In Vivo Biofilm Formation of Pathogenic Leptospira spp. in the Vitreous Humor of Horses with Recurrent Uveitis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Positive Controls
2.2. Sample Selection
2.3. Negative Controls
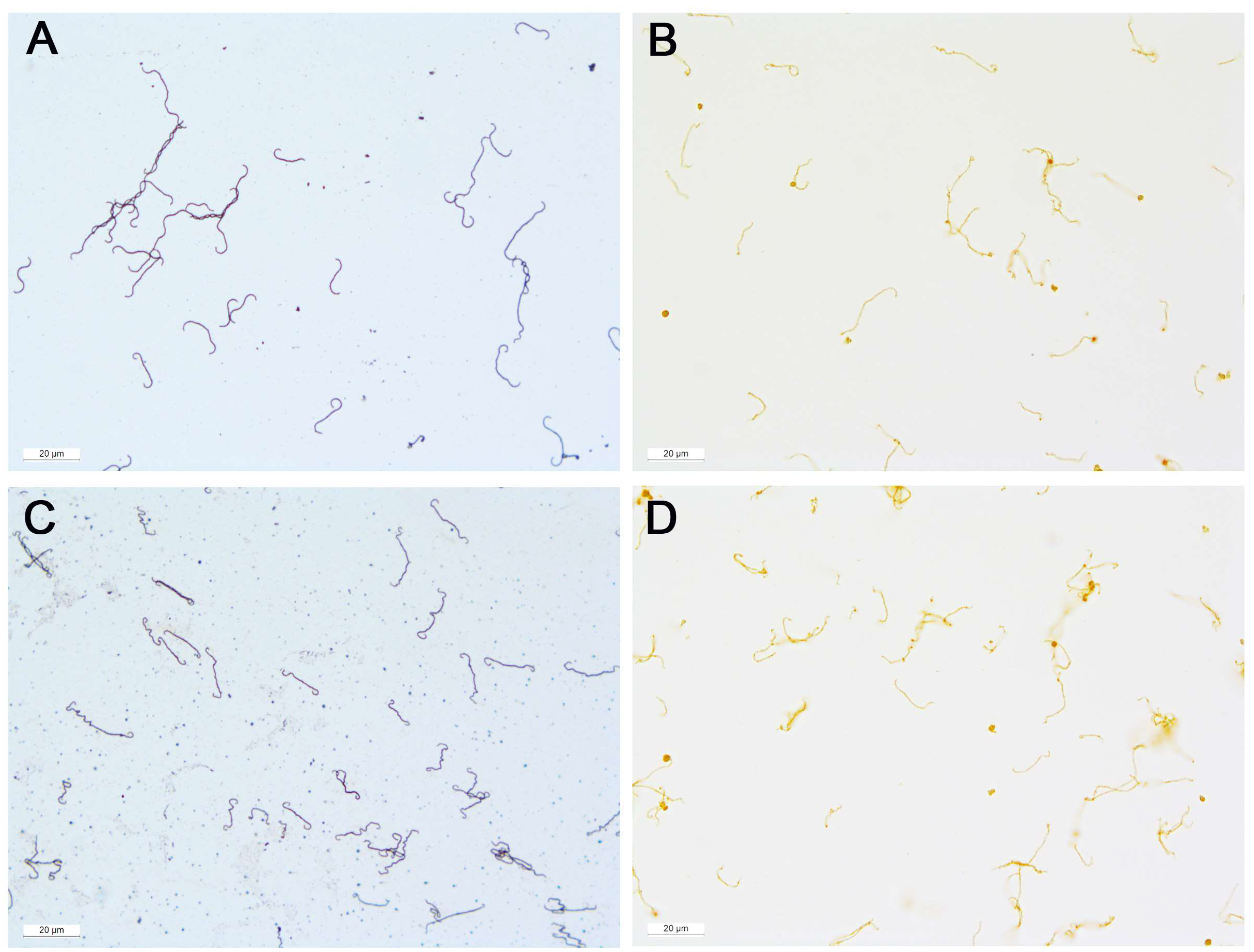
2.4. Warthin-Starry Silver Stain
2.5. Immunohistochemistry
2.6. Microscopy Images Acquisition
- Initial contact/attachment to the surface:Adhesion and cohesion; the attachment to a surface, as well as the attachment of bacteria to each other.
- Microcolony formation:After stable attachment to a surface or other bacteria, the bacteria began to multiply by cell division, as well as forming extracellular polymeric substances (EPS), which led to the formation of microcolonies.
- Maturation and architecture:EPS are the main components of the biofilm. Cell density in the biofilm is controlled by intercellular signaling and cell-to-cell communication (quorum sensing). This leads to the production of EPS and thus to a three-dimensional dense biofilm structure.
- Detachment/dispersion of biofilm:Multiplication of bacteria in the biofilm results in further spreading and reattachment to a surface or other bacteria.
3. Results
3.1. Positive Controls
3.2. Samples from ERU Eyes
3.3. Steps in Biofilm Formation
- Initial contact/attachment to the surface:
- 2.
- Microcolony formation:
- 3.
- Maturation and architecture:
3.4. Negative Controls
4. Discussion
- Infecting bacteria are bound to a substrate or a surface [82]:
- 2.
- The direct examination of infected tissue reveals bacteria living in cell aggregates or microcolonies surrounded by extracellular matrix [82]:
- 3.
- Biofilm-associated infections are generally confined to a specific location (although bacterial dissemination may occur, it is considered a secondary phenomenon) [82]:
- 4.
- Biofilm-associated infections are impossible or difficult to eliminate using antibiotics, to which the responsible organisms are sensitive when in their planktonic or free-living state [82]:
- 5.
- No organism can be cultured despite a strong presumption of infection with the pathogen of interest [83]:
- 6.
- Ineffective immune response as evidenced by bacterial aggregates surrounded by inflammatory cells within host tissue [83]:
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Høiby, N.; Bjarnsholt, T.; Givskov, M.; Molin, S.; Ciofu, O. Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents 2010, 35, 322–332. [Google Scholar] [CrossRef] [Green Version]
- Høiby, N.; Bjarnsholt, T.; Moser, C.; Bassi, G.L.; Coenye, T.; Donelli, G.; Hall-Stoodley, L.; Holá, V.; Imbert, C.; Kirketerp-Møller, K.; et al. ESCMID guideline for the diagnosis and treatment of biofilm infections 2014. Clin. Microbiol. Infect. 2015, 21 (Suppl. 1), S1–S25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Goarant, C.; Trueba, G.; Bierque, E.; Thibeaux, R.; Davis, B.; de la Pena-Moctezuma, A. Part 3: Specific Excreted Pathogens: Environmental and Epidemiology Aspects-Section 2: Bacteria. In Water and Sanitation for the 21st Century: Health and Microbiological Aspects of Excreta and Wastewater Management (Global Water Pathogen Project); UNESCO, Michigan State University: East Lansing, MI, USA, 2019. [Google Scholar] [CrossRef]
- Ristow, P.; Bourhy, P.; Kerneis, S.; Schmitt, C.; Prevost, M.-C.; Lilenbaum, W.; Picardeau, M. Biofilm formation by saprophytic and pathogenic leptospires. Microbiology 2008, 154, 1309–1317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thibeaux, R.; Soupé-Gilbert, M.E.; Kainiu, M.; Girault, D.; Bierque, E.; Fernandes, J.; Bähre, H.; Douyère, A.; Eskenazi, N.; Vinh, J.; et al. The zoonotic pathogen Leptospira interrogans mitigates environmental stress through cyclic-di-GMP-controlled biofilm production. NPJ Biofilms Microbiomes 2020, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Brihuega, B.; Samartino, L.; Auteri, C.; Venzano, A.; Caimi, K. In vivo cell aggregations of a recent swine biofilm-forming isolate of Leptospira interrogans strain from Argentina. Rev. Argent Microbiol. 2012, 44, 138–143. [Google Scholar]
- Yamaguchi, T.; Higa, N.; Okura, N.; Matsumoto, A.; Hermawan, I.; Yamashiro, T.; Suzuki, T.; Toma, C. Characterizing interactions of Leptospira interrogans with proximal renal tubule epithelial cells. BMC Microbiol. 2018, 18, 64. [Google Scholar] [CrossRef] [Green Version]
- Dwyer, A.E.; Crockett, R.; Kalsow, C.M. Association of leptospiral seroreactivity and breed with uveitis and blindness in horses: 372 cases (1986-1993). J. Am. Vet. Med. Assoc. 1995, 207, 1327–1331. [Google Scholar]
- Gerhards, H.; Wollanke, B.; Brem, S. Vitrectomy as a diagnostic and therapeutic approach for equine recurrent uveitis (ERU). In Proceedings of the 45th Annual Convention AAEP, Albuquerque, NM, USA, 8 December 1999; Volume 45, pp. 89–93. [Google Scholar]
- Witmer, R. Periodic ophthalmia in horses. Am. J. Ophthalmol. 1954, 37, 243–253. [Google Scholar] [CrossRef]
- Crowhurst, R.C. Periodic Ophthalmia: Clinical Aspects. Proc. R. Soc. Med. 1954, 47, 236–237. [Google Scholar]
- Wollanke, B. Die Equine Rezidivierende Uveitis (ERU) Als Intraokulare Leptospirose. Habilitation Thesis, Ludwig-Maximilians-University, Munich, Germany, 2002. [Google Scholar]
- Komar, G.; Szutter, L. Die innere periodische Augenentzündung (Mondblindheit) der Pferde (Iridocyclitis recidiva equorum). In Tieraerztliche Augenheilkunde; Paul Parey: Berlin, Germany, 1968; pp. 231–237. [Google Scholar]
- Kulbrock, M.; Von Borstel, M.; Rohn, K.; Distl, O.; Ohnesorge, B. Studie zu Häufigkeit und Schweregrad der equinen rezidivierenden Uveitis bei Warmblütern. Pferdeheilkunde 2013, 29, 27–36. [Google Scholar] [CrossRef] [Green Version]
- Szemes, P.A.; Gerhards, H. Untersuchungen zur Prävalenz der equinen rezidivierenden Uveitis im Großraum Köln-Bonn. Prakt. Tierarzt 2000, 81, 408–420. [Google Scholar]
- Gerding, J.C.; Gilger, B.C. Prognosis and impact of equine recurrent uveitis. Equine Vet. J. 2016, 48, 290–298. [Google Scholar] [CrossRef]
- Gerhards, H.; Wollanke, B. Surgical treatment of equine recurrent uveitis: Trans-pars-plana vitrectomy in horses. In Equine Ophthalmology, 1st ed.; Gilger, B.C., Ed.; Elsevier Saunders: Philadelphia, PA, USA, 2005; pp. 314–319. [Google Scholar]
- Werry, H.; Gerhards, H. Technique and indications for surgical treatment of equine recurrent uveitis. Pferdeheilkunde 1991, 7, 321. [Google Scholar] [CrossRef]
- Werry, H.; Gerhards, H. Surgical treatment of equine recurrent uveitis: A preliminary report. Tierarztl. Prax. 1992, 20, 178–186. [Google Scholar]
- Wollanke, B.; Gerhards, H.; Schinagl, C. Results of 654 trans-pars plana vitrectomies of equine eyes with recurrent uveitis-follow-up until 18 years after surgery. Pferdeheilkunde-Equine Med. 2021, 37, 204–214. [Google Scholar] [CrossRef]
- Baake, E.I.A.; von Borstel, M.; Rohn, K.; Boevé, M.H.; Ohnesorge, B. Long-term ophthalmologic examinations of eyes with equine recurrent uveitis after pars plana vitrectomy. Pferdeheilkunde 2019, 35, 220–233. [Google Scholar] [CrossRef]
- Von Borstel, M.; Von Oppen, T.; Glitz, F.; Frühauf, B.; Deegen, E.; Boevé, M.H.; Ohnesorge, B. Long-term results of pars-plana (double-port) vitrectomy in equine recurrent uveitis. Pferdeheilkunde 2005, 21, 13–18. [Google Scholar] [CrossRef] [Green Version]
- Winterberg, A.; Gerhards, H. Longterm-results of pars-plana-vitrectomy in equine recurrent uveitis. Pferdeheilkunde 1997, 13, 377–383. [Google Scholar] [CrossRef] [Green Version]
- Baumgart, A.; Gerhards, H. Besonderheiten der Tigerschecken-Uveitis und möglicher Cyclosporin A-Einsatz in deren Therapie in Deutschland. Pferdeheilkunde 2014, 30, 626–632. [Google Scholar]
- Bryans, J.T. Studies on equine leptospirosis. Cornell Vet. 1955, 45, 16–50. [Google Scholar]
- Gsell, O.; Rehsteiner, K.; Verrey, F. Iridocyclitis as a late consequence of Leptospirosis Pomona (porter’s disease): Agglutinin and lymphocytosis in the aqueous humor. Ophthalmologica 1946, 112, 320–334. [Google Scholar] [CrossRef] [PubMed]
- Hartwigk, H.; Stoebbe, E. Kultureller Nachweis von Leptospiren bei Hund und Pferd. Berl. Münch. Tierärztl. Wschr. 1952, 65, 188–190. [Google Scholar]
- Morter, R.; Williams, R.; Bolte, H.; Freeman, M.J. Equine leptospirosis. J. Am. Vet. Med. Assoc. 1969, 155, 436–442. [Google Scholar] [PubMed]
- Roberts, S.J. Sequelae of leptospirosis in horses on a small farm. J. Am. Vet. Med. Assoc. 1958, 133, 189–194. [Google Scholar]
- Williams, R.D. The Presence and Duration of Persistence of Leptospira Pomona in Equine Ocular Tissues Following Experimentally Induced Systemic Infection. Master of Science Thesis, Purdue University, West Lafayette, IN, USA, 1968. [Google Scholar]
- Baake, E.; von Borstel, M.; Rohn, K.; Ohnesorge, B. Detection of intraocular leptospiral DNA, antibodies and Leptospira spp. in horses with equine recurrent uveitis in different laboratories. Pferdeheilkunde 2016, 32, 346–356. [Google Scholar] [CrossRef] [Green Version]
- Borstel, M.V.; Oey, L.; Strutzberg-Minder, K.; Boevé, M.H.; Ohnesorge, B. Direct and indirect detection of leptospires in vitreal samples of horses with ERU. Pferdeheilkunde 2010, 26, 219–225. [Google Scholar] [CrossRef]
- Dorrego Keiter, E.; Tóth, J.; Dikker, L.; Sielhorst, J.; Schusser, G.F. Long-term results of pars plana vitrectomy in relationship to leptospiral antibody detection in vitreous humor in 118 horses with equine recurrent uveitis (ERU). Pferdeheilkunde-Equine Med. 2017, 33, 112–118. [Google Scholar] [CrossRef] [Green Version]
- Dorrego-Keiter, E.; Tóth, J.; Dikker, L.; Sielhorst, J.; Schusser, G.F. Detection of leptospira by culture of vitreous humor and detection of antibodies against leptospira in vitreous humor and serum of 225 horses with equine recurrent uveitis. Berl. Munch. Tierarztl. Wochenschr. 2016, 129, 209–215. [Google Scholar]
- Toemoerdy, E.; Haessig, M.; Spiess, B.M. The outcome of pars plana vitrectomy in horses with equine recurrent uveitis with regard to the presence or absence of intravitreal antibodies against various serovars of Leptospira interrogans. Pferdeheilkunde 2010, 26, 251–254. [Google Scholar] [CrossRef] [Green Version]
- Voelter, K.; Vial, Z.; Pot, A.S.; Spiess, B.M. Leptospiral antibody prevalence and surgical treatment outcome in horses with Equine Recurrent Uveitis (ERU) in Switzerland. Vet. Ophthalmol. 2020, 23, 648–658. [Google Scholar] [CrossRef]
- Wollanke, B.; Gerhards, H.; Brem, S.; Kopp, H.; Meyer, P. Intraocular and serum antibody titers to Leptospira in 150 horses with equine recurrent uveitis (ERU) subjected to vitrectomy. Berl. Munch. Tierarztl. Wochenschr. 1998, 111, 134–139. [Google Scholar] [PubMed]
- Wollanke, B.; Gerhards, H.; Brem, S.; Meyer, P.; Kopp, H. Ätiologie der equinen rezidivierenden Uveitis (ERU): Autoimmunkrankheit oder intraokulare Leptospireninfektion. Pferdeheilkunde 2004, 20, 327–340. [Google Scholar] [CrossRef] [Green Version]
- Wollanke, B.; Gerhards, H.; Brem, S.; Wolf, E.; Kopp, H.; Meyer, P. Zur Leptospirenätiologie der equinen rezidivierenden Uveitis (ERU): Ergebnisse der Untersuchungen von Serum-und Glaskörperproben. Tieraerztl. Prax. 2000, 28, 153–158. [Google Scholar]
- Wollanke, B.; Rohrbach, B.W.; Gerhards, H. Serum and vitreous humor antibody titers in and isolation of Leptospira interrogans from horses with recurrent uveitis. J. Am. Vet. Med. Assoc. 2001, 219, 795–800. [Google Scholar] [CrossRef] [PubMed]
- Loibl, J.K.; Gerhards, H.; Brem, S.; Wollanke, B. Improving the laboratory diagnosis of leptospiral uveitis in horses by using an indirect ELISA for the detection of antibodies against Leptospira spp. in intraocular samples. Pferdeheilkunde 2018, 34, 267–277. [Google Scholar] [CrossRef] [Green Version]
- Wollanke, B.; Geiger, T.; Gerhards, H. Evaluation of" SNAP (R) Lepto"-ELISA and comparison with MAT and PCR results for diagnosis of leptospiral uveitis in horses using intraocular samples. Pferdeheilkunde 2018, 34, 508–516. [Google Scholar] [CrossRef]
- Faber, N.A.; Crawford, M.; LeFebvre, R.B.; Buyukmihci, N.C.; Madigan, J.E.; Willits, N.H. Detection of Leptospira spp. in the aqueous humor of horses with naturally acquired recurrent uveitis. J. Clin. Microbiol. 2000, 38, 2731–2733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polle, F.; Storey, E.; Eades, S.; Alt, D.; Hornsby, R.; Zuerner, R.; Carter, R. Role of intraocular Leptospira infections in the pathogenesis of equine recurrent uveitis in the southern United States. J. Equine Vet. Sci. 2014, 34, 1300–1306. [Google Scholar] [CrossRef] [Green Version]
- Popp, M.; Cerhards, H.; Wollanke, B. Enrofloxacin concentrations in the vitreous of horses with equine recurrent uveitis (ERU) after repeated intravenous administration. Pferdeheilkunde 2013, 29, 574–580. [Google Scholar] [CrossRef] [Green Version]
- Brandes, K.; Wollanke, B.; Niedermaier, G.; Brem, S.; Gerhards, H. Recurrent uveitis in horses: Vitreal examinations with ultrastructural detection of leptospires. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2007, 54, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Niedermaier, G.; Wollanke, B.; Hoffmann, R.; Brem, S.; Gerhards, H. Detection of leptospira in the vitreous body of horses without ocular diseases and of horses with equine recurrent uveitis (ERU) using transmission-electron microscopy. Dtsch. Tierarztl. Wschr. 2006, 113, 401–432. [Google Scholar]
- Degroote, R.L.; Deeg, C.A. Immunological Insights in Equine Recurrent Uveitis. Front. Immunol. 2021, 11, 609855. [Google Scholar] [CrossRef]
- Deeg, C.A. Ocular immunology in equine recurrent uveitis. Vet. Ophthalmol. 2008, 11 (Suppl. 1), 61–65. [Google Scholar] [CrossRef] [PubMed]
- Voigt, V.; Wikstrom, M.E.; Kezic, J.M.; Schuster, I.S.; Fleming, P.; Makinen, K.; Daley, S.R.; Andoniou, C.E.; Degli-Esposti, M.A.; Forrester, J.V. Ocular antigen does not cause disease unless presented in the context of inflammation. Sci. Rep. 2017, 7, 14226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavach, J.D. Vitreous. In Large Animal Ophthalmology; Stamathis, G., Ed.; Mosby Company: St Louis, MO, USA, 1990; pp. 202–205. [Google Scholar]
- Niedermaier, G.; Wollanke, B.; Hoffmann, R.; Matiasek, K.; Gerhards, H. Darstellung der Glaskörperstruktur von augengesunden Pferden und von Pferden mit equiner rezidivierender Uveitis (ERU) mittels Transmissions-Elektronenmikroskopie. Dtsch. Tierarztl. Wschr. 2006, 113, 209–248. [Google Scholar]
- Sebag, J. The Vitreous: Structure, Function and Pathobiology, 1st ed.; Springer: New York, NY, USA, 1989. [Google Scholar]
- Geißler, P.; Wollanke, B. Biofilm formation in persistent infections and its role in the pathogenesis of equine recurrent uveitis (ERU)-a literature review. Pferdeheilkunde-Equine Med. 2021, 37, 225–233. [Google Scholar] [CrossRef]
- Riedelsheimer, B.; Buechl-Zimmermann, S. Faerbungen. In Romeis Mikroskopische Technik, 19th ed.; Mulisch, M., Welsch, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 234–235. ISBN 10 3642551890. [Google Scholar]
- Sapi, E.; Bastian, S.L.; Mpoy, C.M.; Scott, S.; Rattelle, A.; Pabbati, N.; Poruri, A.; Burugu, D.; Theophilus, P.A.S.; Pham, T.V.; et al. Characterization of biofilm formation by Borrelia burgdorferi In Vitro. PLoS ONE 2012, 7, e48277. [Google Scholar] [CrossRef]
- Sapi, E.; Balasubramanian, K.; Poruri, A.; Maghsoudlou, J.S.; Socarras, K.M.; Timmaraju, A.V.; Filush, K.R.; Gupta, K.; Shaikh, S.; Theophilus, P.A.; et al. Evidence of In Vivo Existence of Borrelia Biofilm in Borrelial Lymphocytomas. Eur. J. Microbiol. Immunol. 2016, 6, 9–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Domenico, E.G.; Cavallo, I.; Bordignon, V.; D′Agosto, G.; Pontone, M.; Trento, E.; Gallo, M.T.; Prignano, G.; Pimpinelli, F.; Toma, L.; et al. The Emerging Role of Microbial Biofilm in Lyme Neuroborreliosis. Front. Neurol. 2018, 9, 1048. [Google Scholar] [CrossRef]
- Monahan, A.M.; Callanan, J.J.; Nally, J.E. Review paper: Host-pathogen interactions in the kidney during chronic leptospirosis. Vet. Pathol. 2009, 46, 792–799. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Jensen, P.; Fiandaca, M.J.; Pedersen, J.; Hansen, C.R.; Andersen, C.B.; Pressler, T.; Givskov, M.; Høiby, N. Pseudomonas aeruginosa biofilms in the respiratory tract of cystic fibrosis patients. Pediatr. Pulmonol. 2009, 44, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Hall-Stoodley, L.; Stoodley, P.; Kathju, S.; Høiby, N.; Moser, C.; Costerton, W.J.; Moter, A.; Bjarnsholt, T. Towards diagnostic guidelines for biofilm-associated infections. FEMS Immunol. Med. Microbiol. 2012, 65, 127–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Høiby, N. A personal history of research on microbial biofilms and biofilm infections. Pathog. Dis. 2014, 70, 205–211. [Google Scholar] [CrossRef] [Green Version]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dowsett, C. Biofilms: A practice-based approach to identification and treatment. Wounds UK 2013, 9, 68–72. [Google Scholar]
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Prim. 2017, 3, 17038. [Google Scholar] [CrossRef]
- Coffin, D.L. Detection of leptospires by fluorescent antibody. Am. J. Vet. Res. 1962, 23, 159–164. [Google Scholar] [PubMed]
- Cole, J.R., Jr.; Sulzer, C.R.; Pursell, A.R. Improved microtechnique for the leptospiral microscopic agglutination test. Appl. Microbiol. 1973, 25, 976–980. [Google Scholar] [CrossRef]
- Ribotta, M.J.; Higgins, R.; Gottschalk, M.; Lallier, R. Development of an indirect enzyme-linked immunosorbent assay for the detection of leptospiral antibodies in dogs. Can. J. Vet. Res. 2000, 64, 32–37. [Google Scholar]
- Maruoka, T.; Nikaido, Y.; Miyahara, S.; Katafuchi, E.; Inamasu, Y.; Ogawa, M.; Fukuda, K.; Nakayama, T.; Horishita, T.; Saito, M. Correlation between renal distribution of leptospires during the acute phase and chronic renal dysfunction in a hamster model of infection with Leptospira interrogans. PLoS Negl. Trop. Dis. 2021, 15, e0009410. [Google Scholar] [CrossRef] [PubMed]
- Blenden, D.C.; Goldberg, H.S. Silver impregnation stain for Leptospira and Flagella. J. Bacteriol. 1965, 89, 899–900. [Google Scholar] [CrossRef] [Green Version]
- Churukian, C.J.; Schenk, E.A. A Warthin-Starry method for spirochetes and bacteria using a microwave oven. J. Histotechnol. 1988, 11, 149–151. [Google Scholar] [CrossRef]
- Thomas, C. Histopathologie Kompakt: Kursbuch der Allgemeinen und Speziellen Histopathologie, 1st ed.; Schattauer Verlag: Stuttgart, Germany, 2004. [Google Scholar]
- Almeida, D.S.; Paz, L.N. Investigation of chronic infection by Leptospira spp. in asymptomatic sheep slaughtered in slaughterhouse. PLoS ONE. 2019, 14, e0217391. [Google Scholar] [CrossRef]
- Saglam, Y.; Yener, Z. Immunohistochemical detection of leptospiral antigens in cases of naturally occurring abortions in sheep. Small Rumin. Res. 2008, 74, 119–122. [Google Scholar] [CrossRef]
- Santos, A.A.; Figueira, C.P. Heterogenic colonization patterns by Leptospira interrogans in Rattus norvegicus from urban slums. Braz. J. Microbiol. 2015, 46, 1161–1164. [Google Scholar] [CrossRef] [Green Version]
- Sutherland, I.W. The biofilm matrix--an immobilized but dynamic microbial environment. Trends Microbiol. 2001, 9, 222–227. [Google Scholar] [CrossRef]
- Garrett, T.R.; Bhakoo, M.; Zhang, Z. Bacterial adhesion and biofilms on surfaces. Prog. Nat. Sci. 2008, 18, 1049–1056. [Google Scholar] [CrossRef]
- Maric, S.; Vranes, J. Characteristics and significance of microbial biofilm formation. Period. Biol. 2007, 109, 115–121. [Google Scholar]
- Wild, C.J.; Greenlee, J.J.; Bolin, C.A.; Barnett, J.K.; Haake, D.A.; Cheville, N.E. An improved immunohistochemical diagnostic technique for canine leptospirosis using antileptospiral antibodies on renal tissue. J. Vet. Diagn. Investig. 2002, 14, 20–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zilber, A.L.; Belli, P.; Artois, M.; Kodjo, A.; Djelouadji, Z. First Observation of Leptospira interrogans in the Lungs of Rattus norvegicus. Biomed. Res. Int. 2016, 2016, 9656274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parsek, M.R.; Singh, P.K. Bacterial biofilms: An emerging link to disease pathogenesis. Annu. Rev. Microbiol. 2003, 57, 677–701. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Stoodley, P. Evolving concepts in biofilm infections. Cell. Microbiol. 2009, 11, 1034–1043. [Google Scholar] [CrossRef]
- Linke, R.P.; Brandes, K.; Cielewicz, M.-B.; Gerhards, H.; Wollanke, B. Ocular leptospiral infection leads to ciliary induction and local AA-amyloidosis in horses. Amyloid 2019, 26, 127–128. [Google Scholar] [CrossRef]
- Waldner, J.; Gerhards, H.; Wollanke, B. Investigations into the occurrence of serum amyloid A in the equine eye. Pferdeheilkunde–Equine Med. 2018, 34, 461–467. [Google Scholar] [CrossRef]
- Vinod Kumar, K.; Lall, C.; Vimal Raj, R.; Vedhagiri, K.; Vijayachari, P. Molecular detection of pathogenic leptospiral protein encoding gene (lipL32) in environmental aquatic biofilms. Lett. Appl. Microbiol. 2016, 62, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Brem, S.; Gerhards, H.; Wollanke, B.; Meyer, P.; Kopp, H. Demonstration of intraocular leptospira in 4 horses suffering from equine recurrent uveitis (ERU). Berl. Munch. Tierarztl. Wochenschr. 1998, 111, 415–417. [Google Scholar] [PubMed]
- Brem, S. (State Office for Health and Food Safety, 85764 Oberschleißheim, Germany). Personal Communication, 1996. [Google Scholar]
- Bohl, E.; Ferguson, L. Leptospirosis in domestic animals. J. Am. Vet. Med. Assoc. 1952, 121, 421–428. [Google Scholar] [PubMed]
- Halliwell, R.E.; Brim, T.A.; Hines, M.A.; Wolf, D.; White, F.H. Studies on equine recurrent uveitis. II: The role of infection with Leptospira interrogans serovar pomona. Curr. Eye Res. 1985, 4, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Hines, M.T. Immunologically mediated ocular disease in the horse. Vet. Clin. North. Am. Large Anim. Pract. 1984, 6, 264–271. [Google Scholar] [CrossRef]
- Williams, R.D.; Morter, R.L.; Freeman, M.J.; Lavignette, A.M. Experimental chronic uveitis. Ophthalmic signs following equine leptospirosis. Investig. Ophthalmol. 1971, 10, 948–954. [Google Scholar]
- Dubielzig, R.; Render, J.; Morreale, R. Distinctive morphologic features of the ciliary body in equine recurrent uveitis. Vet. Comp. Ophthalmol. 1997, 7, 163–167. [Google Scholar]
- Romeike, A.; Brügmann, M.; Drommer, W. Immunohistochemical studies in equine recurrent uveitis (ERU). Vet. Pathol. 1998, 35, 515–526. [Google Scholar] [CrossRef] [Green Version]
- Roth, T.; Brandes, K.; Gerhards, H.; Giving, E.; Wollanke, B. Histologische Untersuchungen des Glaskörpers bei Pferden mit equiner rezidivierender Uveitis. Pferdeheilkunde 2014, 30, 512–520. [Google Scholar] [CrossRef] [Green Version]
- Fingerhut, L.; Ohnesorge, B.; von Borstel, M.; Schumski, A.; Strutzberg-Minder, K.; Mörgelin, M.; Deeg, C.A.; Haagsman, H.P.; Beineke, A.; von Köckritz-Blickwede, M.; et al. Neutrophil Extracellular Traps in the Pathogenesis of Equine Recurrent Uveitis (ERU). Cells 2019, 8, 1528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Papayannopoulos, V. Neutrophils Facing Biofilms: The Battle of the Barriers. Cell Host Microbe 2019, 25, 477–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schulz, L.-C.; Hermanns, W. Zur Bedeutung der Grenzflächengewebe bei der rheumatoiden Entzündung. In Pathomechanismen Entzündlicher Rheumatischer Erkrankungen Bei Mensch und Tier; Deicher, H., Ed.; DFG, Sonderforschungsbereiche: Berlin, Germany, 1989; pp. 327–341. [Google Scholar]

| Sample | Signalment | Clinical Diagnosis | lipL32 Gene PCR Result (Ct Value) | Vitreal Leptospiral Antibody Titer (MAT) 1 |
|---|---|---|---|---|
| 1 | 4-year-old warmblood gelding | ERU | Positive (Ct 37) | Grippotyphosa 1:3200 |
| 2 | 8-year-old Icelandic horse mare | ERU | Positive (Ct 40) | Grippotyphosa 1:400 |
| 3 | 7-year-old Friesian mare | ERU | Positive (Ct 29) | Grippotyphosa 1:3200 |
| 4 | 7-year-old warmblood mare | ERU | Positive (Ct 37) | Grippotyphosa 1:800 |
| 5 | 15-year-old warmblood gelding | ERU | Positive (Ct 38) | Grippotyphosa 1:400 |
| 6 | 10-year-old Friesian gelding | ERU | Positive (Ct 30) | Altoduro 1:400 |
| 7 | 6-year-old warmblood mare | ERU | Positive (Ct 32) | Grippotyphosa 1:3200 |
| 8 | 7-year-old warmblood mare | ERU | Positive (Ct 35) | Grippotyphosa 1:3200 |
| 9 | 4-year-old thoroughbred gelding | ERU | Positive (Ct 33) | Grippotyphosa 1:3200 |
| 10 | 6-year-old warmblood mare | ERU | Positive (Ct 34) | Grippotyphosa 1:3200 |
| 11 | 5-year-old Icelandic horse gelding | ERU | Positive (Ct 33) | Altoduro 1:100 |
| 12 | 5-year-old warmblood stallion | ERU | Positive (Ct 33) | Grippotyphosa 1:3200 |
| 13 | 5-year-old warmblood gelding | ERU | Positive (Ct 38) | Grippotyphosa 1:3200 |
| 14 | 10-year-old warmblood gelding | ERU | Positive (Ct 39) | Grippotyphosa 1:800 |
| 15 | 8-year-old warmblood mare | ERU | Positive (Ct 38) | Grippotyphosa 1:800 |
| 16 | 5-year-old warmblood mare | ERU | Positive (Ct 35) | Grippotyphosa 1:3200 |
| 17 | 8-year-old warmblood gelding | ERU | Positive (Ct 35) | Grippotyphosa 1:200 |
| 18 | 5-year-old warmblood gelding | ERU | Positive (Ct 32) | Grippotyphosa 1:200 |
| 19 | 5-year-old warmblood mare | ERU | Positive (Ct 33) | Grippotyphosa 1:1600 |
| 20 | 4-year-old warmblood gelding | ERU | Positive (Ct 33) | Grippotyphosa 1:100 |
| 21 | 3-year-old warmblood mare | ERU | Positive (Ct 38) | Negative |
| 22 | 10-year-old warmblood gelding | ERU | Positive (Ct 36) | Grippotyphosa 1:3200 |
| 23 | 6-year-old warmblood mare | ERU | Positive (Ct 36) | Grippotyphosa 1:3200 |
| 24 | 7-year-old purebred Spanish stallion | ERU | Positive (Ct 39) | Grippotyphosa 1:3200 |
| 25 | 10-year-old purebred Spanish mare | ERU | Positive (Ct 39) | Australis 1:3200 |
| 26 | 15-year-old warmblood mare | ERU | Positive (Ct 39) | Grippotyphosa 1:3200 |
| 27 | 8-year-old Welsh pony mare | ERU | Positive (Ct 32) | Grippotyphosa 1:3200 |
| 28 | 7-year-old warmblood gelding | ERU | Positive (Ct 29) | Pomona 1:3200 |
| 29 | 12-year-old warmblood gelding | ERU | Positive (Ct 35) | Grippotyphosa 1:3200 |
| 30 | 8-year-old warmblood gelding | Normal 2 | Negative | Negative |
| 31 | 23-year-old Haflinger mare | Normal 2 | Negative | Negative |
| 32 | 17-year-old warmblood gelding | Normal 2 | Negative | Negative |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ackermann, K.; Kenngott, R.; Settles, M.; Gerhards, H.; Maierl, J.; Wollanke, B. In Vivo Biofilm Formation of Pathogenic Leptospira spp. in the Vitreous Humor of Horses with Recurrent Uveitis. Microorganisms 2021, 9, 1915. https://doi.org/10.3390/microorganisms9091915
Ackermann K, Kenngott R, Settles M, Gerhards H, Maierl J, Wollanke B. In Vivo Biofilm Formation of Pathogenic Leptospira spp. in the Vitreous Humor of Horses with Recurrent Uveitis. Microorganisms. 2021; 9(9):1915. https://doi.org/10.3390/microorganisms9091915
Chicago/Turabian StyleAckermann, Kerstin, Rebecca Kenngott, Monica Settles, Hartmut Gerhards, Johann Maierl, and Bettina Wollanke. 2021. "In Vivo Biofilm Formation of Pathogenic Leptospira spp. in the Vitreous Humor of Horses with Recurrent Uveitis" Microorganisms 9, no. 9: 1915. https://doi.org/10.3390/microorganisms9091915






