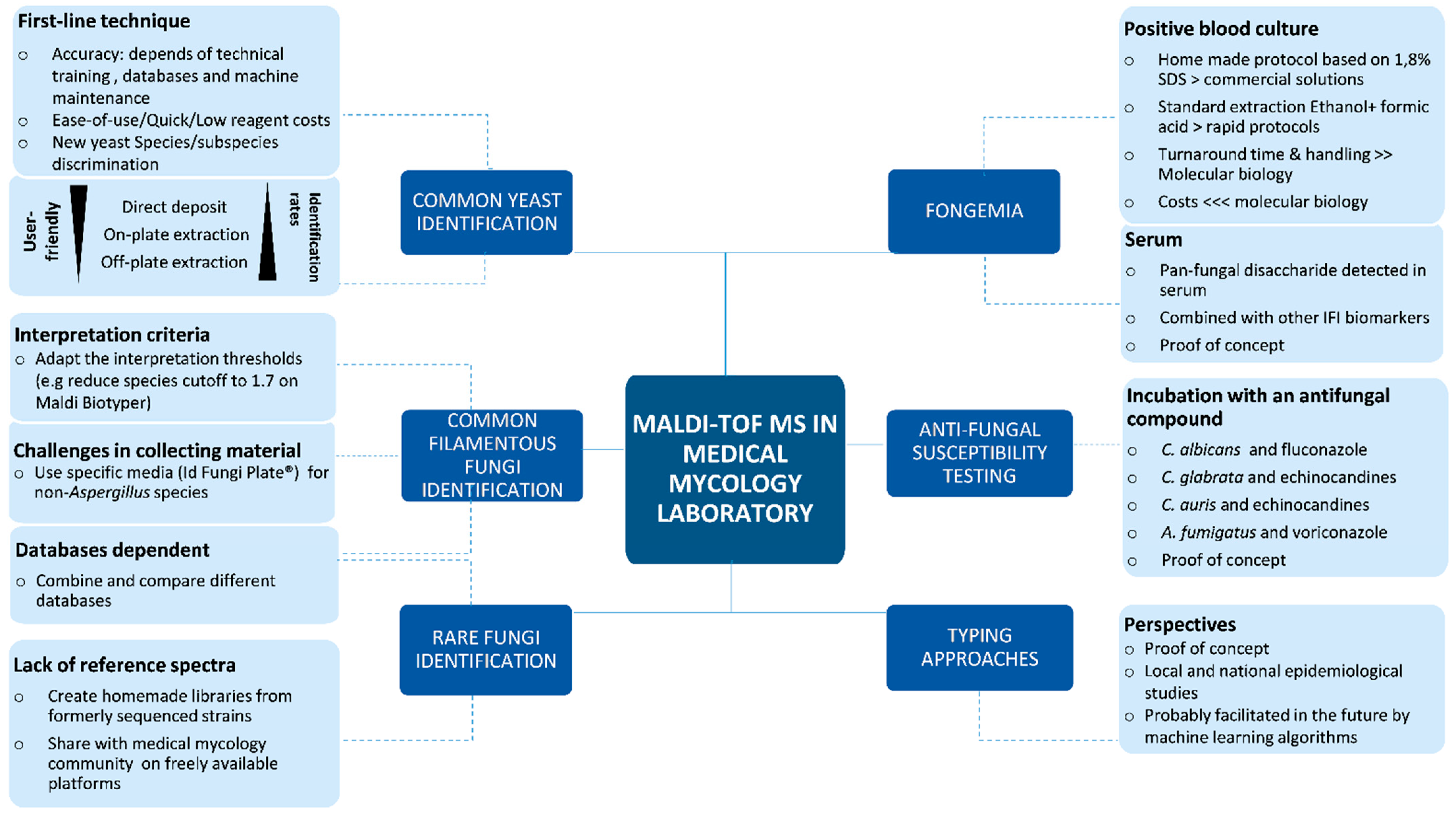
MALDI-TOF MS in a Medical Mycology Laboratory: On Stage and Backstage
Abstract
1. Introduction
2. The Place of MALDI-TOF MS in Yeast Identification
2.1. Routine Use
2.2. Limits and Solutions
3. The Place of MALDI-TOF MS in Common Filamentous Fungi Identification
3.1. Routine Use
3.2. Limits
3.3. Solutions
4. The Place of MALDI-TOF MS in Rare Filamentous Fungi Identification
4.1. Routine Use
4.2. Limits
4.3. Solutions
5. The Place of MALDI-TOF MS in Positive Blood Culture Identification
5.1. Routine Use
5.2. Limits
5.3. Solutions
| Application for | Recommendation/Protocols | Possible Alternatives for FMI | |
|---|---|---|---|
| Yeast identification | Common Yeast identification | Formic acid Off plate extraction On plate extraction Regular maintenance and laser calibration | Direct deposit on biotargets or ground steel targets Score threshold adaptation |
| Rare yeast identification | Check presence in commercial databases | Confront spectra to other databases (MSI, NHI, MicrobNet) | |
| Update databases | Create homemade reference libraries | ||
| Mold identification | Common mold identification | Long protocol extraction, bead beating, subculture in liquid media, EtOH-AF * | On plate extraction Off plate extraction Score threshold adaptation Specific culture or subculture media (IDFP®) Use of MSI, NHI, MicrobNet for accurate identification of cryptic species |
| Rare mold identification | Check presence in commercial databases | Confront spectra to other databases (MSI, NHI, MicrobNet) Homemade reference libraries | |
| Fungemia | On positive blood culture | Commercial kit MBT-Sepsityper® (Bruker Daltonics, Bremen, Germany) Rapid Sepsityper® (Bruker Daltonics, Bremen, Germany) VITEK MS ® blood culture kit (bioMerieux, Marcy l’Etoile, France rapid BACpro® II kit (Nittobo Medical Co., Tokyo, Japan) Sepsityper RUO software (Bruker Daltonics, Bremen, Germany) | In house protocols based on 1.8% SDS solution Score threshold adaptation |
| On patient blood | No commercial solution | Disaccharide detection [62] Proof of concept | |
| Antifungal susceptibility testing | On Strains | Incubation with ATF | Spectra profile changes analysis Growth inhibition analysis (MBT Astra) Strain typing |
| Specific R or S peaks direct detection | Proof of concept on C. glabrata Further development is needed |
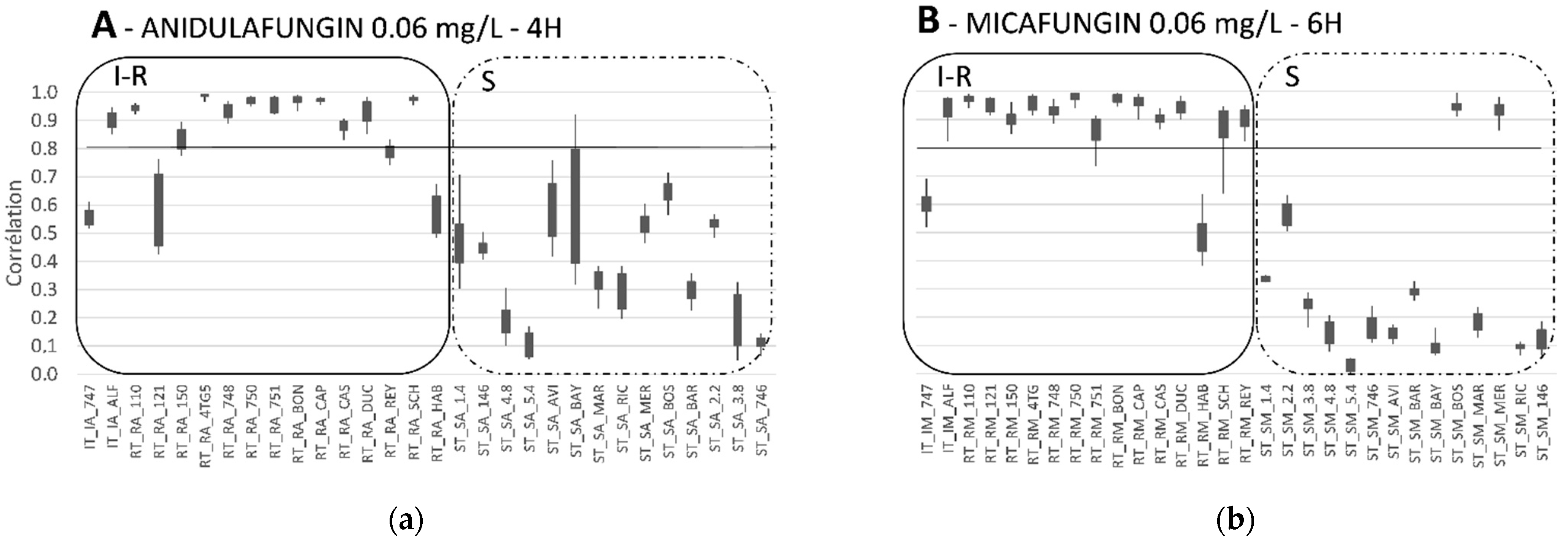
6. The Place of MALDI-TOF MS in Antifungal Susceptibility Testing
6.1. Routine Use
6.2. Limits
6.3. Solutions
7. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Claydon, M.A.; Davey, S.N.; Edwards-Jones, V.; Gordon, D.B. The Rapid Identification of Intact Microorganisms Using Mass Spectrometry. Nat. Biotechnol. 1996, 14, 1584–1586. [Google Scholar] [CrossRef]
- van Veen, S.Q.; Claas, E.C.J.; Kuijper, E.J. High-Throughput Identification of Bacteria and Yeast by Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry in Conventional Medical Microbiology Laboratories. J. Clin. Microbiol. 2010, 48, 900–907. [Google Scholar] [CrossRef]
- Tanaka, K. The Origin of Macromolecule Ionization by Laser Irradiation (Nobel Lecture). Angew Chem. Int. Ed. Engl. 2003, 42, 3860–3870. [Google Scholar] [CrossRef] [PubMed]
- Fenselau, C.; Anhalt, J.P. Identification of Bacteria Using Mass Spectrometry. Anal. Chem. 1975, 47, 219–225. [Google Scholar] [CrossRef]
- Patel, R. A Moldy Application of MALDI: MALDI-ToF Mass Spectrometry for Fungal Identification. J. Fungi 2019, 5, 4. [Google Scholar] [CrossRef]
- Normand, A.-C.; Gabriel, F.; Riat, A.; Cassagne, C.; Bourgeois, N.; Huguenin, A.; Chauvin, P.; De Geyter, D.; Bexkens, M.; Rubio, E.; et al. Optimization of MALDI-ToF Mass Spectrometry for Yeast Identification: A Multicenter Study. Med. Mycol. 2020, 58, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.E.; Ellis, B.C.; Lee, R.; Stamper, P.D.; Zhang, S.X.; Carroll, K.C. Prospective Evaluation of a Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry System in a Hospital Clinical Microbiology Laboratory for Identification of Bacteria and Yeasts: A Bench-by-Bench Study for Assessing the Impact on Time to Identification and Cost-Effectiveness. J. Clin. Microbiol. 2012, 50, 3301–3308. [Google Scholar] [CrossRef] [PubMed]
- Angeletti, S.; Ciccozzi, M. Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry in Clinical Microbiology: An Updating Review. Infect. Genet. Evol. 2019, 76, 104063. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.J.; McCarthy, M.W. The Expanding Use of Matrix-Assisted Laser Desorption/Ionization-Time of Flight Mass Spectroscopy in the Diagnosis of Patients with Mycotic Diseases. Expert Rev. Mol. Diagn. 2019, 19, 241–248. [Google Scholar] [CrossRef]
- Yi, Q.; Xiao, M.; Fan, X.; Zhang, G.; Yang, Y.; Zhang, J.-J.; Duan, S.-M.; Cheng, J.-W.; Li, Y.; Zhou, M.-L.; et al. Evaluation of Autof MS 1000 and Vitek MS MALDI-TOF MS System in Identification of Closely-Related Yeasts Causing Invasive Fungal Diseases. Front. Cell Infect. Microbiol. 2021, 11, 628828. [Google Scholar] [CrossRef]
- Cassagne, C.; Cella, A.-L.; Suchon, P.; Normand, A.-C.; Ranque, S.; Piarroux, R. Evaluation of Four Pretreatment Procedures for MALDI-TOF MS Yeast Identification in the Routine Clinical Laboratory. Med. Mycol. 2013, 51, 371–377. [Google Scholar] [CrossRef]
- Pinto, A.; Halliday, C.; Zahra, M.; van Hal, S.; Olma, T.; Maszewska, K.; Iredell, J.R.; Meyer, W.; Chen, S.C.-A. Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry Identification of Yeasts Is Contingent on Robust Reference Spectra. PLoS ONE 2011, 6, e25712. [Google Scholar] [CrossRef] [PubMed]
- Ceballos-Garzon, A.; Amado, D.; Vélez, N.; Jiménez-A, M.J.; Rodríguez, C.; Parra-Giraldo, C.M. Development and Validation of an In-House Library of Colombian Candida Auris Strains with MALDI-TOF MS to Improve Yeast Identification. J. Fungi 2020, 6, 72. [Google Scholar] [CrossRef]
- Zvezdanova, M.E.; Arroyo, M.J.; Méndez, G.; Guinea, J.; Mancera, L.; Muñoz, P.; Rodríguez-Sánchez, B.; Escribano, P. Implementation of MALDI-TOF Mass Spectrometry and Peak Analysis: Application to the Discrimination of Cryptococcus Neoformans Species Complex and Their Interspecies Hybrids. J. Fungi 2020, 6, 330. [Google Scholar] [CrossRef]
- van Belkum, A.; Welker, M.; Pincus, D.; Charrier, J.-P.; Girard, V. Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry in Clinical Microbiology: What Are the Current Issues? Ann. Lab. Med. 2017, 37, 475–483. [Google Scholar] [CrossRef]
- Arastehfar, A.; Daneshnia, F.; Kord, M.; Roudbary, M.; Zarrinfar, H.; Fang, W.; Hashemi, S.J.; Najafzadeh, M.J.; Khodavaisy, S.; Pan, W.; et al. Comparison of 21-Plex PCR and API 20C AUX, MALDI-TOF MS, and RDNA Sequencing for a Wide Range of Clinically Isolated Yeast Species: Improved Identification by Combining 21-Plex PCR and API 20C AUX as an Alternative Strategy for Developing Countries. Front. Cell Infect. Microbiol. 2019, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Giraud, S.; Pihet, M.; Razafimandimby, B.; Carrere, J.; Degand, N.; Mely, L.; Favennec, L.; Dannaoui, E.; Bouchara, J.-P.; Calenda, A. Geosmithia Argillacea: An Emerging Pathogen in Patients with Cystic Fibrosis. J. Clin. Microbiol. 2010, 48, 2381–2386. [Google Scholar] [CrossRef]
- Alcazar-Fuoli, L.; Mellado, E.; Alastruey-Izquierdo, A.; Cuenca-Estrella, M.; Rodriguez-Tudela, J.L. Aspergillus Section Fumigati: Antifungal Susceptibility Patterns and Sequence-Based Identification. Antimicrob. Agents Chemother. 2008, 52, 1244–1251. [Google Scholar] [CrossRef] [PubMed]
- Imbert, S.; Normand, A.C.; Cassaing, S.; Gabriel, F.; Kristensen, L.; Bonnal, C.; Lachaud, L.; Costa, D.; Guitard, J.; Hasseine, L.; et al. Multicentric Analysis of the Species Distribution and Antifungal Susceptibility of Cryptic Isolates from Aspergillus Section Fumigati. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef]
- Welham, K.J.; Domin, M.A.; Johnson, K.; Jones, L.; Ashton, D.S. Characterization of Fungal Spores by Laser Desorption/Ionization Time-of-Flight Mass Spectrometry. Rapid Commun. Mass Spectrom. 2000, 14, 307–310. [Google Scholar] [CrossRef]
- Cassagne, C.; Ranque, S.; Normand, A.-C.; Fourquet, P.; Thiebault, S.; Planard, C.; Hendrickx, M.; Piarroux, R. Mould Routine Identification in the Clinical Laboratory by Matrix-Assisted Laser Desorption Ionization Time-Of-Flight Mass Spectrometry. PLoS ONE 2011, 6. [Google Scholar] [CrossRef]
- Marklein, G.; Josten, M.; Klanke, U.; Muller, E.; Horre, R.; Maier, T.; Wenzel, T.; Kostrzewa, M.; Bierbaum, G.; Hoerauf, A.; et al. Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry for Fast and Reliable Identification of Clinical Yeast Isolates. J. Clin. Microbiol. 2009, 47, 2912–2917. [Google Scholar] [CrossRef] [PubMed]
- Lau, A.F.; Drake, S.K.; Calhoun, L.B.; Henderson, C.M.; Zelazny, A.M. Development of a Clinically Comprehensive Database and a Simple Procedure for Identification of Molds from Solid Media by Matrix-Assisted Laser Desorption Ionization–Time of Flight Mass Spectrometry. J. Clin. Microbiol. 2013, 51, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Blaize, M.; Normand, A.-C.; Fekkar, A.; Piarroux, R. Identification des moisissures au laboratoire de routine hospitalière. Rev. Francoph. Lab. 2021, 2021, 58–65. [Google Scholar] [CrossRef]
- Schulthess, B.; Ledermann, R.; Mouttet, F.; Zbinden, A.; Bloemberg, G.V.; Böttger, E.C.; Hombach, M. Use of the Bruker MALDI Biotyper for Identification of Molds in the Clinical Mycology Laboratory. J. Clin. Microbiol 2014, 52, 2797–2803. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Zhao, Y.-P.; Xu, Y.-C.; Hsueh, P.-R. Evaluation of the Bruker Biotyper Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry System for Identification of Aspergillus Species Directly from Growth on Solid Agar Media. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Lau, A.F.; Walchak, R.C.; Miller, H.B.; Slechta, E.S.; Kamboj, K.; Riebe, K.; Robertson, A.E.; Gilbreath, J.J.; Mitchell, K.F.; Wallace, M.A.; et al. Multicenter Study Demonstrates Standardization Requirements for Mold Identification by MALDI-TOF MS. Front. Microbiol. 2019, 10, 2098. [Google Scholar] [CrossRef]
- Robert, M.G.; Romero, C.; Dard, C.; Garnaud, C.; Cognet, O.; Girard, T.; Rasamoelina, T.; Cornet, M.; Maubon, D. Evaluation of ID-Fungi-PlatesTM Medium for Identification of Molds by MALDI-BiotyperTM. J. Clin. Microbiol. 2020. [Google Scholar] [CrossRef]
- Heireman, L.; Patteet, S.; Steyaert, S. Performance of the New ID-Fungi Plate Using Two Types of Reference Libraries (Bruker and MSI) to Identify Fungi with the Bruker MALDI Biotyper. Med. Mycol. 2020, 58, 946–957. [Google Scholar] [CrossRef]
- Cardot Martin, E.; Renaux, C.; Catherinot, E.; Limousin, L.; Couderc, L.J.; Vasse, M. Rapid Identification of Fungi from Respiratory Samples by Bruker Biotyper Matrix–Assisted Laser Desorption/Ionisation Time-of-Flight Using ID-FUNGI Plates. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 391–395. [Google Scholar] [CrossRef]
- Normand, A.C.; Becker, P.; Gabriel, F.; Cassagne, C.; Accoceberry, I.; Gari-Toussaint, M.; Hasseine, L.; De Geyter, D.; Pierard, D.; Surmont, I.; et al. Validation of a New Web Application for Identification of Fungi by Use of Matrix-Assisted Laser Desorption Ionization–Time of Flight Mass Spectrometry. J. Clin. Microbiol. 2017, 55, 2661–2670. [Google Scholar] [CrossRef]
- Tavakoli, M.; Rivero-Menendez, O.; Abastabar, M.; Hedayati, M.T.; Sabino, R.; Siopi, M.; Zarrinfar, H.; Nouripour-Sisakht, S.; van der Lee, H.; Valadan, R.; et al. Genetic Diversity and Antifungal Susceptibility Patterns of Aspergillus Nidulans Complex Obtained from Clinical and Environmental Sources. Mycoses 2020, 63, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Suzuki, J.; Watanabe, A.; Matsuki, M.; Higa, K.; Inoue, E.; Akashi, S.; Shimada, M.; Kawashima, M.; Ohshima, N.; et al. Species Identification, Antifungal Susceptibility, and Clinical Feature Association of Aspergillus Section Nigri Isolates from the Lower Respiratory Tract. Med. Mycol 2020, 58, 310–314. [Google Scholar] [CrossRef]
- Stein, M.; Tran, V.; Nichol, K.A.; Lagacé-Wiens, P.; Pieroni, P.; Adam, H.J.; Turenne, C.; Walkty, A.J.; Normand, A.-C.; Hendrickx, M.; et al. Evaluation of Three MALDI-TOF Mass Spectrometry Libraries for the Identification of Filamentous Fungi in Three Clinical Microbiology Laboratories in Manitoba, Canada. Mycoses 2018, 61, 743–753. [Google Scholar] [CrossRef]
- Imbert, S.; Normand, A.C.; Gabriel, F.; Cassaing, S.; Bonnal, C.; Costa, D.; Lachaud, L.; Hasseine, L.; Kristensen, L.; Schuttler, C.; et al. Multi-Centric Evaluation of the Online MSI Platform for the Identification of Cryptic and Rare Species of Aspergillus by MALDI-TOF. Med. Mycol. 2019, 57, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Nabet, C.; Imbert, S.; Normand, A.C.; Blanchet, D.; Chanlin, R.; Becker, P.; Demar, M.; Piarroux, R. Unexpected Mould Diversity in Clinical Isolates from French Guiana and Associated Identification Difficulties. Med. Mycol. 2020, myaa091. [Google Scholar] [CrossRef]
- Michel, J.; Maubon, D.; Varoquaux, D.A.; Boulze, C.; Normand, A.C.; Righini, C.A.; Piarroux, R.; Dessi, P.; Ranque, S. Schizophyllum Commune: An Emergent or Misdiagnosed Fungal Pathogen in Rhinology? Med. Mycol. 2015, 54, 301–309. [Google Scholar] [CrossRef]
- Chowdhary, A.; Randhawa, H.S.; Gaur, S.N.; Agarwal, K.; Kathuria, S.; Roy, P.; Klaassen, C.H.; Meis, J.F. Schizophyllum Commune as an Emerging Fungal Pathogen: A Review and Report of Two Cases. Mycoses 2013, 56, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Buzina, W.; Lang-Loidolt, D.; Braun, H.; Freudenschuss, K.; Stammberger, H. Development of Molecular Methods for Identification of Schizophyllum Commune from Clinical Samples. J. Clin. Microbiol. 2001, 39, 2391–2396. [Google Scholar] [CrossRef]
- Drissner, D.; Freimoser, F.M. MALDI-TOF Mass Spectroscopy of Yeasts and Filamentous Fungi for Research and Diagnostics in the Agricultural Value Chain. Chem. Biol. Technol. Agric. 2017, 4, 13. [Google Scholar] [CrossRef][Green Version]
- Bernheim, D.; Dupont, D.; Aptel, F.; Dard, C.; Chiquet, C.; Normand, A.C.; Piarroux, R.; Cornet, M.; Maubon, D. Pythiosis: Case Report Leading to New Features in Clinical and Diagnostic Management of This Fungal-like Infection. Int. J. Infect. Dis. 2019, 86, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Rasamoelina, T.; Raharolahy, O.; Rakotozandrindrainy, N.; Ranaivo, I.; Andrianarison, M.; Rakotonirina, B.; Maubon, D.; Rakotomalala, F.; Rakoto Andrianarivelo, M.; Andriantsimahavandy, A.; et al. Chromoblastomycosis and Sporotrichosis, Two Endemic but Neglected Fungal Infections in Madagascar. J. Mycol. Med. 2017, 27, 312–324. [Google Scholar] [CrossRef] [PubMed]
- Rasamoelina, T.; Maubon, D.; Andrianarison, M.; Ranaivo, I.; Sendrasoa, F.; Rakotozandrindrainy, N.; Rakotomalala, F.A.; Bailly, S.; Rakotonirina, B.; Andriantsimahavandy, A.; et al. Endemic Chromoblastomycosis Caused Predominantly by Fonsecaea Nubica, Madagascar1. Emerg. Infect. Dis. 2020, 26, 1201–1211. [Google Scholar] [CrossRef] [PubMed]
- Rasamoelina, T.; Maubon, D.; Raharolahy, O.; Razanakoto, H.; Rakotozandrindrainy, N.; Rakotomalala, F.A.; Bailly, S.; Sendrasoa, F.; Ranaivo, I.; Andrianarison, M.; et al. Sporotrichosis in the Highlands of Madagascar, 2013-20171. Emerg. Infect. Dis. 2019, 25, 1893–1902. [Google Scholar] [CrossRef]
- Puig-Asensio, M.; Padilla, B.; Garnacho-Montero, J.; Zaragoza, O.; Aguado, J.M.; Zaragoza, R.; Montejo, M.; Muñoz, P.; Ruiz-Camps, I.; Cuenca-Estrella, M.; et al. Epidemiology and Predictive Factors for Early and Late Mortality in Candida Bloodstream Infections: A Population-Based Surveillance in Spain. Clin. Microbiol. Infect. 2014, 20, O245. [Google Scholar] [CrossRef]
- Posteraro, B.; De Carolis, E.; Vella, A.; Sanguinetti, M. MALDI-TOF Mass Spectrometry in the Clinical Mycology Laboratory: Identification of Fungi and Beyond. Expert Rev. Proteom. 2013, 10, 151–164. [Google Scholar] [CrossRef]
- Marinach, C.; Alanio, A.; Palous, M.; Kwasek, S.; Fekkar, A.; Brossas, J.-Y.; Brun, S.; Snounou, G.; Hennequin, C.; Sanglard, D.; et al. MALDI-TOF MS-Based Drug Susceptibility Testing of Pathogens: The Example of Candida Albicans and Fluconazole. Proteomics 2009, 9, 4627–4631. [Google Scholar] [CrossRef]
- Faron, M.L.; Buchan, B.W.; Ledeboer, N.A. Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry for Use with Positive Blood Cultures: Methodology, Performance, and Optimization. J. Clin. Microbiol. 2017, 55, 3328–3338. [Google Scholar] [CrossRef]
- Bellanger, A.-P.; Gbaguidi-Haore, H.; Liapis, E.; Scherer, E.; Millon, L. Rapid Identification of Candida Sp. by MALDI-TOF Mass Spectrometry Subsequent to Short-Term Incubation on a Solid Medium. APMIS 2019, 127, 217–221. [Google Scholar] [CrossRef]
- Martiny, D.; Dediste, A.; Vandenberg, O. Comparison of an In-House Method and the Commercial SepsityperTM Kit for Bacterial Identification Directly from Positive Blood Culture Broths by Matrix-Assisted Laser Desorption-Ionisation Time-of-Flight Mass Spectrometry. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2269–2281. [Google Scholar] [CrossRef]
- Marinach-Patrice, C.; Fekkar, A.; Atanasova, R.; Gomes, J.; Djamdjian, L.; Brossas, J.-Y.; Meyer, I.; Buffet, P.; Snounou, G.; Datry, A.; et al. Rapid Species Diagnosis for Invasive Candidiasis Using Mass Spectrometry. PLoS ONE 2010, 5, e8862. [Google Scholar] [CrossRef]
- Nomura, F.; Tsuchida, S.; Murata, S.; Satoh, M.; Matsushita, K. Mass Spectrometry-Based Microbiological Testing for Blood Stream Infection. Clin. Proteom. 2020, 17, 14. [Google Scholar] [CrossRef]
- Bidart, M.; Bonnet, I.; Hennebique, A.; Kherraf, Z.E.; Pelloux, H.; Berger, F.; Cornet, M.; Bailly, S.; Maubon, D. An In-House Assay Is Superior to Sepsityper for Direct Matrix-Assisted Laser Desorption Ionization-Time of Flight (MALDI-TOF) Mass Spectrometry Identification of Yeast Species in Blood Cultures. J. Clin. Microbiol. 2015, 53, 1761–1764. [Google Scholar] [CrossRef]
- Ponderand, L.; Pavese, P.; Maubon, D.; Giraudon, E.; Girard, T.; Landelle, C.; Maurin, M.; Caspar, Y. Evaluation of Rapid Sepsityper® Protocol and Specific MBT-Sepsityper Module (Bruker Daltonics) for the Rapid Diagnosis of Bacteremia and Fungemia by MALDI-TOF-MS. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 60. [Google Scholar] [CrossRef]
- Bryant, S.; Almahmoud, I.; Pierre, I.; Bardet, J.; Touati, S.; Maubon, D.; Cornet, M.; Richarme, C.; Maurin, M.; Pavese, P.; et al. Evaluation of Microbiological Performance and the Potential Clinical Impact of the EPlex® Blood Culture Identification Panels for the Rapid Diagnosis of Bacteremia and Fungemia. Front. Cell Infect. Microbiol. 2020, 10. [Google Scholar] [CrossRef]
- Maubon, D.; Dard, C.; Garnaud, C.; Cornet, M. Profile of GenMark’s EPlex® Blood Culture Identification Fungal Pathogen Panel. Expert Rev. Mol. Diagn. 2018, 18, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Maubon, D.; Hamidfar-Roy, R.; Courby, S.; Vesin, A.; Maurin, M.; Pavese, P.; Ravanel, N.; Bulabois, C.-E.; Brion, J.-P.; Pelloux, H.; et al. Therapeutic Impact and Diagnostic Performance of Multiplex PCR in Patients with Malignancies and Suspected Sepsis. J. Infect. 2010, 61, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, M.; Pandor, A.; Martyn-St James, M.; Rafia, R.; Uttley, L.; Stevens, J.; Sanderson, J.; Wong, R.; Perkins, G.D.; McMullan, R.; et al. Sepsis: The LightCycler SeptiFast Test MGRADE®, SepsiTestTM and IRIDICA BAC BSI Assay for Rapidly Identifying Bloodstream Bacteria and Fungi—A Systematic Review and Economic Evaluation. Health Technol. Assess. 2016, 20, 1–246. [Google Scholar] [CrossRef] [PubMed]
- Caspar, Y.; Garnaud, C.; Raykova, M.; Bailly, S.; Bidart, M.; Maubon, D. Superiority of SDS Lysis over Saponin Lysis for Direct Bacterial Identification from Positive Blood Culture Bottle by MALDI-TOF MS. Proteom. Clin. Appl. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.; Sánchez-Juanes, F.; Porras-Guerra, I.; García-García, M.I.; García-Sánchez, J.E.; González-Buitrago, J.M.; Muñoz-Bellido, J.L. Microorganisms Direct Identification from Blood Culture by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry: Blood Culture Pathogens Direct Identification by MALDI-TOF. Clin. Microbiol. Infect. 2011, 17, 546–551. [Google Scholar] [CrossRef]
- La Scola, B.; Raoult, D. Direct Identification of Bacteria in Positive Blood Culture Bottles by Matrix-Assisted Laser Desorption Ionisation Time-of-Flight Mass Spectrometry. PLoS ONE 2009, 4, e8041. [Google Scholar] [CrossRef]
- Mery, A.; Sendid, B.; François, N.; Cornu, M.; Poissy, J.; Guerardel, Y.; Poulain, D. Application of Mass Spectrometry Technology to Early Diagnosis of Invasive Fungal Infections. J. Clin. Microbiol. 2016, 54, 2786–2797. [Google Scholar] [CrossRef]
- Vella, A.; De Carolis, E.; Vaccaro, L.; Posteraro, P.; Perlin, D.S.; Kostrzewa, M.; Posteraro, B.; Sanguinetti, M. Rapid Antifungal Susceptibility Testing by Matrix-Assisted Laser Desorption Ionization–Time of Flight Mass Spectrometry Analysis. J. Clin. Microbiol. 2013, 51, 2964–2969. [Google Scholar] [CrossRef]
- De Carolis, E.; Vella, A.; Florio, A.R.; Posteraro, P.; Perlin, D.S.; Sanguinetti, M.; Posteraro, B. Use of Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry for Caspofungin Susceptibility Testing of Candida and Aspergillus Species. J. Clin. Microbiol. 2012, 50, 2479–2483. [Google Scholar] [CrossRef]
- Saracli, M.A.; Fothergill, A.W.; Sutton, D.A.; Wiederhold, N.P. Detection of Triazole Resistance among Candida Species by Matrix-Assisted Laser Desorption/Ionization-Time of Flight Mass Spectrometry (MALDI-TOF MS). Med. Mycol. 2015, 53, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Singh, P.; Shamanth, A.S.; Rudramurthy, S.M.; Chakrabarti, A.; Ghosh, A.K. Rapid Detection of Fluconazole Resistance in Candida Tropicalis by MALDI-TOF MS. Med. Mycol. 2018, 56, 234–241. [Google Scholar] [CrossRef]
- Roberto, A.E.M.; Xavier, D.E.; Vidal, E.E.; Vidal, C.F.d.L.; Neves, R.P.; Lima-Neto, R.G. Rapid Detection of Echinocandins Resistance by MALDI-TOF MS in Candida Parapsilosis Complex. Microorganisms 2020, 8, 109. [Google Scholar] [CrossRef] [PubMed]
- Vella, A.; De Carolis, E.; Mello, E.; Perlin, D.S.; Sanglard, D.; Sanguinetti, M.; Posteraro, B. Potential Use of MALDI-ToF Mass Spectrometry for Rapid Detection of Antifungal Resistance in the Human Pathogen Candida Glabrata. Sci. Rep. 2017, 7, 9099. [Google Scholar] [CrossRef] [PubMed]
- De Carolis, E.; Marchionni, F.; La Rosa, M.; Meis, J.F.; Chowdhary, A.; Posteraro, B.; Sanguinetti, M. Are We Ready for Nosocomial Candida Auris Infections? Rapid Identification and Antifungal Resistance Detection Using MALDI-TOF Mass Spectrometry May Be the Answer. Front. Cell Infect. Microbiol. 2021, 11, 645049. [Google Scholar] [CrossRef] [PubMed]
- Gitman, M.R.; McTaggart, L.; Spinato, J.; Poopalarajah, R.; Lister, E.; Husain, S.; Kus, J.V. Antifungal Susceptibility Testing of Aspergillus Spp. by Using a Composite Correlation Index (CCI)-Based Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry Method Appears To Not Offer Benefit over Traditional Broth Microdilution Testing. J. Clin. Microbiol. 2017, 55, 2030–2034. [Google Scholar] [CrossRef]
- Vatanshenassan, M.; Arastehfar, A.; Boekhout, T.; Berman, J.; Lass-Flörl, C.; Sparbier, K.; Kostrzewa, M. Anidulafungin Susceptibility Testing of Candida Glabrata Isolates from Blood Cultures by the MALDI Biotyper Antibiotic (Antifungal) Susceptibility Test Rapid Assay. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef]
- Vatanshenassan, M.; Boekhout, T.; Lass-Flörl, C.; Lackner, M.; Schubert, S.; Kostrzewa, M.; Sparbier, K. Proof of Concept for MBT ASTRA, a Rapid Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry (MALDI-TOF MS)-Based Method To Detect Caspofungin Resistance in Candida Albicans and Candida Glabrata. J. Clin. Microbiol. 2018, 56. [Google Scholar] [CrossRef] [PubMed]
- Knoll, M.A.; Ulmer, H.; Lass-Flörl, C. Rapid Antifungal Susceptibility Testing of Yeasts and Molds by MALDI-TOF MS: A Systematic Review and Meta-Analysis. J. Fungi 2021, 7, 63. [Google Scholar] [CrossRef] [PubMed]
- Manukumar, H.M.; Umesha, S. MALDI-TOF-MS Based Identification and Molecular Characterization of Food Associated Methicillin-Resistant Staphylococcus Aureus. Sci. Rep. 2017, 7, 11414. [Google Scholar] [CrossRef] [PubMed]
- Dhieb, C.; Normand, A.C.; Al-Yasiri, M.; Chaker, E.; El Euch, D.; Vranckx, K.; Hendrickx, M.; Sadfi, N.; Piarroux, R.; Ranque, S. MALDI-TOF Typing Highlights Geographical and Fluconazole Resistance Clusters in Candida Glabrata. Med. Mycol. 2015, 53, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Sánchez, B.; Cercenado, E.; Coste, A.T.; Greub, G. Review of the Impact of MALDI-TOF MS in Public Health and Hospital Hygiene, 2018. Euro Surveill. 2019, 24. [Google Scholar] [CrossRef]
- Prakash, A.; Sharma, C.; Singh, A.; Kumar Singh, P.; Kumar, A.; Hagen, F.; Govender, N.P.; Colombo, A.L.; Meis, J.F.; Chowdhary, A. Evidence of Genotypic Diversity among Candida Auris Isolates by Multilocus Sequence Typing, Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry and Amplified Fragment Length Polymorphism. Clin. Microbiol. Infect. 2016, 22, 277.e1–277.e9. [Google Scholar] [CrossRef]
- Bader, O. MALDI-TOF-MS-Based Species Identification and Typing Approaches in Medical Mycology. Proteomics 2013, 13, 788–799. [Google Scholar] [CrossRef]
- Piarroux, R.; Gabriel, F.; Grenouillet, F.; Collombon, P.; Louasse, P.; Piarroux, M.; Normand, A.-C. Using MALDI-ToF Mass Spectrometry to Identify Mushroom Species: Proof of Concept Analysis of Amanita Genus Specimens. Med. Mycol. 2021. [Google Scholar] [CrossRef]
- Weis, C.V.; Jutzeler, C.R.; Borgwardt, K. Machine Learning for Microbial Identification and Antimicrobial Susceptibility Testing on MALDI-TOF Mass Spectra: A Systematic Review. Clin. Microbiol. Infect. 2020, 26, 1310–1317. [Google Scholar] [CrossRef]
- Weis, C.; Horn, M.; Rieck, B.; Cuénod, A.; Egli, A.; Borgwardt, K. Topological and Kernel-Based Microbial Phenotype Prediction from MALDI-TOF Mass Spectra. Bioinformatics 2020, 36, i30–i38. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robert, M.-G.; Cornet, M.; Hennebique, A.; Rasamoelina, T.; Caspar, Y.; Pondérand, L.; Bidart, M.; Durand, H.; Jacquet, M.; Garnaud, C.; et al. MALDI-TOF MS in a Medical Mycology Laboratory: On Stage and Backstage. Microorganisms 2021, 9, 1283. https://doi.org/10.3390/microorganisms9061283
Robert M-G, Cornet M, Hennebique A, Rasamoelina T, Caspar Y, Pondérand L, Bidart M, Durand H, Jacquet M, Garnaud C, et al. MALDI-TOF MS in a Medical Mycology Laboratory: On Stage and Backstage. Microorganisms. 2021; 9(6):1283. https://doi.org/10.3390/microorganisms9061283
Chicago/Turabian StyleRobert, Marie-Gladys, Muriel Cornet, Aurélie Hennebique, Tahinamandranto Rasamoelina, Yvan Caspar, Léa Pondérand, Marie Bidart, Harmonie Durand, Marvin Jacquet, Cécile Garnaud, and et al. 2021. "MALDI-TOF MS in a Medical Mycology Laboratory: On Stage and Backstage" Microorganisms 9, no. 6: 1283. https://doi.org/10.3390/microorganisms9061283
APA StyleRobert, M.-G., Cornet, M., Hennebique, A., Rasamoelina, T., Caspar, Y., Pondérand, L., Bidart, M., Durand, H., Jacquet, M., Garnaud, C., & Maubon, D. (2021). MALDI-TOF MS in a Medical Mycology Laboratory: On Stage and Backstage. Microorganisms, 9(6), 1283. https://doi.org/10.3390/microorganisms9061283






