Immune Transcriptome of Cells Infected with Enterovirus Strains Obtained from Cases of Autoimmune Thyroid Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Samples
2.2. Cells and Virus Strains
2.3. EV Detection in Thyroid Tissue Following Enrichment in Cell Culture
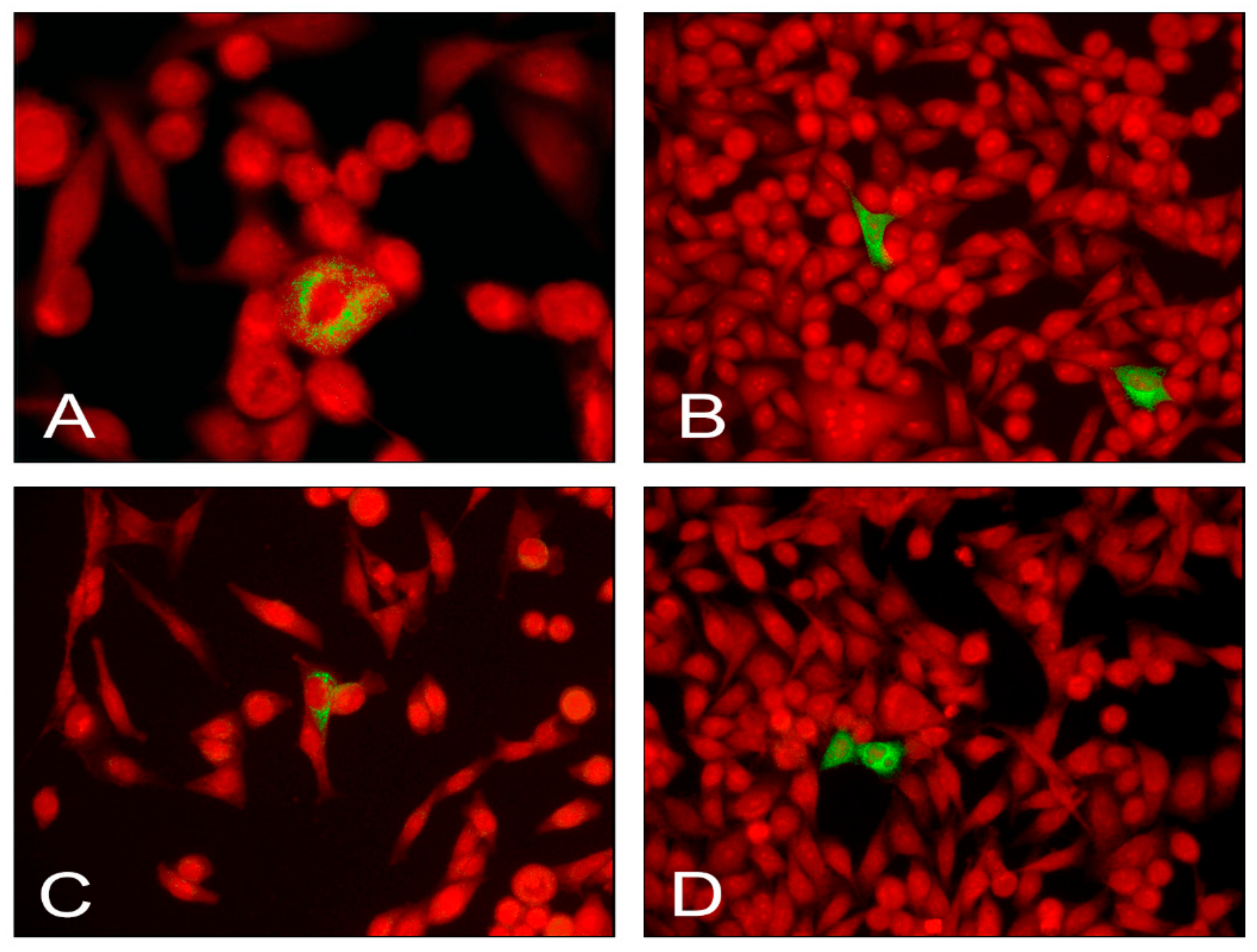
2.4. Detection of EV Antigens in Infected AV3 Cells
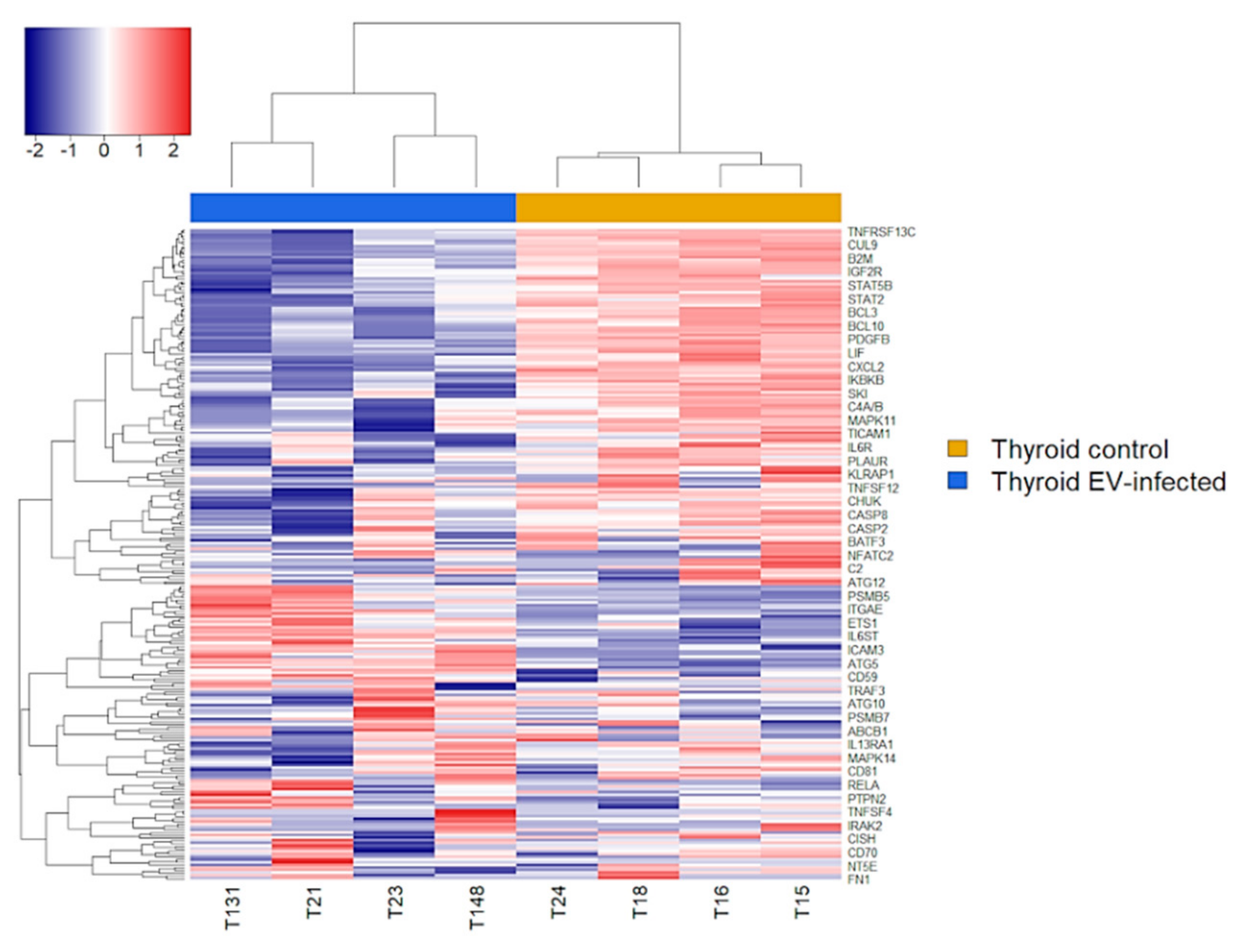
2.5. Immune Transcriptome of Uninfected and Infected AV3 Cells
2.6. Statistical Analysis
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Patil, N.; Rehman, A.; Jialal, I. Hypothyroidism. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Davies, T.F.; Andersen, S.; Latif, R.; Nagayama, Y.; Barbesino, G.; Brito, M.; Eckstein, A.K.; Stagnaro-Green, A.; Kahaly, G.J. Graves’ disease. Nat. Rev. Dis. Prim. 2020, 6, 1–23. [Google Scholar] [CrossRef]
- Li, Q.; Wang, B.; Mu, K.; Zhang, J. The pathogenesis of thyroid autoimmune diseases: New T lymphocytes—Cytokines circuits beyond the Th1−Th2 paradigm. J. Cell. Physiol. 2019, 234, 2204–2216. [Google Scholar] [CrossRef]
- Simmonds, M.J. GWAS in autoimmune thyroid disease: Redefining our understanding of pathogenesis. Nat. Rev. Endocrinol. 2013, 9, 277–287. [Google Scholar] [CrossRef]
- Dunn, N.; Kharlamova, N.; Fogdell-Hahn, A. The role of herpesvirus 6A and 6B in multiple sclerosis and epilepsy. Scand. J. Immunol. 2020, 92, e12984. [Google Scholar] [CrossRef]
- Akhbari, P.; Richardson, S.J.; Morgan, N.G. Type 1 Diabetes: Interferons and the Aftermath of Pancreatic Beta-Cell Enteroviral Infection. Microorganisms 2020, 8, 1419. [Google Scholar] [CrossRef] [PubMed]
- Apaolaza, P.S.; Balcacean, D.; Zapardiel-Gonzalo, J.; Nelson, G.; Lenchik, N.; Akhbari, P.; Gerling, I.; Richardson, S.J.; Rodriguez-Calvo, T.; nPOD-Virus Group. Islet expression of type I interferon response sensors is associated with immune infiltration and viral infection in type 1 diabetes. Sci. Adv. 2021, 7, eabd6527. [Google Scholar] [CrossRef] [PubMed]
- Cuan-Baltazar, Y.; Soto-Vega, E. Microorganisms associated to thyroid autoimmunity. Autoimmun. Rev. 2020, 19, 102614. [Google Scholar] [CrossRef] [PubMed]
- Poma, A.M.; Genoni, A.; Broccolo, F.; Denaro, M.; Pugliese, A.; Basolo, F.; Toniolo, A. Immune Transcriptome of Cells Infected with Enterovirus Strains Obtained from Cases of Type 1 Diabetes. Microorganisms 2020, 8, 1031. [Google Scholar] [CrossRef]
- Krogvold, L.; Edwin, B.; Buanes, T.; Frisk, G.; Skog, O.; Anagandula, M.; Korsgren, O.; Undlien, D.; Eike, M.C.; Richardson, S.J.; et al. Detection of a Low-Grade Enteroviral Infection in the Islets of Langerhans of Living Patients Newly Diagnosed With Type 1 Diabetes. Diabetes 2014, 64, 1682–1687. [Google Scholar] [CrossRef] [PubMed]
- Morgan, N.G.; Richardson, S.J. Enteroviruses as causative agents in type 1 diabetes: Loose ends or lost cause? Trends Endocrinol. Metab. 2014, 25, 611–619. [Google Scholar] [CrossRef]
- Toniolo, A.; Salvatoni, A.; Federico, G.; Maccari, G.; Díaz-Horta, O.; Baj, A. Enteroviruses in Blood. In Diabetes and Viruses; Taylor, K., Hyöty, H., Toniolo, A., Zuckerman, A.J., Eds.; Springer: New York, NY, USA, 2013; pp. 143–155. ISBN 978-1-4614-4051-2. [Google Scholar]
- Oikarinen, M.; Laiho, J.E.; Oikarinen, S.; Richardson, S.J.; Kusmartseva, I.; Campbell-Thompson, M.; Morgan, N.G.; Pugliese, A.; Tauriainen, S.; Toniolo, A.; et al. Detection of Enterovirus Protein and RNA in Multiple Tissues from NPOD Organ Donors with Type 1 Diabetes. bioRxiv 2018, 459347. [Google Scholar] [CrossRef]
- Richardson, S.J.; Rodriguez-Calvo, T.; Gerling, I.C.; Mathews, C.E.; Kaddis, J.S.; Russell, M.A.; Zeissler, M.; Leete, P.; Krogvold, L.; Dahl-Jørgensen, K.; et al. Islet cell hyperexpression of HLA class I antigens: A defining feature in type 1 diabetes. Diabetologia 2016, 59, 2448–2458. [Google Scholar] [CrossRef]
- Shukla, S.K.; Singh, G.; Ahmad, S.; Pant, P. Infections, genetic and environmental factors in pathogenesis of autoimmune thyroid diseases. Microb. Pathog. 2018, 116, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Weider, T.; Richardson, S.J.; Morgan, N.G.; Paulsen, T.H.; Dahl-Jørgensen, K.; Hammerstad, S.S. HLA Class I Upregulation and Antiviral Immune Responses in Graves Disease. J. Clin. Endocrinol. Metab. 2021, 106, e1763–e1774. [Google Scholar] [CrossRef] [PubMed]
- Hammerstad, S.S.; Tauriainen, S.; Hyöty, H.; Paulsen, T.; Norheim, I.; Dahl-Jørgensen, K. Detection of enterovirus in the thyroid tissue of patients with graves’ disease. J. Med Virol. 2012, 85, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Genoni, A.; Canducci, F.; Rossi, A.; Broccolo, F.; Chumakov, K.; Bono, G.; Salerno-Uriarte, J.; Salvatoni, A.; Pugliese, A.; Toniolo, A. Revealing enterovirus infection in chronic human disorders: An integrated diagnostic approach. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nix, W.A.; Oberste, M.S.; Pallansch, M.A. Sensitive, Seminested PCR Amplification of VP1 Sequences for Direct Identification of All Enterovirus Serotypes from Original Clinical Specimens. J. Clin. Microbiol. 2006, 44, 2698–2704. [Google Scholar] [CrossRef] [PubMed]
- Harvala, H.; Broberg, E.; Benschop, K.; Berginc, N.; Ladhani, S.N.; Susi, P.; Christiansen, C.; McKenna, J.; Allen, D.; Makiello, P.; et al. Recommendations for enterovirus diagnostics and characterisation within and beyond Europe. J. Clin. Virol. 2018, 101, 11–17. [Google Scholar] [CrossRef]
- Maccari, G.; Genoni, A.; Sansonno, S.; Toniolo, A. Properties of Two Enterovirus Antibodies that are Utilized in Diabetes Research. Sci. Rep. 2016, 6, 24757. [Google Scholar] [CrossRef]
- Poma, A.M.; Condello, V.; Denaro, M.; Torregrossa, L.; Elisei, R.; Vitti, P.; Basolo, F. DICER1 somatic mutations strongly impair miRNA processing even in benign thyroid lesions. Oncotarget 2019, 10, 1785–1797. [Google Scholar] [CrossRef]
- Pirofski, L.-A.; Casadevall, A. The state of latency in microbial pathogenesis. J. Clin. Investig. 2020, 130, 4525–4531. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, M.A.E.; Van De Schoor, F.R.; Vrijmoeth, H.D.; Netea, M.G.; Joosten, L.A.B. A joint effort: The interplay between the innate and the adaptive immune system in Lyme arthritis. Immunol. Rev. 2020, 294, 63–79. [Google Scholar] [CrossRef] [PubMed]
- Dunne, J.L.; Richardson, S.J.; Atkinson, M.A.; Craig, M.E.; Dahl-Jørgensen, K.; Flodström-Tullberg, M.; Hyöty, H.; Insel, R.A.; Lernmark, Å.; Lloyd, R.E.; et al. Rationale for enteroviral vaccination and antiviral therapies in human type 1 diabetes. Diabetologia 2019, 62, 744–753. [Google Scholar] [CrossRef]
- Oldstone, M.B.A. Lessons learned and concepts formed from study of the pathogenesis of the two negative-strand viruses lymphocytic choriomeningitis and influenza. Proc. Natl. Acad. Sci. USA 2013, 110, 4180–4183. [Google Scholar] [CrossRef]
- Béziat, V. Human genetic dissection of papillomavirus-driven diseases: New insight into their pathogenesis. Qual. Life Res. 2020, 139, 919–939. [Google Scholar] [CrossRef]
- Sarmiento, L.; Frisk, G.; Anagandula, M.; Cabrera-Rode, E.; Roivainen, M.; Cilio, C.M. Expression of Innate Immunity Genes and Damage of Primary Human Pancreatic Islets by Epidemic Strains of Echovirus: Implication for Post-Virus Islet Autoimmunity. PLoS ONE 2013, 8, e77850. [Google Scholar] [CrossRef]
- Sarmiento, L.; Frisk, G.; Anagandula, M.; Hodik, M.; Barchetta, I.; Netanyah, E.; Cabrera-Rode, E.; Cilio, C.M. Echovirus 6 Infects Human Exocrine and Endocrine Pancreatic Cells and Induces Pro-Inflammatory Innate Immune Response. Viruses 2017, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Hammerstad, S.S.; Stefan, M.; Blackard, J.; Owen, R.P.; Lee, H.J.; Concepcion, E.; Yi, Z.; Zhang, W.; Tomer, Y. Hepatitis C Virus E2 Protein Induces Upregulation of IL-8 Pathways and Production of Heat Shock Proteins in Human Thyroid Cells. J. Clin. Endocrinol. Metab. 2016, 102, 689–697. [Google Scholar] [CrossRef]
- Taylor, D.R.; Shi, S.T.; Romano, P.R.; Barber, G.N.; Lai, M.M.C. Inhibition of the Interferon- Inducible Protein Kinase PKR by HCV E2 Protein. Science 1999, 285, 107–110. [Google Scholar] [CrossRef]
- Ferrari, S.M.; Fallahi, P.; Antonelli, A.; Benvenga, S. Environmental Issues in Thyroid Diseases. Front. Endocrinol. 2017, 8, 50. [Google Scholar] [CrossRef] [PubMed]
- Marinko, J.T.; Huang, H.; Penn, W.D.; Capra, J.A.; Schlebach, J.P.; Sanders, C.R. Folding and Misfolding of Human Membrane Proteins in Health and Disease: From Single Molecules to Cellular Proteostasis. Chem. Rev. 2019, 119, 5537–5606. [Google Scholar] [CrossRef]
- Shi, J.; Fung, G.; Piesik, P.; Zhang, J.; Luo, H. Dominant-negative function of the C-terminal fragments of NBR1 and SQSTM1 generated during enteroviral infection. Cell Death Differ. 2014, 21, 1432–1441. [Google Scholar] [CrossRef]
- Saeed, M.; Kapell, S.; Hertz, N.T.; Wu, X.; Bell, K.; Ashbrook, A.W.; Mark, M.T.; Zebroski, H.A.; Neal, M.L.; Flodström-Tullberg, M.; et al. Defining the proteolytic landscape during enterovirus infection. PLoS Pathog. 2020, 16, e1008927. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Pang, Z.; Zhou, Z.; Li, X.; Liu, S.; Cheng, J.; Liu, P.; Tan, W.; Wang, Z.; Wang, T. Enterovirus D68 Protease 2Apro Targets TRAF3 To Subvert Host Innate Immune Responses. J. Virol. 2020, 95. [Google Scholar] [CrossRef] [PubMed]
- Tam, P.E.; Weber-Sanders, M.L.; Messner, R.P. Multiple Viral Determinants Mediate Myopathogenicity in Coxsackievirus B1-Induced Chronic Inflammatory Myopathy. J. Virol. 2003, 77, 11849–11854. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, D.; Su, M.; Sun, P.-P.; Guo, W.-P.; Wang, C.-Y.; Wang, J.-L.; Wang, H.; Zhang, Q.; Du, L.-Y.; Xie, G.-C. Global profiling of the alternative splicing landscape reveals transcriptomic diversity during the early phase of enterovirus 71 infection. Virology 2020, 548, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Giansanti, P.; Strating, J.R.P.M.; Defourny, K.A.Y.; Cesonyte, I.; Bottino, A.M.S.; Post, H.; Viktorova, E.G.; Ho, V.Q.T.; Langereis, M.A.; Belov, G.A.; et al. Dynamic remodelling of the human host cell proteome and phosphoproteome upon enterovirus infection. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef]
- Oikarinen, M.; Bertolet, L.; Toniolo, A.; Oikarinen, S.; Laiho, J.E.; Pugliese, A.; Lloyd, R.E.; Hyöty, H.; The nPOD-V Study Group. Differential Detection of Encapsidated versus Unencapsidated Enterovirus RNA in Samples Containing Pancreatic Enzymes—Relevance for Diabetes Studies. Viruses 2020, 12, 747. [Google Scholar] [CrossRef]
- Stone, V.; Ringqvist, E.; Larsson, P.; Domsgen, E.; Holmlund, U.; Sverremark-Ekström, E.; Flodström-Tullberg, M. Inhibition of Type III Interferon Expression in Intestinal Epithelial Cells—A Strategy Used by Coxsackie B Virus to Evade the Host’s Innate Immune Response at the Primary Site of Infection? Microorganisms 2021, 9, 105. [Google Scholar] [CrossRef]
- Heikki, H.; Knip, M.; Laitinen, O.; Tolonen, O.; Pulkki, M.; Oikarinen, S.; Honkanen, H.; Lecouturier, V.; Almond, J.; Flodström-tullberg, M. Multi-Cbv Vaccine for Preventing or Treating Type I Diabetes. EP2768527A1 2021. [Google Scholar]
| Group | Case No. | Classification | Sex | Age (Years) | TSH | FT4 | FT3 | TPO-Ab | Tg-Ab | TR-Ab |
|---|---|---|---|---|---|---|---|---|---|---|
| No evidence of thyroid autoimmunity | T15 | Ctrl | F | 62 | 0.63 | 13.5 | 7.5 | 34 | 19 | 0.09 |
| T16 | Ctrl | F | 55 | 0.91 | 17.3 | 6.2 | 34 | 19 | 0.09 | |
| T18 | Ctrl | F | 62 | 0.95 | 14.3 | 6.7 | 13 | 19 | 0.09 | |
| T24 | Ctrl | F | 59 | 2.4 | 12.5 | 5.7 | 34 | 19 | 0.09 | |
| Autoimmune thyroid disorders | T21 | GD | F | 69 | 0.029 | 15.7 | 8.1 | 1280 | 422 | 12 |
| T23 | GD | F | 64 | 0.029 | 25.3 | 6.3 | 429 | 269 | 88 | |
| T131 | GD | F | 38 | 0.029 | 31.1 | 17.1 | 106 | 19 | 32.6 | |
| T148 | HT | F | 41 | 1.23 | 14.4 | 5.2 | 927 | 19 | 0.09 |
| Case No. | Representative Sequence | Genome Region | Best Matching EV Species | Identities | Gaps |
|---|---|---|---|---|---|
| T21 | TAATTGGTAGTCCTCCGGCCCCTGAATGCGGCTAATCCTAACTGCGGAGCAGACACCCACGATCCAGTGGGCAGTCTGTCGTAATGGGAAACTCTGCAGCGGAACCGACTACTTTGGGTGTCCGTGTTTCCTTTTATTCTTATACTGGCTGCTTATGGTGACAATCA | 5′ UTR | B (echovirus/enterovirus B) | 161/165 | 0 |
| T23 | TGAATGCGGGCAGACACCCACGTCCAGTGGGCAGTCTGTCGTAATGGGAACTCTGCAGCGGAACCGACTACTTTGGTGTACCGTGTTTCA | 5′ UTR | B (echovirus/EV-B) | 83/89 | 5 |
| T131 | TCGTCCGTTCCCACAGTTGCCCGTTACGACTATTCCACATGGTGGCTTCCATGCAATTTTTCTGTGGGGTAGGATTATCCCGCATTCAGGGGCCGGAGGAAG | 5′ UTR | Rhinovirus C | 88/97 | 5 |
| T148 | TAACTGCAGAGCACATGCCCTCAATCCAGGGGGTGGTGTGTCGTAATGGGCAACtCTGCAGCGGAACCGACTACTTTGGTGtCCGTGTTTCAAT | 5′ UTR | A (coxsackievirus A6) | 90/92 | 1 |
| Gene | Gene Function | Log2 Fold Change | Adjusted p-Value | Up-/Down- Regulation |
|---|---|---|---|---|
| Type I IFN induction pathway (response to viral RNA) | ||||
| IFIH1 (MDA5) | Cytoplasmic sensor of viral nucleic acids. Major role in sensing viral infection and in activating the cascade of antiviral responses including the induction of type I IFNs and proinflammatory cytokines. | −0.19 | 0.436 | - |
| TLR3 | Key component of innate and adaptive immunity, a nucleotide-sensing TLR activated by dsRNA. Acts via the adapter TICAM1. | −0.05 | 0.849 | - |
| TICAM1 | Component of a multi-helicase–TICAM1 complex acting as a cytoplasmic sensor of viral dsRNA. Activates a cascade of antiviral responses, including proinflammatory cytokines. | −0.18 | 0.144 | - |
| TBK1 | Following activation of toll-like receptors by viral or bacterial components, is associated with TRAF3 and TANK and phosphorylates IFN regulatory factors IRF3 and IRF7 and DDX3X. This activity allows subsequent nuclear translocation of IRFs leading to transcriptional activation of type I IFNs and proinflammatory cytokines. Activates IRF3 by phosphorylating innate adapters MAVS, TMEM173/STING, TICAM1, thus leading to recruitment of IRF3. | −0.10 | 0.504 | - |
| IFI16 | After binding to viral DNA in the cytoplasm recruits TMEM173/STING and mediates the induction of IFN-beta. Has anti-inflammatory activity and inhibits activation of the AIM2 inflammasome | −0.33 | 0.032 | ↓ |
| TMEM173 (STING1) | Facilitator of innate immune signaling that acts as a sensor of cytosolic DNA from viruses and bacteria and promotes the production of type I IFN. | −0.19 | 0.054 | - |
| IRAK1 | IL-1 receptor associated kinase 1 phosphorylates interferon regulatory factor 7 (IRF7) to induce its activation and translocation to the nucleus, resulting in transcriptional activation of type I IFN genes. | −0.26 | 0.010 | ↓ |
| TRAF1 | Adapter molecule that regulates activation of NFKB and JNK. | −0.29 | 0.144 | - |
| TRAF2 | Regulates activation of NFKB and JNK. Regulates cell survival and apoptosis. | −0.29 | 0.015 | ↓ |
| TRAF3 | Regulates pathways leading to the activation of NFKB and MAP kinases. Regulates B cell survival. Part of signaling pathways leading to production of IFN and cytokines. Role in T cell-dependent immune responses. | −0.01 | 0.953 | - |
| TRAF4 | Activation of NFKB and JNK in response to signaling through TLRs. Regulates cell survival and apoptosis. | −0.25 | 0.017 | ↓ |
| TRAF5 | Mediates activation of NFKB. | −0.62 | 0.005 | ↓ |
| TRAF6 | Activation of NFKB and JUN. Role in dendritic cells maturation and/or activation. | −0.22 | 0.022 | ↓ |
| IKBKB (IKKB) | Ikappa kinase (IKK) is an enzyme complex that is part of the NFKB signaling pathway. The IKK complex is comprised of three subunits alpha, beta and gamma. Alpha and beta subunits are catalytically active whereas the gamma subunit has regulatory functions. | −0.53 | 0.002 | ↓ |
| IKBKG (NEMO) | −0.26 | 0.019 | ↓ | |
| IKBKE (IKKE) | Noncanonical IKB kinase (IKK) essential for regulating antiviral signaling pathways. | −0.11 | 0.528 | - |
| NFKB1 | NFKB is a homo- or heterodimeric complex formed by the Rel-like domain-containing proteins RELA/p65, RELB, NFKB1/p105, NFKB1/p50, REL and NFKB2/p52 and the heterodimeric p65–p50 complex. Dimers bind at kappa-B sites in the DNA of target genes and individual dimers have distinct preferences for different kappa-B sites. | 0.39 | 0.004 | ↑ |
| NFKB2 | NFKB2 has dual functions such as cytoplasmic retention of attached NFKB proteins by p100 and generation of p52 by cotranslational processing. | −0.41 | 0.003 | ↓ |
| NFKBIA | Member of the NFKB inhibitor family. The protein interacts with REL dimers to inhibit NFKB/REL complexes which are involved in inflammatory responses. | −0.04 | 0.690 | - |
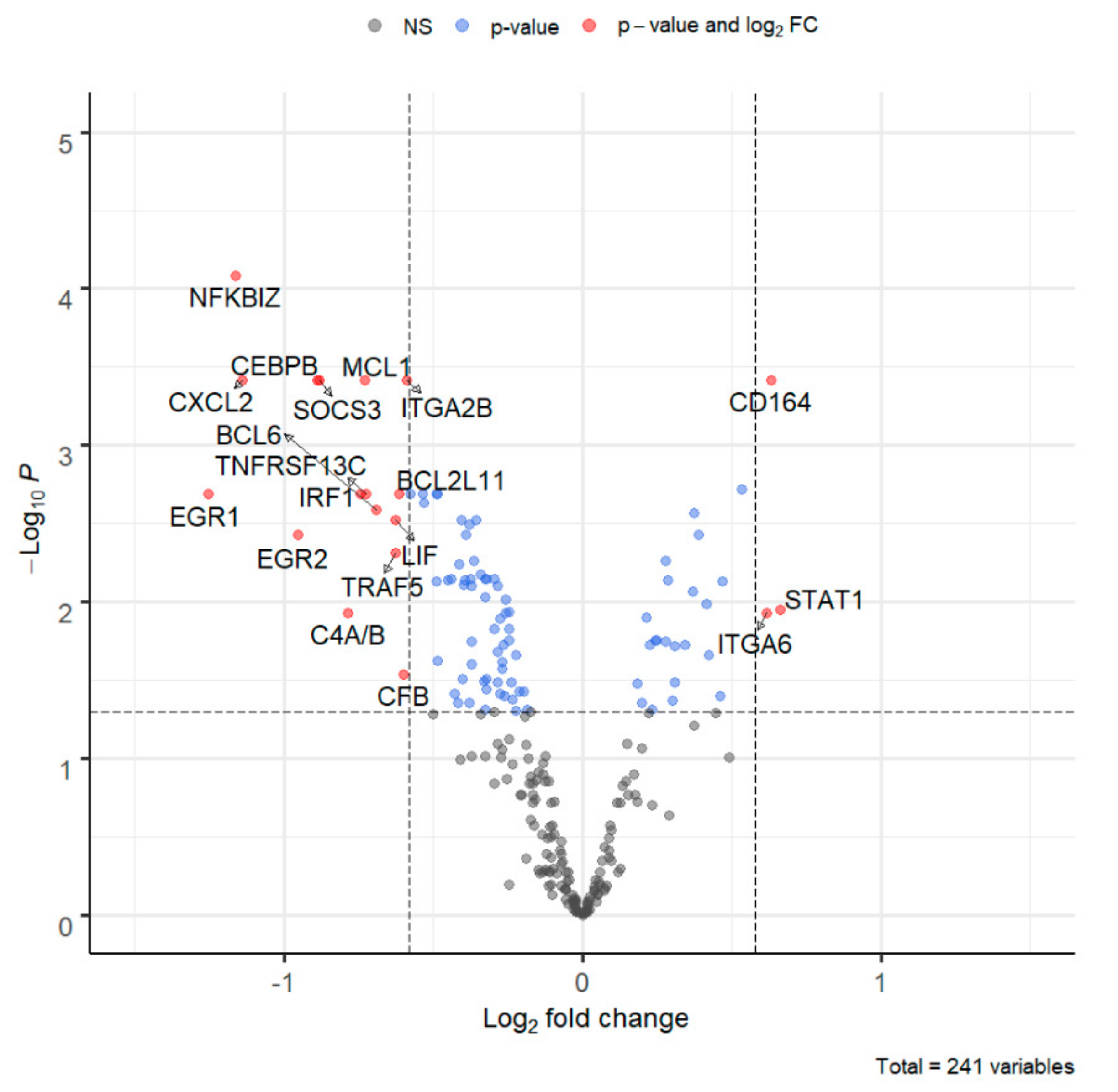
| NFKBIZ | Member of the ankyrin repeat family of proteins known to play a role in inflammatory responses. Activates IL-6 but decreases TNF-alpha production. | −1.16 | <0.001 | ↓ |
| RELA | The NF-kappa-B heterodimeric RELA–NFKB1 and RELA–REL complexes function as transcriptional activators. The NFKB homodimeric RELA–RELA complex activates IL-8 expression. | 0.46 | 0.040 | ↑ |
| RELB | NFKB heterodimeric RelB–p50 and RelB–p52 complexes are transcriptional activators. RELB is required for both T and B lymphocyte maturation and function. | 0.18 | 0.171 | - |
| IRF1 | Transcriptional regulator serving as an activator of genes involved in antiviral response, including as type I IFNs IFN-alpha/beta, DDX58/RIG-I, TNFSF10/TRAIL, OAS1/2, PIAS1/GBP, EIF2AK2/PKR and RSAD2/viperin. | −0.74 | 0.002 | ↓ |
| IRF3 | Key transcriptional regulator of type I IFN-dependent immune responses against viruses. Regulates transcription of type I IFN genes and IFN-stimulated genes by binding to an IFN-stimulated response element (ISRE). | −0.03 | 0.825 | - |
| IFITM1 | IFN-induced antiviral protein which inhibits the entry of viruses to the cytoplasm permitting endocytosis but preventing subsequent viral fusion and release of viral contents into the cytosol. | −0.44 | 0.007 | ↓ |
| Type I IFN signaling pathway (response to IFN) | ||||
| IFNAR1 | Heterodimer with IFNAR2. Type I IFN binding activates the JAK–STAT signaling cascade and triggers tyrosine phosphorylation of proteins including JAKs, TYK2, STAT and the IFNR alpha- and beta-subunits themselves. | 0.37 | 0.041 | ↑ |
| IFNAR2 | Associates with IFNAR1 to form the type I IFN receptor. Involved in IFN-mediated STAT1, STAT2 and STAT3 activation. Isoforms 1 and 2 are involved in signal transduction due to their association with JAK1. | 0.15 | 0.081 | - |
| JAK1 | Tyrosine kinase of the non-receptor type involved in the IFN-alpha/beta/gamma signal pathway. | 0.42 | 0.022 | ↑ |
| JAK2 | Tyrosine kinase of the non-receptor type involved in different processes (cell growth, differentiation, histone modifications). Mediates signaling events in both innate and adaptive immunity. In the cytoplasm, mediates signal transduction via association with type II receptors (IFN-alpha, IFN-beta, IFN-gamma and multiple interleukins). | −0.32 | 0.036 | ↓ |
| TYK2 | Tyrosine kinase that is associated with the cytoplasmic domain of type I and type II cytokine receptors and promulgates cytokine signals by phosphorylating receptor subunits. Component of type I and type III IFN signaling pathways. | −0.41 | 0.006 | ↓ |
| TBK1 | TANK binding kinase 1. TBK1 plays a key role in IRF3 activation. First phosphorylates innate adapter proteins MAVS, STING1, TICAM1 leading to recruitment and phosphorylation of IRF3. Phosphorylated IRF3 enters the nucleus to induce expression of interferons. | −0.10 | 0.504 | - |
| STAT1 | Transcription activator that mediates cellular responses to IFNs, cytokines, growth factors. Following type I IFN binding to cell receptors, signaling via protein kinases leads to activation of TYK2 and JAK1 and to tyrosine phosphorylation of STAT1 and STAT2. Phosphorylated STATs are associated with ISGF3G/IRF-9, the ISGF3 complex transcription factor that enters the nucleus and promotes transcription of IFN-stimulated genes which drive the cell in an antiviral state. | 0.66 | 0.011 | ↑ |
| STAT2 | Signal transducer and activator of transcription that mediates signaling by type I IFNs. | −0.49 | 0.007 | ↓ |
| Gene | Gene Function | Log2 Fold Change | Adjusted p-Value | Up-/Down- Regulation |
|---|---|---|---|---|
| EGR1 | Early growth response 1. Transcriptional regulator of cytokines such as IL-1B and CXCL2 that are involved in inflammatory processes. | −1.25 | 0.002 | ↓ |
| CXCL2 | Chemoattractant active on T lymphocytes and monocytes but not on neutrophils. Activates the C–X–C chemokine receptor CXCR4 to induce an increase in intracellular calcium ions and chemotaxis. | −1.14 | <0.001 | ↓ |
| EGR2 | Early growth response gene 2. Regulatory molecule suppressing excessive immune responses. | −0.95 | 0.004 | ↓ |
| CEBPB | CCAAT-enhancer-binding-protein beta. Binds to regulatory regions of several acute-phase and cytokine genes and plays a role in the regulation of acute-phase reactions and inflammation. | −0.89 | <0.001 | ↓ |
| SOCS3 | STAT-induced STAT inhibitor, suppressor of cytokine signaling. The product of this gene is induced by different cytokines and inhibits the JAK2 kinase. | −0.88 | <0.001 | ↓ |
| LIF | Cytokine of the IL-6 family. Regulates hematopoietic differentiation and neuronal cell differentiation, stimulates synthesis of acute-phase proteins in hepatocytes. | −0.62 | 0.003 | ↓ |
| PDGFB | Platelet-derived growth factor beta: regulation of cell proliferation, cell migration, survival and chemotaxis. Inhibitor of inflammatory responses, mitogen for cells of mesenchymal origin. | −0.58 | 0.002 | ↓ |
| IL6 | Functions in inflammation and maturation of B cells. Produced at sites of acute and chronic inflammation. Endogenous pyrogen in autoimmune diseases and infections. Transcriptional inflammatory response through interleukin 6 receptor-alpha. | −0.50 | 0.050 | ↓ |
| IL20 | Related to IL-10, transduces its signal through STAT3. Related pathways are ERK signaling and TGF-beta pathways. | −0.37 | 0.025 | ↓ |
| TGFB1 | Binds TGF-beta receptors activating the SMAD family transcription factors. The active form consists of a mature peptide homodimer that may form heterodimers with other TGF-beta family members. Regulates cell proliferation, differentiation, growth and expression of other factors including IFN-gamma and TNF-alpha. | −0.36 | 0.005 | ↓ |
| IL1B | Produced by activated macrophages as proprotein that is proteolytically processed to its active form by caspase 1 (CASP1/ICE). It is an important mediator of the inflammatory response and is involved in cell proliferation, differentiation, apoptosis. | −0.34 | 0.007 | ↓ |
| GPI | Glucose phosphate isomerase protein family. Within the cytoplasm, functions as a glycolytic enzyme that interconverts glucose-6-phosphate and fructose-6-phosphate. Extracellularly, GPI is also referred to as a neuroleukin and functions as a neurotrophic factor and as a lymphokine that induces immunoglobulin secretion. | −0.28 | 0.008 | ↓ |
| CCL2 (MCP1) | Chemotactic activity for myeloid and lymphoid cells, not for neutrophils or eosinophils. Binds to chemokine receptors CCR2 and CCR4. Implicated in diseases characterized by monocytic infiltrates. | 0.23 | 0.049 | ↑ |
| IL18 | Proinflammatory cytokine; augments NK cell activity in spleen cells, stimulates IFN-gamma production in T helper cells. | 0.31 | 0.032 | ↑ |
| Pathway | Statistical Mean 1 | p-Value | Number of Genes in the Pathway |
|---|---|---|---|
| hsa04722, neurotrophin signaling pathway | 0.94 | 0.032 | 19 |
| hsa04620, toll-like receptor signaling pathway | 0.88 | 0.040 | 29 |
| hsa04660, T cell receptor signaling pathway | 0.88 | 0.042 | 18 |
| hsa04380, osteoclast differentiation | 0.82 | 0.051 | 32 |
| hsa04650, natural killer cell-mediated cytotoxicity | 0.80 | 0.058 | 19 |
| hsa04662, B cell receptor signaling pathway | 0.73 | 0.075 | 19 |
| hsa04810, regulation of actin cytoskeleton | 0.71 | 0.082 | 12 |
| hsa04622, RIG-I-like receptor signaling pathway | 0.68 | 0.090 | 20 |
| hsa04370, VEGF signaling pathway | 0.65 | 0.104 | 11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poma, A.M.; Hammerstad, S.S.; Genoni, A.; Basolo, A.; Dahl-Jorgensen, K.; Toniolo, A. Immune Transcriptome of Cells Infected with Enterovirus Strains Obtained from Cases of Autoimmune Thyroid Disease. Microorganisms 2021, 9, 876. https://doi.org/10.3390/microorganisms9040876
Poma AM, Hammerstad SS, Genoni A, Basolo A, Dahl-Jorgensen K, Toniolo A. Immune Transcriptome of Cells Infected with Enterovirus Strains Obtained from Cases of Autoimmune Thyroid Disease. Microorganisms. 2021; 9(4):876. https://doi.org/10.3390/microorganisms9040876
Chicago/Turabian StylePoma, Anello Marcello, Sarah Salehi Hammerstad, Angelo Genoni, Alessio Basolo, Knut Dahl-Jorgensen, and Antonio Toniolo. 2021. "Immune Transcriptome of Cells Infected with Enterovirus Strains Obtained from Cases of Autoimmune Thyroid Disease" Microorganisms 9, no. 4: 876. https://doi.org/10.3390/microorganisms9040876
APA StylePoma, A. M., Hammerstad, S. S., Genoni, A., Basolo, A., Dahl-Jorgensen, K., & Toniolo, A. (2021). Immune Transcriptome of Cells Infected with Enterovirus Strains Obtained from Cases of Autoimmune Thyroid Disease. Microorganisms, 9(4), 876. https://doi.org/10.3390/microorganisms9040876