A Selection of Platforms to Evaluate Surface Adhesion and Biofilm Formation in Controlled Hydrodynamic Conditions
Abstract
1. Introduction
2. Effects of Hydrodynamics on Microbial Adhesion and Biofilm Formation
3. Biofilm Platforms
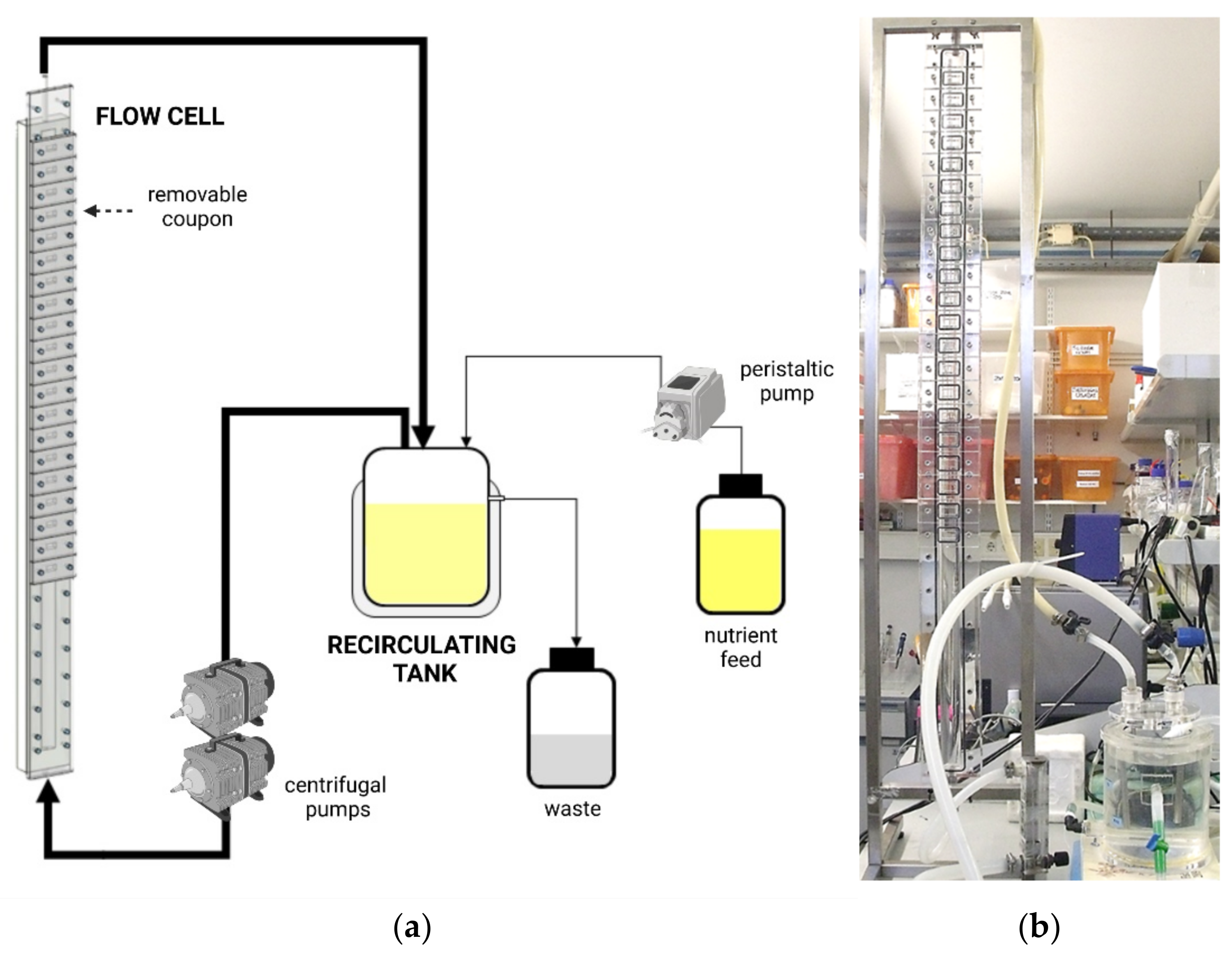
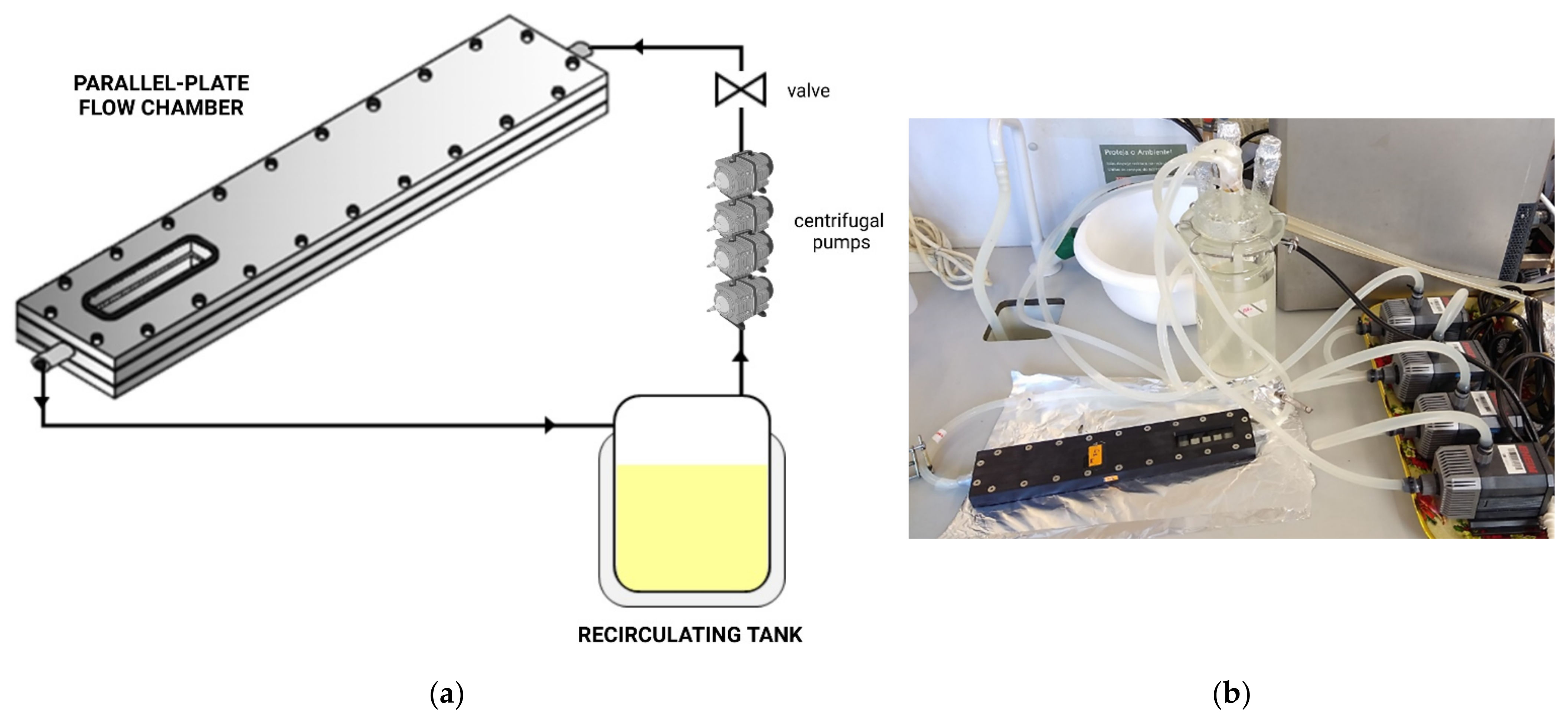
3.1. Flow Cells: Robbins Device and Modifications, and Flow Chambers
3.1.1. Robbins Device and Modifications
3.1.2. Flow Chambers
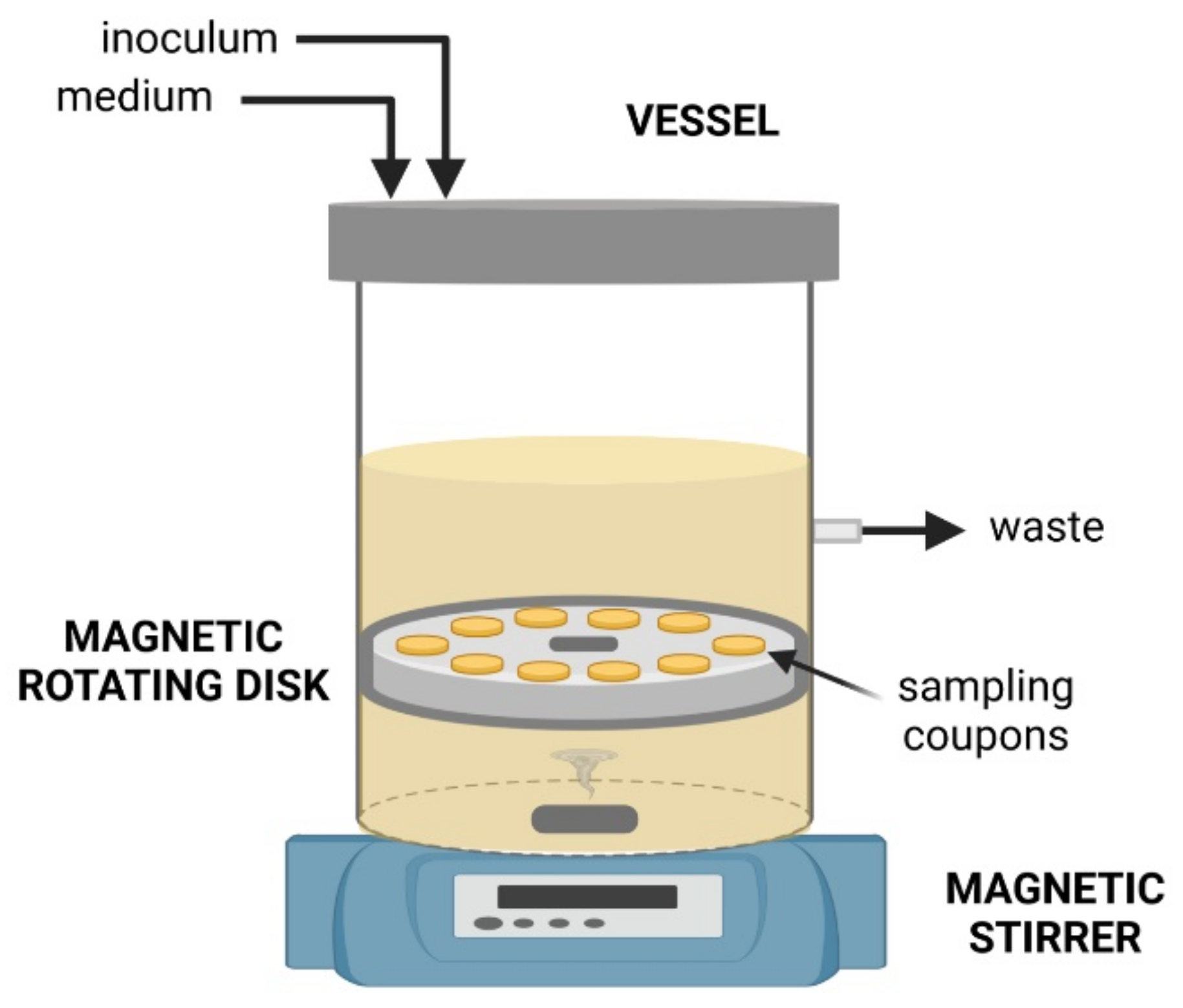
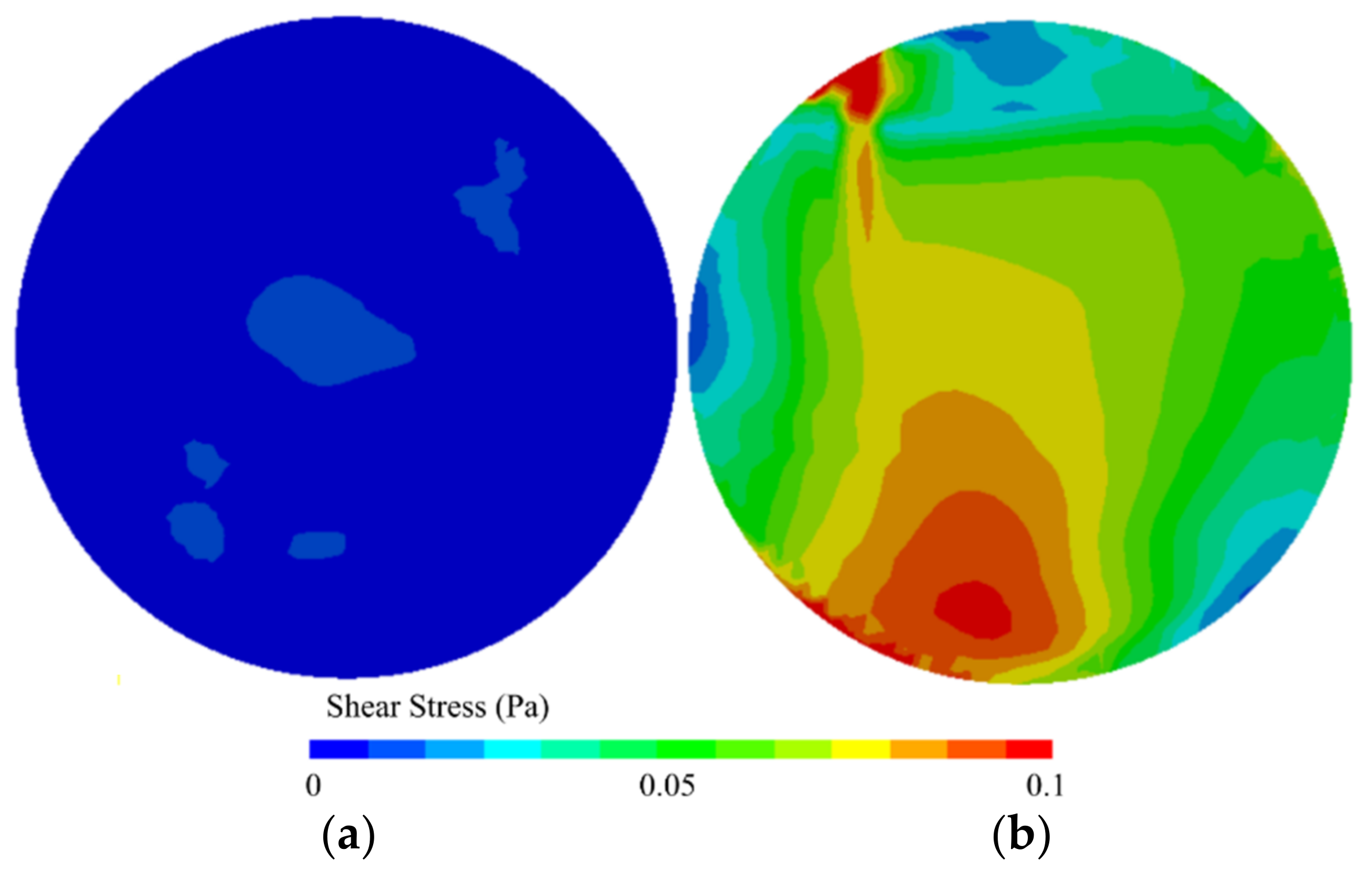
3.2. Rotating Biofilm Devices
3.3. Microfluidic Devices
3.4. Microplates
3.4.1. 96-Well Microplates
3.4.2. 12- and 6-Well Microplates
4. Adhesion and Biofilm Studies Performed under Controlled Hydrodynamics
4.1. Environmental Applications
| Environmental Scenario | Shear Stress (Pa) | Shear Strain (s−1) | References |
|---|---|---|---|
| Drinking-water distribution systems | 0.13–9.10 | n.d. | [79,83] |
| Ship in harbor | n.d. | 50 | [84] |
| Ship navigation (turbulent flow) | n.d. | 125,000 | [84] |
| Marine environments | n.d. | 4 and 40 | [71] |
| Tumbling and pouring | n.d. | 10–100 | [84] |
| Channels within a biofilm | n.d. | 60–300 | [84] |
4.2. Industrial Applications
| Industrial Equipment or Phenomenon | Shear Stress (Pa) | References |
|---|---|---|
| Pipeline elbows | 0.009 | [99] |
| Dead ends | 0.05–18.9 | [100] |
| Removal of deposits from stainless steel tubes | 0.09 | [101] |
| Corners of a washing tank | <0.1 | [102] |
| Angles of a washing tank | 0.1–0.4 | [102] |
| Mix proof valve | 0–0.25 | [103] |
| Three-way valve | 0.4–1.7 | [104] |
| Half-open butterfly valve | 0–190 | [100] |
| Product fill valve | 0–1180 | [105] |
| Milk spray dryer | 0–0.4 | [106] |
| Cleaning-in-place pilot plant | 0–5 | [107] |
| Plate heat exchanger for yoghurt processing | 6.7 and 20–46 | [108,109] |
| Plate heat exchanger of an ice slurry system | 50–100 | [110] |
| Pilot-scale plate heat exchanger for milk treatment | 150–450 | [108] |
4.3. Biomedical Applications
| Human Body or Biomedical Device | Shear Stress (Pa) | Shear Strain (s−1) | References |
|---|---|---|---|
| Blood flow in veins | 0.076–3.4 | 20–800 | [117,118] |
| Blood flow in arteries | 0.2–1 | 50–650 | [117,118] |
| Fluid in the oral cavity | n.d. | 0.1–50 | [84] |
| Kidney collecting duct cells | 0.02–2 | n.d. | [119] |
| Uterus | <0.1 | n.d. | [120] |
| Cerebral circulation | n.d. | >100 | [121] |
| Urinary catheter | n.d. | 15 | [84,122] |
| Hemodialysis catheter | 52.6–88.3 | 20,000–80,000 | [123] |
| Catheter sheath introducer | 0.03 | n.d. | [124] |
| Endovascular stent | 0.22–6.72 | n.d. | [125] |
| Prosthetic valve | 0.06–27.84 | n.d. | [126] |
| Contact lens motion | n.d. | 1000 | [127] |
| Platform | Field | Biofilm Stage | Study Aim | Hydrodynamics | Assay Time | Surface Material | Organisms | Concluding Remarks | References |
| Modified Robbins device | General medical devices | Biofilm formation | Effect of flow rate variation on mass transfer and biofilm development | Flow rates of 374 and 242 L h−1, corresponding to shear stresses between 0.183 and 0.511 Pa | 9 days | Polyvinyl chloride | Escherichia coli | Biofilm formation was favored at the lowest flow rate because shear stress effects were more important than mass transfer limitations. This flow cell system generates wall shear stresses that are similar to those found in some biomedical settings. | [111] |
| Urinary devices | Biofilm formation | Evaluation of the potential of antiadhesive coatings when immobilized onto medical-grade polyurethane | Flow rate of 53 mL s−1, corresponding to 15 s−1 | 48 h | Polyurethane Polyurethane coated with CyanoCoating through a polydopamine layer application, or O2- plasma, N2-plasma, and O3 activation | Escherichia coli | When the coating was produced via O3 activation, CyanoCoating was able to decrease the biofilm biovolume by 88% and the surface coverage by 95%, compared to the uncoated surface. | [50] | |
| Investigation of the role of uncommon bacteria on the Escherichia coli microbial consortium | Flow rate of 300 mL min−1, corresponding to 15 s−1 | 72 h | Silicone rubber | Escherichia coli Delftia tsuruhatensis | E. coli and D. tsuruhatensis were able to form single- and dual-species biofilms. Both bacteria tend to co-aggregate and cooperate over time, persisting in a stable microbial community. | [128] | |||
| Development of new functional coatings using magnetron co-sputtering to deposit triple TiO2/SiO2/Ag nanocomposite thin films | Flow rate of 53 mL s−1, corresponding to 15 s−1 | 48 h | Glass TiO2/SiO2 coated glass with different Ag contents (0 to 19.8 at %) | Escherichia coli | Biofilm formation was reduced down to 92% compared to a control glass surface. The coatings are promising candidates for antimicrobial protection of urinary tract devices for at least 48 h, suggesting benefits over longer periods. | [49] | |||
| Flow chamber | General medical devices | Adhesion | Assessment of interactions of bacteria with specific biomaterial surface chemistries under flow conditions | 50, 500, 1000, and 2000 s−1 | 2 h | Glass Glass with alkyl silane monolayers | Staphylococcus epidermidis | The increase in the ionic strength enhanced adhesion to the different surfaces, in accordance with the Derjaguin–Landau–Verwey–Overbeek (DLVO) theory, under low shear rates. The increase in the shear rate restricted the predictability of the theory. | [129] |
| Effect of shear stress on bacterial adhesion to biomedical materials | Flow rates of 2 and 4 mL s−1, corresponding to shear stresses of 0.01 and 0.022 Pa | 0.5 h | Glass Polydimethylsiloxane Poly(L-lactic acid) | Escherichia coli | Similar adhesion rates were obtained on glass and polydimethylsiloxane. The highest adhesion rates were obtained on glass and polydimethylsiloxane, and the lowest on poly(L-lactic acid). | [40,53] | |||
| Effect of fluid composition and shear conditions on bacterial adhesion to an antifouling peptide-coated surface | Flow rates of 2 and 4 mL s−1, corresponding to 15 and 30 s−1 | 0.5 h | Glass Peptide-coated glass Poly(L-lactic acid) | Escherichia coli | Adhesion reductions of 40–50% were attained at a shear rate of 15 s−1 on the peptide-coated surfaces compared with glass. The performance of the peptide-based antifouling coating was superior to poly(L-lactic acid). | [57] | |||
| Effect of shear stress on bacterial adhesion to antifouling polymer brushes | Flow rates of 2 and 4 mL s−1, corresponding to 0.010 and 0.024 Pa | 0.5 h | Glass Poly[N-(2-hydroxypropyl) methacrylamide] brush Poly[oligo(ethyleneglycol) methyl ether methacrylate] brush | Escherichia coli | Both polymer brushes reduced the initial adhesion up to 90% when compared to glass. | [56] | |||
| Evaluate the antiadhesive activity of carbon nanotube composites | Flow rate of 2 mL s−1, corresponding to 15 s−1 | 0.5 h | Polydimethylsiloxane Carbon nanotube/polydimethylsiloxane composites | Escherichia coli | The introduction of carbon nanotubes composites in the polydimethylsiloxane matrix yielded less bacterial adhesion than the polydimethylsiloxane alone. Less adhesion was obtained on the composites with pristine rather than functionalized carbon nanotubes. Incorporation of higher amounts of carbon nanotubes in polymer composites can affect bacterial adhesion by more than 40%. Composites enabling a 60% reduction in cell adhesion were obtained by carbon nanotube treatment by ball-milling. | [23,130] | |||
| Devices and implants | Adhesion | Prevention of microbial adhesion to silicone rubber using polyacrylamide brush coatings | Flow rate of 0.025 mL s−1, corresponding to 10 s−1 | 4 h | Silicone wafers Silicone rubber Polyacrylamide brushes | Staphylococcus aureus Streptococcus salivarius Escherichia coli Candida albicans | A high reduction (52–92%) in microbial adhesion to the polyacrylamide brushes was observed compared to untreated silicon surfaces. The polymer brush did not cause surface deterioration and discouraged microbial adhesion, even after 1-month of exposure to physiological fluids. | [131,132] | |
| Implanted medical devices | Adhesion | Study of adhesion of bacterial and yeast strains to a poly(ethylene oxide) brush covalently attached to the glass | Flow rate of 0.025 mL s−1, corresponding to 10 s−1 | 4 h | Glass Poly(ethylene oxide) brushes on glass | Staphylococcus epidermidis Staphylococcus aureus Streptococcus salivarius Escherichia coli Pseudomonas aeruginosa Candida albicans Candida tropicalis | The poly(ethylene oxide) brush yielded more than 98% reduction in bacterial adhesion, although for the more hydrophobic P. aeruginosa a smaller reduction was observed. For yeast species, adhesion suppression was less effective than for the bacteria. | [133] | |
| Evaluation of the role of surface free energy on bacterial adhesion to plasma-modified films | 50 and 200 s−1 | 2.5 h | Polyethylene terephthalate Plasma treated polyethylene terephthalate | Staphylococcus epidermidis | Plasma treatments reduced bacterial adhesion, in comparison to the untreated polymer. The ageing effect and the subsequent decrease in the surface free energy seemed to favor bacterial adhesion and aggregation. The increase in the shear rate restricted the predictability of the thermodynamic models. | [134] | |||
| Adhesion and biofilm formation | Evaluation of the effectiveness of different formulations of a biomedical-grade polyetherurethane at inhibiting bacterial colonization under flow conditions | 2.03 Pa | Adhesion: 2, 4 and 6 h Biofilm: 8, 20 and 24 h | Polyetherurethane Polyetherurethane with triglyme Polyetherurethane with poly(butyl methyacrylate) barrier membrane releasing ciprofloxacin | Pseudomonas aeruginosa | The rate of adherent cell accumulation was zero for the polyetherurethane with a poly(butyl methyacrylate) barrier membrane releasing ciprofloxacin. | [135] | ||
| Surgical, catheters, and haemodialysis devices | Adhesion | Evaluation of the adhesion behavior of bacterial strains to hydrophilic and hydrophobic surfaces using theoretical predictions | Flow rate of 0.025 mL s−1, corresponding to 6 s−1 | 2 h | Glass Indium tin oxide-coated glass | Pseudomonas stutzeri Staphylococcus epidermidis | P. stutzeri has a much better adhesion rate than S. epidermidis for both material surfaces. Both bacterial strains adhered better to the hydrophobic indium tin oxide-coated glass than to the hydrophilic glass. | [136] | |
| Orthopedic implants | Adhesion | Study the bacterial adhesion to polymers that show promise as orthopedic materials | Flow rate of 1 mL min−1, corresponding to a shear rate of 1.9 s−1 | 1 h | Poly(orthoester) Poly(L-lactic acid) Poly(ether ether ketone) Polysulfone Polyethylene | Staphylococcus epidermidis Pseudomonas aeruginosa Escherichia coli | Tryptic soy broth decreased adhesion to polymers, when compared to phosphate-buffered saline. The estimated values of the free energy of adhesion correlated with the amount of adherent P. aeruginosa. There was 50% more adhesion of E. coli and P. aeruginosa on poly(orthoester) and poly(L-lactic acid) pre-exposed to hyaluronic acid. P. aeruginosa was the most adherent strain, while S. epidermidis was the least adherent strain. | [137] | |
| Urinary devices | Adhesion | Examination of the ability of probiotic strains to displace adhering cells from hydrophobic and hydrophilic substrata | 15 s−1 | 4.5 h | Glass Fluorinated ethylene propylene | Enterococcus faecalis | Ent. faecalis was displaced by lactobacilli (31%) and streptococci (74%) from fluorinated ethylene propylene in buffer, and that displacement by lactobacilli was even more effective on glass in urine (54%). The passage of an air–liquid interface impacted adhesion, especially when the surface had been challenged with lactobacilli (up to 100%) or streptococci (up to 94%). | [138] | |
| Potential of biosurfactant layer to inhibit adhesion of uropathogens | Flow rate of 0.034 mL s−1, corresponding to 15 s−1 | 4 h | Glass Silicone rubber coated with different concentrations of a biosurfactant | Enterococcus faecalis | Biosurfactant layers inhibited the initial deposition rates (> 30%) and adhesion numbers (≈ 70–100%) in a dose-related way.For urine experiments, biosurfactant coatings caused higher adhesion reductions. | [122] | |||
| Effect of supplementation on human urine and uropathogen adhesion | Flow rate of 0.034 mL s−1, corresponding to 15 s−1 | 4 h | Silicone rubber | Escherichia coli Enterococcus faecalis Staphylococcus epidermidis Pseudomonas aeruginosa Candida albicans | Cranberry and ascorbic acid supplementation can provide a degree of protection against adhesion and colonization of biomaterials by some uropathogens. | [139] | |||
| Effect of combined surface chemistry and topography on bacterial adhesion | Flow rates of 2 and 4 mL s−1, corresponding to 0.010 and 0.024 Pa | 0.5 h | Smooth polydimethylsiloxane Smooth polydimethylsiloxane with peptide coating Micropatterned polydimethylsiloxane Micropatterned polydimethylsiloxane with peptide coating | Escherichia coli | The highest adhesion was obtained on the smooth polydimethylsiloxane, whereas the micropatterned polydimethylsiloxane coated with peptide totally inhibited adhesion. The peptide addition to the smooth surface reduced the adhesion by 43–58%, while the micropatterned surface reduced the adhesion by 99%. | [21] | |||
| Biofilm formation | Impact of temperature and surface on the biofilm-forming capacity of uropathogens | Flow rate of 4 × 10−3 mL s−1, corresponding to 33 s−1 | 20–24 h | Silicone Silicone coated with plasma polymerized vinylpyrrolidone | Escherichia coli | Temperature had a considerable influence upon the adhesion and biofilm-forming capacity of some of the isolates, and the influence of surface chemistry also depended on the temperature. | [140] | ||
| Effect of applying different current densities to platinum electrodes as a possible catheter coating material | Flow rate of 3333 mL s−1, corresponding to 200 s−1 | 6 days | Platinum electrodes | Proteus mirabilis | By applying alternating microcurrent densities, a self-regenerative surface is produced, which removed the conditioning film and reduced bacterial adherence, growth, and survival. | [141] | |||
| Biofilm formation and treatment | Potential of using a polymer brush on the prevention of biofilm formation and susceptibility | Flow rate of 2 mL s−1, corresponding to 15 s−1 | Biofilm: 24 h Treatment: 8 h | Glass Polydimethylsiloxane Poly[oligo(ethyleneglycol) methyl ether methacrylate] brush | Escherichia coli | The polymer brush reduced the surface area and the number of total adhered cells by 57%. The antibiotic treatment potentiated cell death and removal (88%). The polymer brush has the potential to prevent biofilm growth and in eradicating biofilms developed in urinary devices. | [59] | ||
| Effect of using a polymer brush on biofilm cell composition and architecture | Flow rate of 2 mL s−1, corresponding to 15 s−1 | Biofilm: 24 h Treatment: 8 h | Glass Polydimethylsiloxane Poly[N-(2-hydroxypropyl) methacrylamide] brush | Escherichia coli | Initial adhesion and surface coverage decreased on the polymer brush. Viable but nonculturable cells were completely removed from the brush. The polymer brush may reduce biofilm growth and antibiotic resistance in urinary catheters. | [58] | |||
| 96-well microplates | Biomedical scenarios | Biofilm formation | Evaluation of the combined effects of shear forces and nutrient levels on biofilm formation and definition of the operational conditions to be used to simulate relevant biomedical scenarios | Orbital shaking with 25 and 50 mm diameter incubators at 150 rpm (average shear rate of 23 and 46 s−1) | 60 h | Polystyrene | Escherichia coli | Higher glucose concentrations enhanced E. coli adhesion in the first 24 h, but variations in peptone and yeast extract concentrations had no significant impact on biofilm formation. Numerical simulations indicate that 96-well microplates can be used to simulate a variety of biomedical scenarios if the operating conditions are carefully set. | [68] |
| Microfluidic device | General medical devices | Adhesion | Development of a fabrication method to produce a microfluidic device to test cell adhesion | 0.01–1 Pa | 0.5 h | Polyamide Polydimethylsiloxane Polyethylene oxide Poly(L-lactic acid) Polystyrene | Escherichia coli | Bacterial adhesion increased linearly over time. The evaluation performed with polydimethylsiloxane for shear stresses between 0.02 and 1 Pa showed that the lowered surface (inherent weakness of the fabrication method) did not influence adhesion. | [113] |
| Study the initial cell adhesion dependence on local wall shear stress in a microchannel with intercalate zones of constrictions and expansions | 0.2–10 Pa | 0.5 h | Glass | Escherichia coli | Bacterial adhesion increased in locations with a sudden increase in shear stress. | [142] | |||
| Examination of the role of surface properties on bacterial adhesion | 0.002–0.042 Pa | n.d. | Smooth silicone Patterned silicone | Escherichia coli | Cell attachment was observed to be strongly dependent upon the topographical features. The highest attachment density was observed on smooth surfaces. | [114] | |||
| Biofilm formation | Comparison of the biofilm-forming capacities of various Methicillin-resistant Staphylococcus aureus clones | 0.05 Pa | 18 h | Glass | Methicillin-resistant Staphylococcus aureus | From tested isolates, 51% successfully formed biofilms under shear flow. Differences in biofilm formation might also be due to the different adherent surfaces. | [143] | ||
| Study of biofilm formation and host–pathogen interactions | 0.05–1 Pa | 24 h | Glass Eukaryotic cells (HRT-18) | Escherichia coli | Biofilm formation on glass was observed for most strains in M9 medium at 30 °C. HRT-18 cell monolayers enhanced E. coli binding and biofilm formation. | [144] | |||
| Implanted medical device | Biofilm formation | Investigation of how environmental factors, such as surface geometry and chemistry, as well as fluid flow, affect biofilm development | 0.02–1 Pa | 16 h | Uncoated and human blood plasma-coated channels | Staphylococcus aureus | The flow was the major contributor to the shape of biofilm structures, whereas bacterial motility was less significant. | [115] | |
| Mammary environment | Biofilm formation | Evaluation of the effect of coagulase-negative staphylococci isolates with a weak- biofilm phenotype | 0.05 Pa | 24 h | Glass | Coagulase-negative staphylococci | Coagulase-negative staphylococci with a weak biofilm phenotype did not inhibit the growth of isolates with a strong-biofilm phenotype. | [145] | |
| Intravascular catheter | Biofilm formation | Investigation of flow as an environmental signal for biofilm formation | Flow rates of 1–10 mL h−1, corresponding to 0.065–1.14 Pa | 24 h | Channels treated with octyl(tri-ethoxy)silane | Staphylococcus epidermidis | Fluid shear alone induced the formation of polysaccharide intracellular adhesin-positive biofilms and influenced the biofilm structure. | [116] | |
| Urinary devices | Adhesion | Development of microfluidic-based devices replicating the urodynamic field within different configurations of an occluded and stented ureter | Up to 0.175 Pa | 1 h | Polydimethylsiloxane | Pseudomonas fluorescens | The unobstructed device showed no bacterial attachment, including in regions of low shear stress (<0.04 Pa). For the obstructed devices, the cavity region, and the nearby proximal side-hole (shear stresses of 0.131–0.175 Pa) exhibited greater levels of bacterial attachment (18%) compared to other regions of the model. | [63] |
5. Current Challenges and Future Directions of Biofilm Platforms Research
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Flemming, H.-C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef]
- Petrova, O.E.; Sauer, K. Sticky Situations: Key Components That Control Bacterial Surface Attachment. J. Bacteriol. 2012, 194, 2413–2425. [Google Scholar] [CrossRef] [PubMed]
- Nikolaev, Y.A.; Plakunov, V.K. Biofilm—“City of microbes” or an analogue of multicellular organisms? Microbiology 2007, 76, 125–138. [Google Scholar] [CrossRef]
- Monroe, D. Looking for Chinks in the Armor of Bacterial Biofilms. PLoS Biol. 2007, 5, e307. [Google Scholar] [CrossRef] [PubMed]
- Stoodley, P.; Sauer, K.; Davies, D.G.; Costerton, J.W. Biofilms as Complex Differentiated Communities. Annu. Rev. Microbiol. 2002, 56, 187–209. [Google Scholar] [PubMed]
- Slate, A.J.; Wickens, D.; Wilson-Nieuwenhuis, J.; Dempsey-Hibbert, N.; West, G.; Kelly, P.; Verran, J.; Banks, C.E.; Whitehead, K.A. The effects of blood conditioning films on the antimicrobial and retention properties of zirconium-nitride silver surfaces. Colloids Surf. B Biointerfaces 2019, 173, 303–311. [Google Scholar] [CrossRef]
- Moreira, J.M.R.; Gomes, L.C.; Whitehead, K.A.; Lynch, S.; Tetlow, L.A.; Mergulhão, F.J. Effect of surface conditioning with cellular extracts on Escherichia coli adhesion and initial biofilm formation. Food Bioprod. Process. 2017, 104, 1–12. [Google Scholar] [CrossRef]
- Renner, L.D.; Weibel, D.B. Physicochemical regulation of biofilm formation. MRS Bull. 2011, 36, 347–355. [Google Scholar] [CrossRef]
- Donlan, R.M. Biofilms: Microbial life on surfaces. Emerg. Infect. Dis. 2002, 8, 881–890. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, C.C.C.R. Marine Biofilms: A Successful Microbial Strategy with Economic Implications. Front. Mar. Sci. 2018, 5. [Google Scholar] [CrossRef]
- Demirel, Y.K.; Turan, O.; Incecik, A. Predicting the effect of biofouling on ship resistance using CFD. Appl. Ocean. Res. 2017, 62, 100–118. [Google Scholar] [CrossRef]
- Bannister, J.; Sievers, M.; Bush, F.; Bloecher, N. Biofouling in marine aquaculture: A review of recent research and developments. Biofouling 2019, 35, 631–648. [Google Scholar] [CrossRef]
- Bott, T.R. Industrial Biofouling. In Industrial Biofouling; Bott, T.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2011; pp. 1–5. [Google Scholar]
- Flemming, H.-C. Microbial Biofouling: Unsolved Problems, Insufficient Approaches, and Possible Solutions. In Biofilm Highlights; Flemming, H.-C., Wingender, J., Szewzyk, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 81–109. [Google Scholar]
- Srey, S.; Jahid, I.K.; Ha, S.-D. Biofilm formation in food industries: A food safety concern. Food Control 2013, 31, 572–585. [Google Scholar] [CrossRef]
- Shunmugaperumal, T. Introduction and overview of biofilm. In Biofilm Eradication and Prevention; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 36–72. [Google Scholar]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. JCMA 2018, 81, 7–11. [Google Scholar] [CrossRef]
- Marschang, S.; Bernardo, G. Prevention and control of healthcare-associated infection in Europe: A review of patients’ perspectives and existing differences. J. Hosp. Infect. 2015, 89, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Kaali, P.; Strömberg, E.; Karlsson, S. Prevention of biofilm associated infections and degradation of polymeric materials used in biomedical applications. In Biomedical Engineering, Trends in Materials Science; Citeseer: Princeton, NJ, USA, 2011. [Google Scholar]
- Dolid, A.; Gomes, L.C.; Mergulhão, F.J.; Reches, M. Combining chemistry and topography to fight biofilm formation: Fabrication of micropatterned surfaces with a peptide-based coating. Colloids Surf. B Biointerfaces 2020, 196, 111365. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.R.; Tulcidas, A.V.; Ferreira, O.; Bayón, R.; Igartua, A.; Mendoza, G.; Mergulhão, F.J.M.; Faria, S.I.; Gomes, L.C.; Carvalho, S.; et al. Assessment of the environmental compatibility and antifouling performance of an innovative biocidal and foul-release multifunctional marine coating. Environ. Res. 2021, 198, 111219. [Google Scholar] [CrossRef]
- Vagos, M.R.; Gomes, M.; Moreira, J.M.R.; Soares, O.S.G.P.; Pereira, M.F.R.; Mergulhão, F.J. Carbon Nanotube/Poly(dimethylsiloxane) Composite Materials to Reduce Bacterial Adhesion. Antibiotics 2020, 9, 434. [Google Scholar]
- Moreira, J.M.R.; Fulgêncio, R.; Alves, P.; Machado, I.; Bialuch, I.; Melo, L.F.; Simões, M.; Mergulhão, F.J. Evaluation of SICAN performance for biofouling mitigation in the food industry. Food Control 2016, 62, 201–207. [Google Scholar] [CrossRef]
- Martinuzzi, R.J.; Salek, M.M. Numerical Simulation of Fluid Flow and Hydrodynamic Analysis in Commonly Used Biomedical Devices in Biofilm Studies. In Numerical Simulations—Examples and Applications in Computational Fluid Dynamics; Angermann, L., Ed.; InTech: London, UK, 2010; pp. 193–212. [Google Scholar]
- Katsikogianni, M.; Missirlis, Y.F. Concise review of mechanisms of bacterial adhesion to biomaterials and of techniques used in estimating bacteria-material interactions. Eur. Cells Mater. 2004, 8, 37–57. [Google Scholar] [CrossRef]
- Gjersing, E.L.; Codd, S.L.; Seymour, J.D.; Stewart, P.S. Magnetic resonance microscopy analysis of advective transport in a biofilm reactor. Biotechnol. Bioeng. 2005, 89, 822–834. [Google Scholar] [CrossRef]
- Liu, Y.; Tay, J.-H. The essential role of hydrodynamic shear force in the formation of biofilm and granular sludge. Water Res. 2002, 36, 1653–1665. [Google Scholar] [CrossRef]
- Stewart, P.S. A review of experimental measurements of effective diffusive permeabilities and effective diffusion coefficients in biofilms. Biotechnol. Bioeng. 1998, 59, 261–272. [Google Scholar] [CrossRef]
- Stewart, P.S. Diffusion in Biofilms. J. Bacteriol. 2003, 185, 1485–1491. [Google Scholar] [CrossRef]
- Munson, B.R.; Young, D.F.; Okiishi, T.H. Fundamentals of Fluid Mechanics, 4th ed.; John Wiley & Sons, Inc.: Chicago, IL, USA, 2002. [Google Scholar]
- Werner, S.; Kaiser, S.C.; Kraume, M.; Eibl, D. Computational fluid dynamics as a modern tool for engineering characterization of bioreactors. Pharm. Bioprocess. 2014, 2, 85–89. [Google Scholar] [CrossRef]
- Ramírez-Muñoz, J.; Guadarrama-Pérez, R.; Alvarado-Lassman, A.; Valencia-López, J.J.; Márquez-Baños, V.E. CFD study of the hydrodynamics and biofilm growth effect of an anaerobic inverse fluidized bed reactor operating in the laminar regime. J. Environ. Chem. Eng. 2021, 9, 104674. [Google Scholar] [CrossRef]
- Sharma, C.; Malhotra, D.; Rathore, A.S. Review of Computational fluid dynamics applications in biotechnology processes. Biotechnol. Prog. 2011, 27, 1497–1510. [Google Scholar] [CrossRef] [PubMed]
- Salek, M.M.; Jones, S.M.; Martinuzzi, R.J. The influence of flow cell geometry related shear stresses on the distribution, structure and susceptibility of Pseudomonas aeruginosa 01 biofilms. Biofouling 2009, 25, 711–725. [Google Scholar] [CrossRef]
- Azeredo, J.; Azevedo, N.F.; Briandet, R.; Cerca, N.; Coenye, T.; Costa, A.R.; Desvaux, M.; Di Bonaventura, G.; Hébraud, M.; Jaglic, Z.; et al. Critical review on biofilm methods. Crit. Rev. Microbiol. 2017, 43, 313–351. [Google Scholar] [CrossRef] [PubMed]
- Ramstedt, M.; Ribeiro, I.A.C.; Bujdakova, H.; Mergulhao, F.J.M.; Jordao, L.; Thomsen, P.; Alm, M.; Burmolle, M.; Vladkova, T.; Can, F.; et al. Evaluating Efficacy of Antimicrobial and Antifouling Materials for Urinary Tract Medical Devices: Challenges and Recommendations. Macromol. Biosci. 2019, 19, e1800384. [Google Scholar] [CrossRef]
- Gomes, I.B.; Simões, M.; Simões, L.C. An overview on the reactors to study drinking water biofilms. Water Res. 2014, 62, 63–87. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Rayner, J.C.; Stoodley, P.; Lappin-Scott, H.M. Establishment of Experimental Biofilms Using the Modified Robbins Device and Flow Cells. In Environmental Monitoring of Bacteria; Edwards, C., Ed.; Humana Press: Totowa, NJ, USA, 1999; pp. 307–319. [Google Scholar]
- Moreira, J.M.R.; Araújo, J.D.P.; Miranda, J.M.; Simões, M.; Melo, L.F.; Mergulhão, F.J. The effects of surface properties on Escherichia coli adhesion are modulated by shear stress. Colloids Surf. B Biointerfaces 2014, 123, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Moreira, J.M.R.; Fulgêncio, R.; Oliveira, F.; Machado, I.; Bialuch, I.; Melo, L.F.; Simões, M.; Mergulhão, F.J. Evaluation of SICON® surfaces for biofouling mitigation in critical process areas. Food Bioprod. Process. 2016, 98, 173–180. [Google Scholar] [CrossRef]
- Teodósio, J.S.; Simões, M.; Melo, L.F.; Mergulhão, F.J. Flow cell hydrodynamics and their effects on E. coli biofilm formation under different nutrient conditions and turbulent flow. Biofouling 2011, 27, 1–11. [Google Scholar] [CrossRef]
- Teodósio, J.S.; Silva, F.C.; Moreira, J.M.R.; Simões, M.; Melo, L.F.; Alves, M.A.; Mergulhão, F.J. Flow cells as quasi-ideal systems for biofouling simulation of industrial piping systems. Biofouling 2013, 29, 953–966. [Google Scholar] [CrossRef] [PubMed]
- McCoy, W.F.; Bryers, J.D.; Robbins, J.; Costerton, J.W. Observations of fouling biofilm formation. Can. J. Microbiol. 1981, 27, 910–917. [Google Scholar] [CrossRef] [PubMed]
- Stoodley, P.; Warwood, B.K. Use of flow cells an annular reactors to study biofilms. In Biofilms in Medicine, Industry and Environmental Biotechnology: Characteristics, Analysis and Control, 1st ed.; Lens, P., O’Flaherty, V., Moran, A.P., Stoodley, P., Mahony, T., Eds.; IWA Publishing: Cornwall, UK, 2003; pp. 197–213. [Google Scholar]
- Gomes, L.C.; Mergulhão, F.J. Heterologous protein production in Escherichia coli biofilms: A non-conventional form of high cell density cultivation. Process. Biochem. 2017, 57, 1–8. [Google Scholar] [CrossRef]
- Pereira, M.O.; Kuehn, M.; Wuertz, S.; Neu, T.; Melo, L.F. Effect of flow regime on the architecture of a Pseudomonas fluorescens biofilm. Biotechnol. Bioeng. 2002, 78, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Teodósio, J.S.; Simões, M.; Alves, M.A.; Melo, L.F.; Mergulhão, F.J. Setup and Validation of Flow Cell Systems for Biofouling Simulation in Industrial Settings. Sci. World J. 2012, 2012, 361496. [Google Scholar] [CrossRef]
- Vladkova, T.; Angelov, O.; Stoyanova, D.; Gospodinova, D.; Gomes, L.C.; Soares, A.; Mergulhao, F.J.; Ivanova, I. Magnetron co-sputtered TiO2/SiO2/Ag nanocomposite thin coatings inhibiting bacterial adhesion and biofilm formation. Surf. Coat. Technol. 2020, 384, 125322. [Google Scholar] [CrossRef]
- Matinha-Cardoso, J.; Mota, R.; Gomes, L.C.; Gomes, M.; Mergulhão, F.J.; Tamagnini, P.; Martins, M.C.L.; Costa, F. Surface activation of medical grade polyurethane for the covalent immobilization of an anti-adhesive biopolymeric coating. J. Mater. Chem. B 2021, 9, 3705–3715. [Google Scholar] [CrossRef] [PubMed]
- Zou, F.; Bai, L. Using time-lapse fluorescence microscopy to study gene regulation. Methods 2019, 159–160, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Bos, R.; van der Mei, H.C.; Busscher, H.J. Physico-chemistry of initial microbial adhesive interactions—Its mechanisms and methods for study. FEMS Microbiol. Rev. 1999, 23, 179–230. [Google Scholar] [CrossRef]
- Moreira, J.M.R.; Ponmozhi, J.; Campos, J.B.L.M.; Miranda, J.M.; Mergulhão, F.J. Micro- and macro-flow systems to study Escherichia coli adhesion to biomedical materials. Chem. Eng. Sci. 2015, 126, 440–445. [Google Scholar] [CrossRef]
- Alves, P.; Moreira, J.M.; Miranda, J.M.; Mergulhão, F.J. Analysing the Initial Bacterial Adhesion to Evaluate the Performance of Antifouling Surfaces. Antibiotics 2020, 9, 421. [Google Scholar] [CrossRef]
- Busscher, H.J.; van der Mei, H.C. Microbial adhesion in flow displacement systems. Clin. Microbiol. Rev. 2006, 19, 127–141. [Google Scholar] [CrossRef]
- Lopez-Mila, B.; Alves, P.; Riedel, T.; Dittrich, B.; Mergulhão, F.; Rodriguez-Emmenegger, C. Effect of shear stress on the reduction of bacterial adhesion to antifouling polymers. Bioinspir. Biomim. 2018, 13, 065001. [Google Scholar] [CrossRef]
- Alves, P.; Nir, S.; Reches, M.; Mergulhão, F. The effects of fluid composition and shear conditions on bacterial adhesion to an antifouling peptide-coated surface. MRS Commun. 2018, 8, 938–946. [Google Scholar] [CrossRef]
- Alves, P.; Gomes, L.C.; Vorobii, M.; Rodriguez-Emmenegger, C.; Mergulhão, F.J. The potential advantages of using a poly(HPMA) brush in urinary catheters: Effects on biofilm cells and architecture. Colloids Surf. B Biointerfaces 2020, 191, 110976. [Google Scholar] [CrossRef]
- Alves, P.; Gomes, L.C.; Rodríguez-Emmenegger, C.; Mergulhão, F.J. Efficacy of A Poly(MeOEGMA) Brush on the Prevention of Escherichia coli Biofilm Formation and Susceptibility. Antibiotics 2020, 9, 216. [Google Scholar] [CrossRef]
- Cotter, J.J.; O’Gara, J.P.; Stewart, P.S.; Pitts, B.; Casey, E. Characterization of a modified rotating disk reactor for the cultivation of Staphylococcus epidermidis biofilm. J. Appl. Microbiol. 2010, 109, 2105–2117. [Google Scholar] [CrossRef]
- Willcock, L.; Gilbert, P.; Holah, J.; Wirtanen, G.; Allison, D.G. A new technique for the performance evaluation of clean-in-place disinfection of biofilms. J. Ind. Microbiol. Biotechnol. 2000, 25, 235–241. [Google Scholar] [CrossRef]
- Kim, J.; Park, H.D.; Chung, S. Microfluidic approaches to bacterial biofilm formation. Molecules 2012, 17, 9818–9834. [Google Scholar] [CrossRef]
- De Grazia, A.; LuTheryn, G.; Meghdadi, A.; Mosayyebi, A.; Espinosa-Ortiz, E.J.; Gerlach, R.; Carugo, D. A Microfluidic-Based Investigation of Bacterial Attachment in Ureteral Stents. Micromachines 2020, 11, 408. [Google Scholar] [CrossRef]
- Yawata, Y.; Nguyen, J.; Stocker, R.; Rusconi, R. Microfluidic Studies of Biofilm Formation in Dynamic Environments. J. Bacteriol. 2016, 198, 2589–2595. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wippold, J.A.; Stratis-Cullum, D.; Han, A. Eliminating air bubble in microfluidic systems utilizing integrated in-line sloped microstructures. Biomed. Microdevices 2020, 22, 76. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Wittmann, C.; Heinzle, E. Minibioreactors. Biotechnol. Lett. 2004, 26, 1–10. [Google Scholar] [CrossRef]
- Moreira, J.M.R.; Gomes, L.C.; Araújo, J.D.P.; Miranda, J.M.; Simões, M.; Melo, L.F.; Mergulhão, F.J. The effect of glucose concentration and shaking conditions on Escherichia coli biofilm formation in microtiter plates. Chem. Eng. Sci. 2013, 94, 192–199. [Google Scholar] [CrossRef]
- Gomes, L.C.; Moreira, J.M.R.; Teodósio, J.S.; Araújo, J.D.P.; Miranda, J.M.; Simões, M.; Melo, L.F.; Mergulhão, F.J. 96-well microtiter plates for biofouling simulation in biomedical settings. Biofouling 2014, 30, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Stepanovic, S.; Vukovic, D.; Hola, V.; Di Bonaventura, G.; Djukic, S.; Cirkovic, I.; Ruzicka, F. Quantification of biofilm in microtiter plates: Overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS Acta Pathol. Microbiol. Immunol. Scand. 2007, 115, 891–899. [Google Scholar] [CrossRef]
- Faria, S.I.; Teixeira-Santos, R.; Romeu, M.J.; Morais, J.; Jong, E.D.; Sjollema, J.; Vasconcelos, V.; Mergulhão, F.J. Unveiling the Antifouling Performance of Different Marine Surfaces and Their Effect on the Development and Structure of Cyanobacterial Biofilms. Microorganisms 2021, 9, 1102. [Google Scholar] [CrossRef]
- Romeu, M.J.; Alves, P.; Morais, J.; Miranda, J.M.; de Jong, E.D.; Sjollema, J.; Ramos, V.; Vasconcelos, V.; Mergulhão, F.J.M. Biofilm formation behaviour of marine filamentous cyanobacterial strains in controlled hydrodynamic conditions. Environ. Microbiol. 2019, 21, 4411–4424. [Google Scholar] [CrossRef] [PubMed]
- Bridier, A.; Dubois-Brissonnet, F.; Boubetra, A.; Thomas, V.; Briandet, R. The biofilm architecture of sixty opportunistic pathogens deciphered using a high throughput CLSM method. J. Microbiol. Methods 2010, 82, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Moreira, J.M.R.; Gomes, L.C.; Simões, M.; Melo, L.F.; Mergulhão, F.J. The impact of material properties, nutrient load and shear stress on biofouling in food industries. Food Bioprod. Process. 2015, 95, 228–236. [Google Scholar] [CrossRef]
- Gomes, M.; Gomes, L.C.; Teixeira-Santos, R.; Pereira, M.F.R.; Soares, O.S.G.P.; Mergulhão, F.J. Optimizing CNT Loading in Antimicrobial Composites for Urinary Tract Application. Appl. Sci. 2021, 11, 4038. [Google Scholar] [CrossRef]
- Gomes, L.C.; Moreira, J.M.; Miranda, J.M.; Simões, M.; Melo, L.F.; Mergulhão, F.J. Macroscale versus microscale methods for physiological analysis of biofilms formed in 96-well microtiter plates. J. Microbiol. Methods 2013, 95, 342–349. [Google Scholar] [CrossRef]
- Gomes, L.C.; Moreira, J.M.R.; Simões, M.; Melo, L.F.; Mergulhão, F.J. Biofilm Localization in the Vertical Wall of Shaking 96-Well Plates. Scientifica 2014, 2014, 6. [Google Scholar] [CrossRef]
- Gomes, L.C.; Moreira, J.M.R.; Araújo, J.D.P.; Mergulhão, F.J. Surface conditioning with Escherichia coli cell wall components can reduce biofilm formation by decreasing initial adhesion. AIMS Microbiol. 2017, 3, 613–628. [Google Scholar] [CrossRef]
- Gomes, L.C.; Miranda, J.; Mergulhão, F.J. Operation of Biofilm Reactors for the Food Industry Using CFD. In Computational Fluid Dynamics in Food Processing, 2nd ed.; Sun, D.-W., Ed.; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Cowle, M.W.; Webster, G.; Babatunde, A.O.; Bockelmann-Evans, B.N.; Weightman, A.J. Impact of flow hydrodynamics and pipe material properties on biofilm development within drinking water systems. Environ. Technol. 2020, 41, 3732–3744. [Google Scholar] [CrossRef]
- Gomes, I.B.; Lemos, M.; Mathieu, L.; Simões, M.; Simões, L.C. The action of chemical and mechanical stresses on single and dual species biofilm removal of drinking water bacteria. Sci. Total Environ. 2018, 631–632, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, O.; Rijo, P.; Gomes, J.; Santos, R.; Monteiro, S.; Guedes, R.; Serralheiro, M.L.; Gomes, M.; Gomes, L.C.; Mergulhão, F.J.; et al. Antimicrobial Ceramic Filters for Water Bio-Decontamination. Coatings 2021, 11, 323. [Google Scholar] [CrossRef]
- Faria, S.I.; Teixeira-Santos, R.; Gomes, L.C.; Silva, E.R.; Morais, J.; Vasconcelos, V.; Mergulhão, F.J.M. Experimental Assessment of the Performance of Two Marine Coatings to Curb Biofilm Formation of Microfoulers. Coatings 2020, 10, 893. [Google Scholar] [CrossRef]
- Mathieu, L.; Bertrand, I.; Abe, Y.; Angel, E.; Block, J.C.; Skali-Lami, S.; Francius, G. Drinking water biofilm cohesiveness changes under chlorination or hydrodynamic stress. Water Res. 2014, 55, 175–184. [Google Scholar] [CrossRef]
- Bakker, D.P.; Plaats, A.v.d.; Verkerke, G.J.; Busscher, H.J.; Mei, H.C.v.d. Comparison of Velocity Profiles for Different Flow Chamber Designs Used in Studies of Microbial Adhesion to Surfaces. Appl. Environ. Microbiol. 2003, 69, 6280–6287. [Google Scholar] [CrossRef]
- Liu, C.; Zhao, Q. The CQ ratio of surface energy components influences adhesion and removal of fouling bacteria. Biofouling 2011, 27, 275–285. [Google Scholar] [CrossRef]
- Liu, C.; Zhao, Q. Influence of Surface-Energy Components of Ni–P–TiO2–PTFE Nanocomposite Coatings on Bacterial Adhesion. Langmuir 2011, 27, 9512–9519. [Google Scholar] [CrossRef]
- Gomes, I.B.; Simões, L.C.; Simões, M. Influence of surface copper content on Stenotrophomonas maltophilia biofilm control using chlorine and mechanical stress. Biofouling 2020, 36, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, N.F.; Pinto, A.R.; Reis, N.M.; Vieira, M.J.; Keevil, C.W. Shear stress, temperature, and inoculation concentration influence the adhesion of water-stressed Helicobacter pylori to stainless steel 304 and polypropylene. Appl. Environ. Microbiol. 2006, 72, 2936–2941. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Romeu, M.J.; Alves, P.; Morais, J.; Ramos, V.; Vasconcelos, V.; Mergulhão, F.J. Biofilm formation by a cyanobacterial strain belonging to a distinct Leptolyngbya phylotype: Surface effect. In Biofilms: Characterization, Applications and Recent Advances; Henderson, J., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2016; pp. 153–174. [Google Scholar]
- Faria, S.I.; Teixeira-Santos, R.; Romeu, M.J.; Morais, J.; Vasconcelos, V.; Mergulhão, F.J. The Relative Importance of Shear Forces and Surface Hydrophobicity on Biofilm Formation by Coccoid Cyanobacteria. Polymers 2020, 12, 653. [Google Scholar] [CrossRef] [PubMed]
- Romeu, M.J.L.; Domínguez-Pérez, D.; Almeida, D.; Morais, J.; Campos, A.; Vasconcelos, V.; Mergulhão, F.J.M. Characterization of planktonic and biofilm cells from two filamentous cyanobacteria using a shotgun proteomic approach. Biofouling 2020, 36, 631–645. [Google Scholar] [CrossRef] [PubMed]
- Faria, S.I.; Teixeira-Santos, R.; Morais, J.; Vasconcelos, V.; Mergulhão, F.J. The association between initial adhesion and cyanobacterial biofilm development. FEMS Microbiol. Ecol. 2021, 97, fiab052. [Google Scholar] [CrossRef] [PubMed]
- Romeu, M.J.; Domínguez-Pérez, D.; Almeida, D.; Morais, J.; Araújo, M.J.; Osório, H.; Campos, A.; Vasconcelos, V.; Mergulhão, F.J. Quantitative proteomic analysis of marine biofilms formed by filamentous cyanobacterium. Environ. Res. 2021, 201, 111566. [Google Scholar] [CrossRef]
- Perrin, A.; Herbelin, P.; Jorand, F.P.A.; Skali-Lami, S.; Mathieu, L. Design of a rotating disk reactor to assess the colonization of biofilms by free-living amoebae under high shear rates. Biofouling 2018, 34, 368–377. [Google Scholar] [CrossRef]
- Lemos, M.; Mergulhão, F.; Melo, L.; Simões, M. The effect of shear stress on the formation and removal of Bacillus cereus biofilms. Food Bioprod. Process. 2015, 93, 242–248. [Google Scholar] [CrossRef]
- Brugnoni, L.I.; Cubitto, M.A.; Lozano, J.E. Role of shear stress on biofilm formation of Candida krusei in a rotating disk system. J. Food Eng. 2011, 102, 266–271. [Google Scholar] [CrossRef]
- Szlavik, J.; Paiva, D.S.; Mørk, N.; van den Berg, F.; Verran, J.; Whitehead, K.; Knøchel, S.; Nielsen, D.S. Initial adhesion of Listeria monocytogenes to solid surfaces under liquid flow. Int. J. Food Microbiol. 2012, 152, 181–188. [Google Scholar] [CrossRef]
- Skovager, A.; Whitehead, K.; Siegumfeldt, H.; Ingmer, H.; Verran, J.; Arneborg, N. Influence of flow direction and flow rate on the initial adhesion of seven Listeria monocytogenes strains to fine polished stainless steel. Int. J. Food Microbiol. 2012, 157, 174–181. [Google Scholar] [CrossRef]
- Piepiórka-Stepuk, J.; Mierzejewska, S. Simulation tests of liquid flow in the pipeline elements. Agric. Eng. 2014, 1, 175–184. [Google Scholar]
- Jensen, B.B.B.; Friis, A. CFD Prediction of Hygiene in Food Processing Equipment. In Computational Fluid Dynamics in Food Processing; Sun, D.-W., Ed.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Bergman, B.-O.; Tragardh, C. An approach to study and model the hydrodynamic cleaning effect. J. Food Process. Eng. 1990, 13, 135–154. [Google Scholar] [CrossRef]
- Cunault, C.; Faille, C.; Bouvier, L.; Föste, H.; Augustin, W.; Scholl, S.; Debreyne, P.; Benezech, T. A novel set-up and a CFD approach to study the biofilm dynamics as a function of local flow conditions encountered in fresh-cut food processing equipments. Food Bioprod. Process. 2015, 93, 217–223. [Google Scholar] [CrossRef]
- Jensen, B.B.B.; Stenby, M.; Nielsen, D.F. Improving the cleaning effect by changing average velocity. Trends Food Sci. Technol. 2007, 18 (Suppl. 1), S58–S63. [Google Scholar] [CrossRef]
- Lelièvre, C.; Legentilhomme, P.; Legrand, J.; Faille, C.; Bénézech, T. Hygienic Design: Influence of the Local Wall Shear Stress Variations on the Cleanability of a Three-Way Valve. Chem. Eng. Res. Des. 2003, 81, 1071–1076. [Google Scholar] [CrossRef]
- Rahaman, M.F.; Bari, S.; Veale, D. Flow investigation of the product fill valve of filling machine for packaging liquid products. J. Food Eng. 2008, 85, 252–258. [Google Scholar] [CrossRef]
- Jin, Y.; Chen, X.D. A fundamental model of particle deposition incorporated in CFD simulations of an industrial milk spray dryer. Dry. Technol. 2010, 28, 960–971. [Google Scholar] [CrossRef]
- Lelièvre, C.; Legentilhomme, P.; Gaucher, C.; Legrand, J.; Faille, C.; Bénézech, T. Cleaning in place: Effect of local wall shear stress variation on bacterial removal from stainless steel equipment. Chem. Eng. Sci. 2002, 57, 1287–1297. [Google Scholar] [CrossRef]
- Afonso, I.M.; Hes, L.; Maia, J.M.; Melo, L.F. Heat transfer and rheology of stirred yoghurt during cooling in plate heat exchangers. J. Food Eng. 2003, 57, 179–187. [Google Scholar] [CrossRef][Green Version]
- Fernandes, C.S.; Dias, R.; Nóbrega, J.M.; Afonso, I.M.; Melo, L.F.; Maia, J.M. Simulation of stirred yoghurt processing in plate heat exchangers. J. Food Eng. 2005, 69, 281–290. [Google Scholar] [CrossRef]
- Nørgaard, E.; Sørensen, T.A.; Hansen, T.M.; Kauffeld, M. Performance of components of ice slurry systems: Pumps, plate heat exchangers, and fittings. Int. J. Refrig. 2005, 28, 83–91. [Google Scholar] [CrossRef]
- Moreira, J.M.R.; Teodósio, J.S.; Silva, F.C.; Simões, M.; Melo, L.F.; Mergulhão, F.J. Influence of flow rate variation on the development of Escherichia coli biofilms. Bioprocess. Biosyst. Eng. 2013, 36, 1787–1796. [Google Scholar] [CrossRef]
- Moreira, J.M.R.; Simões, M.; Melo, L.F.; Mergulhão, F.J. The combined effects of shear stress and mass transfer on the balance between biofilm and suspended cell dynamics. Desalination Water Treat. 2015, 53, 3348–3354. [Google Scholar] [CrossRef]
- Ponmozhi, J.; Moreira, J.M.R.; Mergulhão, F.J.; Campos, J.B.L.M.; Miranda, J.M. Fabrication and Hydrodynamic Characterization of a Microfluidic Device for Cell Adhesion Tests in Polymeric Surfaces. Micromachines 2019, 10, 303. [Google Scholar] [CrossRef]
- Graham, M.V.; Mosier, A.P.; Kiehl, T.R.; Kaloyeros, A.E.; Cady, N.C. Development of antifouling surfaces to reduce bacterial attachment. Soft Matter 2013, 9, 6235–6244. [Google Scholar] [CrossRef]
- Kim, M.K.; Drescher, K.; Pak, O.S.; Bassler, B.L.; Stone, H.A. Filaments in curved streamlines: Rapid formation of Staphylococcus aureus biofilm streamers. New J. Phys. 2014, 16, 065024. [Google Scholar]
- Weaver, W.M.; Milisavljevic, V.; Miller, J.F.; Di Carlo, D. Fluid flow induces biofilm formation in Staphylococcus epidermidis polysaccharide intracellular adhesin-positive clinical isolates. Appl. Environ. Microbiol. 2012, 78, 5890–5896. [Google Scholar] [CrossRef]
- Inauen, W.; Baumgartner, H.R.; Bombeli, T.; Haeberli, A.; Straub, P.W. Dose- and shear rate-dependent effects of heparin on thrombogenesis induced by rabbit aorta subendothelium exposed to flowing human blood. Arterioscler. Off. J. Am. Heart Assoc. Inc. 1990, 10, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Michelson, A. Platelets, 2nd ed.; Academic Press: New York, NY, USA, 2002. [Google Scholar]
- Cai, Z.; Xin, J.; Pollock, D.M.; Pollock, J.S. Shear stress-mediated NO production in inner medullary collecting duct cells. Am. J. Physiology. Ren. Physiol. 2000, 279, F270–F274. [Google Scholar] [CrossRef]
- Nauman, E.A.; Ott, C.M.; Sander, E.; Tucker, D.L.; Pierson, D.; Wilson, J.W.; Nickerson, C.A. Novel Quantitative Biosystem for Modeling Physiological Fluid Shear Stress on Cells. Appl. Environ. Microbiol. 2007, 73, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.K.; Marzo, A.; Howard, B.; Rufenacht, D.A.; Bijlenga, P.; Frangi, A.F.; Lawford, P.V.; Coley, S.C.; Hose, D.R.; Patel, U.J. Effects of smoking and hypertension on wall shear stress and oscillatory shear index at the site of intracranial aneurysm formation. Clin. Neurol. Neurosurg. 2010, 112, 306–313. [Google Scholar] [CrossRef]
- Velraeds, M.M.C.; Van Der Mei, H.C.; Reid, G.; Busscher, H.J. Inhibition of initial adhesion of uropathogenic Enterococcus faecalis to solid substrata by an adsorbed biosurfactant layer from Lactobacillus acidophilus. Urology 1997, 49, 790–794. [Google Scholar] [CrossRef]
- Mareels, G.; De Wachter, D.S.; Verdonck, P.R. Computational fluid dynamics-analysis of the Niagara hemodialysis catheter in a right heart model. Artif. Organs 2004, 28, 639–648. [Google Scholar] [CrossRef]
- Frumento, R.J.; Hirsh, A.L.; Parides, M.K.; Bennett-Guerrero, E. Differences in arterial and venous thromboelastography parameters: Potential roles of shear stress and oxygen content. J. Cardiothorac. Vasc. Anesth. 2002, 16, 551–554. [Google Scholar] [CrossRef]
- Nicoud, F.; Vernhet, H.; Dauzat, M. A numerical assessment of wall shear stress changes after endovascular stenting. J. Biomech. 2005, 38, 2019–2027. [Google Scholar] [CrossRef] [PubMed]
- Morsi, Y.; Kogure, M.; Umezu, M. Relative blood damage index of the jellyfish valve and the Bjork-Shiley tilting-disk valve. J. Artif. Organs 1999, 2, 163–169. [Google Scholar] [CrossRef]
- Tran, V.B.; Fleiszig, S.M.J.; Evans, D.J.; Radke, C.J. Dynamics of flagellum- and pilus-mediated association of Pseudomonas aeruginosa with contact lens surfaces. Appl. Environ. Microbiol. 2011, 77, 3644–3652. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, A.S.; Almeida, C.; Gomes, L.C.; Ferreira, C.; Mergulhão, F.J.; Melo, L.F.; Azevedo, N.F. An in vitro model of catheter-associated urinary tract infections to investigate the role of uncommon bacteria on the Escherichia coli microbial consortium. Biochem. Eng. J. 2017, 118, 64–69. [Google Scholar] [CrossRef][Green Version]
- Katsikogianni, M.G.; Missirlis, Y.F. Interactions of bacteria with specific biomaterial surface chemistries under flow conditions. Acta Biomater. 2010, 6, 1107–1118. [Google Scholar] [CrossRef]
- Vagos, M.R.; Moreira, J.M.R.; Soares, O.S.G.P.; Pereira, M.F.R.; Mergulhão, F.J. Incorporation of carbon nanotubes in polydimethylsiloxane to control Escherichia coli adhesion. Polym. Compos. 2019, 40, E1697–E1704. [Google Scholar] [CrossRef]
- Cringus-Fundeanu, I.; Luijten, J.; van der Mei, H.C.; Busscher, H.J.; Schouten, A.J. Synthesis and Characterization of Surface-Grafted Polyacrylamide Brushes and Their Inhibition of Microbial Adhesion. Langmuir 2007, 23, 5120–5126. [Google Scholar] [CrossRef] [PubMed]
- Fundeanu, I.; van der Mei, H.C.; Schouten, A.J.; Busscher, H.J. Polyacrylamide brush coatings preventing microbial adhesion to silicone rubber. Colloids Surf. B Biointerfaces 2008, 64, 297–301. [Google Scholar] [CrossRef]
- Roosjen, A.; Kaper, H.J.; van der Mei, H.C.; Norde, W.; Busscher, H.J. Inhibition of adhesion of yeasts and bacteria by poly(ethylene oxide)-brushes on glass in a parallel plate flow chamber. Microbiology 2003, 149, 3239–3246. [Google Scholar] [CrossRef]
- Katsikogianni, M.; Amanatides, E.; Mataras, D.; Missirlis, Y.F. Staphylococcus epidermidis adhesion to He, He/O2 plasma treated PET films and aged materials: Contributions of surface free energy and shear rate. Colloids Surf. B Biointerfaces 2008, 65, 257–268. [Google Scholar] [CrossRef]
- Hendricks, S.K.; Kwok, C.; Shen, M.; Horbett, T.A.; Ratner, B.D.; Bryers, J.D. Plasma-deposited membranes for controlled release of antibiotic to prevent bacterial adhesion and biofilm formation. J. Biomed. Mater. Res. 2000, 50, 160–170. [Google Scholar] [CrossRef]
- Bayoudh, S.; Othmane, A.; Mora, L.; Ben Ouada, H. Assessing bacterial adhesion using DLVO and XDLVO theories and the jet impingement technique. Colloids Surf. B Biointerfaces 2009, 73, 1–9. [Google Scholar] [CrossRef]
- Barton, A.J.; Sagers, R.D.; Pitt, W.G. Bacterial adhesion to orthopedic implant polymers. J. Biomed. Mater. Res. 1996, 30, 403–410. [Google Scholar] [CrossRef]
- Millsap, K.; Reid, G.; van der Mei, H.C.; Busscher, H.J. Displacement of Enterococcus faecalis from hydrophobic and hydrophilic substrata by Lactobacillus and Streptococcus spp. as studied in a parallel plate flow chamber. Appl. Environ. Microbiol. 1994, 60, 1867–1874. [Google Scholar] [CrossRef] [PubMed]
- Habash, M.B.; Mei, H.C.V.d.; Busscher, H.J.; Reid, G. The effect of water, ascorbic acid, and cranberry derived supplementation on human urine and uropathogen adhesion to silicone rubber. Can. J. Microbiol. 1999, 45, 691–694. [Google Scholar] [CrossRef]
- Andersen, T.E.; Kingshott, P.; Palarasah, Y.; Benter, M.; Alei, M.; Kolmos, H.J. A flow chamber assay for quantitative evaluation of bacterial surface colonization used to investigate the influence of temperature and surface hydrophilicity on the biofilm forming capacity of uropathogenic Escherichia coli. J. Microbiol. Methods 2010, 81, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Gabi, M.; Hefermehl, L.; Lukic, D.; Zahn, R.; Vörös, J.; Eberli, D. Electrical microcurrent to prevent conditioning film and bacterial adhesion to urological stents. Urol. Res. 2011, 39, 81–88. [Google Scholar] [CrossRef]
- Neves, S.F.; Ponmozhi, J.; Mergulhão, F.J.; Campos, J.B.L.M.; Miranda, J.M. Cell adhesion in microchannel multiple constrictions—Evidence of mass transport limitations. Colloids Surf. B Biointerfaces 2020, 198, 111490. [Google Scholar] [CrossRef]
- Vanhommerig, E.; Moons, P.; Pirici, D.; Lammens, C.; Hernalsteens, J.-P.; De Greve, H.; Kumar-Singh, S.; Goossens, H.; Malhotra-Kumar, S. Comparison of Biofilm Formation between Major Clonal Lineages of Methicillin Resistant Staphylococcus aureus. PLoS ONE 2014, 9, e104561. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, Y.D.; Vogeleer, P.; Jacques, M.; Harel, J. High-throughput microfluidic method to study biofilm formation and host-pathogen interactions in pathogenic Escherichia coli. Appl. Environ. 2015, 81, 2827–2840. [Google Scholar] [CrossRef]
- Goetz, C.; Tremblay, Y.D.N.; Lamarche, D.; Blondeau, A.; Gaudreau, A.M.; Labrie, J.; Malouin, F.; Jacques, M. Coagulase-negative staphylococci species affect biofilm formation of other coagulase-negative and coagulase-positive staphylococci. J. Dairy Sci. 2017, 100, 6454–6464. [Google Scholar] [CrossRef] [PubMed]
- Brackman, G.; Coenye, T. In Vitro and In Vivo Biofilm Wound Models and Their Application. Adv. Exp. Med. Biol. 2016, 897, 15–32. [Google Scholar]
- Goeres, D.M.; Walker, D.K.; Buckingham-Meyer, K.; Lorenz, L.; Summers, J.; Fritz, B.; Goveia, D.; Dickerman, G.; Schultz, J.; Parker, A.E. Development, standardization, and validation of a biofilm efficacy test: The single tube method. J. Microbiol. Methods 2019, 165, 105694. [Google Scholar] [CrossRef]
- Johnson, E.; Petersen, T.; Goeres, D.M. Characterizing the Shearing Stresses within the CDC Biofilm Reactor Using Computational Fluid Dynamics. Microorganisms 2021, 9, 1709. [Google Scholar] [CrossRef]
- Wu, B. Advances in the use of CFD to characterize, design and optimize bioenergy systems. Comput. Electron. Agric. 2013, 93, 195–208. [Google Scholar] [CrossRef]
- Benoit, M.R.; Conant, C.G.; Ionescu-Zanetti, C.; Schwartz, M.; Matin, A. New device for high-throughput viability screening of flow biofilms. Appl. Environ. Microbiol. 2010, 76, 4136–4142. [Google Scholar] [CrossRef]
- Straub, H.; Eberl, L.; Zinn, M.; Rossi, R.M.; Maniura-Weber, K.; Ren, Q. A microfluidic platform for in situ investigation of biofilm formation and its treatment under controlled conditions. J. Nanobiotechnol. 2020, 18, 166. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Sun, K.; Abulimiti, A.; Xu, P.P.; Li, Z.Y. Microfluidic System for Observation of Bacterial Culture and Effects on Biofilm Formation at Microscale. Micromachines 2019, 10, 606. [Google Scholar] [CrossRef]
- Blanco-Cabra, N.; López-Martínez, M.J.; Arévalo-Jaimes, B.V.; Martin-Gómez, M.T.; Samitier, J.; Torrents, E. A new BiofilmChip device for testing biofilm formation and antibiotic susceptibility. NPJ Biofilms Microbiomes 2021, 7, 62. [Google Scholar] [CrossRef]

| Platform | Advantages | Disadvantages |
|---|---|---|
| Modified Robbins device | Large amount of biomass is produced | Complex setup |
| High/moderate throughput | Entry effects | |
| Allows periodical sampling | Sampling can affect the biofilm | |
| Can be run for very long periods without intervention | Limited in situ biofilm visualization | |
| Large dynamic range | Biofilm destruction for most quantitative analysis | |
| Flow chamber | Optimized for online in situ microscopy | Low throughput |
| Allows direct and nondestructive observation of biofilm development | Inability to study adhesion to nontransparent surfaces | |
| Complex setup | ||
| Rotating biofilm devices | Possibility to study different materials in the same run | The flow pattern changes in the boundaries of the coupons |
| Shear stress and feed flow rate can be set independently | Lack of sampling surface area | |
| Easy to control the operational conditions | Complex setup | |
| Expensive | ||
| Microplates | High-throughput analysis | Direct observation under the microscope can be difficult |
| Simple to run | Batch system | |
| Needs small space | Loosely attached biofilm may not be correctly quantified | |
| Inexpensive | Operator dependent | |
| Microfluidic devices | Noninvasive technique | Requires special equipment for manufacturing and running systems |
| Allows real-time visualization of biofilm development | Clogging can occur due to small dimensions | |
| Requires small volumes | Laborious operation | |
| Can be custom made for specific purposes | Air bubbles may be an issue | |
| Rapid and precise analysis | Viscosity effects may arise | |
| Compatible with single-cell analysis |
| Platform | Field | Biofilm Stage | Study Aim | Hydrodynamics | Assay Time | Surface Material | Organisms | Concluding Remarks | References |
| Modified Robbins device | Drinking- water distribution systems | Biofilm formation | Investigate the combined impact of flow hydrodynamics and pipe material | 0.13 and 0.24 Pa | 100 days | Polyvinyl chloride Polypropylene Structured wall high-density polyethylene Solid wall high-density polyethylene | Natural flora present in drinking water | The biomass amount was greater for the biofilms formed at lower shear stress. The opportunistic pathogens have limited ability to propagate within biofilms under high shear conditions without protection (surface roughness). | [79] |
| Water treatment | Biofilm formation | Evaluate the application of non-biocide release coatings as coated filters for biofouling prevention | Flow rate of 300 L h−1, corresponding to an average shear stress of 0.25 Pa | 2 days | Polyurethane coating Polyurethane coating with incorporated Econea Polyurethane coating with grafted Econea | Enterococcus faecalis | Biocidal polyurethane-based surfaces were less prone to biofilm formation, with an average reduction of 60%, compared to pristine polyurethane. | [81] | |
| Flow chamber | Man-made equipment (heat exchangers, ship hulls, and pipelines) | Biofilm formation | Study the influence of surface energy components on the adhesion and removal of fouling | 9.8 × 10−4, 4.6 × 10−4, and 2.1 × 10−4 Pa | 10 days | 316 L Stainless steel Ni–P–TiO2–polytetrafluoroethylene nanocomposite coatings | Pseudomonas fluorescences Cobetia marina Vibrio alginolyticus | Coatings with the lowest ratio between the Lifshitz van der Waals apolar component and the electron donor component had the lowest bacterial adhesion or the highest bacterial removal. | [85,86] |
| Rotating cylinder reactor | Drinking- water distribution systems | Biofilm formation and treatment | Effect of chemical and mechanical stresses on single and dual- species biofilm removal | Biofilm formation: 1 Pa Treatment: 1–23 Pa | 7 days | Polyvinyl chloride | Acinectobacter calcoaceticus Stenotrophomonas maltophilia | Dual species biofilms were the most susceptible to chemical and mechanical removal. Stenotrophomonas maltophilia biofilms demonstrated high tolerance to chemical and mechanical stress. | [80] |
| Biofilm formation | Action of copper materials on biofilm formation and control by chemical and mechanical stress | 0.1 Pa | 7 days | Stainless steel Copper alloys (100, 96, and 57%) | Stenotrophomonas maltophilia | Chemical, mechanical, and combined shocks were not effective in biofilm control. Copper surfaces were found to reduce the number of non-damaged cells. | [87] | ||
| 6-well microplates | Drinking- water distribution systems | Adhesion and biofilm formation | Influence of shear stress, temperature, and inoculation concentration on water-stressed Helicobacter pylori | 0, 60, and 120 rpm corresponding to 0, 0.138, and 0.317 Pa | 2, 6, 12, 24, 48, 96, and 192 h | 304 stainless steel Polypropylene | Helicobacter pylori | High shear stresses negatively influenced the adhesion to the substrata. However, the temperature and inoculation concentration appeared to not affect adhesion. | [88] |
| 12-well microplates | Marine environment | Biofilm formation | Effect of surface hydrophobicity on biofilm development by a filamentous cyanobacterium | Orbital shaking with a 25 mm diameter incubator at 185 rpm (average shear stress of 0.07 Pa) | 3 weeks | Glass Perspex | Leptolyngbya mycoidea LEGE 06118 | Higher biofilm growth was observed on perspex, the most hydrophobic surface. | [89] |
| Effect of different marine coatings on biofilm formation by microfoulers | Orbital shaking with a 25 mm diameter incubator at 185 rpm (average shear rate of 40 s−1) | 7 weeks | Epoxy-coated glass Silicone hydrogel coating | Cyanobium sp. LEGE 10375 Pseudoalteromonas tunicata (marine bacterium) | Epoxy-coated surface was effective in inhibiting biofilm formation at the initial stages, while silicone coating showed high antibiofilm efficacy during maturation. Silicone coating was less prone to biofilm formation. The efficacy of silicone may be dependent on the organism, while the performance of epoxy-coated surface was strongly influenced by a combined effect of surface and microorganism. | [82] | |||
| Effect of different materials on biofilm structure | 7 weeks | Glass Perspex Polystyrene Epoxy-coated glass Silicone hydrogel coating | Synechocystis salina LEGE 00041 Cyanobium sp. LEGE 06098 Cyanobium sp. LEGE 10375 | Silicone coating was effective in inhibiting cyanobacterial biofilm formation. Cyanobacterial biofilms formed on silicone coating showed a lower percentage and size of empty spaces among all surfaces. | [70] | ||||
| Study the environmental compatibility of an innovative biocidal foul-release multifunctional coating | 7 weeks | Polydimethylsiloxane Polydimethylsiloxane coating with grafted Econea | Pseudoalteromonas tunicata | Polydimethylsiloxane coating with grafted Econea was more effective in inhibiting biofilm formation than the bare polydimethylsiloxane (reductions of 77%, 60%, and 73% on biovolume, thickness, and substratum coverage, respectively). Long-lasting antifouling performances were observed in simulated and real scenarios. | [22] | ||||
| Effect of shear forces on biofilm development by filamentous cyanobacteria | Orbital shaking with a 25 mm diameter incubator at 40 rpm (average shear rate of 4 s−1) and 185 rpm (average shear rate of 40 s−1) | 7 weeks | Glass Perspex | Nodosilinea sp. LEGE 06020 Nodosilinea sp. LEGE 06022 Unidentified filamentous Synechococcales LEGE 07185 | Biofilm formation was higher under low shear conditions. The hydrodynamics was more effective on biofilm maturation than during initial cell adhesion. Different shear rates affected biofilm architecture. | [71] | |||
| Effect of shear forces and surface hydrophobicity on biofilm development by coccoid cyanobacteria with different biofilm formation capacities | 6 weeks | Glass Epoxy-coated glass | Synechocystis salina LEGE 00041 Cyanobium sp. LEGE 06097 | Biofilms developed in both surfaces at lower shear conditions had a higher number of cells, wet weight, thickness, and chlorophyll a content. The impact of hydrodynamics was generally stronger than the impact of surface hydrophobicity. The antibiofilm performance of the polymeric coating was confirmed. | [90] | ||||
| Qualitative proteomic analyses of filamentous cyanobacterial biofilms formed under different shear rates | 7 weeks | Glass Perspex | Nodosilinea sp. LEGE 06145 Nodosilinea sp. LEGE 0611 | Biofilm formation was higher under low shear conditions. Biofilm development of Nodosilinea sp. LEGE 06145 was higher than LEGE 06119, but no significant differences were found between surfaces. | [91] | ||||
| Adhesion and biofilm formation | Potential of adhesion assays on the estimation of biofilm development behavior at different hydrodynamic conditions | Adhesion: 7.5 h Biofilm: 6 weeks | Glass Epoxy-coated glass | Synechocystis salina LEGE 00041 Synechocystis salina LEGE 06155 Cyanobium sp. LEGE 06097 | For both adhesion and biofilm assays, the number of adhered cells was higher under low shear conditions. Higher biofilm development at 4 s−1 was confirmed by biofilm wet weight, thickness, and chlorophyll a content. Initial adhesion assays can be used to estimate marine biofilm development. | [92] | |||
| Quantitative proteomic analyses of biofilms formed on different surfaces | 7 weeks | Glass Perspex | Unidentified filamentous cyanobacterium LEGE 06007 | After 7 weeks, high biofilm thickness was observed in biofilms formed at 4 s−1 on glass when compared to perspex. Differences in protein expression were more noticeable in biofilms formed under low shear conditions. Proteomic analysis revealed differentially expressed proteins between surfaces. | [93] |
| Platform | Biofilm Stage | Study Aim | Hydrodynamics | Assay Time | Surface Material | Organisms | Concluding Remarks | References |
| Modified Robbins device | Biofilm formation | Effect of flow rate/shear stress variation on mass transfer and biofilm development in a flow cell that mimics industrial piping | Flow rates of 374 and 242 L h−1, corresponding to wall shear stresses between 0.183 and 0.511 Pa | 9 days | Polyvinyl chloride | Escherichia coli | Biofilm formation was favored at the lowest flow rate. Shear stress effects were more important than mass transfer limitations. This flow cell system generates wall shear stresses that are similar to those found in some industrial settings. | [111,112] |
| Biofilm formation and treatment | Evaluation of a modified diamond-like carbon surface for biofouling mitigation in critical process areas | Flow rate of 300 L h−1, corresponding to an average shear stress of 0.25 Pa | Biofilm formation: 5 days Treatment: 6, 18, and 24 h | 316 L Stainless steel SICON® | Escherichia coli Natural flora present in the water from an industrial salad washing line | Biofilm formation was reduced on SICON® (1–2 Log). Biofilm cleaning with chlorine was more efficient when SICON® was used (3.5-Log reduction and 15% removal). Industries with cleaning frequencies up to 6 h may benefit from the use of SICON®. | [41] | |
| Biofilm formation and treatment | Evaluation of SICAN for biofouling mitigation in the food industry | Biofilm formation: 5 days Treatment: 6, 18 and 24 h | 316 L Stainless Steel SICAN | Escherichia coli Natural flora present in the water from an industrial salad washing line | Biofilm formation on SICAN and stainless steel were similar. Processes with cleaning intervals of about 6 h could potentially use SICAN surfaces on critical areas. | [24] | ||
| Flow chamber | Adhesion | Effect of strain, shear stress, surface soiling, and growth conditions on Listeria monocytogenes adhesion | Flow rates of 0.76 and 10.9 mL min−1, corresponding to wall shear stresses of 0.0505 and 0.7620 Pa | 30 min | Glass Polyvinyl chloride Glass coated with beef extract, casein, and milk | Listeria monocytogenes | Strain differences influenced the initial adhesion rate to all the surfaces at both low and high shear stress. There was a significant effect of the surfaces on the adhesion ability of almost all strains. The initial adhesion rate decreased at high shear stress for most strains. | [97] |
| Effect of flow direction and flow rate on the initial adhesion of Listeria monocytogenes strains | Flow rates of 0.75 and 8.40 mL min−1, corresponding to wall shear stresses of 0.10 and 1.20 Pa | 15 min | Fine polished stainless steel | Listeria monocytogenes | Initial adhesion rates were influenced by flow rate and strain specificity. The flow direction, in relation to the orientation of surface features, could be disregarded. | [98] | ||
| Biofilm formation | Effect of surface conditioning on adhesion and biofilm formation under conditions that are prevalent in the food industry | Flow rate of 11 mL s−1, corresponding to an average shear stress of 0.07 Pa | 24 h | Polystyrene Polystyrene conditioned with cell extracts and cell wall components | Escherichia coli | Under flow conditions, all conditioning films reduced biofilm formation, except mannose. Surface conditioning affected the amount and clustering of bacteria on surfaces. | [8,77] | |
| Rotating cylinder reactor | Biofilm formation and treatment | Effect of shear stress on the formation and removal of biofilms | 0.02, 0.12, and 0.17 Pa | Biofilm formation: 7 days Treatment: 0.5 h | AISI 316 Stainless steel | Bacillus cereus | Biofilm density increased with the shear stress, while the thickness decreased. The biocide treatment promoted the higher removal of biofilms formed under higher shear stress. Biofilms formed under higher shear stress were more resistant to the mechanical and combined biocide and mechanical treatments. | [95] |
| Rotating disk reactor | Biofilm formation | Effect of shear stress on biofilm formation | Rotational speeds of 350 and 800 rpm, corresponding to shear stresses between 0 and 91 Pa | 4 days | AISI 304 2B food grade stainless steel | Candida krusei | The early development of a biofilm (24 h) was unaffected by shear stress. In a mature biofilm, shear stress determined the disposition of biofilm cells onto the surface. Biofilms formed under higher shear stress differ in their arrangement, as compared with those formed under lower shear conditions. | [96] |
| Assessment of the colonization of biofilms by free-living amoebae | Shear rates between 31,000 and 85,000 s−1, representative of cooling circuits | 10 days | Stainless steel | Freshwater containing free-living amoebae and bacteria | Free-living amoebae were able to establish in biofilms under shear rate as high as 85,000 s−1. The developed reactor seems to be ideal for studying the effects of high shear stress on surface colonization by microorganisms. | [94] | ||
| 96-well microplates | Adhesion | Effect of surface conditioning on adhesion and biofilm formation under conditions that are prevalent in the food industry | Orbital shaking with a 50 mm diameter incubator at 150 rpm (average shear stress of 0.07 Pa) | 1 h | Polystyrene Polystyrene conditioned with cell extracts and cell wall components | Escherichia coli | Total cell extract, cytoplasm with cellular debris, myristic, and palmitic acid decreased initial adhesion. Adhesion increased when periplasmic extract was used. Adhesion was dependent on the conditioning film concentration. | [8,77] |
| 6-well microplates | Adhesion | Evaluation of the antiadhesive activity of SICON® | Orbital shaking with a 25 mm diameter incubator (average shear stress of 0.25 Pa) | 0.5, 2, and 6 h | 316 L Stainless steel SICON® | Escherichia coli Natural flora present in the water from an industrial salad washing line | Bacterial adhesion on SICON® and stainless steel were similar. | [41] |
| Adhesion | Evaluation of the antiadhesive activity of SICAN | Orbital shaking with a 25 mm diameter incubator (average shear stress of 0.25 Pa) | 0.5, 2, and 6 h | 316 L Stainless steel SICAN | Escherichia coli Natural flora present in the water from an industrial salad washing line | Adhesion on SICAN and stainless steel were similar. Escherichia coli and the flora from industrial water had similar adhesion behaviour. | [24] | |
| Adhesion and biofilm formation | Assessment of the impact of material properties, nutrient load, and shear stress on biofouling in food industries | Static and orbital shaking with a 25 mm diameter incubator at 115 rpm (average shear stress of 0.27 Pa) | Adhesion: 0.5 h Biofilm: 6 h | Glass Copper Stainless steel | Escherichia coli | Surface material was the most important factor in adhesion and biofilm formation. Adhesion and biofilm formation were correlated with surface hydrophobicity. The effect of surface properties was dependent on the nutrient load and shear stress. Initial adhesion performance may be a good predictor for biofilm formation. | [73] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomes, L.C.; Mergulhão, F.J.M. A Selection of Platforms to Evaluate Surface Adhesion and Biofilm Formation in Controlled Hydrodynamic Conditions. Microorganisms 2021, 9, 1993. https://doi.org/10.3390/microorganisms9091993
Gomes LC, Mergulhão FJM. A Selection of Platforms to Evaluate Surface Adhesion and Biofilm Formation in Controlled Hydrodynamic Conditions. Microorganisms. 2021; 9(9):1993. https://doi.org/10.3390/microorganisms9091993
Chicago/Turabian StyleGomes, Luciana C., and Filipe J. M. Mergulhão. 2021. "A Selection of Platforms to Evaluate Surface Adhesion and Biofilm Formation in Controlled Hydrodynamic Conditions" Microorganisms 9, no. 9: 1993. https://doi.org/10.3390/microorganisms9091993
APA StyleGomes, L. C., & Mergulhão, F. J. M. (2021). A Selection of Platforms to Evaluate Surface Adhesion and Biofilm Formation in Controlled Hydrodynamic Conditions. Microorganisms, 9(9), 1993. https://doi.org/10.3390/microorganisms9091993






