Effects of Atrazine on Chernozem Microbial Communities Evaluated by Traditional Detection and Modern Sequencing Technology
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design and Soil Samples
2.2. Determination of Atrazine Residue in Soil
2.3. Determination of Soil Enzyme Activity
2.4. Functional Diversity of Soil Microbial Community
2.5. Determination of Bacterial Population and Abundance in Soil
2.6. Statistical Analysis
3. Results
3.1. Residue Dynamics of Atrazine in Different Plough Layers of Chernozem in Cold Region
3.1.1. Determination of Standard Addition Recovery Test
3.1.2. Residue Changes of Atrazine in Different Plough Layers of Chernozem in Cold Region
3.2. Effect of Atrazine on Enzyme Activity in Chernozem
3.3. Effect of Atrazine on Carbon Utilization of Microbial Community in Chernozem in Cold Region
3.3.1. Average Color Change Rate (AWCD) of Soil Microorganisms in Plough Layers in Different Periods
3.3.2. Correlation between Soil Microbial Community, Enzyme Activity, Carbon Source Utilization, and Atrazine Residue
3.4. Effects of Atrazine on Bacterial Diversity in Cultivated Soil Layers
3.4.1. Effects of Atrazine on Bacterial Composition in Tillage Soil Layers of Chernozem in Cold Region
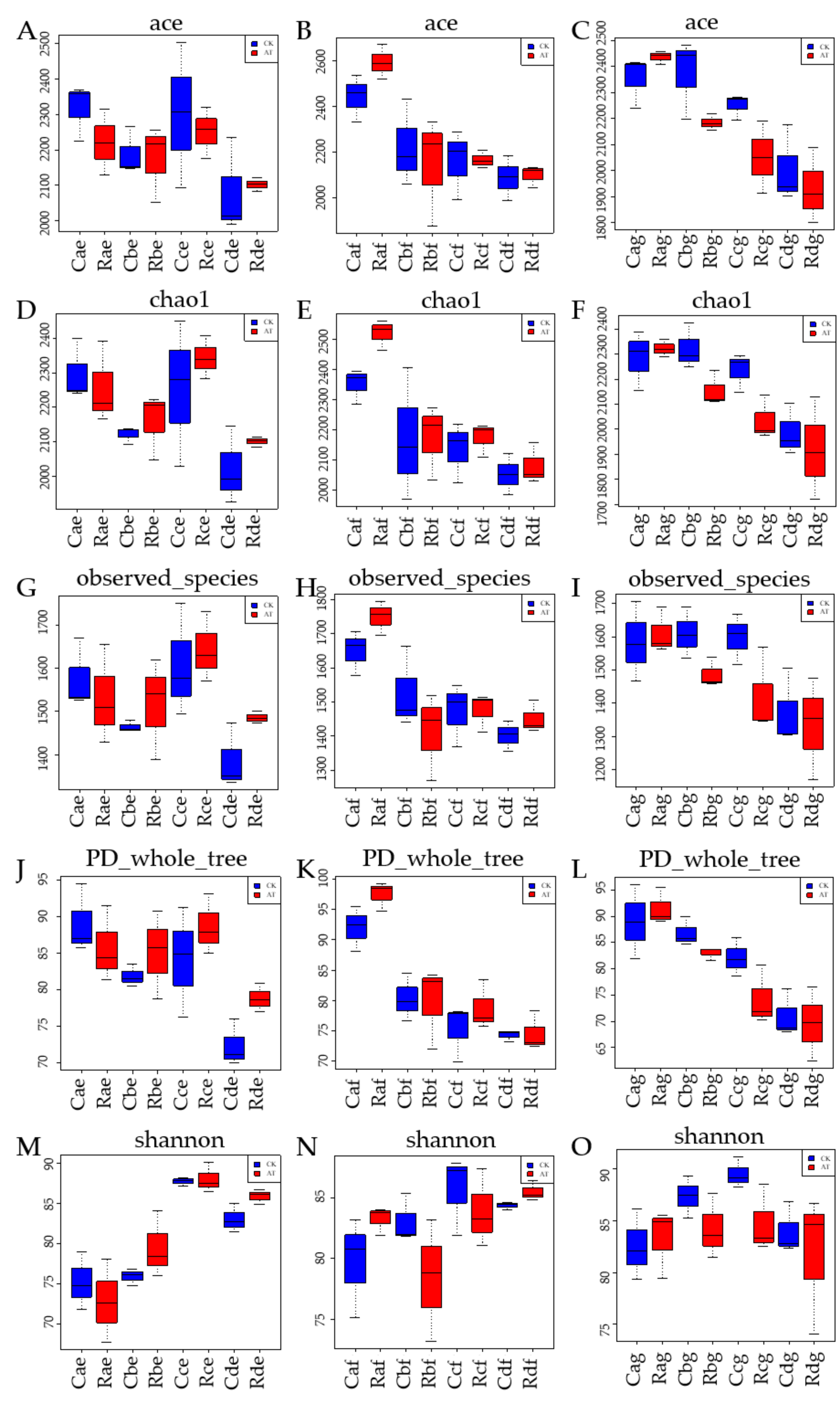
3.4.2. Effect of Atrazine on Diversity of Single Samples in Different Plough Layers of Chernozem in Cold Region
3.4.3. Effects of Atrazine on Diversity of Multiple Samples in Different Plough Layers of Chernozem in Cold Region
4. Discussion
4.1. Dynamic Residue of Atrazine in Different Plough Layers of Chernozem in Cold Region
4.2. Effect of Atrazine on Enzyme Activity in Cultivated Soil Layers of Chernozem in Cold Region
4.3. Effects of Atrazine on Carbon Source Utilization of Microbial Community in Cold Chernozem
4.4. Effects of Atrazine on Bacterial Population and Abundance in Cultivated Soil Layers
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Solomon, K.R.; Giesy, J.P.; LaPoint, T.W.; Giddings, J.M.; Richards, R.P. Ecological risk assessment of atrazine in North American surface waters. Environ. Toxicol. Chem. 2013, 32, 10–11. [Google Scholar] [CrossRef] [PubMed]
- He, X.M.; Ge, H.B.; Li, A.M.; Ma, X.F.; Song, G.Q. Determination of carbofuran, carbaryl and atrazine in water by SPE–HPLC. Adm. Tech. Environ. Monit. 2011, 23, 46–48. [Google Scholar]
- Mecozzi, R.; Di Palma, L.; Merli, C. Experimental in situ chemical peroxidation of atrazine in contaminated soil. Chemosphere 2006, 62, 1481–1489. [Google Scholar] [CrossRef]
- Tao, Q.H. Effect of coexisting pollutants on atrazine adsorption on natural sediments. J. Environ. Sci. 2004, 24, 696–701. [Google Scholar]
- Qu, M.J.; Li, H.D.; Liu, W.; Li, N.; Zhu, D.W. A review on remediation of atrazine in soil and water systems. Asian J. Ecotoxicol. 2017, 4, 119–128. [Google Scholar]
- Adnan, M.; Xu, H.; Muhammad, M.A.; Syed, A.A.S.; Sun, N.; Qudsia, S.; Muhammad, K.; Muhammad, N.; Manuel, C.; Gao, H.J.; et al. Long-term fertilization enhanced carbon mineralization and maize biomass through physical protection of organic carbon in fractions under continuous maize cropping. Appl. Soil Ecol. 2021, 165, 103971. [Google Scholar]
- Muhammad, M.A.; Xu, M.G.; Syed, A.A.S.; Muhammad, W.A.; Tariq, A.; Adnan, M.; Muhammad, N.A.; Zhou, B.K.; Ma, X.Z. Variations in the profile distribution and protection mechanisms of organic carbon under long-term fertilization in a Chinese Mollisol. Sci. Total. Environ. 2020, 723, 138181. [Google Scholar]
- Liu, X.X.; Hui, C.L.; Bi, L.Z.; Romantschuk, M.; Kontro, M.; Strommer, R.; Hui, N. Bacterial community structure in atrazine treated reforested farmland in Wuying China. Appl. Soil Ecol. 2016, 98, 39–46. [Google Scholar] [CrossRef]
- Li, L.X.; Gao, Z.G.; Yang, R.X. Effects of common herbicides on microorganisms in maize rhizosphere soil. Liaoning Agric. Sci. 2015, 2015, 14–16. [Google Scholar]
- Fang, H.; Han, Y.L.; Yin, Y.M.; Jin, X.X.; Wang, S.Y.; Tang, F.F.; Cai, L.; Yu, Y.L. Microbial response to repeated treatments of manure containing sulfadiazine and chlortetracycline in soil. J. Environ. Sci. Health Part B 2014, 8, 609–615. [Google Scholar] [CrossRef]
- Bharati, K.; Rakesh, P.; Anand, V.; Garima, D.; Ashok, P.; Suresh, K.C.; Santosh, R.M. Nitrous oxide production from soybean and maize under the influence of weedicides and zero tillage conservation agriculture. J. Hazard. Mater. 2021, 402, 123572. [Google Scholar]
- Chen, Q.; Yang, B.; Wang, H.; He, F.; Gao, Y.; Scheel, R.A. Soil microbial community toxic response to atrazine and its residues under atrazine and lead contamination. Environ. Sci. Pollut. Res. 2015, 22, 996–1007. [Google Scholar] [CrossRef]
- Cao, B.; Zhang, Y.; Wang, Z.Y.; Li, M.Y.; Yang, F.; Jiang, D.; Jiang, Z. Insight into the variation of bacterial structure in atrazine-contaminated soil regulating by potential phytoremediator: Pennisetum americanum (L.) K. Schum. Front. Microbiol. 2018, 9, 00864. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.F.; Fan, X.X.; Zhang, T.; He, W.Y.; Song, F.Q. Effects of the long-term application of atrazine on soil enzyme activity and bacterial community structure in farmlands in China. Environ. Pollut. 2020, 262, 114264. [Google Scholar] [CrossRef]
- Ellappan, T.; Balasubramaian, P.; Chidambaram, K.; Subhash, C.M. α–Glucosidase and α–amylase inhibitory activity of Senna surattensis. J. Acupunct. Meridian. Stud. 2013, 6, 24–30. [Google Scholar]
- Konstantinou, C.; Wang, Y.; Biscontin, G.; Soga, K. The role of bacterial urease activity on the uniformity of carbonate precipitation profiles of bio-treated coarse sand specimens. Sci. Rep. 2021, 11, 6161. [Google Scholar] [CrossRef]
- Lin, W.; Liu, J.Y.; Feng, Y.X.; Zhang, C.S.; Zhao, Q.; Lin, Q.; Wang, J. First report of tobacco bacterial hollow stalk caused by Dickeya chrysanthemi (syn. Erwinia chrysanthemi) in Nanping, China. Plant Dis. 2020, 104, 3248. [Google Scholar] [CrossRef] [PubMed]
- Batisson, I.; Crouzet, O.; Besse-Hoggan, P.; Sancelme, M.; Mangot, J.-F.; Mallet, C.; Bohatier, J. Isolation and characterization of mesotrione–degrading Bacillus sp from soil. Environ. Pollut. 2009, 157, 1195–1201. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.N.; Gao, Z.G.; Sun, Y.Q.; Liu, X.; Li, W.Y. Dynamic analysis of atrazine residue in Shenyang soil. Mod. Pestic. 2014, 13, 40–43, 51. [Google Scholar]
- Fang, L.P.; Li, H.D.; Ding, R.Y.; Wang, F.G. Residues and application safety assessment of atrazine in corn and soil. Mod. Agrochem. 2012, 11, 33–36. [Google Scholar]
- Popov, V.H.; Cornish, P.S.; Sultana, K.; Morris, E.C. Atrazine degradation in soils: The role of microbial communities, atrazine application history, and soil carbon. Soil Res. 2005, 7, 861–871. [Google Scholar] [CrossRef]
- Muhammad, M.A.; Xu, H.; Tariq, A.; Nan, S.; Adnan, M.; Muhammad, W.A.; Syed, A.A.S.; Khalid, M.; Zhou, B.K.; Ma, X.Z.; et al. Carbon, nitrogen, and phosphorus stoichiometry mediate sensitivity of carbon stabilization mechanisms along with surface layers of a Mollisol after long-term fertilization in Northeast China. J. Soils Sediments 2021, 2, 705–723. [Google Scholar]
- Gong, A.; Ye, C.; Wang, X.; Lei, Z.; Liu, J. Dynamics and mechanism of ultraviolet photolysis of atrazine on soil surface. Pest. Managt. Sci. 2001, 57, 380–385. [Google Scholar] [CrossRef]
- Mohiuddin, M.; Mohammed, M.K. Fungicide (carbendazim) and herbicides (2,4–D and atrazine) influence on soil microorganisms and soil enzymes of rhizospheric soil of groundnut crop. Int. J. Recent Sci. Res. 2014, 5, 585–589. [Google Scholar]
- Emurotu, M.O.; Anyanwu, C.U. Effect of atrazine and butachlor on some soil enzymes activities at different concentrations. Pelagia Res. Libr. 2016, 2, 9–15. [Google Scholar]
- Xu, J.B.; Wang, Y.L.; Liu, M.; Chen, M.J.; Lin, X.G. Use of BIOLOG and microcalorimetry in combination to study factors of carbon sources stimulating metabolic activity of soil microbe in red soil. Acta Pedol. Sin. 2018, 55, 203–212. [Google Scholar]
- Wang, J.H.; Zhu, L.S.; Sun, R.L.; Zhao, B.Q. Effect of atrazine on soil urease under two different fertilization conditions. J. Agro-Environ. Sci. 2004, 23, 162–166. [Google Scholar]
- Zhang, S.; Wang, H.F.; Lin, P.; Luo, Z.J.; Zhang, F.J. Effect of atrazine on soil urease activity. Tianjin Agric. Sci. 2015, 21, 93–95. [Google Scholar]
- Yang, N.; Liu, Y.P.; Zhu, J.; Wang, Z.Q.; Li, J.W. Study on the efficacy and mechanism of Fe-TiO2 visible heterogeneous Fenton catalytic degradation of atrazine. Chemosphere 2020, 252, 126333. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Liu, R.; Currie, R.S.; Lee, Y.B.; Lee, Y.H. Cross-resistance to atrazine and metribuzin in multiple herbicide-resistant kochia accessions: Confirmation, mechanism, and management. Weed Technol. 2020, 49, 480–488. [Google Scholar]
- Chen, P.; Na, R.S.; Zhang, C.L.; Qin, H.; Zhang, Y.; Zhang, F.J. Effect of atrazine on soil phosphatase activity. Tianjin Agric. Sci. 2014, 20, 23–25. [Google Scholar]
- Shen, H.Q.; Duan, Y.X.; Chen, L.J.; Zhu, X.F.; Wang, Y.Y.; Huang, S.S. Defense enzymatic reaction of different tolerant soybean varieties after treated with atrazine. Soybean Sci. 2011, 30, 259–262. [Google Scholar]
- Qi, L.; Zhou, P.; Yang, L.H.; Gao, M. Effects of land reclamation on the physical, chemical, and microbial quantity and enzyme activity properties of degraded agricultural soils. J. Soil. Sediment 2020, 20, 973–981. [Google Scholar] [CrossRef]
- Yang, Y.; Li, T.; Wang, Y. Negative effects of multiple global change factors on soil microbial diversity. Soil. Biol. Biochem. 2021, 156, 108229. [Google Scholar] [CrossRef]
- Chen, Y.L.; Zhang, Z.S.; Huang, L.; Zhao, Y.; Hu, Y.G.; Zhang, P.; Zhang, D.H.; Zhang, H. Co–variation of fine–root distribution with vegetation and soil properties along a revegetation chronosequence in a desert area in northwestern China. Catena 2017, 151, 16–25. [Google Scholar] [CrossRef]
- Ning, Y.; Zou, D.S.; Yang, M.Y.; Lin, Z.G. Variations in soil microbial biomass carbon and soil dissolved organic carbon in the re-vegetation of hilly slopes with purple soil. PLoS ONE 2016, 11, e0166536. [Google Scholar]
- Wang, Z.; Han, X.Z.; Zhang, Z.M.; Hao, X.X. Carbon source metabolism characteristics of microbial community in black soil profile of Northeast China. Acta Ecol. Sin. 2016, 36, 7740–7748. [Google Scholar]
- Kang, U.G.; Shin, W.C.; Choi, J.S.; Lee, Y.B. Impacts of cropping systems on the distribution of soil microorganisms in mid-mountainous paddy. South Korea Land Fertil. Soc. 2016, 49, 480–488. [Google Scholar] [CrossRef] [Green Version]
- Woloszczyk, P.; Fiencke, C.; Elsner, D.C.; Cordsen, E.; Pfeiffer, E.M. Spatial and temporal patterns in soil organic carbon, microbial biomass and activity under different land–use types in a long-term soil-monitoring network. Pedobiologia 2020, 80, 150642. [Google Scholar] [CrossRef]
- Li, C.; Yan, L.; Tang, L. The effects of long-term fertilization on the accumulation of organic carbon in the deep soil profile of an oasis farmland. Plant Soil 2013, 369, 645–656. [Google Scholar] [CrossRef]
- Li, C.; Yan, K.; Tang, L.; Jia, Z.; Li, Y. Change in deep soil microbial communities due to long-term fertilization. Soil Biol. Biochem. 2014, 75, 264–272. [Google Scholar] [CrossRef]
- Eilers, K.G.; Debenport, S.; Anderson, S.; Fierer, N. Digging deeper to find unique microbial communities: The strong effect of depth on the structure of bacterial and archaeal communities in soil. Soil Biol. Biochem. 2012, 50, 58–65. [Google Scholar] [CrossRef]
- Ding, P.Y.; Chu, L.B.; Zhang, N.; Wang, X.; Wang, J.L. Effects of dissolved oxygen in the oxic parts of A/O reactor on degradation of organic pollutants and analysis of microbial community for treating petrochemical wastewater. Huan Jing Ke Xue 2015, 36, 604–611. [Google Scholar]
- Xiao, S.Q.; Zhang, Q.; Chen, X.M.; Dong, F.Q.; Chen, H.; Liu, M.X.; Ali, I. Speciation distribution of heavy metals in uranium mining impacted soils and impact on bacterial community revealed by high-throughput sequencing. Front. Microbiol. 2019, 10, 1867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, N.; Xu, H.C.; Yan, Z.S.; Yang, T.; Wang, C.H.; Jiang, H.L. Improved lignin degradation through distinct microbial community in subsurface sediments of one eutrophic lake. Renew. Energy 2019, 138, 861–869. [Google Scholar] [CrossRef]
- Hao, J.; Wang, P.H.; Kang, Y.F.; He, H.T.; Luo, H.H.; Kim, S.; Niu, L.L.; Jiang, H.Z.; Ma, K.S. Degradation of perfluorooctane sulfonamide by Acinetobacter sp. M and its extracellular enzymes. Chem.—Asian J. 2019, 14, 14–16. [Google Scholar] [CrossRef]
- Wu, M.-L.; Nie, M.Q.; Wang, X.C.; Su, J.M.S. Effect of Mn2+ on pyrene degradation by Flavobacterium sp. FCN2. Environ. Sci. 2008, 29, 1982–1985. [Google Scholar]
- Soares, F.L.; Melo, I.S.; Dias, A.C.F.; Andreote, F.D. Cellulolytic bacteria from soils in harsh environments. World J. Microbiol. Biotechnol. 2012, 28, 2195–2203. [Google Scholar] [CrossRef] [PubMed]
- Papale, M.; Giannarelli, S.; Francesconi, S.; Di Marco, G.; Mikkonen, A.; Conte, A.; Rizzo, C.; De Domenico, E.; Michaud, L.; Lo Giudice, A.; et al. Enrichment, isolation and biodegradation potential of psychrotolerant polychlorinated-biphenyl degrading bacteria from the Kongsfjorden (Svalbard Islands, High Arctic Norway). Mar. Pollut. Bull. 2017, 114, 849. [Google Scholar] [CrossRef]







| A | B | C | D | E | F | G | H | J | K | L | M | N | O | P | |
| A | 1 | ||||||||||||||
| B | −0.023 | 1 | |||||||||||||
| C | −0.242 | 0.261 | 1 | ||||||||||||
| D | 0.387 | 0.542 ** | 0.032 | 1 | |||||||||||
| E | 0.216 | 0.613 ** | 0.13 | 0.728 ** | 1 | ||||||||||
| F | −0.161 | −0.112 | 0.306 | −0.439 * | −0.0242 | 1 | |||||||||
| G | 0.096 | 0.228 | 0.104 | 0.532 ** | 0.417 | −0.620 ** | 1 | ||||||||
| H | 0.227 | 0.088 | −0.456 * | 0.174 | 0.043 | −0.443 * | −0.084 | 1 | |||||||
| J | −0.093 | −0.218 | −0.268 | −0.048 | −0.212 | −0.221 | −0.1 | −0.255 | 1 | ||||||
| K | −0.219 | 0.055 | 0.124 | −0.165 | 0.049 | −0.153 | −0.328 | 0.295 | −0.029 | 1 | |||||
| L | 0.209 | 0.009 | 0.069 | −0.044 | −0.007 | 0.065 | 0.041 | −0.045 | −0.583 ** | −0.272 | 1 | ||||
| M | 0.410 * | 0.32 | 0.074 | 0.638 ** | 0.271 | −0.324 | 0.269 | 0.261 | 0.054 | −0.281 | 0.016 | 1 | |||
| N | −0.486 * | −0.551 ** | 0.022 | −0.723 ** | −0.620 ** | 0.304 | −0.582 ** | 0.053 | 0.103 | 0.342 | −0.107 | −0.394 | 1 | ||
| O | 0.006 | −0.363 | −0.092 | −0.38 | −0.523 ** | 0.036 | −0.488 * | 0.32 | 0.115 | 0.187 | 0.056 | 0.099 | 0.682 ** | 1 | |
| P | 0.411* | −0.29 | −0.235 | −0.2 | −0.337 | −0.045 | −0.432 * | 0.515 * | −0.083 | 0.308 | 0.087 | 0.116 | 0.375 | 0.608 ** | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, F.; Gao, M.; Lu, H.; Wei, Y.; Chi, H.; Yang, T.; Yuan, M.; Fu, H.; Zeng, W.; Liu, C. Effects of Atrazine on Chernozem Microbial Communities Evaluated by Traditional Detection and Modern Sequencing Technology. Microorganisms 2021, 9, 1832. https://doi.org/10.3390/microorganisms9091832
Yang F, Gao M, Lu H, Wei Y, Chi H, Yang T, Yuan M, Fu H, Zeng W, Liu C. Effects of Atrazine on Chernozem Microbial Communities Evaluated by Traditional Detection and Modern Sequencing Technology. Microorganisms. 2021; 9(9):1832. https://doi.org/10.3390/microorganisms9091832
Chicago/Turabian StyleYang, Fengshan, Mengying Gao, Honggang Lu, Yuning Wei, Huiting Chi, Tai Yang, Mingrui Yuan, Haiyan Fu, Weimin Zeng, and Chunguang Liu. 2021. "Effects of Atrazine on Chernozem Microbial Communities Evaluated by Traditional Detection and Modern Sequencing Technology" Microorganisms 9, no. 9: 1832. https://doi.org/10.3390/microorganisms9091832
APA StyleYang, F., Gao, M., Lu, H., Wei, Y., Chi, H., Yang, T., Yuan, M., Fu, H., Zeng, W., & Liu, C. (2021). Effects of Atrazine on Chernozem Microbial Communities Evaluated by Traditional Detection and Modern Sequencing Technology. Microorganisms, 9(9), 1832. https://doi.org/10.3390/microorganisms9091832





