The Influence of Essential Oils on Gut Microbial Profiles in Pigs
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Preparation of Oregano Herb Extract
2.3. Formulation of “Liquid/Solid” Phase with Essential Oils
2.4. EEOs Combination
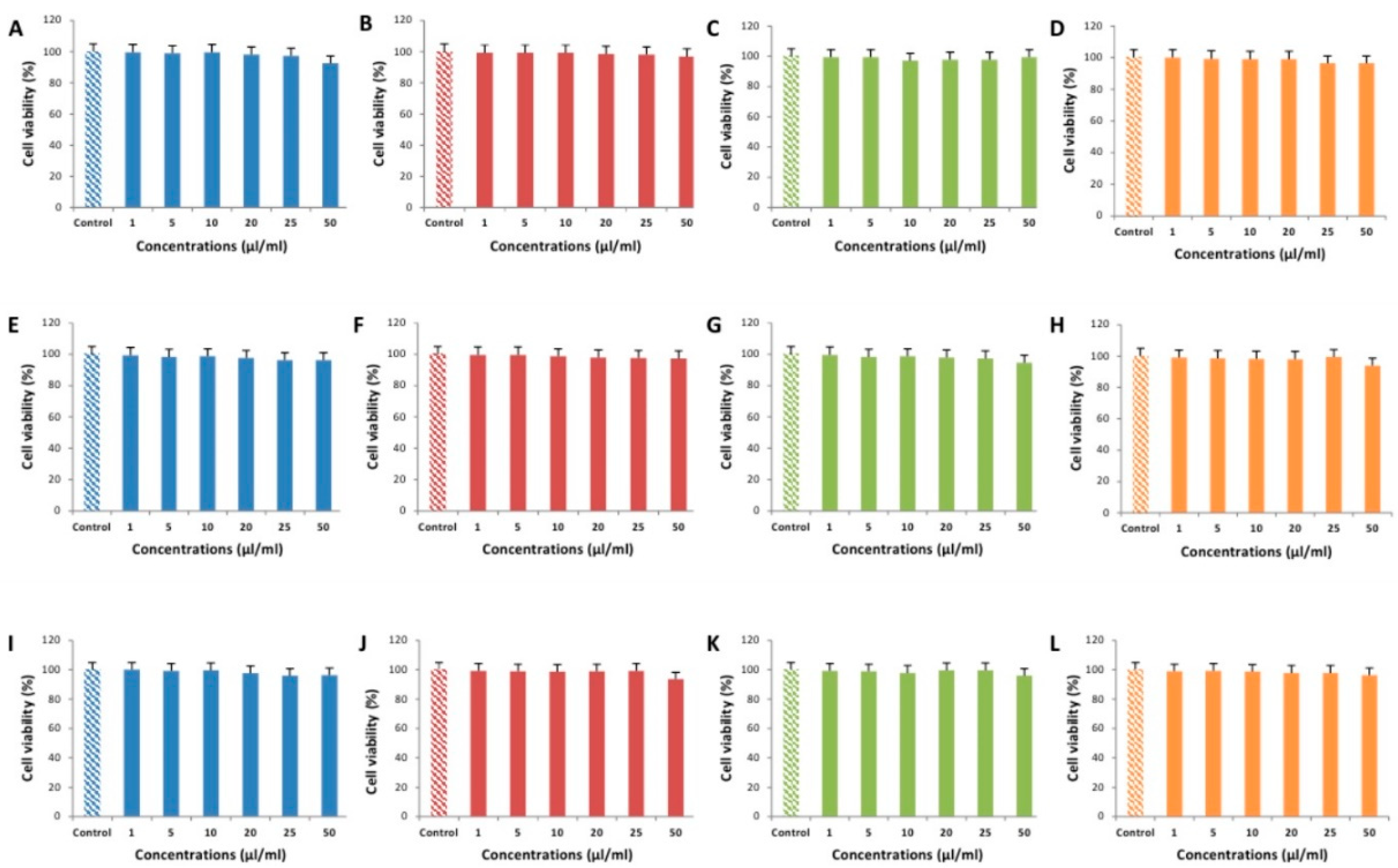
2.5. Cytotoxicity Testing of EEOs
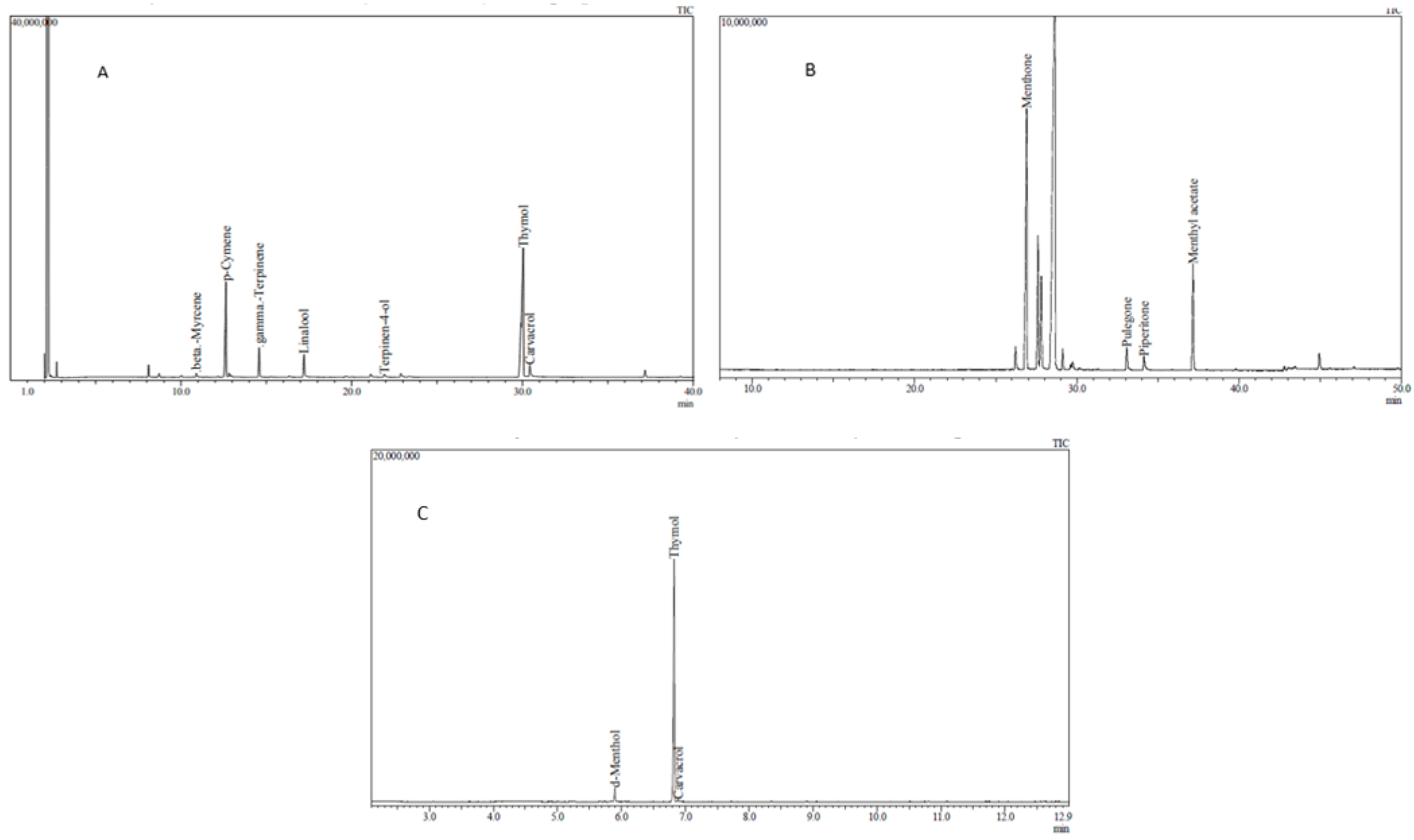
2.6. Chemical Analysis of Essential Oils and Tablets
2.7. In Vivo Experiment Design
2.8. Metagenomics and Microbial Profiling
2.9. Statistical Analysis
3. Results
3.1. Chemical Analysis of EOs
3.2. Cytotoxic Effects of EEOs
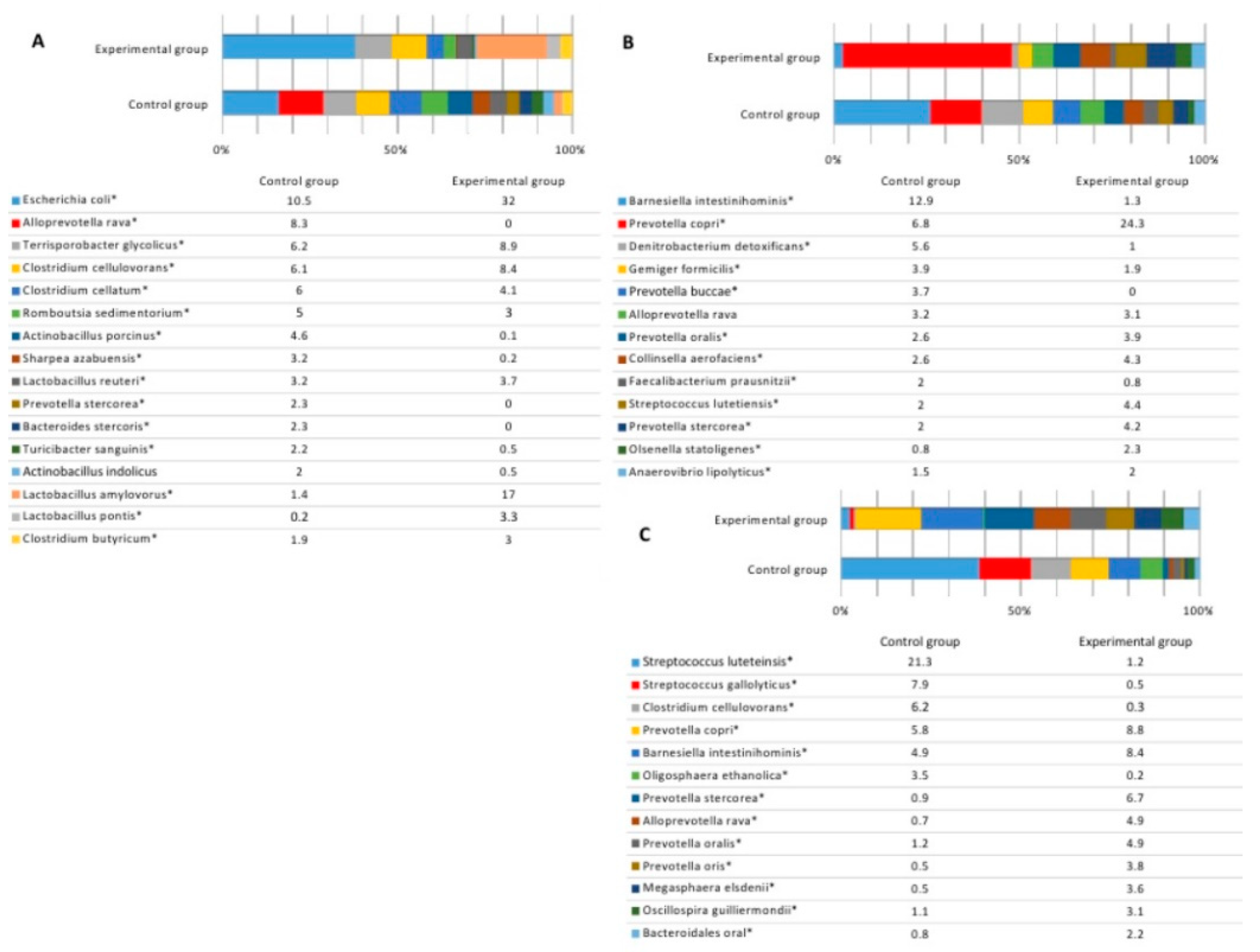
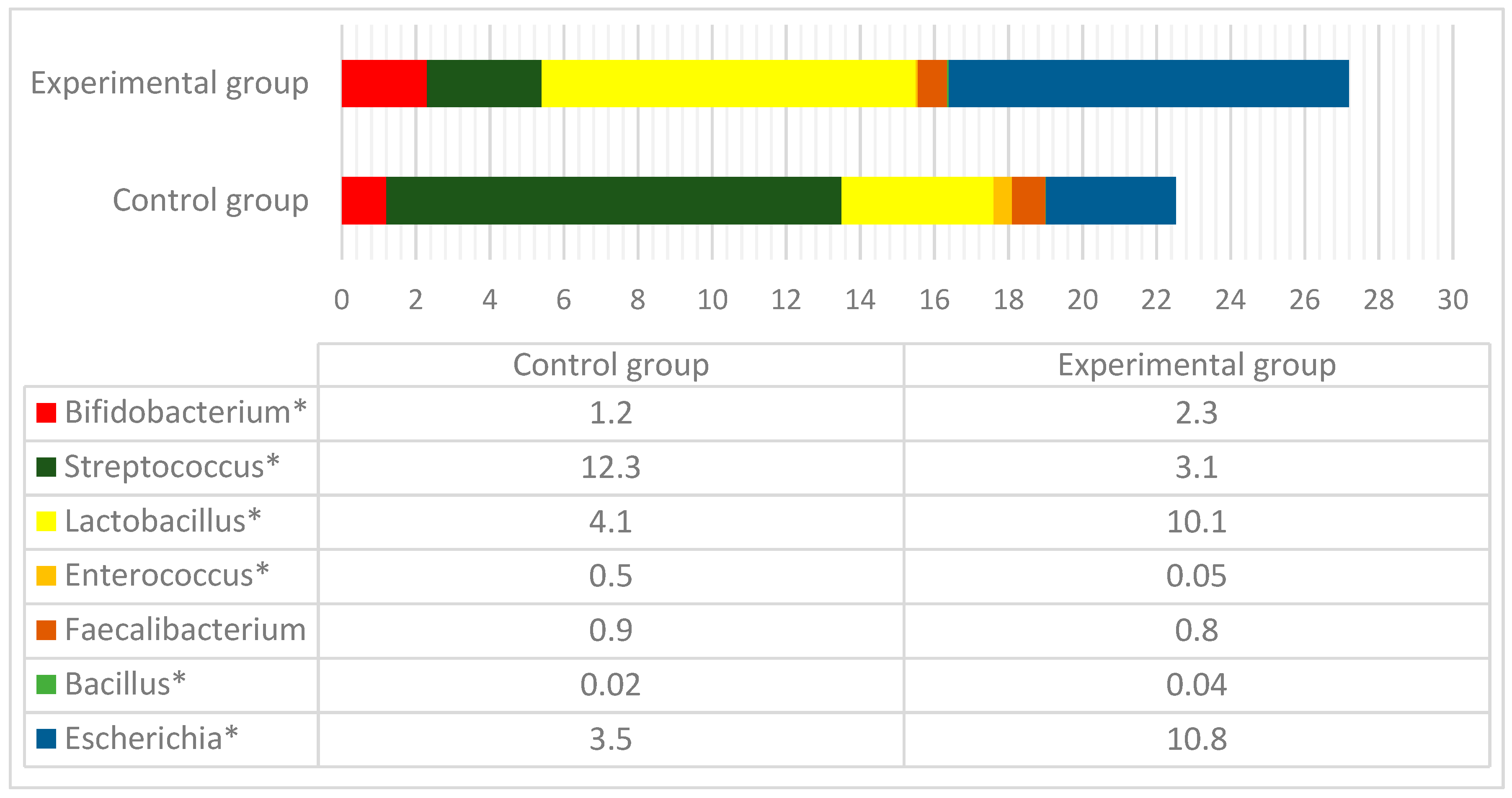
3.3. Microbial Profiles in the Gut of Experimental Animals
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Maggini, S.; Pierre, A.; Calder, P.C. Immune function and micronutrient requirements change over the life course. Nutrients 2018, 10, 1531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Childs, C.E.; Calder, P.C.; Miles, E.A. Diet and immune function. Nutrients 2019, 11, 1933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szliszka, E.; Krol, W. The role of dietary polyphenols in tumor necrosis factor-related apoptosis inducing ligand (trail)-induced apoptosis for cancer chemoprevention. Eur. J. Cancer Prev. 2011, 20, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Lewis, E.D.; Pae, M.; Meudani, S.N. Nutritional modulation of immune function: Analysis of evidence, mechanisms, and clinical relevance. Front Immunol. 2018, 9, 3160. [Google Scholar] [CrossRef]
- Bartkiene, E.; Ruzauskas, M.; Lele, V.; Zavistanaviciute, P.; Bernatoniene, J.; Jakstas, V.; Ivanauskas, L.; Zadeike, D.; Klupsaite, D.; Viskelis, P.; et al. Development of antimicrobial gummy candies with addition of bovine colostrum, essential oils and probiotics. Int. J. Food Sci. Technol. 2018, 53, 1227–1235. [Google Scholar] [CrossRef]
- Bartkiene, E.; Lele, V.; Sakiene, V.; Zavistanaviciute, P.; Ruzauskas, M.; Stankevicius, A.; Grigas, J.; Pautienius, A.; Bernatonienė, J.; Jakštas, V.; et al. Fermented, ultrasonicated, and dehydrated bovine colostrum: Changes in antimicrobial properties and immunoglobulin content. J. Dairy Sci. 2020, 103, 1315–1323. [Google Scholar] [CrossRef] [Green Version]
- Bartkiene, E.; Lele, V.; Starkute, V.; Zavistanaviciute, P.; Zokaityte, E.; Varinauskaite, I.; Pileckaite, G.; Rutkauskaite, G.; Kanaporis, T.; Dmitrijeva, L.; et al. Plants and lactic acid bacteria combination for new antimicrobial and antioxidant properties product development in a sustainable manner. Foods 2020, 9, 433. [Google Scholar] [CrossRef] [Green Version]
- Matulyte, I.; Jekabsone, A.; Jankauskaitė, L.; Zavistanaviciute, P.; Sakiene, V.; Bartkienė, E.; Ružauskas, M.; Kopustinskiene, D.M.; Santini, A.; Bernatoniene, J. The essential oil and hydrolats from myristica fragrans seeds with magnesium aluminometasilicate as excipient: Antioxidant, antibacterial, and antiinflammatory activity. Foods 2020, 9, 37. [Google Scholar] [CrossRef] [Green Version]
- Franz, C.; Novak, J. Sources of essential oils. In Handbook of Essential Oils: Science, Technology, and Applications; Baser, K.H.C., Buchbauer, G., Eds.; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Abingdon, UK, 2010; pp. 39–82. [Google Scholar]
- Gong, J.; Yin, F.; Hou, R.; Yin, Y.L. Review: Chinese herbs as alternatives to antibiotics in feed for swine and poultry production: Potential and challenges in application. Can. J. Anim. Sci. 2014, 94, 223–241. [Google Scholar] [CrossRef]
- Khalil, A.A.; Rahman, U.; Khan, M.R.; Sahar, A.; Mehmood, T.; Khan, M. Essential oil eugenol: Sources, extraction techniques and nutraceutical perspectives. RSC Adv. 2017, 7, 32669–32681. [Google Scholar] [CrossRef] [Green Version]
- Farhath, S.; Vijaya, D.; Vimal, D. Immunomodulatory activity of geranial, geranial acetate, gingerol and eugenol essential oils: Evidence for humoral and cell-mediated responses. Avicenna J. Phytomed. 2013, 3, 224–230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patterson, J.E.; McElmeel, L.; Wiederhold, N.P. In vitro activity of essential oils against gram-positive and gram-negative clinical isolates, including carbepenem-resistant Enterobacteriaceae. Open Forum Infect. Dis. 2019, 12, ofz502. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, J.K.R.; Figueiredo, P.L.B.; Byler, K.G.; Setzer, W.N. Essential oils as antiviral agents, potential of essential oils to treat sars-cov-2 infection: An in-silico investigation. Int. J. Mol. Sci. 2020, 21, 3426. [Google Scholar] [CrossRef] [PubMed]
- Peterfalvi, A.; Miko, E.; Nagy, T.; Reger, B.; Simon, D.; Miseta, A.; Czéh, B.; Szereday, L. Much More Than a Pleasant Scent: A Review on Essential Oils Supporting the Immune System. Molecules 2019, 24, 4530. [Google Scholar] [CrossRef] [Green Version]
- Sander, G.; Heckmann, M.; Weghuber, J. Immunomodulatory activities of selected essential oils. Biomolecules 2020, 10, 1139. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Bolwell, P.G.; Bramley, P.M.; Pridham, J.B. The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic. Res. 1995, 22, 375–383. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [Google Scholar] [CrossRef]
- Simitzis, P.E. Enrichment of animal diets with essential oils-a great perspective on improving animal performance and quality characteristics of the derived products. Medicines 2017, 4, 35. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez, L.M.; Moeser, A.J.; Blikslager, A.T. Porcine models of digestive disease: The future of large animal translational research. Transl. Res. 2015, 166, 12–27. [Google Scholar] [CrossRef] [Green Version]
- Barone, F.; Laghi, L.; Gianotti, A.; Ventrella, D.; Saa, D.L.T.; Bordoni, A.; Forni, M.; Brigidi, P.; Bacci, M.L.; Turroni, S. In vivo effects of einkorn wheat (Triticum monococcum) bread on the intestinal microbiota, metabolome, and on the glycemic and insulinemic response in the pig model. Nutrients 2018, 11, 16. [Google Scholar] [CrossRef] [Green Version]
- Li, P.; Piao, X.; Ru, Y.; Han, X.; Xue, L.; Zhang, H. Effects of adding essential oil to the diet of weaned pigs on performance, nutrient utilization, immune response and intestinal health. Asian-Aust. J. Anim. Sci. 2012, 25, 1617–1626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, Z.; Xu, X.; Zhang, Q.; Li, P.; Zhao, P.; Li, Q.; Liu, J.; Piao, X. Effects of essential oil supplementation of a low-energy diet on performance, intestinal morphology and microflora, immune properties and antioxidant activities in weaned pigs. Anim. Sci. J. 2015, 86, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Maenner, K.; Vahjen, W.; Simon, O. Studies on the effects of essential-oil-based feed additives on performance, ileal nutrient digestibility, and selected bacterial groups in the gastrointestinal tract of piglets. J. Anim. Sci. 2011, 89, 2106–2112. [Google Scholar] [CrossRef] [Green Version]
- Platel, K.; Srinivasan, K. Digestive stimulant action of spices: A myth or reality? Indian J. Med. Res. 2004, 119, 167–179. [Google Scholar] [PubMed]
- Li, Y.; Fu, X.; Ma, X.; Geng, S.; Jiang, X.; Huang, Q.; Hu, C.; Han, X. Intestinal microbiome-metabolome responses to essential oils in piglets. Front. Microbiol. 2018, 9, 1988. [Google Scholar] [CrossRef] [PubMed]
- Michiels, J.; Missotten, J.; Dierick, N.; Freumaut, D.; Maene, P.; Smet, S. In vitro degradation and in vivo passage kinetics of carvacrol, thymol, eugenol and trans-cinnamaldehyde along the gastrointestinal tract of piglets. J. Sci. Food Agric. 2008, 13, 2371–2381. [Google Scholar] [CrossRef]
- Peddi, M.G. Novel drug delivery system: Liquid/solid compacts. J. Mol. Pharm. Org. Process Res. 2013, 1, 3. [Google Scholar] [CrossRef]
- Zhai, H.; Liu, H.; Wang, S.; Wu, J.; Anna-Maria, K. Potential of essential oils for poultry and pigs. Anim. Nutr. 2018, 4, 179–186. [Google Scholar] [CrossRef]
- Belkaid, Y.; Hand, T.W. Role of the microbiota in immunity and inflammation. Cell 2014, 157, 121–141. [Google Scholar] [CrossRef] [Green Version]
- Dieterich, W.; Schink, M.; Zopf, Y. Microbiota in the gastrointestinal tract. Med. Sci. 2018, 6, 116. [Google Scholar] [CrossRef] [Green Version]
- Bustos Fernandez, L.M.; Lasa, J.S.; Man, F. Intestinal microbiota: It’s role in digestive diseases. J. Clin. Gastroenterol. 2014, 48, 657–666. [Google Scholar] [CrossRef] [PubMed]
- O’Keefe, S.J. Nutrition and colonic health: The critical role of the microbiota. Curr. Opin. Gastroenterol. 2008, 24, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Valdivieso-Ugarte, M.; Gomez-Llorente, C.; Plaza-Díaz, J.; Gil, Á. Antimicrobial, antioxidant, and immunomodulatory properties of essential oils: A systematic review. Nutrients 2019, 11, 2786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, W.Z.; Lu, C.H. Carvacrol-induced [Ca2+] i rise and apoptosis in human glioblastoma cells. Life Sci. 2012, 90, 703–711. [Google Scholar] [CrossRef]
- Chou, T.H.; Ding, H.Y.; Hung, W.J.; Liang, C.H. Antioxidative characteristics and inhibition of α-melanocyte-stimulating hormone-stimulated melanogenesis of vanillin and vanillic acid from Origanum vulgare. Exp. Dermatol. 2010, 19, 742–750. [Google Scholar] [CrossRef]
- Suntres, Z.E.; Coccimiglio, J.; Alipour, M. The bioactivity and toxicological actions of carvacrol. Crit. Rev. Food Sci. Nutr. 2015, 55, 304–318. [Google Scholar] [CrossRef]
- Therdthai, N.; Zhou, W. Characterization of microwave vacuum drying and hot air drying of mint leaves (Mentha cordifolia Opiz ex Fresen). J. Food Eng. 2009, 91, 482–489. [Google Scholar] [CrossRef]
- Akpinar, E.K. Drying of mint leaves in a solar dryer and under open sun: Modelling, performance analyses. Energ. Convers. Manag. 2010, 51, 2407–2418. [Google Scholar] [CrossRef]
- Nayak, S.; Kumar, A.; Mishra, J.; Tiwari, G.N. Drying and testing of mint (Mentha piperita) by a hybrid photovoltaic-thermal (PVT)-based greenhouse dryer. Dry. Technol. 2011, 29, 1002–1009. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Sun, M.; Corke, H. Antioxidant capacity of 26 spice extracts and characterization of their phenolic constituents. J. Agric. Food Chem. 2005, 53, 7749–7759. [Google Scholar] [CrossRef]
- Nabigol, A.; Morshedi, H. Evaluation of the antifungal activity of the Iranian thyme essential oils on the postharvest pathogens of strawberry fruits. Afr. J. Biotechnol. 2011, 10, 9864–9869. [Google Scholar] [CrossRef] [Green Version]
- Mendivil, E.A.; Rodriguez, J.F.; Spinora, M.; Garcia-Fajardo, J.A.; Obledo-Vazquez, E.N. Chemical composition and fungicidal activity of the essential oil of Thymus vulgaris against alternaria citri. e-Gnosis 2006, 4, 16. [Google Scholar]
- De Assis Alves, T.; Pinheiro, P.F.; Praça-Fontes, M.M.; Andrade-Vieira, L.F.; Corrêa, K.B.; de Assis Alves, T.; Soares, T.C.B. Toxicity of thymol, carvacrol and their respective phenoxyacetic acids in Lactuca sativa and Sorghum bicolor. Ind. Crops Prod. 2018, 114, 59–67. [Google Scholar] [CrossRef]
- Park, J.; Jeon, Y.; Lee, C.; Chung, N.; Lee, H. Insecticidal toxicities of carvacrol and thymol derived from Thymus vulgaris Lin. against Pochazia shantungensis Chou & Lu., newly recorded pest. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [Green Version]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- The European Parliament and of the Council EUR-Lex-32010L0063-EN-EUR-Lex. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32010L0063 (accessed on 18 August 2020).
- Lithuanian Director of the State Food and Veterinary Service. B1-866 Dėl Mokslo ir Mokymo Tikslais Naudojamų Gyvūnų Laikymo, Priežiūros ir Naudojimo Reikalavimų Patvirtinimo. Available online: https://e-seimas.lrs.lt/portal/legalAct/lt/TAD/TAIS.437081?positionInSearchResults=0&searchModelUUID=64a1f51f-6356-4b60-a21e-3a67ac3b1107 (accessed on 18 August 2020).
- Fijan, S. Microorganisms with claimed probiotic properties: An overview of recent literature. Int. J. Environ. Res. Public Health 2014, 11, 4745–4767. [Google Scholar] [CrossRef]
- California Daily Research Foundation. Probiotic Basics. Available online: http://cdrf.org/home/checkoff-investments/usprobiotics/probiotics-basics/#:~:text=Most%20probiotic%20products%20contain%20bacteria,have%20been%20developed%20as%20probiotic (accessed on 2 September 2020).
- Martin, R.; Miquel, S.; Benevides, L.; Bridonneau, C.; Robert, V.; Hudault, S.; Chain, F.; Berteau, O.; Azevedo, V.; Chatel, J.M.; et al. Functional characterization of novel Faecalibacterium prausnitzii strains isolated from healthy volunteers: A step forward in the use of F. prausnitzii as a next-generation probiotic. Front. Microbiol. 2017, 8, 1226. [Google Scholar] [CrossRef]
- Social Science Statistics. Available online: https://www.socscistatistics.com/tests/ztest/default.aspx (accessed on 2 September 2020).
- Pajarillo, E.A.B.; Chae, J.P.; Balolong, M.P.; Bum Kim, H.; Kang, D.K. Assessment of fecal bacterial diversity among healthy piglets during the weaning transition. J. Gen. Appl. Microbiol. 2014, 60, 140–146. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Tsai, T.; Deng, F.; Wei, X.; Chai, J.; Knapp, J.; Apple, J.; Maxwell, C.V.; Lee, J.A.; Li, Y.; et al. Longitudinal investigation of the swine gut microbiome from birth to market reveals stage and growth performance associated bacteria. Microbiome 2019, 7, 109. [Google Scholar] [CrossRef] [Green Version]
- Holman, D.B.; Brunelle, B.W.; Trachsel, J.; Allen, H.K. Meta-analysis to define a core microbiota in the swine gut. MSystems 2017, 2, e00004-17. [Google Scholar] [CrossRef] [Green Version]
- Crespo-Piazuelo, D.; Estellé, J.; Revilla, M.; Criado_Mesas, L.; Ramyao-Caldas, Y.; Ovilo, C.; Fernandez, A.; Ballester, M.; Folch, J.M. Characterization of bacterial microbiota compositions along the intestinal tract in pigs and their interactions and functions. Sci. Rep. 2018, 8, 12727. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.F.; Nyachoti, M. Using probiotics to improve swine gut health and nutrient utilization. Anim. Nutr. 2017, 3, 331–343. [Google Scholar] [CrossRef] [PubMed]
- Guevarra, R.B.; Hong, S.H.; Cho, J.H.; Kim, B.R.; Shin, J.; Lee, J.H.; Kang, B.N.; Kim, Y.H.; Wattanaphansak, S.; Isaacson, R.; et al. The Dynamics of the piglet gut microbiome during the weaning transition in association with health and nutrition. J. Anim. Sci. Biotechnol. 2018, 9, 54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Q.; Huang, X.; Zhao, S.; Sun, W.; Yan, Z.; Wang, P.; Li, S.; Huang, W.; Zhang, S.; Liu, L.; et al. Structure and function of the fecal microbiota in diarrheic neonatal piglets. Front. Microbiol. 2017, 8, 502. [Google Scholar] [CrossRef] [Green Version]
- Hynonen, U.; Kant, R.; Lahteinen, T.; Pietila, T.E.; Beganovic, J.; Smidt, H.; Uroic, K.; Avall-Jaaskelainen, S.; Palva, A. Functional characterization of probiotic surface layer protein-carrying lactobacillus amylovorus strains. BMC Microbiol. 2014, 14, 199. [Google Scholar] [CrossRef] [Green Version]
- Tett, A.; Huang, K.D.; Asnicar, F.; Fehlner-Peach, H.; Pasolli, E.; Karcher, N.; Armanini, F.; Manghi, P.; Bonharn, K.; Zolfo, M.; et al. The Prevotella copri complex comprises four distinct clades underrepresented in westernized populations. Cell Host Microbe 2019, 26, 666–679. [Google Scholar] [CrossRef] [Green Version]
- De Filippis, F.; Pasolli, E.; Tett, A.; Tarallo, S.; Naccarati, A.; De Angelis, M.; Neviani, E.; Cocolin, L.; Gobbetti, M.; Segata, N.; et al. Distinct genetic and functional traits of human intestinal prevotella copri strains are associated with different habitual diets. Cell Host Microbe 2019, 25, 444–453. [Google Scholar] [CrossRef] [Green Version]
- Almuzara, M.; Bonofiglio, L.; Cittadini, R.; Vera Ocampo, C.; Montilla, A.; del Castillo, M.; Ramirez, M.S.; Mollerach, M.; Vay, C. First case of Streptococcus lutetiensis bacteremia involving a clindamycin-resistant isolate carrying the lnuB gene. J. Clin. Microbiol. 2013, 51, 4259–4261. [Google Scholar] [CrossRef] [Green Version]
- Piva, S.; Pietra, M.; Serraino, A.; Merialdi, G.; Magarotto, J.; Giacometti, F. First description of Streptococcus lutetiensis from a diseased cat. Lett. Appl. Microbiol. 2019, 69, 96–99. [Google Scholar] [CrossRef]
- Ghali, M.B.; Scott, P.T.; Al Jassim, R.A.M. Characterization of Streptococcus bovis from the rumen of the dromedary camel and rusa deer. Lett. Appl. Microbiol. 2004, 39, 341–346. [Google Scholar] [CrossRef]
- Dumke, J.; Hinse, D.; Vollmer, T.; Schulz, J.; Knabbe, C.; Dreier, J. Potential transmission pathways of Streptococcus gallolyticus subsp. Gallolyticus. PLoS ONE 2015, 10, e0126507. [Google Scholar] [CrossRef] [Green Version]
- Goyette-Desjardins, G.; Auger, J.P.; Xu, J.; Segura, M.; Gottschalk, M. Streptococcus suis, an important pig pathogen and emerging zoonotic agent-an update on the worldwide distribution based on serotyping and sequence typing. Emerg. Microbes Infect. 2014, 3, e45. [Google Scholar] [CrossRef]
- Gottschalk, M.; Lacouture, S.; Bonifait, L.; Roy, D.; Fittipaldi, N.; Grenier, D. Characterization of Streptococcus suis isolates recovered between 2008 and 2011 from diseased pigs in Quebec, Canada. Vet. Microbiol. 2013, 162, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Tien Le, H.T.; Sugiyama, N.; Duangsonk, K.; Tharavichitkul, P.; Osawa, R. Phenotypic and PCR-based identification of bacterial strains isolated from patients with suspected Streptococcus suis infection in northern thailand. Jpn. J. Infect. Dis. 2012, 65, 171–174. [Google Scholar] [PubMed]
- Górska, A.; Przystupski, D.; Niemczura, M.J.; Kulbacka, J. Probiotic bacteria: A promising tool in cancer prevention and therapy. Curr. Microbiol. 2019, 76, 939–949. [Google Scholar] [CrossRef] [Green Version]
- Choi, S.S.; Kim, Y.; Han, K.S.; You, S.; Oh, S.; Kim, S.H. Effects of Lactobacillus strains on cancer cell proliferation and oxidative stress in vitro. Lett. Appl. Microbiol. 2006, 42, 452–458. [Google Scholar] [CrossRef]
- Gautam, N.; Mantha, A.K.; Mittal, S. Essential oils and their constituents as anticancer agents: A mechanistic view. BioMed Res. Int. 2014, 2014, 154106. [Google Scholar] [CrossRef] [Green Version]
- Bartkiene, E.; Lele, V.; Sakiene, V.; Zavistanaviciute, P.; Ruzauskas, M.; Bernatoniene, J.; Jakstas, V.; Viskelis, P.; Zadeike, D.; Juodeikiene, G. Improvement of the antimicrobial activity of lactic acid bacteria in combination with berries/fruits and dairy industry by-products. J. Sci. Food Agric. 2019, 99, 3992–4002. [Google Scholar] [CrossRef]
- Bartkiene, E.; Bartkevics, V.; Ikkere, L.E.; Pugajeva, I.; Zavistanaviciute, P.; Lele, V.; Ruzauskas, M.; Bernatoniene, J.; Jakstas, V.; Klupsaite, D.; et al. The effects of ultrasonication, fermentation with Lactobacillus sp., and dehydration on the chemical composition and microbial contamination of bovine colostrum. J. Dairy Sci. 2018, 8, 6787–6798. [Google Scholar] [CrossRef]
- Juodeikiene, G.; Zadeike, D.; Bartkiene, E.; Lele, V.; Bernatoniene, J.; akstas, V. A new delivery system based on apple pomace-pectin gels to encourage the viability of antimicrobials strains. Food Sci. Technol. Int. 2020, 26, 242–253. [Google Scholar] [CrossRef]


| Samples | Thymol mg/mL | Carvacrol mg/mL | Menthol mg/mL |
|---|---|---|---|
| Tablets before (19 May 2019) | 1.20206 | 0.0145364 | 0.56 |
| Tablets after (19 May 2019) | 1.02065 | 0.0112584 | 0.225 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruzauskas, M.; Bartkiene, E.; Stankevicius, A.; Bernatoniene, J.; Zadeike, D.; Lele, V.; Starkute, V.; Zavistanaviciute, P.; Grigas, J.; Zokaityte, E.; et al. The Influence of Essential Oils on Gut Microbial Profiles in Pigs. Animals 2020, 10, 1734. https://doi.org/10.3390/ani10101734
Ruzauskas M, Bartkiene E, Stankevicius A, Bernatoniene J, Zadeike D, Lele V, Starkute V, Zavistanaviciute P, Grigas J, Zokaityte E, et al. The Influence of Essential Oils on Gut Microbial Profiles in Pigs. Animals. 2020; 10(10):1734. https://doi.org/10.3390/ani10101734
Chicago/Turabian StyleRuzauskas, Modestas, Elena Bartkiene, Arunas Stankevicius, Jurga Bernatoniene, Daiva Zadeike, Vita Lele, Vytaute Starkute, Paulina Zavistanaviciute, Juozas Grigas, Egle Zokaityte, and et al. 2020. "The Influence of Essential Oils on Gut Microbial Profiles in Pigs" Animals 10, no. 10: 1734. https://doi.org/10.3390/ani10101734
APA StyleRuzauskas, M., Bartkiene, E., Stankevicius, A., Bernatoniene, J., Zadeike, D., Lele, V., Starkute, V., Zavistanaviciute, P., Grigas, J., Zokaityte, E., Pautienius, A., Juodeikiene, G., & Jakstas, V. (2020). The Influence of Essential Oils on Gut Microbial Profiles in Pigs. Animals, 10(10), 1734. https://doi.org/10.3390/ani10101734











