Lactobacillus johnsonii L531 Alleviates the Damage Caused by Salmonella Typhimurium via Inhibiting TLR4, NF-κB, and NLRP3 Inflammasome Signaling Pathways
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Antibodies
2.2. Bacterial Strains and Growth Conditions
2.3. Cell Culture
2.4. Western Blotting
2.5. Immunofluorescence
2.6. Real-Time Quantitative PCR
2.7. Statistical Analysis
3. Results
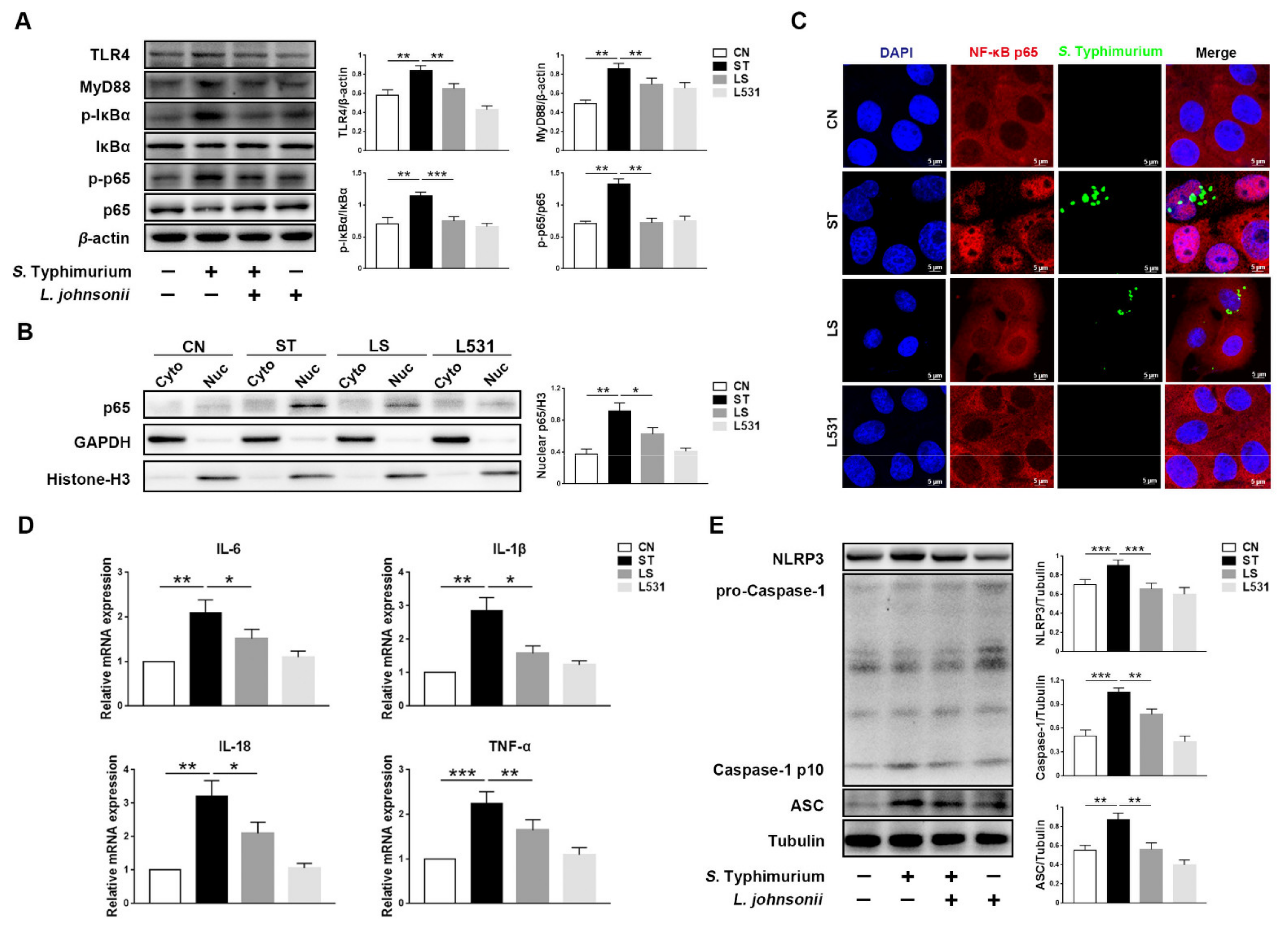
3.1. L. johnsonii L531 Attenuates S. Typhimurium-Induced Inflammatory Response in IPEC-J2 Cells
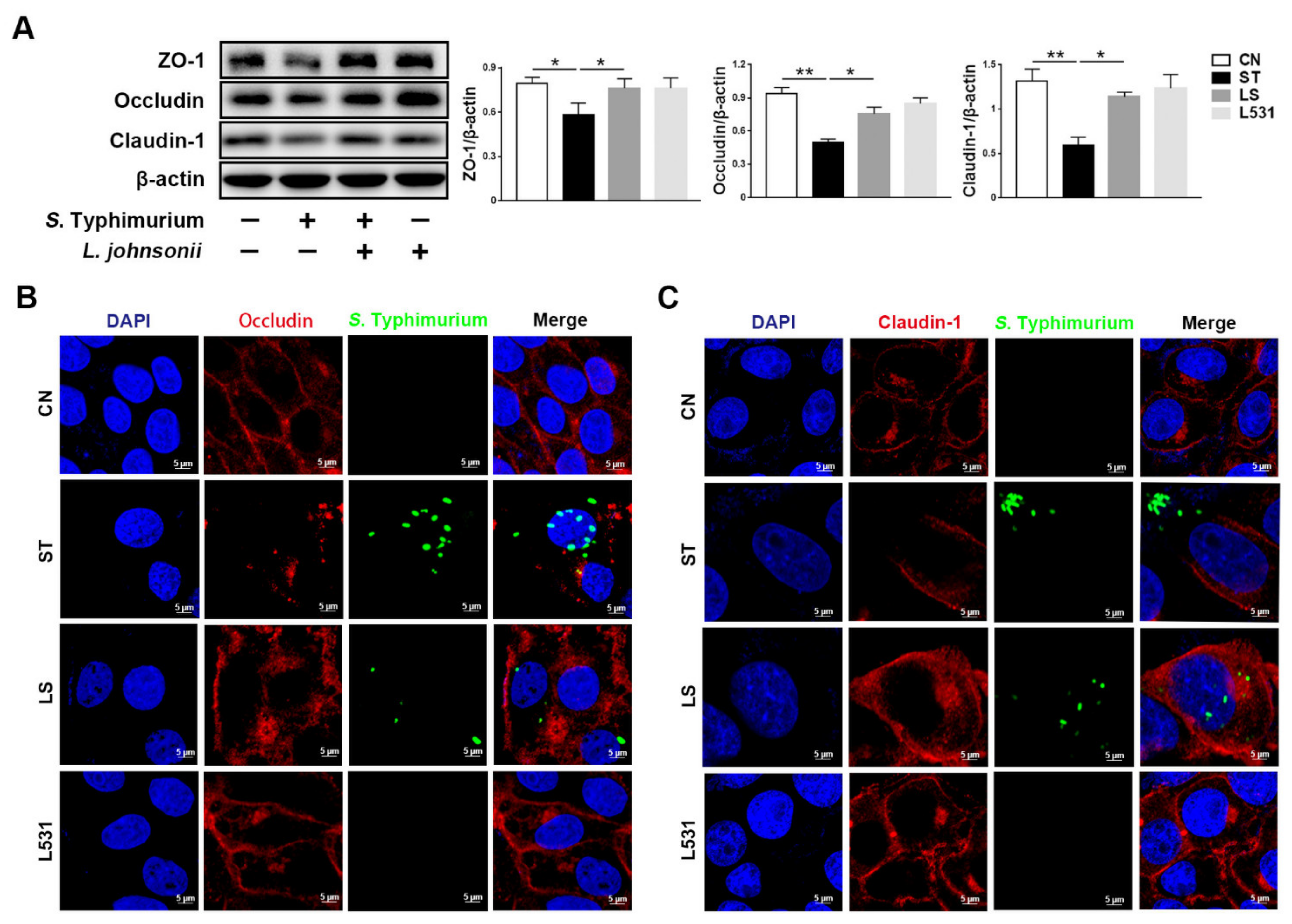
3.2. L. johnsonii L531 Ameliorates S. Typhimurium-Induced Damage of Tight Junctions in IPEC-J2 Cells
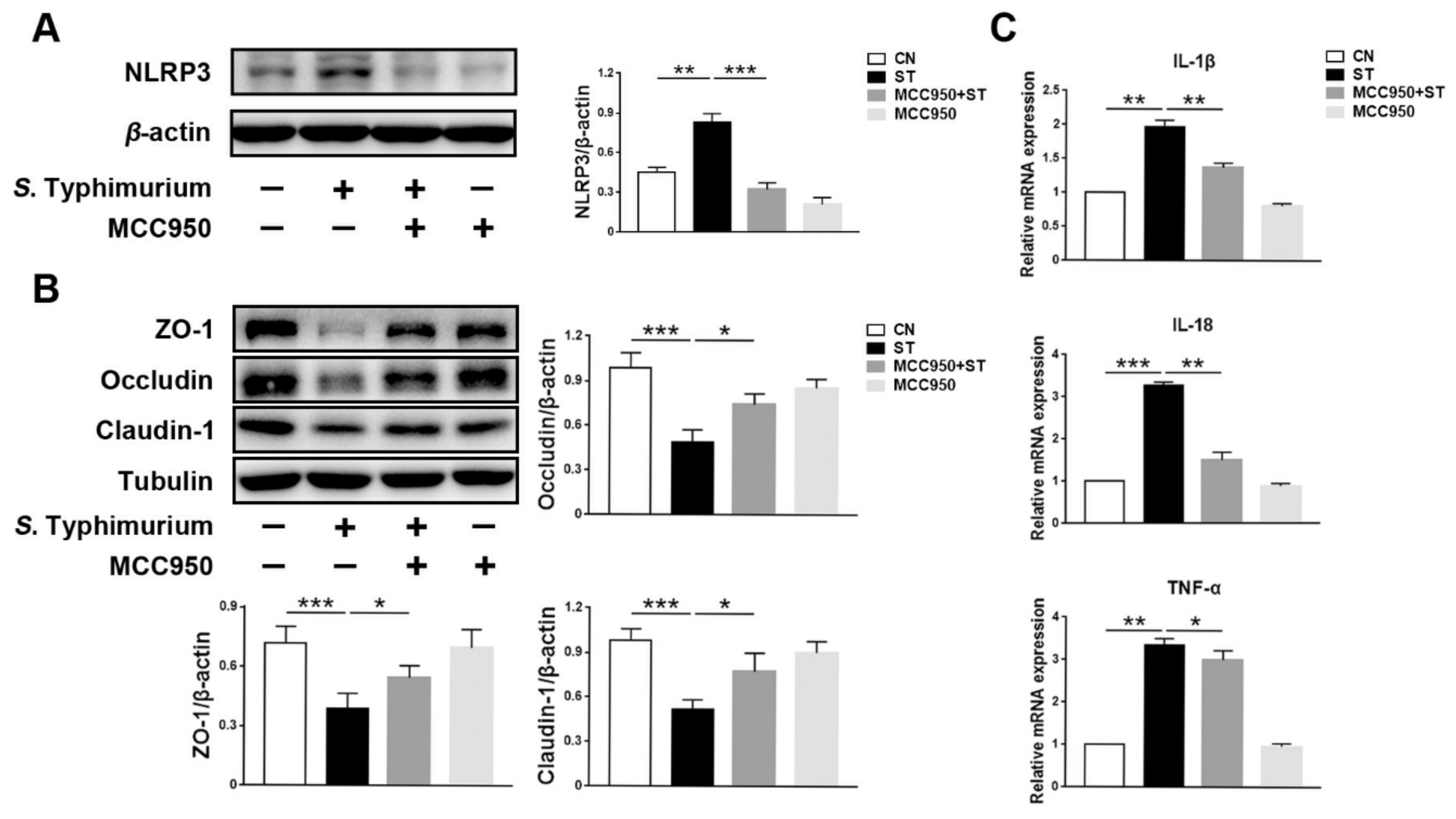
3.3. L. johnsonii L531 Inhibits S. Typhimurium-Induced Assembly of the NLRP3 Inflammasome to Maintain Tight Junctions
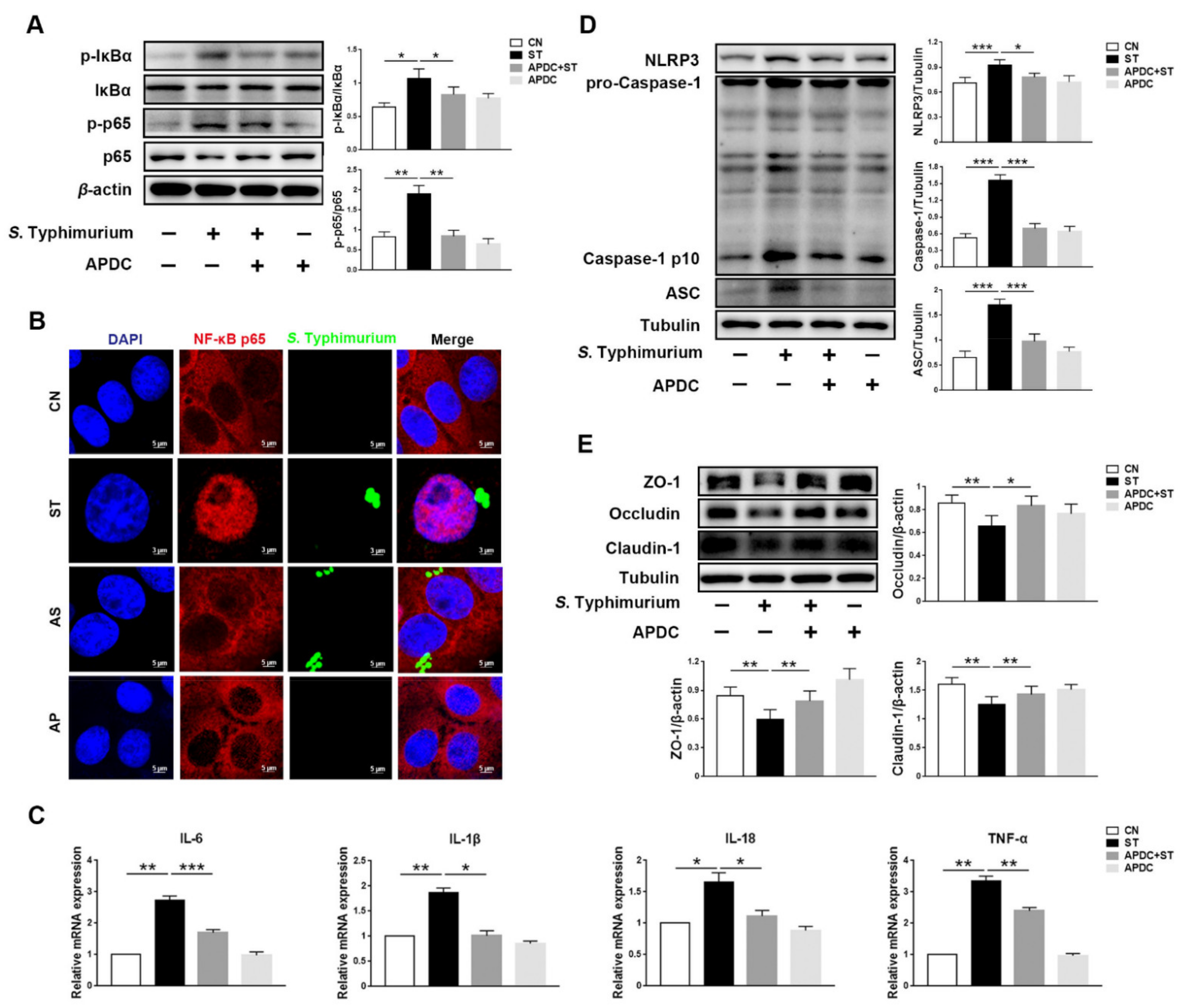
3.4. L. johnsonii L531 Inhibits S. Typhimurium-Induced Activation of NF-κB Pathway to Maintain Tight Junctions
3.5. L. johnsonii L531 Inhibits S. Typhimurium-Induced Activation of TLR4 Pathway to Maintain Tight Junctions
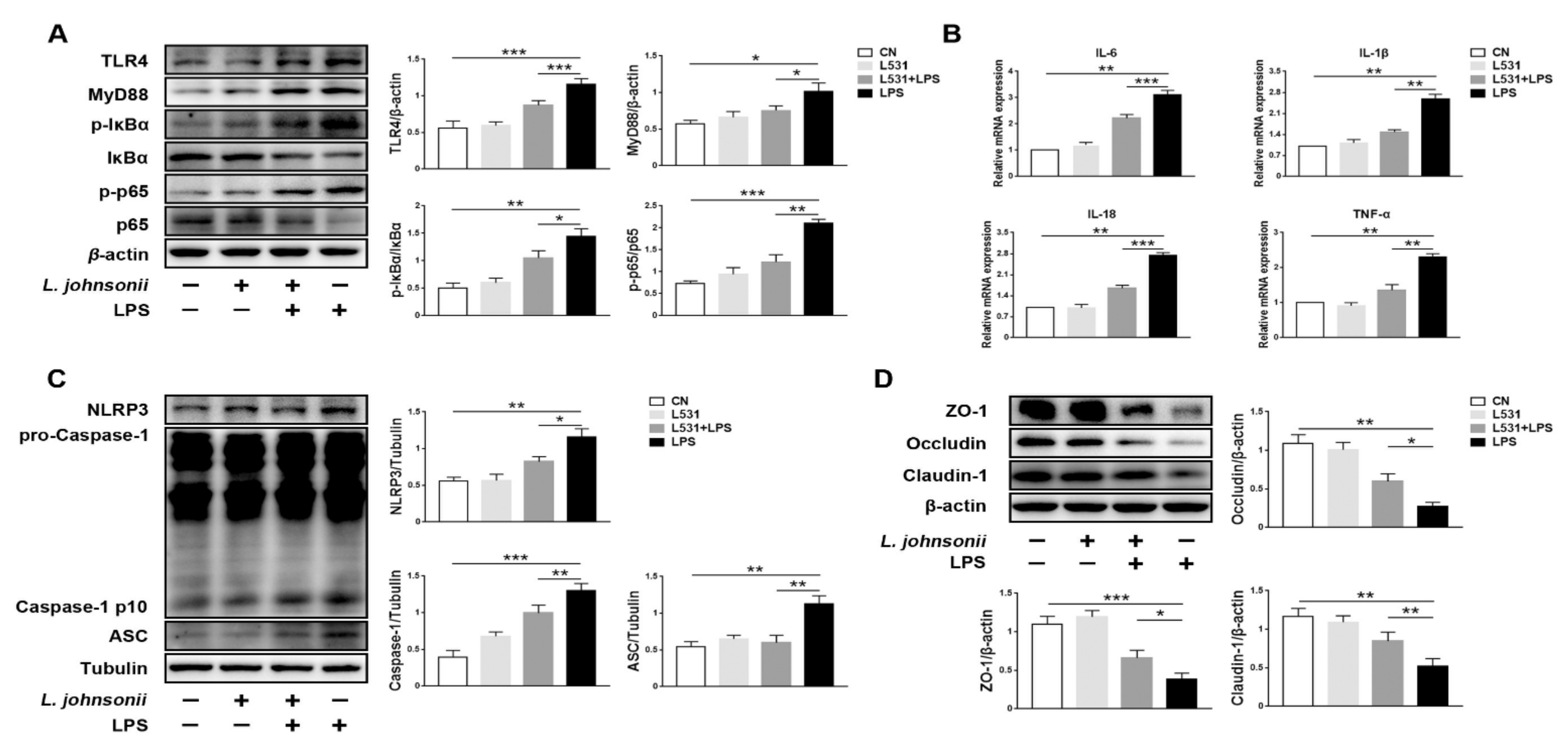
3.6. L. johnsonii L531 Alleviates LPS-Induced Inflammatory Response in IPEC-J2 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ferrari, R.G.; Rosario, D.K.A.; Cunha-Neto, A.; Mano, S.B.; Figueiredo, E.E.S.; Conte-Junior, C.A. Worldwide Epidemiology of Salmonella Serovars in Animal-Based Foods: A Meta-analysis. Appl. Environ. Microbiol. 2019, 85, e00591–e00619. [Google Scholar] [CrossRef] [Green Version]
- Patterson, S.K.; Kim, H.B.; Borewicz, K.; Isaacson, R.E. Towards an understanding of Salmonella enterica serovar Typhimurium persistence in swine. Anim. Health Res. Rev. 2016, 17, 159–168. [Google Scholar] [CrossRef]
- Gut, A.M.; Vasiljevic, T.; Yeager, T.; Donkor, O.N. Salmonella infection-prevention and treatment by antibiotics and probiotic yeasts: A review. Microbiology 2018, 164, 1327–1344. [Google Scholar] [CrossRef]
- Su, J.H.; Zhu, Y.H.; Ren, T.Y.; Guo, L.; Yang, G.Y.; Jiao, L.G.; Wang, J.F. Distribution and Antimicrobial Resistance of Salmonella Isolated from Pigs with Diarrhea in China. Microorganisms 2018, 6, 117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allen, S.J.; Martinez, E.G.; Gregorio, G.V.; Dans, L.F. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst. Rev. 2010, 11, CD003048. [Google Scholar] [CrossRef] [PubMed]
- Bereswill, S.; Ekmekciu, I.; Escher, U.; Fiebiger, U.; Stingl, K.; Heimesaat, M.M. Lactobacillus johnsonii ameliorates intestinal, extra-intestinal and systemic pro-inflammatory immune responses following murine Campylobacter jejuni infection. Sci. Rep. 2017, 7, 2138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, G.Y.; Xia, B.; Su, J.H.; He, T.; Liu, X.; Guo, L.; Zhang, S.; Zhu, Y.H.; Wang, J.F. Anti-inflammatory effects of Lactobacillus johnsonii L531 in a pig model of Salmonella Infantis infection involves modulation of CCR6(+) T cell responses and ER stress. Vet. Res. 2020, 51, 26. [Google Scholar] [CrossRef] [Green Version]
- Slifer, Z.M.; Blikslager, A.T. The Integral Role of Tight Junction Proteins in the Repair of Injured Intestinal Epithelium. Int. J. Mol. Sci. 2020, 21, 972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takeuchi, O.; Akira, S. Pattern recognition receptors and inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef] [Green Version]
- Bruno, V.M.; Hannemann, S.; Lara-Tejero, M.; Flavell, R.A.; Kleinstein, S.H.; Galan, J.E. Salmonella Typhimurium type III secretion effectors stimulate innate immune responses in cultured epithelial cells. PLoS Pathog. 2009, 5, e1000538. [Google Scholar] [CrossRef] [Green Version]
- He, Y.; Hara, H.; Nunez, G. Mechanism and Regulation of NLRP3 Inflammasome Activation. Trends Biochem. Sci. 2016, 41, 1012–1021. [Google Scholar] [CrossRef] [Green Version]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [Green Version]
- Dong, N.; Xue, C.; Zhang, L.; Zhang, T.; Wang, C.; Bi, C.; Shan, A. Oleanolic acid enhances tight junctions and ameliorates inflammation in Salmonella Typhimurium-induced diarrhea in mice via the TLR4/NF-kappaB and MAPK pathway. Food Funct. 2020, 11, 1122–1132. [Google Scholar] [CrossRef]
- He, T.; Zhu, Y.; Yu, J.; Xia, B.; Liu, X.; Yang, G.; Su, J.; Guo, L.; Wang, M.; Wang, J. Lactobacillus johnsonii L531 reduces pathogen load and helps maintain short-chain fatty acid levels in the intestines of pigs challenged with Salmonella enterica Infantis. Vet. Microbiol. 2019, 230, 187–194. [Google Scholar] [CrossRef]
- Chu, B.; Zhu, Y.; Su, J.; Xia, B.; Zou, Y.; Nie, J.; Zhang, W.; Wang, J. Butyrate-mediated autophagy inhibition limits cytosolic Salmonella Infantis replication in the colon of pigs treated with a mixture of Lactobacillus and Bacillus. Vet. Res. 2020, 51, 99. [Google Scholar] [CrossRef]
- Winter, S.E.; Thiennimitr, P.; Winter, M.G.; Butler, B.P.; Huseby, D.L.; Crawford, R.W.; Russell, J.M.; Bevins, C.L.; Adams, L.G.; Tsolis, R.M.; et al. Gut inflammation provides a respiratory electron acceptor for Salmonella. Nature 2010, 467, 426–429. [Google Scholar] [CrossRef] [PubMed]
- Thiennimitr, P.; Winter, S.E.; Winter, M.G.; Xavier, M.N.; Tolstikov, V.; Huseby, D.L.; Sterzenbach, T.; Tsolis, R.M.; Roth, J.R.; Baumler, A.J. Intestinal inflammation allows Salmonella to use ethanolamine to compete with the microbiota. Proc. Natl. Acad. Sci. USA 2011, 108, 17480–17485. [Google Scholar] [CrossRef] [Green Version]
- Taguchi, T.; Mukai, K. Innate immunity signalling and membrane trafficking. Curr. Opin. Cell Biol. 2019, 59, 1–7. [Google Scholar] [CrossRef]
- Galan, J.E. Salmonella Typhimurium and inflammation: A pathogen-centric affair. Nat. Rev. Microbiol. 2021. online ahead of print. [Google Scholar] [CrossRef]
- Wen, L.; Duffy, A. Factors Influencing the Gut Microbiota, Inflammation, and Type 2 Diabetes. J. Nutr. 2017, 147, 1468S–1475S. [Google Scholar] [CrossRef] [Green Version]
- Ulluwishewa, D.; Anderson, R.C.; McNabb, W.C.; Moughan, P.J.; Wells, J.M.; Roy, N.C. Regulation of tight junction permeability by intestinal bacteria and dietary components. J. Nutr. 2011, 141, 769–776. [Google Scholar] [CrossRef] [Green Version]
- Paradis, T.; Begue, H.; Basmaciyan, L.; Dalle, F.; Bon, F. Tight Junctions as a Key for Pathogens Invasion in Intestinal Epithelial Cells. Int. J. Mol. Sci. 2021, 22, 2506. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wu, Y.; Ye, L.; Wang, Y.; Zhang, K.; Wang, L.; Huang, Y.; Wang, L.; Xian, S.; Zhang, Y.; et al. Aspirin alleviates endothelial gap junction dysfunction through inhibition of NLRP3 inflammasome activation in LPS-induced vascular injury. Acta Pharm. Sin. B 2019, 9, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.V.; Deng, M.; Ting, J.P. The NLRP3 inflammasome: Molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019, 19, 477–489. [Google Scholar] [CrossRef]
- Wullaert, A.; Bonnet, M.C.; Pasparakis, M. NF-kappaB in the regulation of epithelial homeostasis and inflammation. Cell Res. 2011, 21, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.S.; Ghosh, S. Shared principles in NF-kappaB signaling. Cell 2008, 132, 344–362. [Google Scholar] [CrossRef] [Green Version]
- Wang, Q.; Jiang, H.; Li, Y.; Chen, W.; Li, H.; Peng, K.; Zhang, Z.; Sun, X. Targeting NF-kB signaling with polymeric hybrid micelles that co-deliver siRNA and dexamethasone for arthritis therapy. Biomaterials 2017, 122, 10–22. [Google Scholar] [CrossRef]
- He, C.; Deng, J.; Hu, X.; Zhou, S.; Wu, J.; Xiao, D.; Darko, K.O.; Huang, Y.; Tao, T.; Peng, M.; et al. Vitamin A inhibits the action of LPS on the intestinal epithelial barrier function and tight junction proteins. Food Funct. 2019, 10, 1235–1242. [Google Scholar] [CrossRef]
- Fu, C.; Hao, S.; Xu, X.; Zhou, J.; Liu, Z.; Lu, H.; Wang, L.; Jin, W.; Li, S. Activation of SIRT1 ameliorates LPS-induced lung injury in mice via decreasing endothelial tight junction permeability. Acta Pharmacol. Sin. 2019, 40, 630–641. [Google Scholar] [CrossRef]

| Gene Product | Primer | GenBank Accession | |
|---|---|---|---|
| Direction | Sequences (5′ to 3′) | ||
| IL-6 | F | GGGAAATGTGGAGGCTGTG | NM_214399 |
| R | AGGGGTGGAGGCTTTGTCT | ||
| IL-1β | F | GGCCGCCAAGGTATAATGA | NM_214055 |
| R | GGACCTCTGGGCATGGCTTTC | ||
| IL-18 | F | GCTGCTGAACGGGAAGACAA | NM_213997.1 |
| R | AAACACGGCTAGATGTCCCT | ||
| TNF-α | F | GCCCACGTTGTGGCCAATGTCAAA | NM_214002 |
| R | GTTGTCTTTCAGGTTCACGCCGTT | ||
| GAPDH | F | GCTGCTGAACGGGAAGACAA | NM_001206359 |
| R | AAACACGGCTAGATGTCCCT | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Li, Y.; Chu, B.; Yuan, L.; Liu, N.; Zhu, Y.; Wang, J. Lactobacillus johnsonii L531 Alleviates the Damage Caused by Salmonella Typhimurium via Inhibiting TLR4, NF-κB, and NLRP3 Inflammasome Signaling Pathways. Microorganisms 2021, 9, 1983. https://doi.org/10.3390/microorganisms9091983
Chen S, Li Y, Chu B, Yuan L, Liu N, Zhu Y, Wang J. Lactobacillus johnsonii L531 Alleviates the Damage Caused by Salmonella Typhimurium via Inhibiting TLR4, NF-κB, and NLRP3 Inflammasome Signaling Pathways. Microorganisms. 2021; 9(9):1983. https://doi.org/10.3390/microorganisms9091983
Chicago/Turabian StyleChen, Shiyan, Yanan Li, Bingxin Chu, Lanxin Yuan, Ning Liu, Yaohong Zhu, and Jiufeng Wang. 2021. "Lactobacillus johnsonii L531 Alleviates the Damage Caused by Salmonella Typhimurium via Inhibiting TLR4, NF-κB, and NLRP3 Inflammasome Signaling Pathways" Microorganisms 9, no. 9: 1983. https://doi.org/10.3390/microorganisms9091983
APA StyleChen, S., Li, Y., Chu, B., Yuan, L., Liu, N., Zhu, Y., & Wang, J. (2021). Lactobacillus johnsonii L531 Alleviates the Damage Caused by Salmonella Typhimurium via Inhibiting TLR4, NF-κB, and NLRP3 Inflammasome Signaling Pathways. Microorganisms, 9(9), 1983. https://doi.org/10.3390/microorganisms9091983






