Effects of the Appropriate Addition of Antioxidants from Pinus densiflora and Mentha canadensis Extracts on Methane Emission and Rumen Fermentation
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. In Vitro Batch Fermentation
2.3. Total Phenolic and Flavonoid Content of Pinus densiflora and Mentha canadensis Extracts
2.4. Total DPPH, ABTS and HO, NO Radical Assay
2.5. Analysis of In Vitro End-Products
2.6. Relative Quantification of Specific Ruminal Microbes
2.7. Statistical Analysis
3. Results
3.1. Total Phenol and Flavonoid Contents, DPPH and ABTS Assays, OH and NO Scavenging Activities
3.2. Effect of Terpene-Based Plant Extracts on Rumen Fermentation and Dry Matter Degradability
3.3. Effect of Terpene-Based Plant Extracts on Gas Profiles
3.4. Effect of Terpene-Based Plant Extracts on VFA Profiles
3.5. Relative Quantification of Specific Ruminal Microbes
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moss, A.R.; Jouany, J.P.; Newbold, J. Methane production by ruminants: Its contribution to global warming. Ann. Zootech. 2000, 49, 231–253. [Google Scholar] [CrossRef]
- Calabrò, S.; Infascelli, F.; Tudisco, R.; Musco, N.; Grossi, M.; Monastra, G.; Cutrignelli, M.I. Estimation of in vitro methane production in buffalo and cow. Buffalo Bull. 2013, 32, 924–927. Available online: https://www.researchgate.net/publication/282576097 (accessed on 10 April 2013).
- Cummings, J.H.; Macfarlane, G.T. Gastrointestinal effects of prebiotics. Br. J. Nutr. 2002, 87, S145–S151. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.Y.; Zhang, C.H.; He, X.L.; Huang, L.; Yin, Z.J. Growth performance responses and indicators of gastrointestinal health in early weaned pigs fed Chinese herbal medicine additives-supplemented diets. J. Anim. Vet. Adv. 2011, 10, 1580–1587. [Google Scholar] [CrossRef][Green Version]
- Kwak, C.S.; Moon, S.C.; Lee, M.S. Antioxidant, antimutagenic, and antitumor effects of pine needles (Pinus densiflora). Nutr. Cancer 2006, 56, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Won, S.B.; Jung, G.Y.; Kim, J.; Chung, Y.S.; Hong, E.K.; Kwon, Y.H. Protective effect of Pinus koraiensis needle water extract against oxidative stress in HepG2 cells and obese mice. J. Med. Food 2013, 16, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.J.; Wi, H.R.; Kim, H.R.; Park, K.W.; Hwang, K.A. Pinus densiflora Sieb. et Zucc. alleviates lipogenesis and oxidative stress during oleic acid-induced steatosis in HepG2 cells. Nutrients 2014, 6, 2956–2972. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, H.G.; Lee, H.W.; Kim, W.Y.; Ahn, Y.C.; Son, C.G. Pine needle extract prevents hippocampal memory impairment in acute restraint stress mouse model. J. Ethnopharmacol. 2017, 207, 226–236. [Google Scholar] [CrossRef]
- Koukos, P.K.; Papadopoulou, K.I.; Patiaka, D.T.; Papagiannopoulos, A.D. Chemical composition of essential oils from needles and twigs of balkan pine (Pinus peuce Grisebach) grown in Northern Greece. J. Agric. Food Chem. 2000, 48, 1266–1268. [Google Scholar] [CrossRef]
- Kim, Y.J.; Cho, B.J.; Ko, M.S.; Jung, M.J.; Kim, H.R.; Song, H.S.; Lee, J.Y.; Sim, S.S.; Kim, S.J. Anti-oxidant and Anti-aging Activities of Essential Oils of Pinus densiflora Needles and Twigs. Pharm. Soc. Korea 2010, 54, 215–225. [Google Scholar]
- Hong, E.J.; Na, K.J.; Choi, I.G.; Choi, K.C.; Jeung, E.B. Antibacterial and Antifungal Effects of Essential Oils from Coniferous Trees. Biol. Pharm. Bull. 2004, 27, 863–866. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.H.; Kim, H.J.; Lee, J.Y.; Cho, Y.J.; An, B.J. Major Compound Analysis and Assessment of Natural Essential Oil on Anti-Oxidative and Anti-Microbial Effects. J. Life Sci. 2012, 22, 1344–1351. [Google Scholar] [CrossRef]
- Brahmi, F.; Khodir, M.; Mohamed, C.; Pierre, D. Chemical composition and biological activities of Mentha species. Aromat. Med. Plants-Back Nat. 2017, 47–78. [Google Scholar] [CrossRef]
- Snoussi, M.; Noumi, E.; Trabelsi, N.; Flamini, G.; Papetti, A.; Feo, V.D. Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules 2015, 20, 4402–4424. [Google Scholar] [CrossRef]
- Kim, D.H.; Lee, S.J.; Oh, D.S.; Lee, I.D.; Eom, J.S.; Park, H.Y.; Choi, S.H.; Lee, S.S. In vitro evaluation of Rhus succedanea extracts for ruminants. Asian-Australas. J. Anim. Sci. 2018, 31, 1635–1642. [Google Scholar] [CrossRef]
- McDougall, E.I. Studies on ruminant saliva. 1. The composition and output of sheep’s saliva. Biochem. J. 1948, 43, 99–109. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.R. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Kang, S.N.; Semeneh, S.; Lee, J.S.; Lee, S.; Lee, H.J.; Kim, G.R.; Yeo, J.H.; Kim, J.Y. Evaluation of Antioxidant and Antimicrobial Activities of Ethanol Extracts of Three Kinds of Strawberries. Prev. Nutr. Food Sci. 2017, 22, 203–210. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Elizabeth, K.; Rao, M.N.A. Oxygen radical scavenging activity of curcumin. Int. J. Pharm. 1990, 58, 237–240. [Google Scholar]
- Lee, H.S. Antioxidative activity of browning reaction products isolated from storage-aged orange juice. J. Agric. Food Chem. 1992, 40, 550–552. [Google Scholar] [CrossRef]
- Lee, S.J.; Jeong, J.S.; Shin, N.H.; Lee, S.K.; Kim, H.S.; Eom, J.S.; Lee, S.S. Impact of Ecklonia stolonifera extract on in vitro ruminal fermentation characteristics, methanogenesis, and microbial populations. Asian Australas. J. Anim. Sci. 2019, 32, 1864–1872. [Google Scholar] [CrossRef]
- Lee, S.J.; Shin, N.H.; Jeong, J.S.; Kim, E.T.; Lee, S.K.; Lee, I.D.; Lee, S.S. Effects of Gelidium amansii extracts on in vitro ruminal fermentation characteristics, methanogenesis, and microbial populations. Asian Australas. J. Anim. Sci. 2018, 31, 71–79. [Google Scholar] [CrossRef]
- Denman, S.E.; McSweeney, C.S. Development of a real-time PCR assay for monitoring anaerobic fungal and cellulolytic bacterial populations within the rumen. FEMS Microbiol. Ecol. 2006, 58, 572–582. [Google Scholar] [CrossRef]
- Koike, S.; Kobayashi, Y. Development and use of competitive PCR assays for the rumen cellulolytic bacteria: Fibrobacter succinogenes, Ruminococcus albus and Ruminococcus flavefaciens. FEMS Microbiol. Lett. 2001, 204, 361–366. [Google Scholar] [CrossRef]
- Skillman, L.C.; Toovey, A.F.; Williams, A.J.; Wright, A.D.G. Development and validation of a real-time PCR method to quantify rumen protozoa and examination of variability between Entodinium populations in sheep offered a hay-based diet. Appl. Environ. Microb. 2006, 72, 200–2006. [Google Scholar] [CrossRef]
- Luton, P.E.; Wayne, J.M.; Sharp, R.J.; Riley, P.W. The mcrA gene as an alternative to 16S rRNA in the phylogenetic analysis of methanogen populations in landfill. Microbiology 2002, 148, 3521–3530. [Google Scholar] [CrossRef]
- SAS Institute Inc. SAS/STAT User’s Guide: Version 9.2; SAS Institute Inc.: Cary, NC, USA, 2002. [Google Scholar]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef]
- Arigò, A.; Česla, P.; Šilarová, P.; Calabrò, M.L.; Česlová, L. Development of extraction method for characterization of free and bonded polyphenols in barley (Hordeum vulgare L.) grown in Czech Republic using liquid chromatography-tandem mass spectrometry. Food Chem. 2018, 245, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R.; Yang, R. Optimization of a new mobile phase to know the complex and real polyphenolic composition: Towards a total phenolic index using high-performance liquid chromatography. J. Chromatogr. A 2003, 1018, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, K. Occurrence and content of hydroxycinnamic and hydroxy-benzoic acid compounds in foods. Crit. Rev. Food Sci. Nutr. 1989, 28, 315–347. [Google Scholar] [CrossRef]
- Yusof, S.; Ghazali, H.M.; King, G.S. Naringin content in local citrus fruits. Food Chem. 1990, 37, 113–121. [Google Scholar] [CrossRef]
- McCartney-Francis, N.; Allen, J.B.; Mizel, D.E.; Albina, J.E.; Xie, Q.W.; Nathan, C.F.; Wahl, S.M. Supperession of arthritis by an inhibitor of nitric oxide synthase. J. Exp. Med. 1993, 178, 749–754. [Google Scholar] [CrossRef]
- McCullough, M.E.; Smart, J.W.W.G. Effects of intake of forge level on ruminal turnover rate, bacterial protein synthesis and duodenal amino acid flow in sheep. J. Anim. Sci. 1968, 62, 216. [Google Scholar]
- Lee, J.H.; Chung, H.S.; Kim, I.H.; Kim, S.H. Antioxidant Properties of Pinus koraiensis Needle Powder Extracts as Influenced by Drying Methods. Food Eng. Prog. 2013, 17, 396–400. [Google Scholar] [CrossRef]
- Oh, M.H.; Whang, H.J. Chemical Composition of Several Herb Plants. Korean J. Food Sci. Technol. 2003, 35, 1–6. [Google Scholar]
- Salem, A.Z.M.; Kholif, A.E.; Olivares, M.; Elghandour, M.M.; Mellado, M.; Arece, J. Influence of S. babylonica extract on feed intake, growth performance and diet in vitro gas production profile in young lambs. Trop. Anim. Health Prod. 2014, 46, 213–219. [Google Scholar] [CrossRef]
- Boussaada, A.; Arhab, R.; Calabrò, S.; Grazioli, R.; Ferrara, M.; Musco, N.; Thlidjane, M.; Cutrignelli, M.I. Effect of Eucalyptus globulus leaves extracts on in vitro rumen fermentation, methanogenesis, degradability and protozoa population. Ann. Anim. Sci. 2018, 18, 753–767. [Google Scholar] [CrossRef]
- Oskoueian, E.; Abdullah, N.; Oskoueian, A. Effects of flavonoids on rumen fermentation activity, methane production, and microbial population. BioMed Res. Int. 2013, 2013, 349129. [Google Scholar] [CrossRef] [PubMed]
- Aderao, G.N.; Sahoo, A.; Bhatt, R.S.; Kumawat, P.K.; Soni, L. In vitro rumen fermentation kinetics, metabolite production, methane and substrate degradability of polyphenol rich plant leaves and their component complete feed blocks. J. Anim. Sci. Technol. 2018, 60, 26. [Google Scholar] [CrossRef] [PubMed]
- Akanmu, A.M.; Hassen, A.; Adejoro, F.A. Gas Production, Digestibility and Efficacy of Stored or Fresh Plant Extracts to Reduce Methane Production on Different Substrates. Animals 2020, 10, 146. [Google Scholar] [CrossRef] [PubMed]
- Lowry, J.B.; Kennedy, P.M. Fermentation of flavonols by rumen organisms. Proc. Aust. Soc. Anim. Prod. 1996, 21, 366. [Google Scholar]
- Olagaray, K.E.; Bradford, B.J. Plant flavonoids to improve productivity of ruminants. Anim. Feed Sci. Technol. 2019, 251, 21–36. [Google Scholar] [CrossRef]
- Broudiscou, L.P.; Papon, Y.; Broudiscou, A.F. Effects of dry plant extracts on fermentation and methanogenesis in continuous culture of rumen microbes. Anim. Feed Sci. Technol. 2000, 87, 263–277. [Google Scholar] [CrossRef]

| Content | Terpene-Based Plant Extract | |
|---|---|---|
| Pinus densiflora | Mentha canadensis | |
| Total polyphenol content (µg GAE/g) | 925 ± 113.24 | 469 ± 14.27 |
| Total flavonoid content (µg QE/g) | 680.90 ± 67.49 | 715.77 ± 61.01 |
| Total antioxidant capacity | ||
| DPPH | ||
| 200 (µg/mL) | 49.38 ± 2.54 | 48.25 ± 1.28 |
| 100 (µg/mL) | 43.27 ± 2.66 | 47.56 ± 1.63 |
| 50 (µg/mL) | 21.90 ± 1.09 | 37.95 ± 4.63 |
| 10 (µg/mL) | 16.63 ± 3.39 | 17.38 ± 1.53 |
| ABTS | ||
| 200 (µg/mL) | 78.85 ± 0.98 | 91.36 ± 0.29 |
| 100 (µg/mL) | 67.34 ± 0.85 | 90.51 ± 0.68 |
| 50 (µg/mL) | 52.20 ± 3.41 | 82.68 ± 1.20 |
| 10 (µg/mL) | 23.11 ± 1.16 | 79.50 ± 1.97 |
| HO | ||
| 200 (µg/mL) | 2336.36 ± 100.39 | 299.97 ± 43.00 |
| 100 (µg/mL) | 2251.17 ± 137.16 | 179.81 ± 30.84 |
| 50 (µg/mL) | 1707.47 ± 115.93 | 102.46 ± 7.81 |
| 10 (µg/mL) | 790.53 ± 50.97 | 37.11 ± 6.80 |
| NO | ||
| 200 (µg/mL) | 59.31 ± 1.36 | 54.93 ± 2.92 |
| 100 (µg/mL) | 32.42 ± 2.01 | 32.50 ± 2.52 |
| 50 (µg/mL) | 20.59 ± 3.14 | 20.35 ± 3.71 |
| 10 (µg/mL) | 7.94 ± 2.59 | 12.04 ± 3.74 |
| Incubation Time (h) | Treatment | Extract Concentration (1) (mg/L) | SEM (2) | p Value (3) | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 30 | 50 | 70 | T | L | Q | |||
| pH | |||||||||
| 12 | Herb (4) | 6.58 a | 6.60 a | 6.48 b | 6.62 a | 0.01 | 0.0003 | 0.7580 | 0.0099 |
| Pine (5) | 6.79 a | 6.80 a | 6.69 b | 6.82 a | 0.02 | 0.0055 | 0.7644 | 0.0338 | |
| 24 | Herb | 6.24 | 6.53 | 6.49 | 6.51 | 0.11 | 0.2922 | 0.1268 | 0.3111 |
| Pine | 6.70 a | 6.73 a | 6.56 b | 6.71 a | 0.03 | 0.0136 | 0.2929 | 0.2027 | |
| Microbial growth rate, OD at 550 nm | |||||||||
| 12 | Herb | 0.42 a | 0.30 b | 0.41 a | 0.39 ab | 0.03 | 0.0792 | 0.7914 | 0.0901 |
| Pine | 0.34 | 0.34 | 0.40 | 0.35 | 0.06 | 0.8686 | 0.7922 | 0.7290 | |
| 24 | Herb | 0.35 ab | 0.37 ab | 0.37 a | 0.31 b | 0.02 | 0.1179 | 0.2543 | 0.0433 |
| Pine | 0.35 | 0.29 | 0.38 | 0.35 | 0.03 | 0.1862 | 0.5431 | 0.3383 | |
| Glucose (mg∙mL−1) | |||||||||
| 12 | Herb | 0.07 a | 0.06 bc | 0.06 c | 0.07 ab | 0.00 | 0.0244 | 0.1329 | 0.0086 |
| Pine | 0.06 | 0.08 | 0.06 | 0.05 | 0.01 | 0.2376 | 0.1892 | 0.1667 | |
| 24 | Herb | 0.05 b | 0.07 a | 0.05 b | 0.06 ab | 0.01 | 0.0242 | 0.2306 | 0.0446 |
| Pine | 0.06 | 0.06 | 0.05 | 0.05 | 0.01 | 0.3502 | 0.1135 | 0.4862 | |
| Protein (mg∙mL−1) | |||||||||
| 12 | Herb | 0.20 a | 0.17 ab | 0.18 ab | 0.16 b | 0.01 | 0.0962 | 0.0273 | 0.8446 |
| Pine | 0.20 | 0.20 | 0.23 | 0.21 | 0.01 | 0.1537 | 0.2807 | 0.4698 | |
| 24 | Herb | 0.19 b | 0.19 b | 0.20 ab | 0.21 a | 0.01 | 0.0473 | 0.0105 | 0.2879 |
| Pine | 0.22 | 0.22 | 0.23 | 0.21 | 0.01 | 0.3240 | 0.7081 | 0.1946 | |
| Ammonia nitrogen (mg∙mL−1) | |||||||||
| 12 | Herb | 3.18 a | 3.93 a | 1.18 b | 2.93 a | 0.44 | 0.0123 | 0.1272 | 0.6570 |
| Pine | 3.98 | 7.78 | 4.16 | 6.33 | 1.15 | 0.1324 | 0.4231 | 0.3784 | |
| 24 | Herb | 2.60 ab | 2.27 b | 4.31 a | 2.89 ab | 0.52 | 0.0983 | 0.2542 | 0.5618 |
| Pine | 4.80 b | 4.78 b | 5.51 ab | 6.53 a | 0.46 | 0.0854 | 0.0260 | 0.1910 | |
| Incubation Time (h) | Treatment | Extract Concentration (1) (mg/L) | SEM (2) | p Value (3) | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 30 | 50 | 70 | T | L | Q | |||
| DM degradability, % | |||||||||
| 3 | Herb (4) | 19.33 | 18.67 | 17.89 | 19.56 | 0.76 | 0.4468 | 0.8850 | 0.1900 |
| Pine (5) | 22.11 | 21.22 | 23.00 | 21.33 | 1.29 | 0.7519 | 0.9133 | 0.8601 | |
| 6 | Herb | 20.11 | 19.78 | 19.00 | 19.67 | 1.14 | 0.9159 | 0.6732 | 0.7375 |
| Pine | 23.00 | 22.00 | 23.11 | 22.22 | 0.82 | 0.7196 | 0.7093 | 0.8722 | |
| 9 | Herb | 21.44 | 21.22 | 21.22 | 20.00 | 1.01 | 0.7416 | 0.3842 | 0.5983 |
| Pine | 23.00 | 23.89 | 23.44 | 23.11 | 1.14 | 0.9444 | 0.9673 | 0.5882 | |
| 12 | Herb | 27.11 | 26.33 | 27.11 | 27.33 | 0.38 | 0.332 | 0.5261 | 0.1647 |
| Pine | 29.89 | 29.78 | 29.11 | 31.00 | 0.85 | 0.505 | 0.5529 | 0.2887 | |
| 24 | Herb | 30.78 | 33.78 | 34.67 | 29.44 | 2.00 | 0.2816 | 0.8716 | 0.0784 |
| Pine | 32.44 | 33.22 | 31.44 | 33.33 | 1.25 | 0.6992 | 0.8741 | 0.7543 | |
| 48 | Herb | 41.22 | 38.33 | 39.78 | 33.78 | 1.77 | 0.0776 | 0.0314 | 0.3655 |
| Pine | 36.89 | 37.67 | 39.11 | 36.56 | 0.97 | 0.3167 | 0.8298 | 0.1564 | |
| 72 | Herb | 49.37 | 52.46 | 50.79 | 50.96 | 1.43 | 0.5362 | 0.5485 | 0.3099 |
| Pine | 48.67 | 48.56 | 55.22 | 51.89 | 1.63 | 0.0594 | 0.0568 | 0.6463 | |
| Incubation Time (h) | Treatment | Extract Concentration (1) (mg/L) | SEM (2) | p Value (3) | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 30 | 50 | 70 | T | L | Q | |||
| Total gas, mL/g DM | |||||||||
| 12 | Herb (4) | 190.27 b | 189.64 b | 204.58 a | 187.32 b | 2.01 | 0.0011 | 0.4233 | 0.0094 |
| Pine (5) | 167.94 ab | 163.24 bc | 175.01 a | 159.49 c | 2.37 | 0.0088 | 0.2539 | 0.0950 | |
| 24 | Herb | 212.13 | 207.75 | 214.03 | 205.00 | 4.75 | 0.5535 | 0.4962 | 0.6923 |
| Pine | 176.97 b | 177.92 b | 187.32 a | 176.97 b | 2.79 | 0.0811 | 0.4221 | 0.1367 | |
| Methane, mL/g DM | |||||||||
| 12 | Herb | 10.83 b | 9.40 b | 13.33 a | 9.49 b | 0.54 | 0.0029 | 0.9572 | 0.1585 |
| Pine | 11.95 a | 8.68 b | 11.13 a | 8.36 b | 0.51 | 0.0024 | 0.0048 | 0.5125 | |
| 24 | Herb | 19.33 | 16.84 | 18.39 | 17.24 | 1.10 | 0.4242 | 0.3173 | 0.5270 |
| Pine | 17.22 | 12.90 | 19.83 | 11.30 | 3.23 | 0.2955 | 0.4683 | 0.6308 | |
| Carbon dioxide, mL/g DM | |||||||||
| 12 | Herb | 61.30 b | 64.26 b | 88.00 a | 64.66 b | 3.20 | 0.0012 | 0.0348 | 0.0116 |
| Pine | 45.47 b | 41.36 bc | 52.44 a | 37.38 c | 2.06 | 0.0047 | 0.2098 | 0.0553 | |
| 24 | Herb | 89.69 | 93.75 | 99.07 | 94.21 | 4.37 | 0.5405 | 0.3307 | 0.4248 |
| Pine | 56.73 | 50.79 | 57.75 | 48.01 | 4.72 | 0.4465 | 0.3809 | 0.7546 | |
| Incubation Time (h) | Treatment | Extract Concentration (1) (mg/L) | SEM (2) | p Value (3) | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 30 | 50 | 70 | T | L | Q | |||
| Total VFA, mM | |||||||||
| 12 | Herb (4) | 53.85 c | 54.74 b | 59.77 a | 54.79 b | 0.27 | <0.0001 | 0.0001 | <0.0001 |
| Pine (5) | 53.98 | 60.36 | 59.37 | 55.70 | 1.95 | 0.1458 | 0.4766 | 0.0339 | |
| 24 | Herb | 62.40 | 63.15 | 65.17 | 62.46 | 1.50 | 0.5527 | 0.6974 | 0.3463 |
| Pine | 61.52 | 60.88 | 64.91 | 62.49 | 1.22 | 0.1815 | 0.2500 | 0.7364 | |
| Acetate, mM | |||||||||
| 12 | Herb | 36.58 b | 36.90 b | 40.80 a | 37.04 b | 0.28 | <0.0001 | 0.0019 | 0.0004 |
| Pine | 36.20 | 39.68 | 39.22 | 36.40 | 1.79 | 0.4255 | 0.8482 | 0.1172 | |
| 24 | Herb | 42.62 | 42.91 | 44.49 | 42.60 | 0.91 | 0.4475 | 0.6737 | 0.3417 |
| Pine | 40.90 | 40.99 | 42.46 | 41.48 | 0.93 | 0.6371 | 0.4530 | 0.7138 | |
| Propionate, mM | |||||||||
| 12 | Herb | 10.56 b | 10.77 b | 11.88 a | 10.61 b | 0.13 | 0.0003 | 0.0367 | 0.0015 |
| Pine | 10.93 b | 11.74 a | 11.92 a | 10.92 b | 0.17 | 0.0049 | 0.5144 | 0.0009 | |
| 24 | Herb | 12.59 | 12.64 | 13.28 | 12.47 | 0.36 | 0.4306 | 0.8262 | 0.3357 |
| Pine | 12.52 b | 12.04 b | 13.44 a | 12.17 b | 0.28 | 0.0269 | 0.7993 | 0.3740 | |
| Butyrate, mM | |||||||||
| 12 | Herb | 6.71 | 7.07 | 7.09 | 7.14 | 0.21 | 0.4762 | 0.1712 | 0.5345 |
| Pine | 6.84 | 8.94 | 8.23 | 8.38 | 0.48 | 0.0750 | 0.0744 | 0.0800 | |
| 24 | Herb | 7.20 b | 7.59 a | 7.41 ab | 7.40 ab | 0.24 | 0.7228 | 0.6249 | 0.4097 |
| Pine | 8.11 | 7.85 | 9.01 | 8.84 | 0.36 | 0.1424 | 0.0828 | 0.6160 | |
| Item | Extract Concentration (1) (mg/L) | SEM (2) | p Value (3) | |||||
|---|---|---|---|---|---|---|---|---|
| 0 | 30 | 50 | 70 | T | L | Q | ||
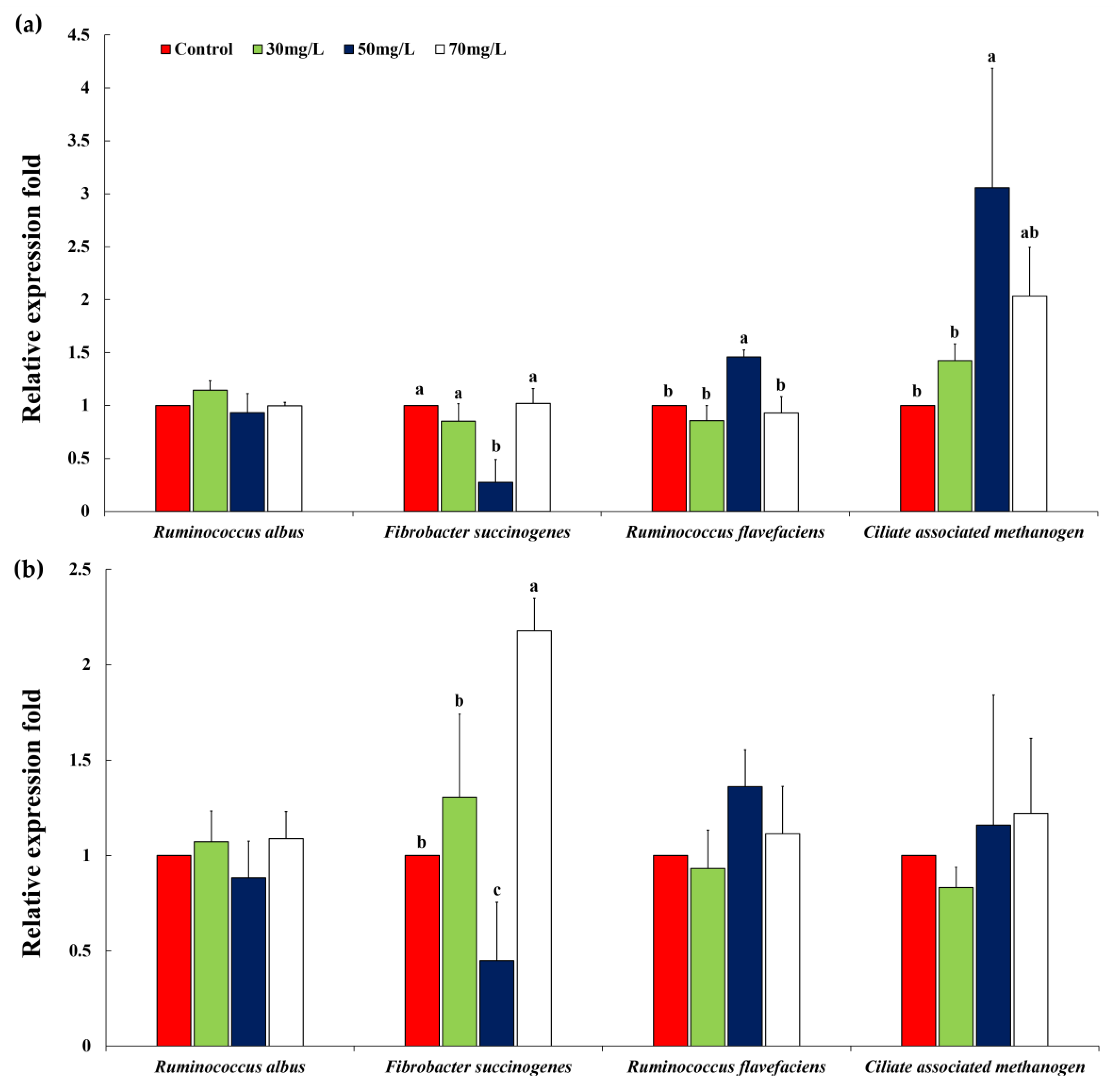
| Pinus densiflora (12 h) | ||||||||
| Ruminococcus albus | 1.00 | 1.15 | 0.93 | 1.00 | 0.07 | 0.1930 | 0.5809 | 0.3892 |
| Fibrobacter succinogenes | 1.00 a | 0.85 a | 0.27 b | 1.02 a | 0.09 | 0.0010 | 0.1474 | 0.0025 |
| Ruminococcus flavefaciens | 1.00 b | 0.86 b | 1.46 a | 0.93 b | 0.07 | 0.0032 | 0.2266 | 0.0945 |
| Ciliate associated methanogen | 1.00 b | 1.42 b | 3.06 a | 2.03 ab | 0.35 | 0.0165 | 0.0155 | 0.1927 |
| Pinus densiflora (24 h) | ||||||||
| Ruminococcus albus | 1.00 | 1.07 | 0.88 | 1.09 | 0.09 | 0.3989 | 0.8606 | 0.5979 |
| Fibrobacter succinogenes | 1.00 b | 1.31 b | 0.45 c | 2.18 a | 0.17 | 0.0020 | 0.0143 | 0.0085 |
| Ruminococcus flavefaciens | 1.00 | 0.93 | 1.36 | 1.11 | 0.11 | 0.1553 | 0.1643 | 0.7306 |
| Ciliate associated methanogen | 1.00 | 0.83 | 1.16 | 1.22 | 0.23 | 0.6440 | 0.4032 | 0.5144 |
| Mentha Canadensis (12 h) | ||||||||
| Ruminococcus albus | 1.00 | 1.17 | 1.22 | 1.03 | 0.13 | 0.6194 | 0.7263 | 0.2397 |
| Fibrobacter succinogenes | 1.00 a | 0.88 a | 0.17 b | 0.94 a | 0.06 | 0.0001 | 0.0061 | 0.0004 |
| Ruminococcus flavefaciens | 1.00 | 1.02 | 1.35 | 0.95 | 0.14 | 0.2357 | 0.7238 | 0.2457 |
| Ciliate associated methanogen | 1.00 b | 1.27 b | 3.39 a | 1.60 b | 0.26 | 0.0008 | 0.0083 | 0.0192 |
| Mentha Canadensis (24 h) | ||||||||
| Ruminococcus albus | 1.00 | 1.06 | 0.99 | 1.2 | 0.0766 | 0.2605 | 0.1699 | 0.3500 |
| Fibrobacter succinogenes | 1.00 a | 0.93 a | 0.15 c | 0.49 b | 0.0691 | 0.0003 | 0.0003 | 0.2000 |
| Ruminococcus flavefaciens | 1.00 | 1.16 | 2.39 | 1.34 | 0.202 | 0.0050 | 0.0313 | 0.0500 |
| Ciliate associated methanogen | 1.00 b | 0.82 b | 1.12 a | 0.92 b | 0.2288 | 0.8152 | 0.9909 | 0.9500 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.J.; Lee, Y.J.; Eom, J.S.; Kim, H.S.; Choi, Y.Y.; Jo, S.U.; Kang, S.N.; Park, H.Y.; Kim, D.H.; Lee, S.S. Effects of the Appropriate Addition of Antioxidants from Pinus densiflora and Mentha canadensis Extracts on Methane Emission and Rumen Fermentation. Animals 2020, 10, 1888. https://doi.org/10.3390/ani10101888
Lee SJ, Lee YJ, Eom JS, Kim HS, Choi YY, Jo SU, Kang SN, Park HY, Kim DH, Lee SS. Effects of the Appropriate Addition of Antioxidants from Pinus densiflora and Mentha canadensis Extracts on Methane Emission and Rumen Fermentation. Animals. 2020; 10(10):1888. https://doi.org/10.3390/ani10101888
Chicago/Turabian StyleLee, Shin Ja, Ye Jun Lee, Jun Sik Eom, Hyun Sang Kim, You Young Choi, Seong Uk Jo, Suk Nam Kang, Ha Young Park, Do Hyung Kim, and Sung Sill Lee. 2020. "Effects of the Appropriate Addition of Antioxidants from Pinus densiflora and Mentha canadensis Extracts on Methane Emission and Rumen Fermentation" Animals 10, no. 10: 1888. https://doi.org/10.3390/ani10101888
APA StyleLee, S. J., Lee, Y. J., Eom, J. S., Kim, H. S., Choi, Y. Y., Jo, S. U., Kang, S. N., Park, H. Y., Kim, D. H., & Lee, S. S. (2020). Effects of the Appropriate Addition of Antioxidants from Pinus densiflora and Mentha canadensis Extracts on Methane Emission and Rumen Fermentation. Animals, 10(10), 1888. https://doi.org/10.3390/ani10101888





