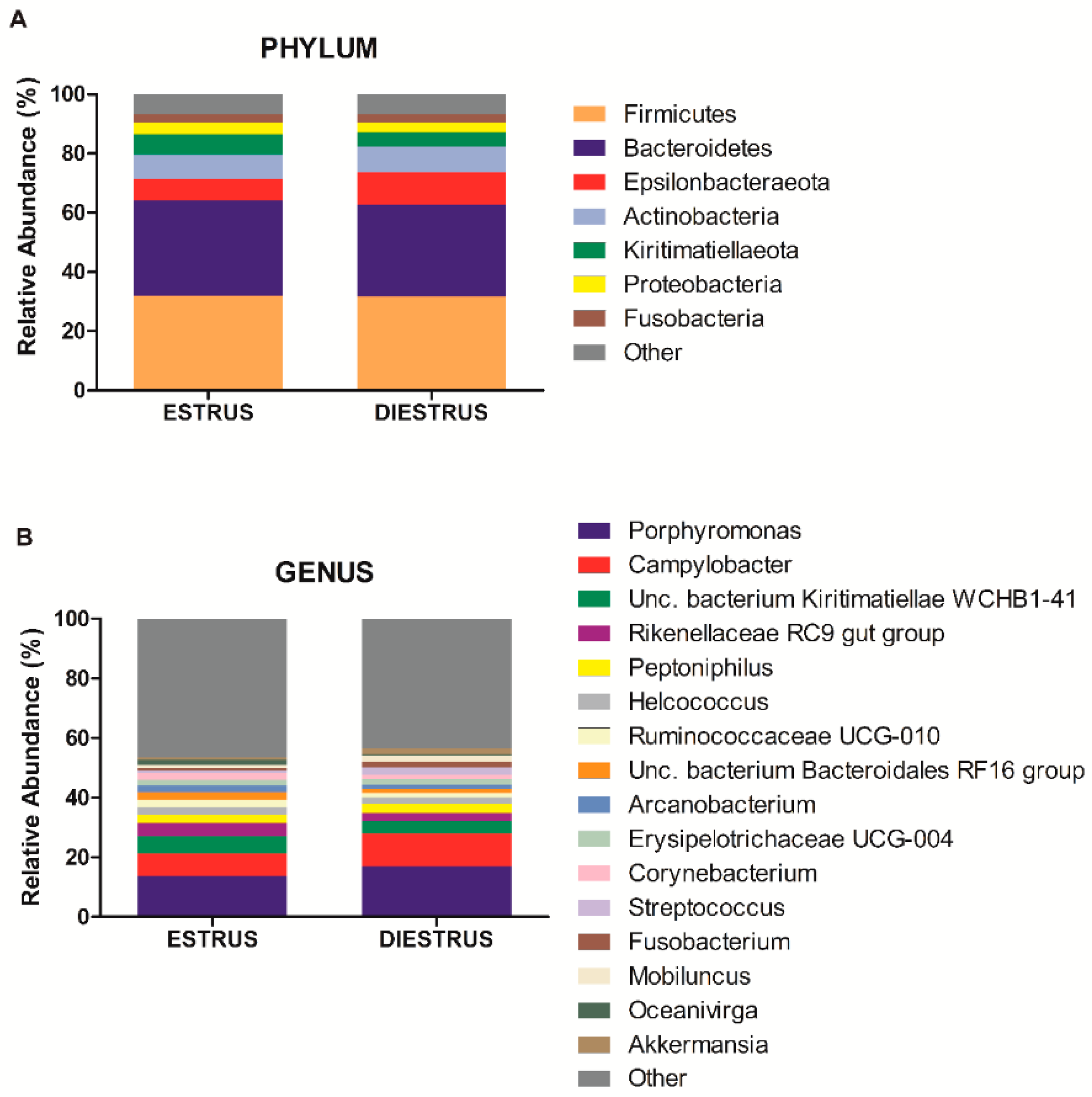
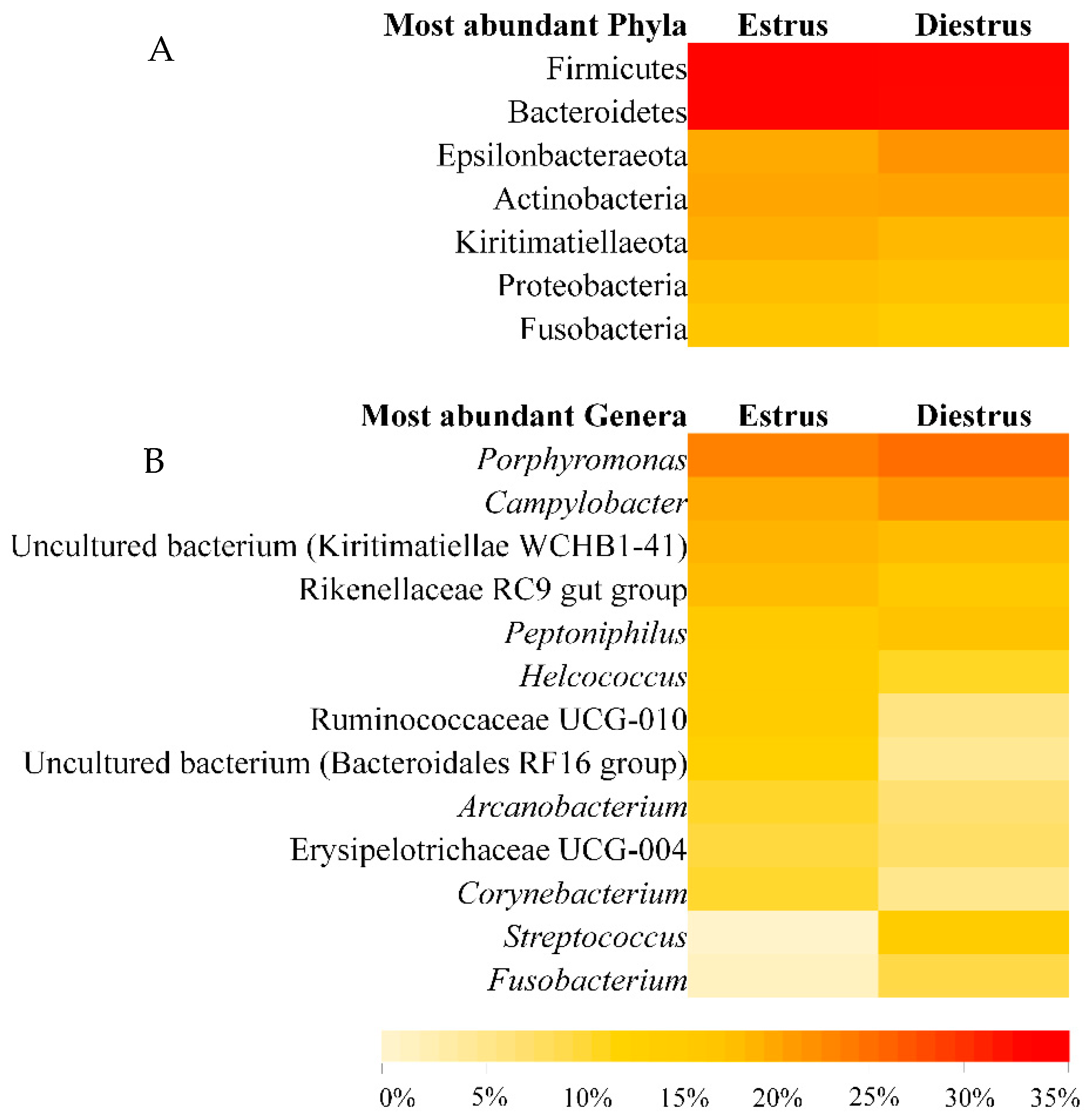
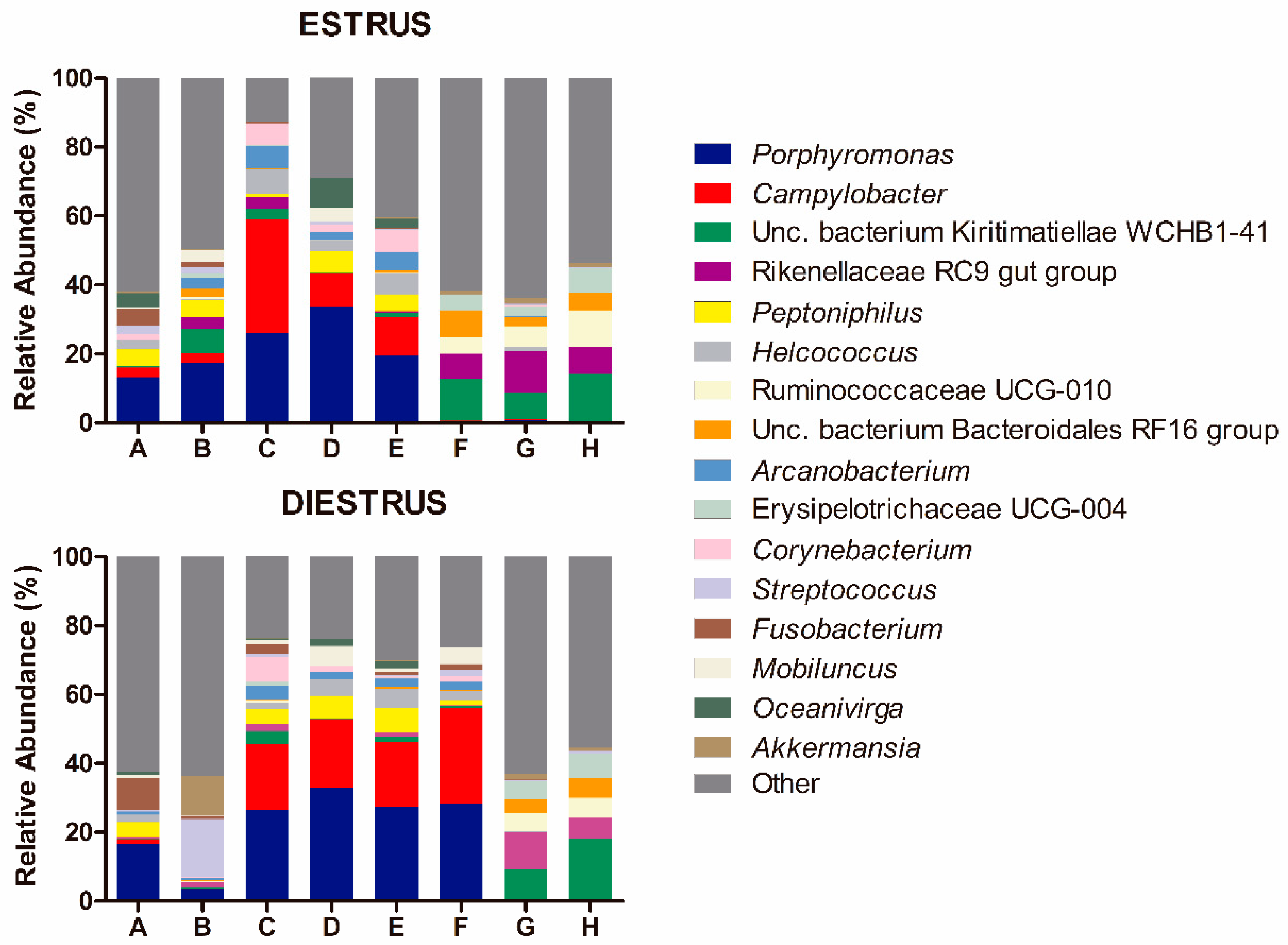
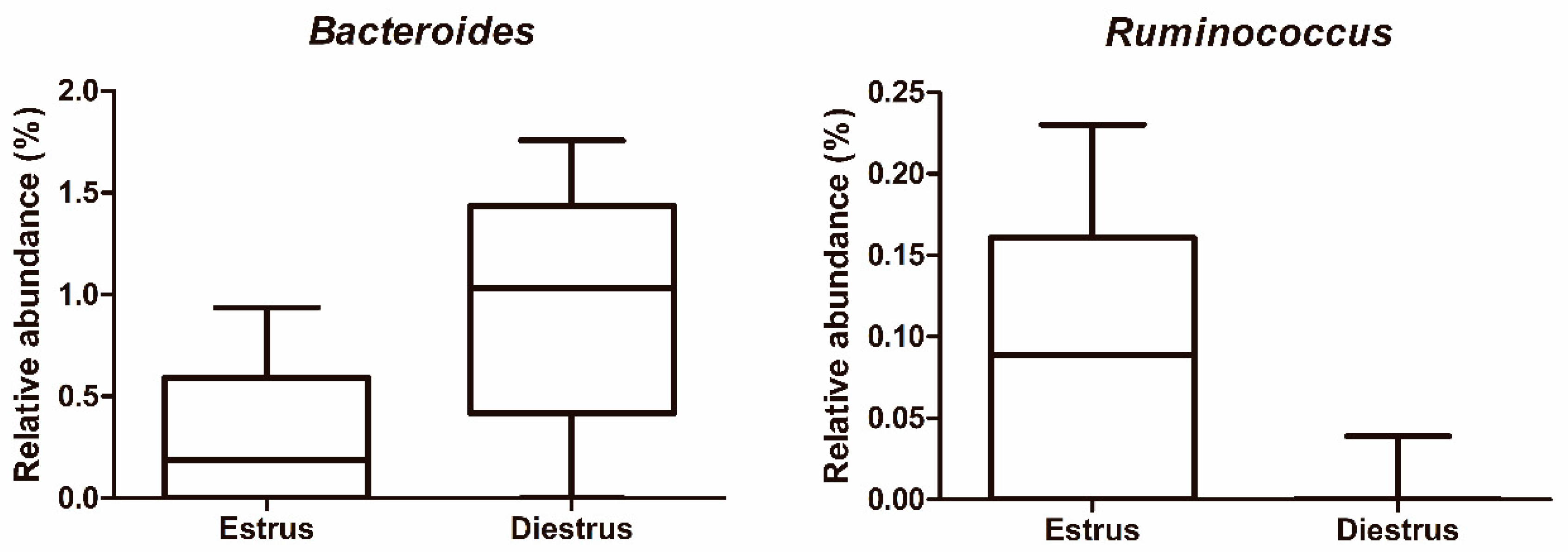
3.2.3. Taxonomic Composition of Caudal Vagina (Vestibule) in Mares in Estrus and Diestrus
3.3. Vaginal Cytological Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Christoffersen, M.; Troedsson, M. Inflammation and fertility in the mare. Reprod. Domest. Anim. 2017, 52, 14–20. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, M.; McKinnon, A. Breeding the problem mare. In Equine Reproduction, 2nd ed.; McKinnon, A., Vaala, W., Varner, D., Eds.; Wiley-Blackwell: Oxford, UK, 2011; Volume 2, pp. 2620–2642. [Google Scholar]
- Nelson, D.B.; Rockwell, L.C.; Prioleau, M.D.; Goetzl, L. The role of the bacterial microbiota on reproductive and pregnancy health. Anaerobe 2016, 42, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Pandya, S.; Ravi, K.; Srinivas, V.; Jadhav, S.; Khan, A.; Arun, A.; Riley, L.W.; Madhivanan, P. Comparison of culture-dependent and culture-independent molecular methods for characterization of vaginal microflora. J. Med. Microbiol. 2017, 66, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Holyoak, G.R.; Lyman, C.C.; Wieneke, X.D.; DeSilva, U. The equine endometrial microbiome. Clin. Theriogenol. Web Arch. 2018, 10, 273–278. [Google Scholar]
- Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.K.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; et al. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. USA 2011, 108, 4680–4687. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.D.; Lee, R.A.; Balen, A.; Rutherford, A.J. Bacterial vaginal flora in relation to changing oestrogen levels. Int. J. STD AIDS 2007, 18, 308–311. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.A.; Livermore, J.A.; Alberts, S.C.; Tung, J.; Archie, E.A. Ovarian cycling and reproductive state shape the vaginal microbiota in wild baboons. Microbiome 2017, 5, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lorenzen, E.; Kudirkienė, E.; Gutman, N.; Grossi, A.B.; Agerholm, J.S.; Erneholm, K.; Skytte, C.; Dalgaard, M.D.; Bojesen, A.M. The vaginal microbiome is stable in prepubertal and sexually mature Ellegaard Göttingen Minipigs throughout an estrous cycle. Veter Res. 2015, 46, 125. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ametaj, B.N.; Ambrose, D.J.; Gänzle, M.G. Characterisation of the bacterial microbiota of the vagina of dairy cows and isolation of pediocin-producing Pediococcus acidilactici. BMC Microbiol. 2013, 13, 19. [Google Scholar] [CrossRef]
- Quereda, J.J.; Barba, M.; Mocé, M.L.; Gomis, J.; Jiménez-Trigos, E.; García-Muñoz, Á.; Gómez-Martín, Á.; González-Torres, P.; Carbonetto, B.; García-Roselló, E. Vaginal Microbiota Changes During Estrous Cycle in Dairy Heifers. Front. Veter Sci. 2020, 7, 371. [Google Scholar] [CrossRef]
- Swartz, J.D.; Elachman, M.; Ewestveer, K.; OâNeill, T.; Egeary, T.; Kott, R.W.; Berardinelli, J.G.; Hatfield, P.G.; Thomson, J.M.; Eroberts, A.; et al. Characterization of the Vaginal Microbiota of Ewes and Cows Reveals a Unique Microbiota with Low Levels of Lactobacilli and Near-Neutral pH. Front. Veter Sci. 2014, 1, 19. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.A.; Beasley, D.E.; Dunn, R.R.; Archie, E.A. Lactobacilli Dominance and Vaginal pH: Why Is the Human Vaginal Microbiome Unique? Front. Microbiol. 2016, 7, 1936. [Google Scholar] [CrossRef]
- Fraga, M.; Perelmuter, K.; Delucchi, L.; Cidade, E.; Zunino, P. Vaginal lactic acid bacteria in the mare: Evaluation of the probiotic potential of native Lactobacillus spp. and Enterococcus spp. strains. Antonie Leeuwenhoek 2007, 93, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Heil, B.; Thompson, S.; Kearns, T.; Davolli, G.; King, G.; Sones, J. Metagenetic Characterization of the Resident Equine Uterine Microbiome Using Multiple Techniques. J. Equine Veter Sci. 2018, 66, 111. [Google Scholar] [CrossRef]
- Ginther, O.; Utt, M.; Bergfelt, D.; Beg, M. Controlling interrelationships of progesterone/LH and estradiol/LH in mares. Anim. Reprod. Sci. 2006, 95, 144–150. [Google Scholar] [CrossRef]
- Groppetti, D.; Pecile, A.; Barbero, C.; Martino, P. Vaginal bacterial flora and cytology in proestrous bitches: Role on fertility. Theriogenology 2012, 77, 1549–1556. [Google Scholar] [CrossRef]
- Manes, J.; Campero, C.; Hozbor, F.; Alberio, R.; Ungerfeld, R. Vaginal Histological Changes after Using Intravaginal Sponges for Oestrous Synchronization in Anoestrous Ewes. Reprod. Domest. Anim. 2015, 50, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Quereda, J.J.; García-Roselló, E.; Barba, M.; Mocé, M.L.; Gomis, J.; Jiménez-Trigos, E.; Bataller, E.; Martínez-Boví, R.; García-Muñoz, Á.; Gómez-Martín, Á. Use of Probiotics in Intravaginal Sponges in Sheep: A Pilot Study. Animals 2020, 10, 719. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Price, M.N.; Dehal, P.S.; Arkin, A.P. FastTree: Computing Large Minimum Evolution Trees with Profiles instead of a Distance Matrix. Mol. Biol. Evol. 2009, 26, 1641–1650. [Google Scholar] [CrossRef]
- Lozupone, C.; Knight, R. UniFrac: A New Phylogenetic Method for Comparing Microbial Communities. Appl. Environ. Microbiol. 2005, 71, 8228–8235. [Google Scholar] [CrossRef]
- Abraham, A.; Pedregosa, F.; Eickenberg, M.; Gervais, P.; Mueller, A.; Kossaifi, J.; Gramfort, A.; Thirion, B.; Varoquaux, G. Machine learning for neuroimaging with scikit-learn. Front. Aging Neurosci. 2014, 8, 14. [Google Scholar] [CrossRef]
- Mandal, S.; Van Treuren, W.; White, R.A.; Eggesbø, M.Å.; Knight, R.T.; Peddada, S.D. Analysis of composition of microbiomes: A novel method for studying microbial composition. Microb. Ecol. Health Dis. 2015, 26, 27663. [Google Scholar] [CrossRef]
- Anderson, M.J.; Walsh, D.C.I. Permanova, anosim, and the Mantel test in the face of heterogeneous dispersions: What null hypothesis are you testing? Ecol. Monogr. 2013, 83, 557–574. [Google Scholar] [CrossRef]
- Li, K.; Bihan, M.; Methé, B.A. Analyses of the Stability and Core Taxonomic Memberships of the Human Microbiome. PLoS ONE 2013, 8, e63139. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Martin, D.H.; Marrazzo, J.M. The Vaginal Microbiome: Current Understanding and Future Directions. J. Infect. Dis. 2016, 214, S36–S41. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, S.; Yeoman, C.J.; Janga, S.C.; Thomas, S.M.; Ho, M.; Leigh, S.R.; White, B.A.; Wilson, B.A.; Stumpf, R.M.; Primate Microbiome Consortium. Primate vaginal microbiomes exhibit species specificity without universal Lactobacillus dominance. ISME J. 2014, 8, 2431–2444. [Google Scholar] [CrossRef] [PubMed]
- Barfod, K.K.; Roggenbuck, M.; Hansen, L.H.; Schjørring, S.; Larsen, S.T.; Sørensen, S.; Krogfelt, K.A. The murine lung microbiome in relation to the intestinal and vaginal bacterial communities. BMC Microbiol. 2013, 13, 303. [Google Scholar] [CrossRef]
- Neuendorf, E.; Gajer, P.; Bowlin, A.K.; Marques, P.X.; Ma, B.; Yang, H.; Fu, L.; Humphrys, M.S.; Forney, L.J.; Myers, G.S.; et al. Chlamydia caviae infection alters abundance but not composition of the guinea pig vaginal microbiota. Pathog. Dis. 2015, 73. [Google Scholar] [CrossRef]
- Clemmons, B.A.; Reese, S.T.; Dantas, F.G.; Franco, G.A.; Smith, T.P.L.; Adeyosoye, O.I.; Pohler, K.G.; Myer, P.R. Vaginal and Uterine Bacterial Communities in Postpartum Lactating Cows. Front. Microbiol. 2017, 8, 1047. [Google Scholar] [CrossRef] [PubMed]
- Lyman, C.C.; Holyoak, G.R.; Meinkoth, K.; Wieneke, X.; Chillemi, K.A.; DeSilva, U. Canine endometrial and vaginal microbiomes reveal distinct and complex ecosystems. PLoS ONE 2019, 14, e0210157. [Google Scholar] [CrossRef]
- Yang, X.; Cheng, G.; Li, C.; Yang, J.; Li, J.; Chen, D.; Zou, W.; Jin, S.; Zhang, H.; Li, D.; et al. The normal vaginal and uterine bacterial microbiome in giant pandas (Ailuropoda melanoleu ca). Microbiol. Res. 2017, 199, 1–9. [Google Scholar] [CrossRef]
- Heil, B.A.; Paccamonti, D.L.; Douglas, N.C. Role for the mammalian female reproductive tract microbiome in pregnancy outcomes. Physiol. Genom. 2019, 51, 390–399. [Google Scholar] [CrossRef]
- Rock, K.S.; Love, B.C.; DeSilva, U.; Rezabek, G.B.; Meijer, W.G.; Carrington, S.D. Detectable differences in the endometrial microbiome between normal and susceptible mares using metagenomic profiling and conventional bacterial culture. Clin. Theriogenol. 2011, 3. Available online: https://www.ivis.org/library/sft/sft-theriogenology-annual-conference-milwaukee-2011/detectable-differences-endometrial-microbiome-between-normal-and-susceptible-mares-using-metagenomic (accessed on 16 June 2020).
- Sathe, S.; Leiken, A.; Plummer, P. Metagenomic sequencing of the uterine microbial environment during estrus and early pregnancy in mares. Clin. Theriogenol. 2017, 9, 453. [Google Scholar]
- Husso, A.; Jalanka, J.; Alipour, M.J.; Huhti, P.; Kareskoski, M.; Pessa-Morikawa, T.; Iivanainen, A.; Niku, M. The composition of the perinatal intestinal microbiota in horse. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Kamińska, D.; Gajecka, M. Is the role of human female reproductive tract microbiota underestimated? Benef. Microbes 2017, 8, 327–343. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.C.; Weese, J.S. Understanding the Intestinal Microbiome in Health and Disease. Veter Clin. N. Am. Equine Pr. 2018, 34, 1–12. [Google Scholar] [CrossRef]
- Xia, Y.W.; Cornelius, A.J.; Donnelly, C.G.; Bicalho, R.C.; Cheong, S.H.; Sonesc, J.L. Metagenomic analysis of the equine placental microbiome. Clin. Theriogenol. 2017, 9, 452. [Google Scholar]
- Laguardia-Nascimento, M.; Branco, K.M.G.R.; Gasparini, M.R.; Giannattasio-Ferraz, S.; Leite, L.R.; Araújo, F.M.G.; Salim, A.C.D.M.; Nicoli, J.R.; Oliveira, G.C.; Barbosa-Stancioli, E.F. Vaginal Microbiome Characterization of Nellore Cattle Using Metagenomic Analysis. PLoS ONE 2015, 10, e0143294. [Google Scholar] [CrossRef]
- Ata, B.; Yildiz, S.; Turkgeldi, E.; Brocal, V.P.; Dinleyici, E.C.; Moya, A.; Urman, B. The Endobiota Study: Comparison of Vaginal, Cervical and Gut Microbiota between Women with Stage 3/4 Endometriosis and Healthy Controls. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Flores, R.; Shi, J.; Fuhrman, B.; Xu, X.; Veenstra, T.D.; Gail, M.H.; Gajer, P.; Ravel, J.; Goedert, J.J. Fecal microbial determinants of fecal and systemic estrogens and estrogen metabolites: A cross-sectional study. J. Transl. Med. 2012, 10, 253. [Google Scholar] [CrossRef]
- Quaranta, G.; Sanguinetti, M.; Masucci, L. Fecal Microbiota Transplantation: A Potential Tool for Treatment of Human Female Reproductive Tract Diseases. Front. Immunol. 2019, 10, 2653. [Google Scholar] [CrossRef]
- Bulmer, L.S.; Murray, J.-A.; Burns, N.M.; Garber, A.; Wemelsfelder, F.; McEwan, N.R.; Hastie, P.M. High-starch diets alter equine faecal microbiota and increase behavioural reactivity. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Salem, S.E.; Maddox, T.W.; Berg, A.; Antczak, P.; Ketley, J.M.; Williams, N.J.; Archer, D.C. Variation in faecal microbiota in a group of horses managed at pasture over a 12-month period. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Huang, B.; Fettweis, J.M.; Brooks, J.P.; Jefferson, K.K.; Buck, G.A. The Changing Landscape of the Vaginal Microbiome. Clin. Lab. Med. 2014, 34, 747–761. [Google Scholar] [CrossRef]
- Prince, A.L.; Chu, D.M.; Seferovic, M.D.; Antony, K.M.; Ma, J.; Aagaard, K.M. The Perinatal Microbiome and Pregnancy: Moving Beyond the Vaginal Microbiome. Cold Spring Harb. Perspect. Med. 2015, 5, a023051. [Google Scholar] [CrossRef] [PubMed]
- Polak, K.L.; Kammlade, W.G. Vaginal pH during estrus in mares. Theriogenology 1981, 15, 271–276. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barba, M.; Martínez-Boví, R.; Quereda, J.J.; Mocé, M.L.; Plaza-Dávila, M.; Jiménez-Trigos, E.; Gómez-Martín, Á.; González-Torres, P.; Carbonetto, B.; García-Roselló, E. Vaginal Microbiota Is Stable throughout the Estrous Cycle in Arabian Mares. Animals 2020, 10, 2020. https://doi.org/10.3390/ani10112020
Barba M, Martínez-Boví R, Quereda JJ, Mocé ML, Plaza-Dávila M, Jiménez-Trigos E, Gómez-Martín Á, González-Torres P, Carbonetto B, García-Roselló E. Vaginal Microbiota Is Stable throughout the Estrous Cycle in Arabian Mares. Animals. 2020; 10(11):2020. https://doi.org/10.3390/ani10112020
Chicago/Turabian StyleBarba, Marta, Rebeca Martínez-Boví, Juan José Quereda, María Lorena Mocé, María Plaza-Dávila, Estrella Jiménez-Trigos, Ángel Gómez-Martín, Pedro González-Torres, Belén Carbonetto, and Empar García-Roselló. 2020. "Vaginal Microbiota Is Stable throughout the Estrous Cycle in Arabian Mares" Animals 10, no. 11: 2020. https://doi.org/10.3390/ani10112020
APA StyleBarba, M., Martínez-Boví, R., Quereda, J. J., Mocé, M. L., Plaza-Dávila, M., Jiménez-Trigos, E., Gómez-Martín, Á., González-Torres, P., Carbonetto, B., & García-Roselló, E. (2020). Vaginal Microbiota Is Stable throughout the Estrous Cycle in Arabian Mares. Animals, 10(11), 2020. https://doi.org/10.3390/ani10112020





