1. Introduction
Genetic diversity of domestic hens existing across Ecuador is not only promoted by climatic stratification but also natural and human driven selection. From the red junglefowl (
Gallus gallus), the most likely ancestor of these avian populations [
1], the effects of natural selection may have resulted in a high heterogeneity and variability of the morphological characteristics of fowl, with a high potential to adapt to the different environmental conditions [
2,
3,
4,
5,
6].
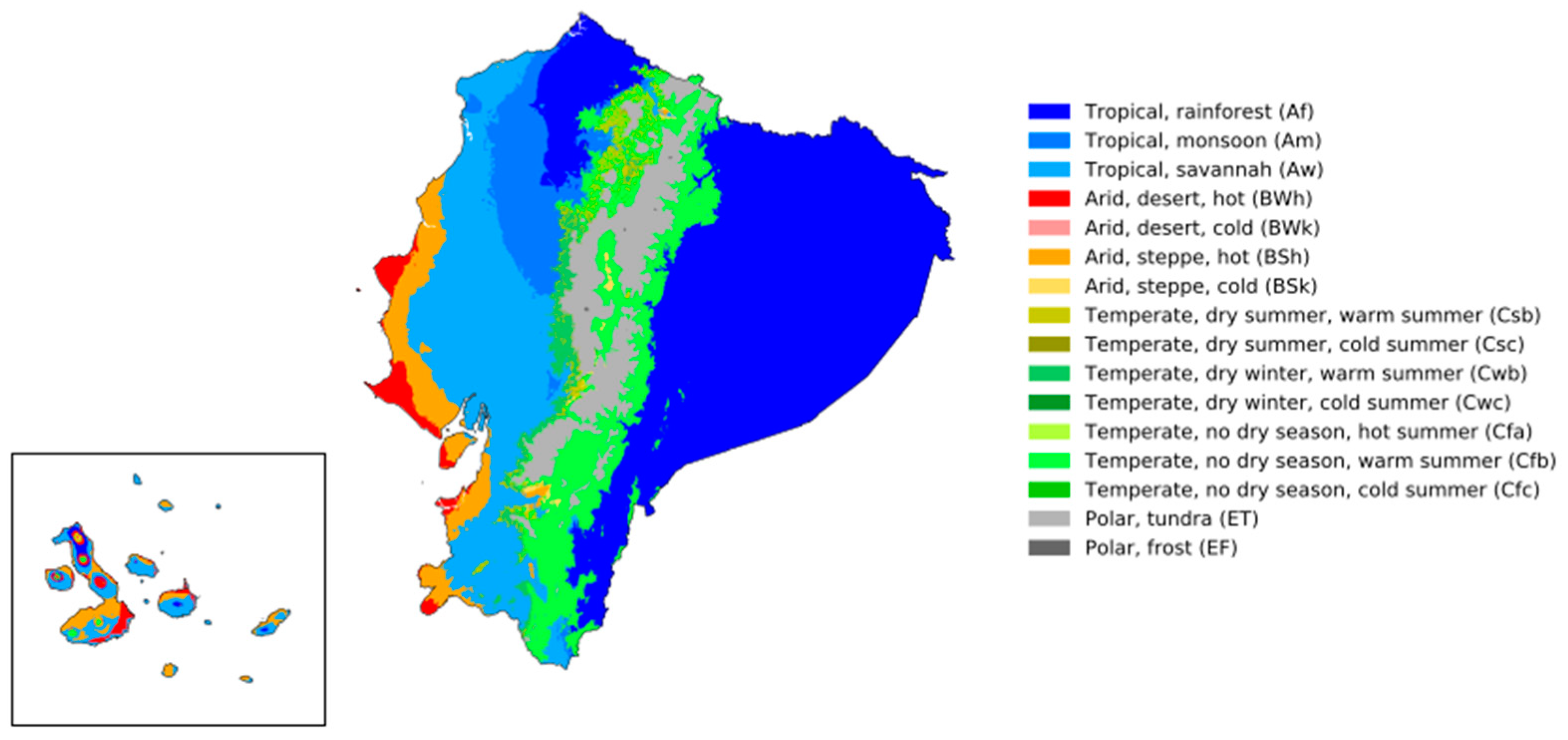
Contextually, Continental Ecuador comprises three agroecological regions, which are the Sierra (Andean highlands), the Amazonian Oriente (eastern rainforests), and the Costa (Pacific coastal lowlands), with five thermal or climatic floors: the warm floor (0–1000 m above mean sea level (mamsl) at 25 °C); the temperate floor (1000 to 2000 mamsl, at temperatures that range from 16 to 23 °C); the cold floor (2000 to 3000 mamsl at 12 °C); the paramo/moor floor (from 3000 to 4000 mamsl close to 0 °C); and the glacial floor (4000 mamsl < 0 °C).
Apart from naturally driven selection processes and natural migratory movements, genetic variability in local chicken populations may have been conducted as a result of human action. For instance, human-made migration processes [
7] brought about the widespread distribution of poultry genetic material, given the size of animals was convenient and facilitated transport, favoring the expansion of these fowl across the different agroecological levels [
3].
These factors led to genetic divergences contributing to poultry production under a family-run backyard system usually developed by each household’s women [
3]. Husbandry practices characterized by the use of rustic animals in free range conditions with a low capital investment, which enables assuming a relatively low economic risk to implement an efficient productive management to produce high-biological-value protein sources such as meat and eggs [
8,
9]. Additionally, these products are preferred among consumers because of their pigmentation, taste, and lean quality of meat [
10,
11]; which translates into acceptable income that returns to each family, closing the cycle [
12,
13,
14,
15,
16,
17].
Breeds originating in the Old World were introduced to Latin American territories by the Spanish colonists and adapted to the different agroecological areas and conditions that they found, forming what has traditionally been addressed as Creole hen populations. For decades, these creole populations occupied local productive niches and evolved towards their current state, but still lacked the necessary characterization actions that may help consolidating and protecting them. In parallel, current breed development and formation until the XVII century provided the basic elements for the directed selection of our days and for the pursuit of concrete characteristics of interest to the farmer or producer. In this context, a new conglomerate of breeds and commercial lines formed in the first world were introduced into developing countries in an attempt to fulfil the growing market demands at a lower cost [
18].
This global situation resulted in an alarming loss in the biodiversity of animals of zootechnical interest that the region faces nowadays. According to Food and Agriculture Organization (FAO), the endangerment risk that 81 percent of Latin America and the Caribbean avian breeds are exposed to is unknown [
19], as even censuses are not appropriately registered. The increased risk of a population whose endangerment status is unknown is based on the fact that measures towards its protection are not implemented. In this regard, efforts are being made to maintain, conserve and, in turn, benefit from their most profitable or useful traits, such as disease or stress resistance, in commercial breeding plans [
18].
Not only local hen breeds face a serious risk of extinction, but there is also a simultaneous loss of the traits that made them survive after the evolutionary process that they followed when they arrived in and adapted to the lands to which they were introduced. Creole hens present a good ability to scavenge and forage, have good maternal qualities, and are hardier than exotic breeds with higher survival rates and minimal care and attention requirements. This rusticity is one of those traits to positively influence avian zootechnical production, given its implication with the adaptation ability of animals to the environment in which they are produced.
After a period characterized by a lack of actions regarding local genetic resources conservation, with policies more likely focusing on intensive production, morphological characterization studies in fowl started being run again in Ecuador. These studies lay the basis for local resources conservation and breeding plans. Zoometric traits have widely been reported to depend on an inherited basis and to be suitable means of prediction for the live weight of the individuals [
20,
21,
22]. Thus, they may play an important role in the subsequent performance of animal carcasses [
23]; a relationship that translates to new potential selection criteria, seeking the maximization of the profitability of the products derived from such local genetic resources.
Despite the fact that research projects seeking the zoometrical characterization of Ecuadorian local hen breeds started being implemented using univariate analysis, there is still a patent lack of knowledge regarding the differentiation of such local populations, and hence policies towards the protection of such genetic resources cannot be implemented properly. Therefore, the aim of this study was to perform differentiated zoometric characterization of Creole hens through the application of a canonical discriminant analysis (CDA) to provide insights on the possible clustering patterns described by the population and into which subpopulations can be distinguished using Ecuadorian provinces as the criteria of origin [
24]. Conclusively, this approach will enable quantification of the large existing phenotypic variability in the Ecuadorian creole hen population as a strategy to facilitate the rational development of such productively important avian local resources and their use, and to direct the implementation of conservation strategies aimed at ensuring their survival in the competitive world of poultry production and future consolidation as breeds.
4. Discussion
The morphometric measurements show highly significant differences in relation to sex, as reported by Yakubu and Salako [
62] in indigenous chickens in Nigeria, which reported such differences to be based on the hormonal effects of sex that condition growth. These results were consistent with similar results in the literature [
6,
63].
The high coefficient of variation observed in the results is similar to that reported in different populations of chickens in Mexico [
64], and also in indigenous chickens in Nigeria [
61], which demonstrates the variability of the morphometry in the birds studied, which may be due to genetic divergence processes followed by the populations, such as migration [
65], which resulted in the morphological modification of the populations to adapt to the characteristics of the different environments and the orography to which the birds were introduced [
63,
66,
67,
68].
Measurements for head length are higher than those measured in Batsi Alak Hens of Mexico whose mean for males and females varied from 4.16 to 4.6 cm [
67], and lower than those reported for Yoruba ecotypes of Nigeria with an average of 9.90 cm [
68].
In terms of crest length, the values are lower than those measured in indigenous Nigerian roosters and higher than those indicated for hens of the same country [
62]. Likewise, Yoruba and Fulani ecotypes [
67] reported a similar value to that reported for the province of Cotopaxi when comparing fowl in general, without considering males and females separately [
69]. However, in autochthonous Catalonian chicken breeds, such as Patridged Penedesenca and Blonde Empordanesa, we observe crest sizes double those measured in the present study [
70].
For beak length, the values are analogous to those found in Botswana hens. This could be supported by the fact that both studies were conducted across three agroecological regions [
71]. However, higher values were reported by Yakubu and Salako [
62] in indigenous Nigerian fowl for males and females, as well as in native Catalonian breeds Partridged Penedesenca and Blonde Empordanesa. The opposite situation was described by Batsi Alak hens from Mexico [
67] and Fulani ecotypes of Nigeria [
68], which reported similar mean values to those of Bolivar, Guayas, Morona Santiago. This could be ascribed to the similarity between the climates of the locations in which the study took place.
For the neck length trait, the values found were equivalent to those found in Partridged Penedesenca and Blonde Empordanesa [
70] and Yoruba and Fulani Nigerian ecotypes [
68]; while lower values were found in indigenous Nigerian hens for both males and females [
62]. The highest values in literature were reported for Batsi Alak Hens from Mexico with measures of 19 to 17 cm in males and females, respectively [
67].
Body and dorsal lengths along with head length have been related in literature to the potential of animals for egg production [
68]. When these data are compared with those reported by Moazami-Goudarzi [
5] and their studies of local Tanzanian chicken ecotypes, the values for males of the Singamagazi ecotype were slightly higher than the average reported for Morona Santiago hens, but comparable to the males of the Kuchi ecotype. However, for the male of Mbeya ecotype and female of the Singamagazi ecotypes [
5], these values were similar to those reported for the Patridged Penedesenca hens and for the Blonde Empordanesa [
70] and equivalent to those found in Bolívar, Chimborazo, and Guayas. Studies conducted on Botswanan hens across three different agroecological areas found similar values for females and males [
71] to those measured in Cotopaxi and Tungurahua. In turn, Yakubu and Salako [
62] reported higher average measurements than those of Ecuadorian creole hens found in this study or those found for the Fulani and Yoruba.
Higher values for the ventral length variable are found in native breeds of Partridged Penedesenca, Blonde Empordanesa hens [
5], and Yarubi and Fulani ecotypes [
68]. The thoracic perimeter variable is a good indicator of meat yield in most species of poultry [
68]. Higher values were found than those obtained for Nigerian males and females [
62]. However, native Nigerian birds bred for research purposes belonging to the Anak Titan ecotype [
72] and the Yoruba and Fulani ecotypes [
68] reported similar values to those found for Chimborazo individuals.
The Batsi Alak hens in Mexico reached values similar to those reported in Bolívar, Chimborazo and Cotopaxi [
67]. These values were common in backyard hens in Mexico [
63]. The measurements for half wing radius ulna length (hwrul) were shorter than those for the males and females of the Batsi Alak hen breed from Mexico [
67]. However, the values for distal phalanx wing length (dpwl), were similar to those observed in Batsi Alak hens from Mexico for both males and females. This may be due to the fact that the study was carried out at an altitude of 1200 to 2760 m above mean sea level, similar conditions found at the location where our study took place [
67]. Thigh length in Partridged Penedesenca and Blonde Empordanesa hens [
70] were similar to those from Ecuadorian fowl from Cotopaxi and Tungurahua provinces, but shorter than the Nigerian Yoruba ecotype hens [
68].
Regarding the circumference of the leg, the indigenous hens of Nigeria reported similar values to those measured in Bolívar and Chimborazo and slightly similar ones to those reported for the hens of Cotopaxi. The dimensions of the leg have been related in literature with the type of production, with those animals presenting higher dimensions (both in width and length), being more appropriate to suit the requirements for meat production and characteristic of carnic breeds [
68]. Tanzanian local chicken ecotypes presented higher tarso-metatarsal lengths [
5] for Singamagazi and Kuchi than those of Ecuadorian Creole hens. However, females of the same ecotypes reported similar average values [
73] to those measured in Morona Santiago hens.
Similarly, males of the Ching’wekwe ecotype and females of the same ecotype and female of the Morogo ecotype presented similar values to those of the provinces of Guayas and Tungurahua, but much higher than those of the province of Cotopaxi. Likewise, this value was similar to that reported by Nigerian birds [
62], which may support the fact that ecotype, may be strongly conditioned by the agroecological conditions in the area in which these avian population are based.
Similarly, larger sizes were recorded for breeds such as the Partridged Penedesenca and Blonde Empordanesa with an average of 8 cm, which may be because the birds studied also come from four different eco-climates, homologous to those of the areas considered in this research.
For birds from India [
74], average values were similar to those measured in Bolívar and Chimborazo. Long tarsi have been associated with dry regions and flat topographies, as they allow birds to travel long distances in search for food, unlike birds with short tarsi, which could be attributed to the effects of natural selection [
72].
Short tarsi have been identified with a greater ability to escape from predators [
74], hence, they have been directly related to processes of adaptation and improvement of survival. Functionally, from a productive point of view, tall animals tend to be destined for meat production and small animals for egg production [
68]. In addition, the length of tarsi may be related to the prediction of live weight in the field, as reported by some authors [
75,
76,
77]. Ecuadorian creole hens present morphological traits which would made them more prone to produce eggs. However, the dimensions of certain morphological variables could make them suitable for meat production, so it is essential to implement breeding programs to select and direct crosses that allow ecotypes to be obtained by classifying individuals depending on to their productive potential.
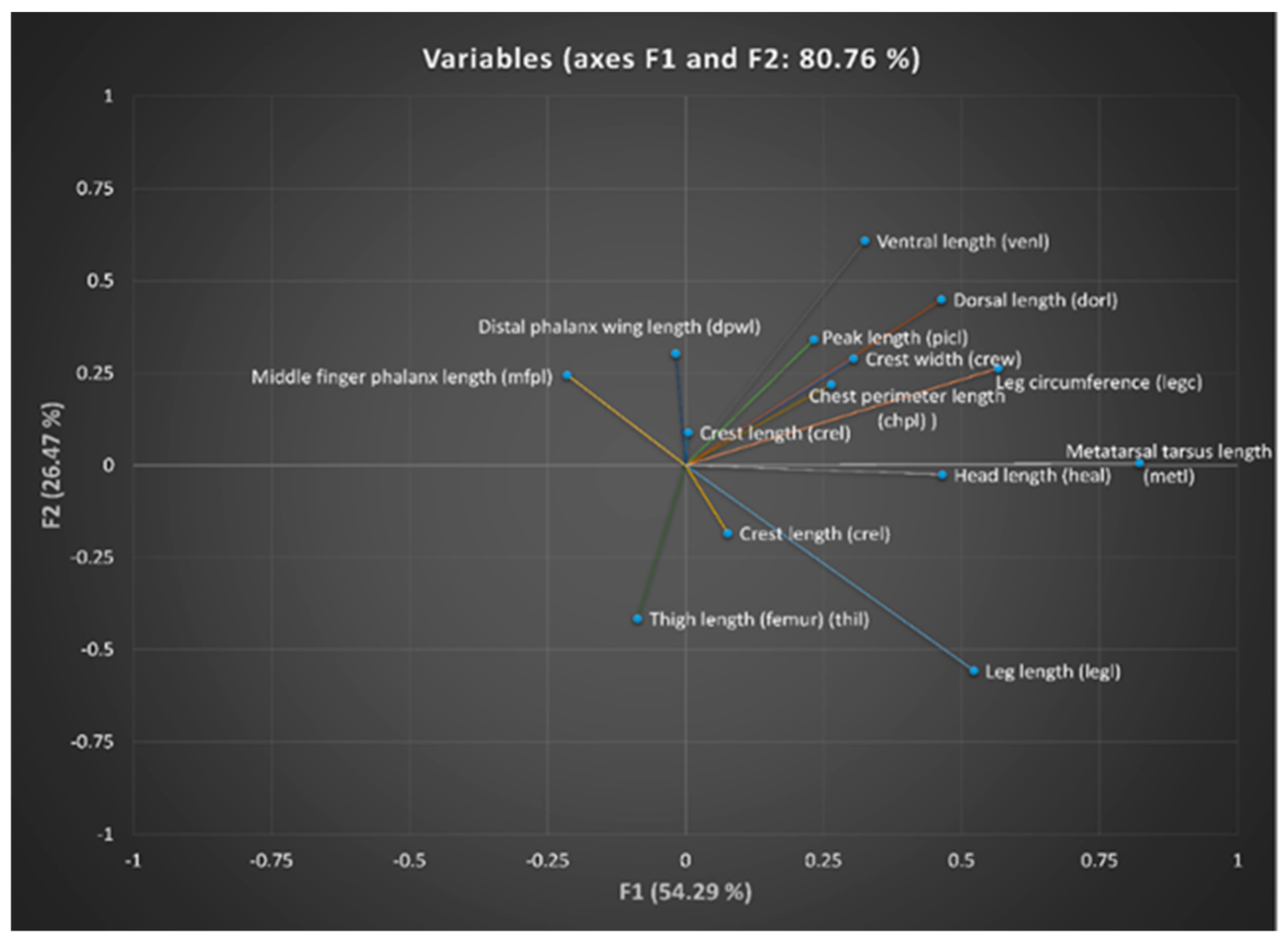
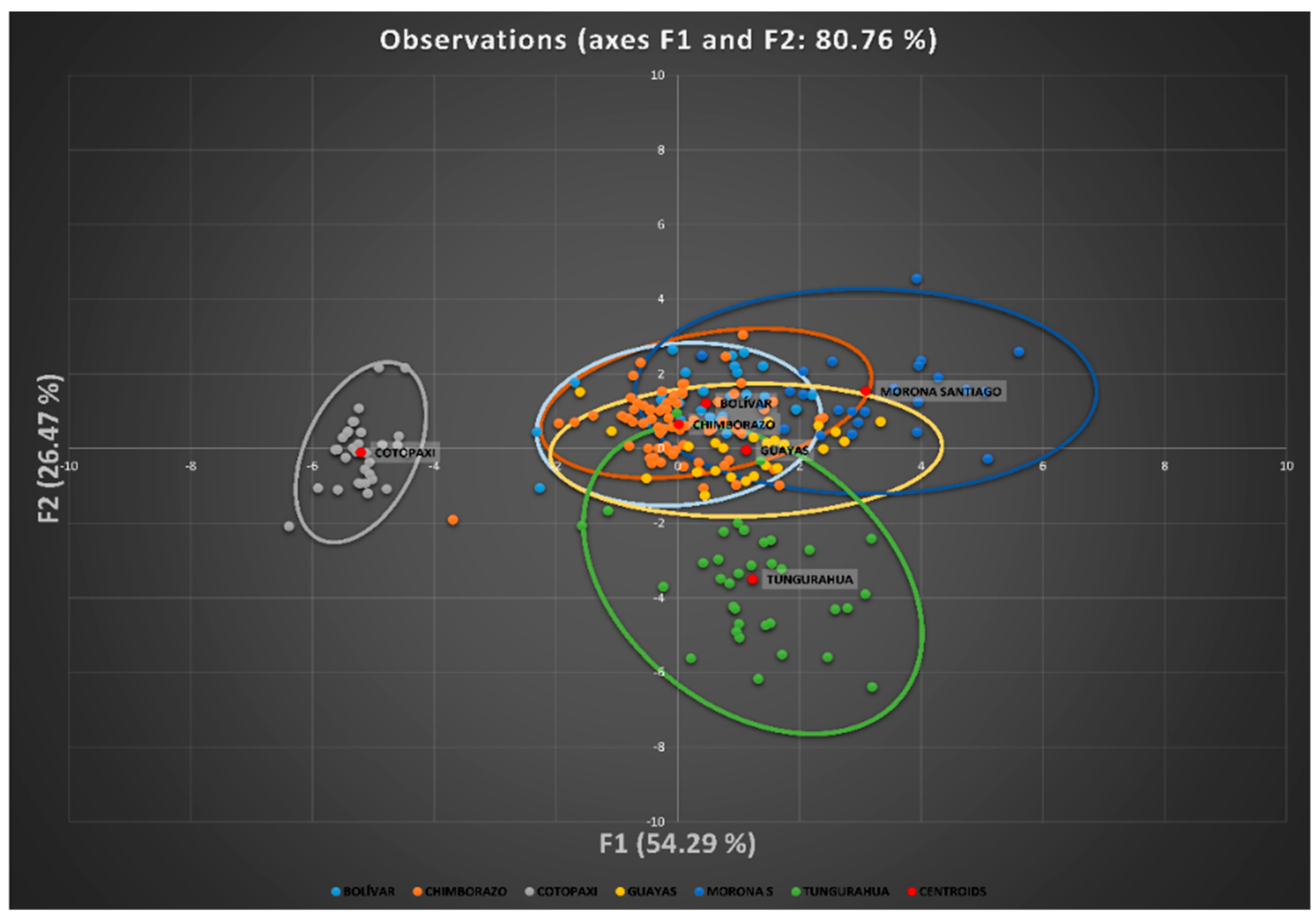
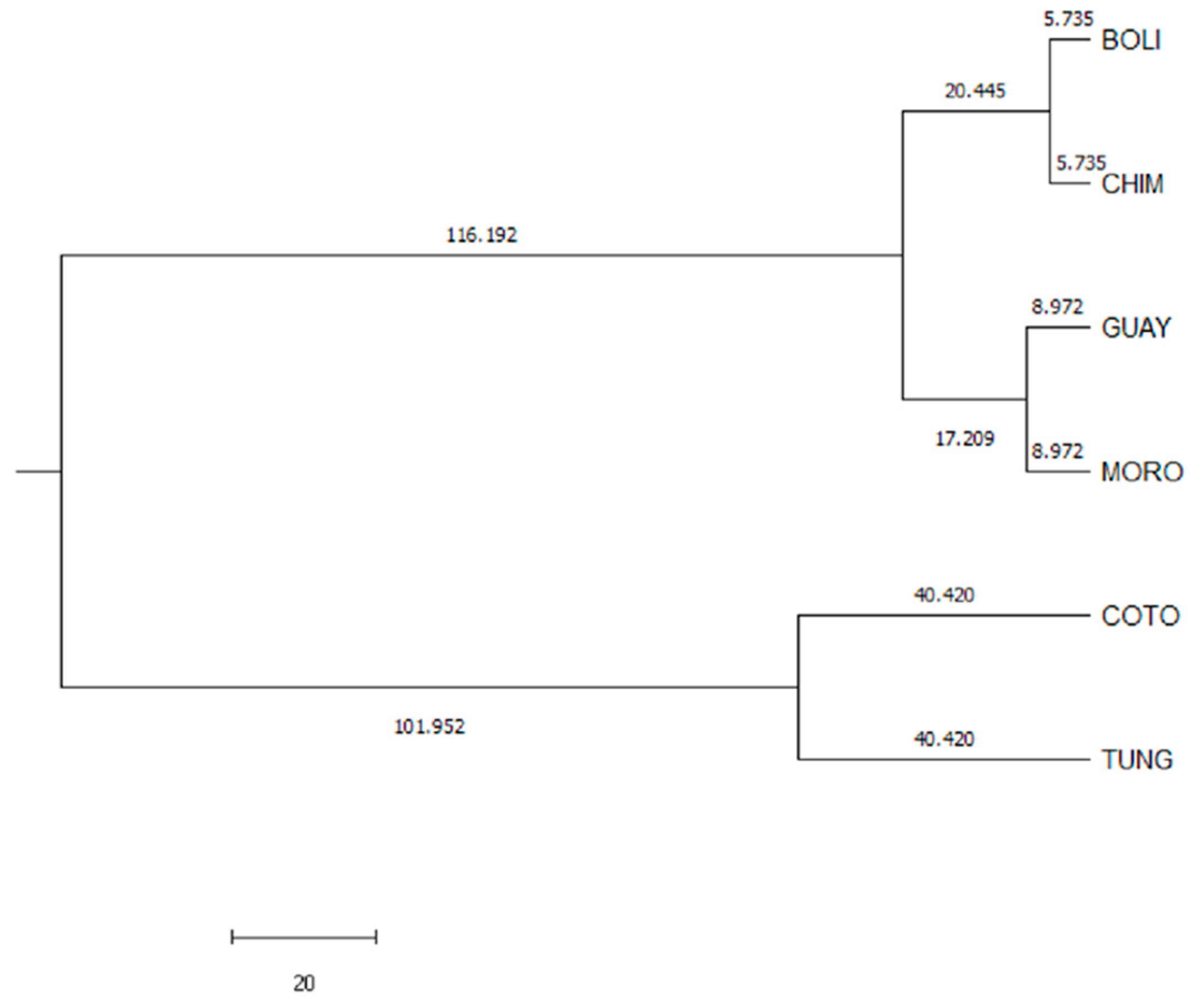
Canonical discriminant analysis suggests that the variables perimeter of the leg (legc), metatarsus tarsus length (metl) and middle finger phalanx length (mfpl) were the ones that had the greatest discriminatory capacity between provinces. The results revealed the presence of wide ranges of variation within and among Creole hens in Ecuador. However, four large population blocs could be identified, namely Cotopaxi, Tungurahua, Morona Santiago, and that comprising the populations of Bolivar, Chimborazo and Guayas, a fact that could be attributed to the different conditions found across the various agroecological zones in the country, ethnic groups handling these resources and cultural implications that they have, along with the huge migration events suffered by these resources when facing natural and/or man-made challenges.
Toalombo, et al. [
78] identified a common pattern of haplogroups of mitochondrial DNA for Ecuadorian chickens reared across Ecuadorian agroecological systems, which suggests these animals may belong to the same maternal lineages. In fact, the maternal origin for these populations could presumably be attributed to pre-Columbian Asiatic matrilines or Iberian matrilines arriving during the Spanish colonization. Furthermore, mitochondrial findings support the fact that current Ecuadorian local chickens do not show maternal influences from commercial lines and maintain high levels of genetic diversity without evidence of genetic drift and/or population bottlenecks. This high diversity may be owing to internal heterogeneity, which, as suggested by the results in the present study, may be promoted by the breeding policies that are carried out. Additionally, the patterns found by canonical discriminant analysis are supported by the results reported by Toalombo et al. [
78], as a certain internal substructure can be found. However, the absence of any breeding program, registers, or zootechnic management produces a high fragmentation of the potential Ecuadorian breeds.
The most likely reason for which Morona Santiago, Bolívar, Chimborazo, and Guayas populations present a mixed structure which does not permit their complete segregation from one another may have its basis on the fact that poultry farmers in this province are prone to preserve their birds, avoiding the introduction of individuals from external populations, despite the fact that poultry production has already been developed in their areas.
The provinces of Morona Santiago, Bolívar, and Chimborazo are nearby to each other, sin contrast to the province of Guayas, located in the Costa Region. However, there is a provincial road that joins Chimborazo (Sierra) and Guayas (Costa). It should be noted that agricultural fairs are likely to take place along the road connecting both provinces. These events act as exchange centers of genetic material, which is mainly performed with minor species such as birds, given the considerable ease to transport such resources. The same would happen with Morona Santiago, which is located in the Ecuadorian Amazon, but near Chimborazo, which is geographically located in the center of the country. The provinces in question are characterized by their agricultural and livestock background, such that 13% of the population is engaged in poultry production activity; hence, it could be assumed that the populations maintain the genetics of their fowl over time.
The connection with Guayas, as stated by the General Secretariat of the Andean Community in 2009, lays on the fact that during the seventies, there was a reduction in domestic agricultural-livestock production, which led to the migration of the inhabitants of Nabuzo-Penipe (Chimborazo) to the coast of Ecuador. This social movement implied people carried easily-used animal species such as hens along with them. Complementarily, as a result of the eruptions of the Tungurahua volcano, constant since the late 1990s [
79], the Canton Penipe experienced immigration from the surrounding populations, which was also noticeable in Nabuzo (Chimborazo), the area least affected by volcanic ash. This confirms that the genetic material did not suffer such migratory pressure and hence its resources remain intact, which in turn explains the clustering revealed by the canonical discriminant analysis and which may outline the same genetic structure.