Fecal Cortisol Metabolites in Dairy Cows: A Cross-Sectional Exploration of Associations with Animal, Stockperson, and Farm Characteristics
Abstract
:Simple Summary
Abstract
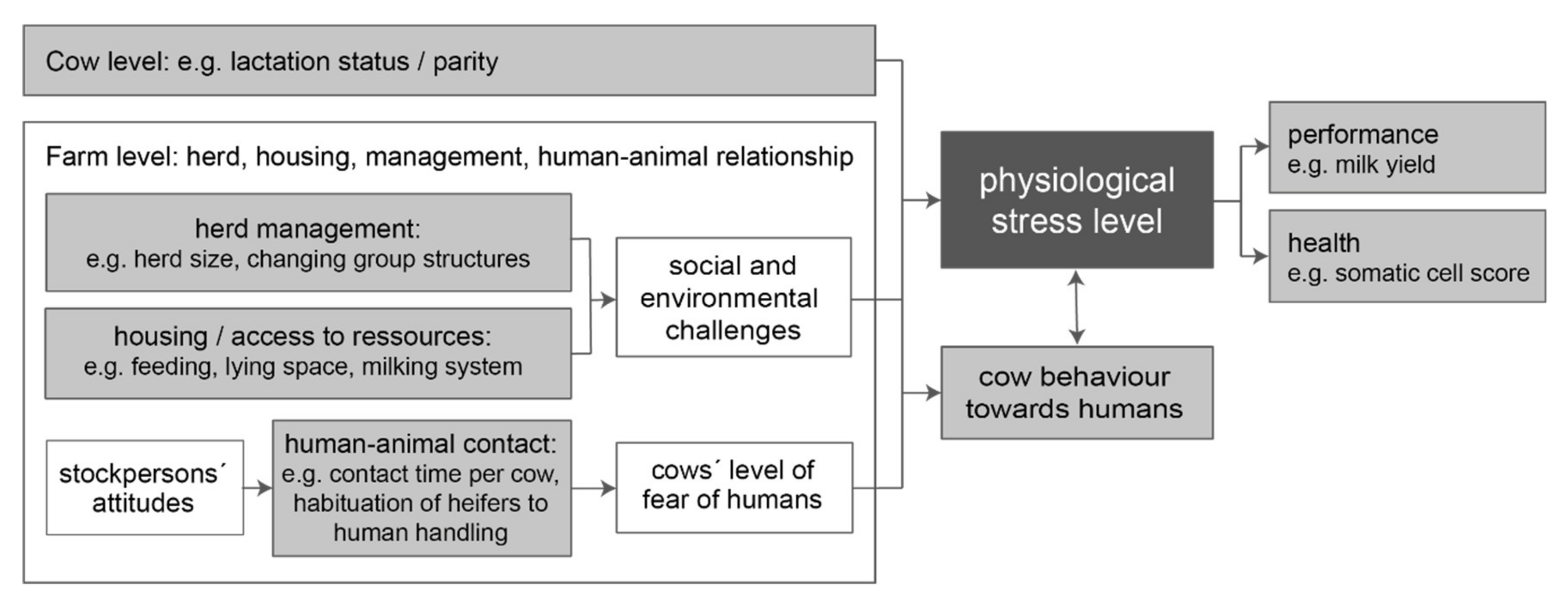
1. Introduction
2. Materials and Methods
2.1. Farms and Animals
2.2. Data Collection
2.2.1. Herd, Housing, Management, and Human–Animal Contact
2.2.2. Cows’ Physiological Stress Levels (FCMs)
2.2.3. Cows’ Behaviors towards Humans
2.2.4. Cows’ Milk Yield and Udder Health
2.3. Statistical Analyses
3. Results
3.1. Descriptive Data
3.2. Influences on FCM Levels
3.3. Associations of FCM Levels with Cows’ Behaviors towards Humans, Cows’ Milk Yield and Udder Health
4. Discussion
4.1. Level and Range of FCM Levels in the Investigated Sample
4.2. Identified Factors Influencing FCM Measures
4.3. Associations between FCMs and Cow Behavior
4.4. Associations between FCMs and Milk Yield and Udder Health
4.5. Limitations of the Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Variables | Max. No of Cows per Group | Concentrates (kg/Cow*Year) | No of Cows per Stockperson | Contact Time per Cow | Voluntary Contact to Cows (%) | Voluntary Contact to Dry Cows (%) | |
|---|---|---|---|---|---|---|---|
| Herd size | rs | 0.895 | 0.106 | 0.458 | −0.249 | −0.146 | −0.238 |
| p | 0.000 | 0.615 | 0.021 | 0.229 | 0.487 | 0.253 | |
| Max. no of cows per group | rs | 0.104 | 0.445 | −0.360 | −0.209 | −0.274 | |
| p | 0.620 | 0.026 | 0.077 | 0.316 | 0.185 | ||
| Concentrates (kg/cow*year) | rs | 0.315 | −0.008 | −0.047 | −0.014 | ||
| p | 0.125 | 0.968 | 0.824 | 0.946 | |||
| No of cows per stockperson | rs | −0.544 | −0.252 | −0.357 | |||
| p | 0.005 | 0.225 | 0.080 | ||||
| Contact time per cow | rs | 0.389 | 0.456 | ||||
| p | 0.055 | 0.022 | |||||
| Voluntary contact to cows (%) | rs | 0.884 | |||||
| p | 0.000 | ||||||
| References | Cubicles (Milking Parlor) (Cow:Cubicle Ratio) | Cubicles (AMS) (Cow:Cubicle Ratio) | Straw Yards (Hornless Cows) (m2/Cow) | Straw Yards (Horned Cows) (m2/Cow) |
|---|---|---|---|---|
| [59] | 1:1 | 4.5–5 m2 | 7–9 m2 | |
| [60] | 1:1 | 6 m2 | 8 m2 | |
| [61] | 1:1 | 1:0.981 | 8–9 m2 | |
| [62] | ≥5 m2 | 8 m2 | ||
| [63] | 8 m2 | |||
| [64] | 1:0.902 |
| Categories | Cubicles (Milking Parlor) (Cow:Cubicle Ratio) | Cubicles (AMS) (Cow:Cubicle Ratio) | Straw Yards (Hornless Cows) (m2/Cow) | Straw Yards (Horned Cows) (m2/Cow) |
|---|---|---|---|---|
| Suboptimal | <1:1 | <1:0.95 | <4.5 m2 | <7 m2 |
| According to recommendations | 1:1–1:1.05 | 1:0.95–1:1 | 4.5–6.5 m2 | 7–9 m2 |
| Generous | >1:1.05 | >1:1 | >6.5 m2 | >9 m2 |
| References | Feeding Gates (Milking Parlor) (Cow:Feeding Place Ratio) | Feeding Gates (AMS) (Cow:Feeding Place Ratio) | Feed Rails (Hornless) (Feeding Place Width) | Feed Rails (Horned) (Feeding Place Width) |
|---|---|---|---|---|
| [59] | 1:1 | 70–75 cm | ||
| [60,65] | 75 cm | 85–90 cm | ||
| [61] | 1:1 | 1.5:1 | 65–75 cm | |
| [62] | 75 cm | 85 cm | ||
| [63] | 1:1–1:1.1 | 85–100 cm | ||
| [64] | 1.5:1 | |||
| [66] | 1:1.1 | 85 cm |
| Categories | Feeding Gates (Milking Parlor) (Cow:Feeding Place Ratio) | Feeding Gates (AMS) (Cow:Feeding Place Ratio) | Feed Rails (Hornless, Milking Parlor) (Feeding Place Width) | Feed Rails (Hornless, AMS) (Feeding Place Width) | Feed Rails (Horned, Milking Parlor) (Feeding Place Width) | Feed Rails (Horned, AMS) (Feeding Place Width) |
|---|---|---|---|---|---|---|
| Suboptimal | <1:0.95 | <1:0.83 | <70 cm | <62 cm | <80 cm | <70 cm |
| According to recommendations | 1:0.95–1:1.05 | 1:0.83–1:1 | 70–75 cm | 62–75 cm | 80–90 cm | 70–90 cm |
| Generous | >1:1.05 | >1:1 | >75 cm | >75 cm | >90 cm | >90 cm |
References
- Waiblinger, S.; Menke, C.; Korff, J.; Bucher, A. Previous handling and gentle interactions affect behaviour and heart rate of dairy cows during a veterinary procedure. Appl. Anim. Behav. Sci. 2004, 85, 31–42. [Google Scholar] [CrossRef]
- Lindahl, C.; Pinzke, S.; Herlin, A.; Keeling, L.J. Human-animal interactions and safety during dairy cattle handling—Comparing moving cows to milking and hoof trimming. J. Dairy Sci. 2016, 99, 2131–2141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pesenhofer, G.; Palme, R.; Pesenhofer, R.M.; Kofler, J. Comparison of two methods of fixation during functional claw trimming-walk-in crush versus tilt table-in dairy cows using faecal cortisol metabolite concentrations and daily milk yield as parameters. Wien. Tierarztl. Monatsschr. 2006, 93, 288–294. [Google Scholar]
- Möstl, E.; Palme, R. Hormones as indicators of stress. Domest. Anim. Endocrinol. 2002, 23, 67–74. [Google Scholar] [CrossRef]
- Holtenius, K.; Waller, K.P.; Essen-Gustavsson, B.; Holtenius, P.; Sandgren, C.H. Metabolic parameters and blood leukocyte profiles in cows from herds with high or low mastitis incidence. Vet. J. 2004, 168, 65–73. [Google Scholar] [CrossRef]
- Ivemeyer, S.; Simantke, C.; Ebinghaus, A.; Poulsen, P.H.; Sorensen, J.T.; Rousing, T.; Palme, R.; Knierim, U. Herd-level associations between human–animal relationship, management, fecal cortisol metabolites, and udder health of organic dairy cows. J. Dairy Sci. 2018, 101. [Google Scholar] [CrossRef] [Green Version]
- Palme, R. Non-invasive measurement of glucocorticoids: Advances and problems. Physiol. Behav. 2019, 199, 229–243. [Google Scholar] [CrossRef]
- Palme, R. Monitoring stress hormone metabolites as a useful, non-invasive tool for welfare assessment in farm animals. Anim. Welf. 2012, 21, 331–337. [Google Scholar] [CrossRef]
- Mormède, P.; Andanson, S.; Aupérin, B.; Beerda, B.; Guémené, D.; Malmkvist, J.; Manteca, X.; Manteuffel, G.; Prunet, P.; van Reenen, C.G.; et al. Exploration of the hypothalamic–pituitary–adrenal function as a tool to evaluate animal welfare. Physiol. Behav. 2007, 92, 317–339. [Google Scholar] [CrossRef]
- Palme, R.; Möstl, E. Measurement of cortisol metabolites in faeces of sheep as a parameter of cortisol concentration in blood. Int. J. Mammal. Biol. 1997, 62, 192–197. [Google Scholar]
- Palme, R.; Robia, C.; Baumgartner, W.; Mostl, E. Transport stress in caftle as reflected by an increase in faecal cortisol metabolite concentrations. Vet. Rec. 2000, 146, 108–109. [Google Scholar] [CrossRef] [PubMed]
- Möstl, E.; Maggs, J.L.; Schrötter, G.; Besenfelder, U.; Palme, R. Measurement of cortisol metabolites in faeces of ruminants. Vet. Res. Commun. 2002, 26, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Huzzey, J.M.; Nydam, D.V.; Grant, R.J.; Overton, T.R. The effects of overstocking Holstein dairy cattle during the dry period on cortisol secretion and energy metabolism. J. Dairy Sci. 2012, 95, 4421–4433. [Google Scholar] [CrossRef]
- Fisher, A.D.; Stewart, M.; Verkerk, G.A.; Morrow, C.J.; Matthews, L.R. The effects of surface type on lying behaviour and stress responses of dairy cows during periodic weather-induced removal from pasture. Appl. Anim. Behav. Sci. 2003, 81, 1–11. [Google Scholar] [CrossRef]
- Rouha-Mülleder, C.; Palme, R.; Waiblinger, S. Erhebungen zur Tiergerechtheit in 80 Milchkuhbetrieben mit Boxenlaufställen—Tiergesundheit und andere tierbezogene Indikatoren. Wien. Tierarztl. Monatsschr. 2010, 97, 231–241. [Google Scholar]
- Waiblinger, S.; Menke, C.; Fölsch, D.W. Influences on the avoidance and approach behaviour of dairy cows towards humans on 35 farms. Appl. Anim. Behav. Sci. 2003, 84, 23–39. [Google Scholar] [CrossRef]
- Ebinghaus, A.; Ivemeyer, S.; Knierim, U. Human and farm influences on dairy cows’ responsiveness towards humans—A cross-sectional study. PLoS ONE 2018, 13, e0209817. [Google Scholar] [CrossRef]
- Breuer, K.; Hemsworth, P.H.; Barnett, J.L.; Matthews, L.R.; Coleman, G.J. Behavioural response to humans and the productivity of commercial dairy cows. Appl. Anim. Behav. Sci. 2000, 66, 273–288. [Google Scholar] [CrossRef]
- Rushen, J.; de Passillé, A.M.B.; Munksgaard, L. Fear of people by cows and effects on milk yield, behavior, and heart rate at milking. J. Dairy Sci. 1999, 82, 720–727. [Google Scholar] [CrossRef]
- Waiblinger, S.; Menke, C.; Coleman, G. The relationship between attitudes, personal characteristics and behaviour of stockpeople and subsequent behaviour and production of dairy cows. Appl. Anim. Behav. Sci. 2002, 79, 195–219. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Coleman, G.J.; Barnett, J.L.; Borg, S. Relationships between human-animal interactions and productivity of commercial dairy cows. J. Anim. Sci. 2000, 78, 2821–2831. [Google Scholar] [CrossRef] [PubMed]
- Ivemeyer, S.; Knierim, U.; Waiblinger, S. Effect of human-animal relationship and management on udder health in Swiss dairy herds. J. Dairy Sci. 2011, 94, 5890–5902. [Google Scholar] [CrossRef] [PubMed]
- Hemsworth, P.H.; Barnett, J.L.; Tilbrook, A.J.; Hansen, C. The effects of handling by humans at calving and during milking on the behaviour and milk cortisol concentrations of primiparous dairy cows. Appl. Anim. Behav. Sci. 1989, 22, 313–326. [Google Scholar] [CrossRef]
- Breuer, K.; Hemsworth, P.H.; Coleman, G.J. The effect of positive or negative handling on the behavioural and physiological responses of nonlactating heifers. Appl. Anim. Behav. Sci. 2003, 84, 3–22. [Google Scholar] [CrossRef]
- Lürzel, S.; Windschnurer, I.; Futschik, A.; Palme, R.; Waiblinger, S. Effects of gentle interactions on the relationship with humans and on stress-related parameters in group-housed calves. Anim. Welf. 2015, 24, 475–484. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Barnett, J.L. The effects of aversively handling pigs, either individually or in groups, on their behaviour, growth and corticosteroids. Appl. Anim. Behav. Sci. 1991, 30, 61–72. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Barnett, J.L.; Hansen, C. The influence of handling by humans on the behavior, growth, and corticosteroids in the juvenile female pig. Horm. Behav. 1981. [Google Scholar] [CrossRef]
- Palme, R.; Robia, C.H.; Messmann, S.; Hofer, J.; Mostl, E. Measurement of faecal cortisol metabolites in ruminants: A non-invasive parameter of adrenocortical function. Wien. Tierarztl. Monatsschr. 1999, 86, 237–241. [Google Scholar]
- Ebinghaus, A.; Ivemeyer, S.; Rupp, J.; Knierim, U. Identification and development of measures suitable as potential breeding traits regarding dairy cows´ reactivity towards humans. Appl. Anim. Behav. Sci. 2016, 185, 30–38. [Google Scholar] [CrossRef]
- Wemelsfelder, F.; Millard, F.; De Rosa, G.; Napolitano, F. Qualitative behaviour assessment. In Assessment of Animal Welfare Measures for Dairy Cattle, Beef Bulls and Veal Calves; Forkman, B., Keeling, L., Eds.; Cardiff University: Cardiff, UK, 2009; pp. 215–224. [Google Scholar]
- Rosenthal, R.; Cooper, H.; Hedges, L. Parametric measures of effect size. Handb. Res. Synth. 1994, 621, 231–244. [Google Scholar]
- Crawley, M.J. Statistik mit R.; John Wiley & Sons: West Sussex, UK, 2012; ISBN 3527506993. [Google Scholar]
- Palme, R. Measuring fecal steroids: Guidelines for practical application. Ann. N. Y. Acad. Sci. 2005, 1046, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Bertulat, S.; Fischer-Tenhagen, C.; Suthar, V.; Möstl, E.; Isaka, N.; Heuwieser, W. Measurement of fecal glucocorticoid metabolites and evaluation of udder characteristics to estimate stress after sudden dry-off in dairy cows with different milk yields. J. Dairy Sci. 2013, 96, 3774–3787. [Google Scholar] [CrossRef] [PubMed]
- Weiss, D.; Helmreich, S.; Möstl, E.; Dzidic, A.; Bruckmaier, R.M. Coping capacity of dairy cows during the change from conventional to automatic milking1. J. Anim. Sci. 2004, 82, 563–570. [Google Scholar] [CrossRef]
- Fukasawa, M.; Tsukada, H.; Kosako, T.; Yamada, A. Effect of lactation stage, season and parity on milk cortisol concentration in Holstein cows. Livest. Sci. 2008, 113, 280–284. [Google Scholar] [CrossRef]
- des Roches, A.d.B.; Veissier, I.; Boivin, X.; Gilot-Fromont, E.; Mounier, L. A prospective exploration of farm, farmer, and animal characteristics in human-animal relationships: An epidemiological survey. J. Dairy Sci. 2016, 99, 5573–5585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ivemeyer, S. Einfluss der Mensch–Tier–Beziehung auf die Eutergesundheit von Milchkühen. Ph.D. Thesis, University of Kassel, Witzenhausen, Germany, 2010. [Google Scholar]
- Schmitz, L.; Ebinghaus, A.; Ivemeyer, S.; Domas, L.; Knierim, U. Validity aspects of behavioural measures to assess cows’ responsiveness towards humans. Appl. Anim. Behav. Sci. 2020. [Google Scholar] [CrossRef]
- Krumrych, W.; Gołda, R.; Gołyński, M.; Markiewicz, H.; Buzała, M. Effect of physical exercise on cortisol concentration and neutrophil oxygen metabolism in peripheral blood of horses. Ann. Anim. Sci. 2018, 18, 53–68. [Google Scholar] [CrossRef] [Green Version]
- von Keyserlingk, M.A.G.; Olenick, D.; Weary, D.M. Acute behavioral effects of regrouping dairy cows. J. Dairy Sci. 2008, 91, 1011–1016. [Google Scholar] [CrossRef]
- Palme, R.; Wetscher, F.; Winckler, C. Measuring faecal cortisol metabolites: A noninvasive tool to assess animal welfare in cattle. In Proceedings of the 4th Central European Buiatric Congress, Lovran, Croatia, 23–27 April 2003; pp. 145–150. [Google Scholar]
- Pawluski, J.; Jego, P.; Henry, S.; Bruchet, A.; Palme, R.; Coste, C.; Hausberger, M. Low plasma cortisol and fecal cortisol metabolite measures as indicators of compromised welfare in domestic horses (Equus caballus). PLoS ONE 2017, 12, e0182257. [Google Scholar] [CrossRef] [Green Version]
- Busch, D.S.; Hayward, L.S. Stress in a conservation context: A discussion of glucocorticoid actions and how levels change with conservation-relevant variables. Biol. Conserv. 2009, 142, 2844–2853. [Google Scholar] [CrossRef]
- Hill, W.G.; Edwards, M.R.; Ahmed, M.-K.; Thompson, R. Heritability of milk yield and composition at different levels and variability of production. Anim. Sci. 1983, 36, 59–68. [Google Scholar] [CrossRef]
- Logfren, D.L.; Vinson, W.E.; Pearson, R.E.; Powell, R.L. Heritability of milk yield at different herds means and variances for production. J. Dairy Sci. 1985, 68, 2737–2739. [Google Scholar] [CrossRef]
- Dillon, P.; Crosse, S.; O’Brien, B. Effect of concentrate supplementation of grazing dairy cows in early lactation on milk production and milk processing quality. Irish J. Agric. Food Res. 1997, 145–159. [Google Scholar]
- Macdonald, K.A.; Penno, J.W.; Bryant, A.M.; Roche, J.R. Effect of feeding level pre- and post-puberty and body weight at first calving on growth, milk production, and fertility in grazing dairy cows. J. Dairy Sci. 2005. [Google Scholar] [CrossRef] [Green Version]
- Kennelly, J.J.; Robinson, B.; Khorasani, G.R. Influence of carbohydrate source and buffer on rumen fermentation characteristics, milk yield, and milk composition in early-lactation Holstein cows. J. Dairy Sci. 1999. [Google Scholar] [CrossRef]
- Moorby, J.M.; Lee, M.R.F.; Davies, D.R.; Kim, E.J.; Nute, G.R.; Ellis, N.M.; Scollan, N.D. Assessment of dietary ratios of red clover and grass silages on milk production and milk quality in dairy cows. J. Dairy Sci. 2009. [Google Scholar] [CrossRef]
- Hansen, J.V.; Friggens, N.C.; Højsgaard, S. The influence of breed and parity on milk yield, and milk yield acceleration curves. Livest. Sci. 2006, 104, 53–62. [Google Scholar] [CrossRef]
- Hernandez, J.; Shearer, J.K.; Webb, D.W. Effect of lameness on milk yield in dairy cows. J. Am. Vet. Med. Assoc. 2002, 220, 640–644. [Google Scholar] [CrossRef] [Green Version]
- Rajala-Schultz, P.J.; Gröhn, Y.T.; McCulloch, C.E. Effects of milk fever, ketosis, and lameness on milk yield in dairy cows. J. Dairy Sci. 1999, 82, 288–294. [Google Scholar] [CrossRef]
- Moberg, G.P. Biological response to stress: Implications for animal welfare. In The Biology of Animal Stress; Moberg, G.P., Mench, J.A., Eds.; CAB International: Wallingford, UK, 2005. [Google Scholar]
- Borg, K.E.; Esbenshade, K.L.; Johnson, B.H. Cortisol, growth hormone, and testosterone concentrations during mating behavior in the bull and boar. J. Anim. Sci. 1991. [Google Scholar] [CrossRef] [Green Version]
- Borg, K.E.; Esbenshade, K.L.; Johnson, B.H. Effects of the peri-pubertal rearing environment on endocrine and behavioural responses to oestrous female exposure in the mature bull. Appl. Anim. Behav. Sci. 1993. [Google Scholar] [CrossRef]
- Colborn, D.R.; Thompson, D.L.; Roth, T.L.; Capehart, J.S.; White, K.L. Responses of cortisol and prolactin to sexual excitement and stress in stallions and geldings. J. Anim. Sci. 1991, 69, 2556–2562. [Google Scholar] [CrossRef] [PubMed]
- De Jong, I.C.; Prelle, I.T.; Van De Burgwal, J.A.; Lambooij, E.; Korte, S.M.; Blokhuis, H.J.; Koolhaas, J.M. Effects of environmental enrichment on behavioral responses to novelty, learning, and memory, and the circadian rhythm in cortisol in growing pigs. Physiol. Behav. 2000. [Google Scholar] [CrossRef]
- Laves Tierschutzdienst. Tierschutzleitlinie für die Milchkuhhaltung; Niedersächsisches Ministerium für den ländlichen Raum, Ernährung, Landwirtschaft und Verbraucherschutz: Hannover, Germany, 2007. [Google Scholar]
- Eilers, U. Planungshilfen für den Rinder-Stallbau; Landwirtschaftliches Zentrum für Rinderhaltung, Grünlandwirtschaft, Milchwirtschaft, Wild und Fischerei Baden-Württemberg: Aulendorf, Germany, 2018. [Google Scholar]
- Pelzer, A. NRW-Bauschrift Milchviehhaltung: Beratungsempfehlungen für den Bau und die Ausstattung von Milchviehställen; Landwirtschaftskammer Nordrhein-Westfalen: Münster, Germany, 2012. [Google Scholar]
- Bachinger, J.; Becherer, U.; Bee, W.; Belau, T.; Blum, H.; Blumschein, A.; Brinkmann, J.; Deerberg, F.; Dreyer, W.; Euen, S.; et al. Faustzahlen für den Ökologischen Landbau; Kuratorium für Technik und Bauwesen in der Landwirtschaft e.V. (KTBL): Darmstadt, Germany, 2015. [Google Scholar]
- Schneider, C. Laufställe für Horntragende Milchkühe, 2nd ed.; Forschungsinstitut für biologischen Landbau (FiBL): Frick, Switzerland, 2011. [Google Scholar]
- Alberti, J.H. 100 Antworten zu Automatischen Melksystemen: Praxisempfehlungen zu Anforderungen an Mensch und Tier-Management-Haltung-Fütterung-Gesundheit; DLG-Verlag: Frankfurt, Germany, 2010. [Google Scholar]
- Eilers, U.; Euler, F.; Wagner, H.U.R. Laufstallhaltung von Rindern im Ökologischen Landbau—Merkblatt für die Umweltgerechte Landbewirtschaftung Nr. 32; Landwirtschaftliches Zentrum für Rinderhaltung, Grünlandwirtschaft, Milchwirtschaft, Wild und Fischerei Baden-Württemberg: Karlsruhe, Germany, 2012. [Google Scholar]
- Klöble, U.; Meyer, B. Investitionsbedarf von Milchviehställen für horntragende Kühe; Kuratorium für Technik und Bauwesen in der Landwirtschaft e.V. (KTBL): Darmstadt, Germany, 2014. [Google Scholar]
| Variables | Recording 1 | Descriptive Data | |
|---|---|---|---|
| Herd Characteristics | Mean ± sd | Median (Min–Max) | |
| Parity (n) | MRD | 3.2 ± 2.1 | 3.0 (1.0–12.0) |
| DIM (d) | MRD | 92.3 ± 51.5 | 89.5 (1.0–200.0) |
| Herd size (n) | MRD | 69.4 ± 30.8 | 68 (29–161) |
| Max. number of cows per group (n) | o | 56.2 ± 20.8 | 57 (29–95) |
| Groups | Number (%) | ||
| Cow groups (n) | o | 1 (all cows in one group) | 5 (20) |
| 2 (dry cows separated) | 13 (52) | ||
| >2 groups | 7 (28) | ||
| Housing Characteristics | Groups | Number (%) | |
| Housing type | o | Raised cubicles | 8 (32) |
| Deep bedded cubicles | 8 (32) | ||
| Straw yards or mixed 2 | 9 (36) | ||
| Cow:cubicle ratio or lying space 3 (m2/cow) | o | Suboptimal | 10 (40) |
| According to recommendations | 11 (44) | ||
| Generous | 4 (16) | ||
| Cow:feeding place ratio 4 | o | Suboptimal | 7 (28) |
| According to recommendations | 9 (36) | ||
| Generous | 9 (36) | ||
| Management Characteristics | Mean ± sd | Median (min–max) | |
| Concentrates (kg/cow*year) | i | 1203 ± 521 | 1200 (0000–2000) |
| Groups | Number (%) | ||
| Routine fixation for feeding | i | No | 11 (44) |
| Yes | 14 (56) | ||
| Concentrates station (in the barn) | o | No | 13 (52) |
| Yes | 12 (48) | ||
| Separation of dry cows | i | No | 6 (24) |
| Yes | 19 (76) | ||
| Separation of cows in heat | i | No (or rarely) | 16 (64) |
| Yes | 9 (36) | ||
| Separation of diseased or lame cows | i | No | 7 (28) |
| Sometimes | 11 (44) | ||
| Yes | 7 (28) | ||
| Milking system | o | AMS | 5 (20) |
| Fishbone parlor | 16 (64) | ||
| Tandem parlor | 4 (16) | ||
| Selection of cows for docility | i | No | 18 (72) |
| Yes | 7 (28) | ||
| Human–Animal Contact | Mean ± sd | Median (min–max) | |
| Number of cows per stockperson 5 (n) | i | 19.4 ± 10.0 | 18 (4.4–40.5) |
| Contact time per cow ‘on foot’ 6 (min/d) | i | 9.0 ± 7.6 | 6.0 (1.7–32.6) |
| Voluntary contact to cows (%) 7 | i | 48.3 ± 13.1 | 50.0 (12.5–68.8) |
| Voluntary contact to dry cows (%) 7 | i | 50.0 ± 18.7 | 50.0 (18.75–81.3) |
| Groups | Number (%) | ||
| Active habituation of heifers to human handling | i | No | 15 (60) |
| Yes | 10 (40) | ||
| Active habituation of heifers to the milking parlor / AMS | i | No | 14 (56) |
| Yes | 11 (44) | ||
| Frequency of control rounds in the barn | i | At maximum once a day | 7 (28) |
| Several times a day | 18 (72) | ||
| Roughage provision | i | Only by machine | 9 (36) |
| (Also) manually | 16 (64) | ||
| Concentrates provision (in the barn) | i | Only by machine/station | 15 (60) |
| (Also) manually | 10 (40) | ||
| Stockpersons know individual cows | o | Not all cows | 15 (60) |
| Yes | 10 (40) | ||
| FCManimal | n | Mean ± sd | Median | Min–Max | 25–75% Percentile |
|---|---|---|---|---|---|
| a. | 671 | 13.95 ± 13.45 | 10.36 | 2.20–159.50 | 4.95–19.69 |
| b. | 674 | 12.20 ± 6.90 | 11.03 | 2.20–47.60 | 6.86–15.82 |
| FCMherd | n | Mean ± sd | Median | Min–Max | 25–75% Percentile |
| a. | 25 | 11.42 ± 6.13 | 12.53 | 2.20–26.21 | 6.35–15.33 |
| b. | 25 | 11.15 ± 3.90 | 10.79 | 4.42–17.53 | 7.91–15.45 |
| Fixed Effects | Estimate | CI 2.5% | CI 97.5% | p-Value | Effect Size |
|---|---|---|---|---|---|
| Day time of sampling (ref: 05:00–08:59 am) | |||||
| 09:00–12:59 am | 0.030 | −0.005 | 0.065 | 0.087 | 0.03 |
| 01:00–04:59 pm | 0.074 | 0.038 | 0.110 | <0.001 | 0.08 |
| 05:00–09:00 pm | 0.086 | 0.043 | 0.130 | <0.001 | 0.09 |
| Days in milk (DIM) | <0.001 | <0.001 | 0.001 | <0.001 | 0.14 |
| Separation of diseased/lame cows (ref: never) | |||||
| Sometimes | 0.103 | 0.052 | 0.152 | <0.001 | 0.65 |
| Always | 0.176 | 0.117 | 0.234 | <0.001 | 0.76 |
| Housing type (ref: raised cubicles) | |||||
| Deep bedded cubicles | −0.049 | −0.106 | 0.009 | 0.096 | 0.30 |
| Straw yard 1 | −0.084 | −0.136 | −0.031 | 0.003 | 0.53 |
| Active habituation of heifers to milking/AMS | 0.085 | 0.040 | 0.129 | 0.001 | 0.61 |
| Cow:cubicle ratio or lying area 2 (m2/cow) (ref. suboptimal) | |||||
| According to recommendations | 0.060 | 0.009 | 0.112 | 0.025 | 0.43 |
| Generous | −0.132 | −0.209 | −0.055 | 0.002 | 0.58 |
| (Also) manual concentrates provision | 0.054 | 0.003 | 0.106 | 0.042 | 0.39 |
| Contact time per cow 3 (min/d) | −0.006 | −0.010 | −0.002 | 0.004 | 0.54 |
| VIF = 1.018–2.453 | |||||
| Variables | Descriptive Data | Associations with the Cows’ FCM Levels | ||||
|---|---|---|---|---|---|---|
| Cow Behavior 1 | n | Mean ± sd | Median | Min–Max | rs | p |
| AD (cm) | 569 | 26.8 ± 35.1 | 10.0 | 0.0–170.0 | −0.069 | 0.102 |
| QBAanimal (PC1) 2 | 315 | −0.090 ± 1.025 | −0.178 | –1.713–2.355 | −0.149 | 0.008 |
| Milk Recording Data 3 | n | Mean ± sd | Median | Min–Max | rs | p |
| ECM (kg) | 674 | 25.22 ± 6.70 | 25.11 | 7.93–43.60 | −0.034 | 0.385 |
| SCS | 674 | 3.08 ± 1.70 | 2.90 | −0.64–9.25 | 0.109 | 0.005 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ebinghaus, A.; Knierim, U.; Simantke, C.; Palme, R.; Ivemeyer, S. Fecal Cortisol Metabolites in Dairy Cows: A Cross-Sectional Exploration of Associations with Animal, Stockperson, and Farm Characteristics. Animals 2020, 10, 1787. https://doi.org/10.3390/ani10101787
Ebinghaus A, Knierim U, Simantke C, Palme R, Ivemeyer S. Fecal Cortisol Metabolites in Dairy Cows: A Cross-Sectional Exploration of Associations with Animal, Stockperson, and Farm Characteristics. Animals. 2020; 10(10):1787. https://doi.org/10.3390/ani10101787
Chicago/Turabian StyleEbinghaus, Asja, Ute Knierim, Christel Simantke, Rupert Palme, and Silvia Ivemeyer. 2020. "Fecal Cortisol Metabolites in Dairy Cows: A Cross-Sectional Exploration of Associations with Animal, Stockperson, and Farm Characteristics" Animals 10, no. 10: 1787. https://doi.org/10.3390/ani10101787
APA StyleEbinghaus, A., Knierim, U., Simantke, C., Palme, R., & Ivemeyer, S. (2020). Fecal Cortisol Metabolites in Dairy Cows: A Cross-Sectional Exploration of Associations with Animal, Stockperson, and Farm Characteristics. Animals, 10(10), 1787. https://doi.org/10.3390/ani10101787






