Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas sp. AAP5
Abstract
1. Introduction
2. Materials and Methods
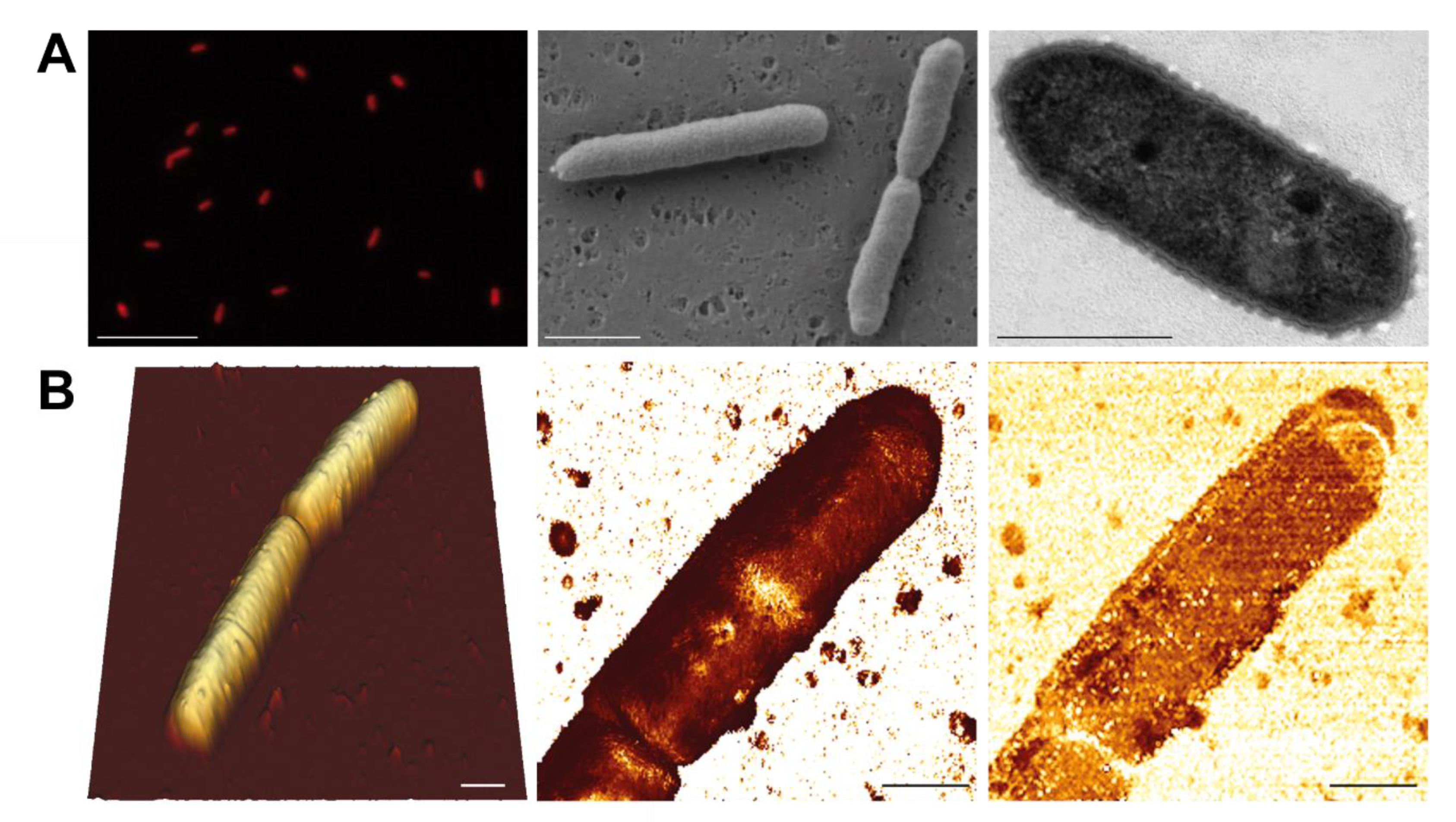
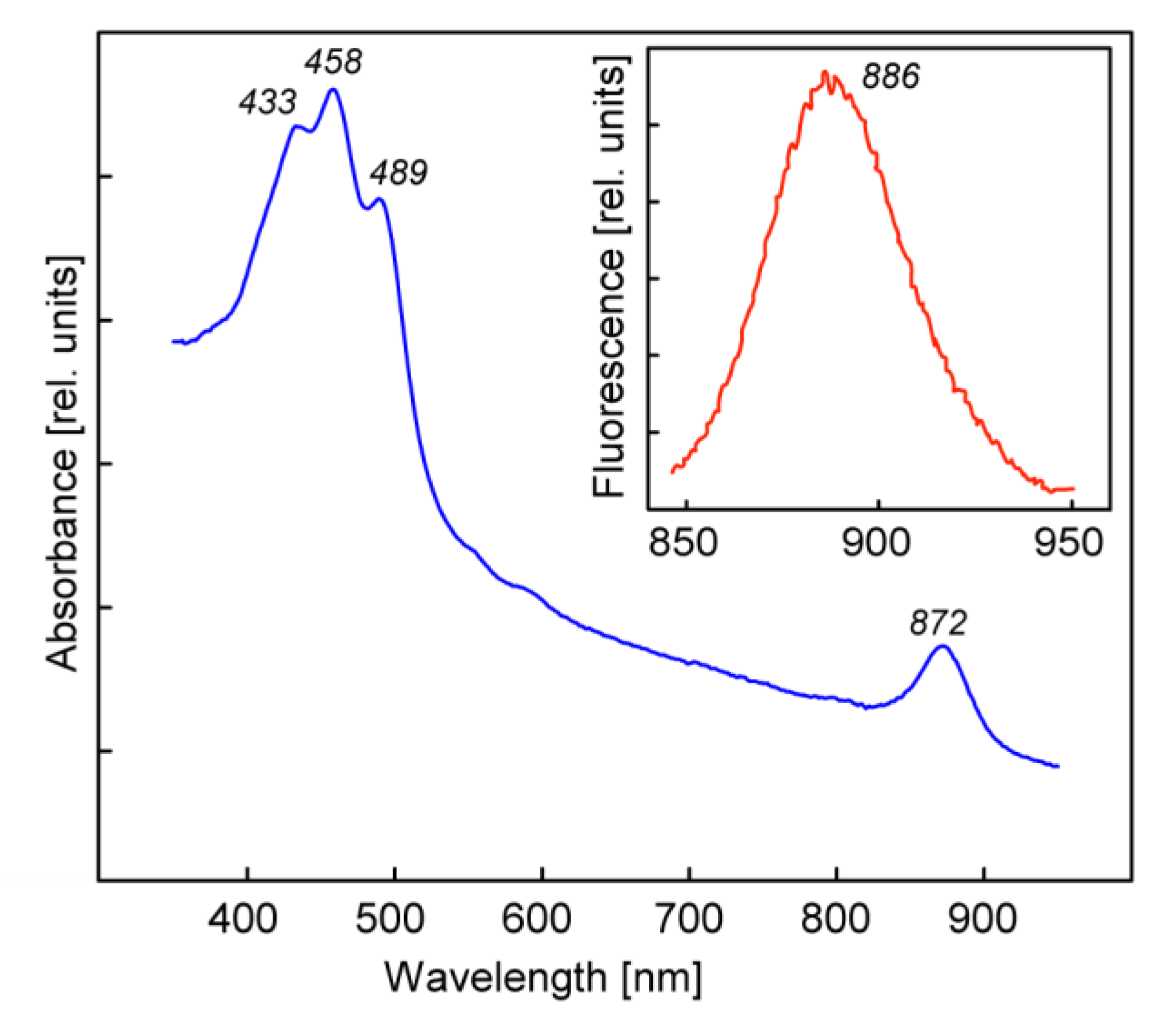
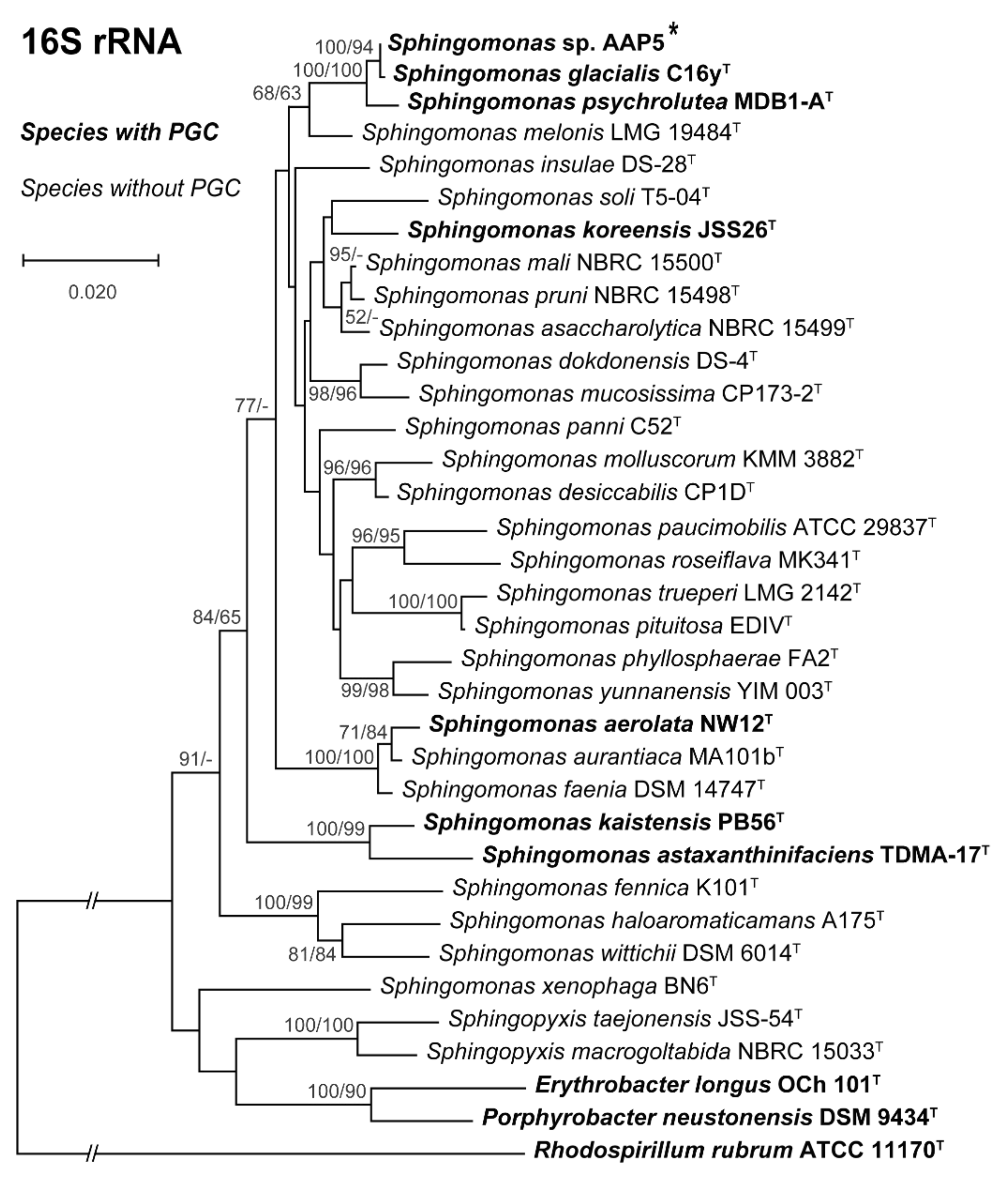
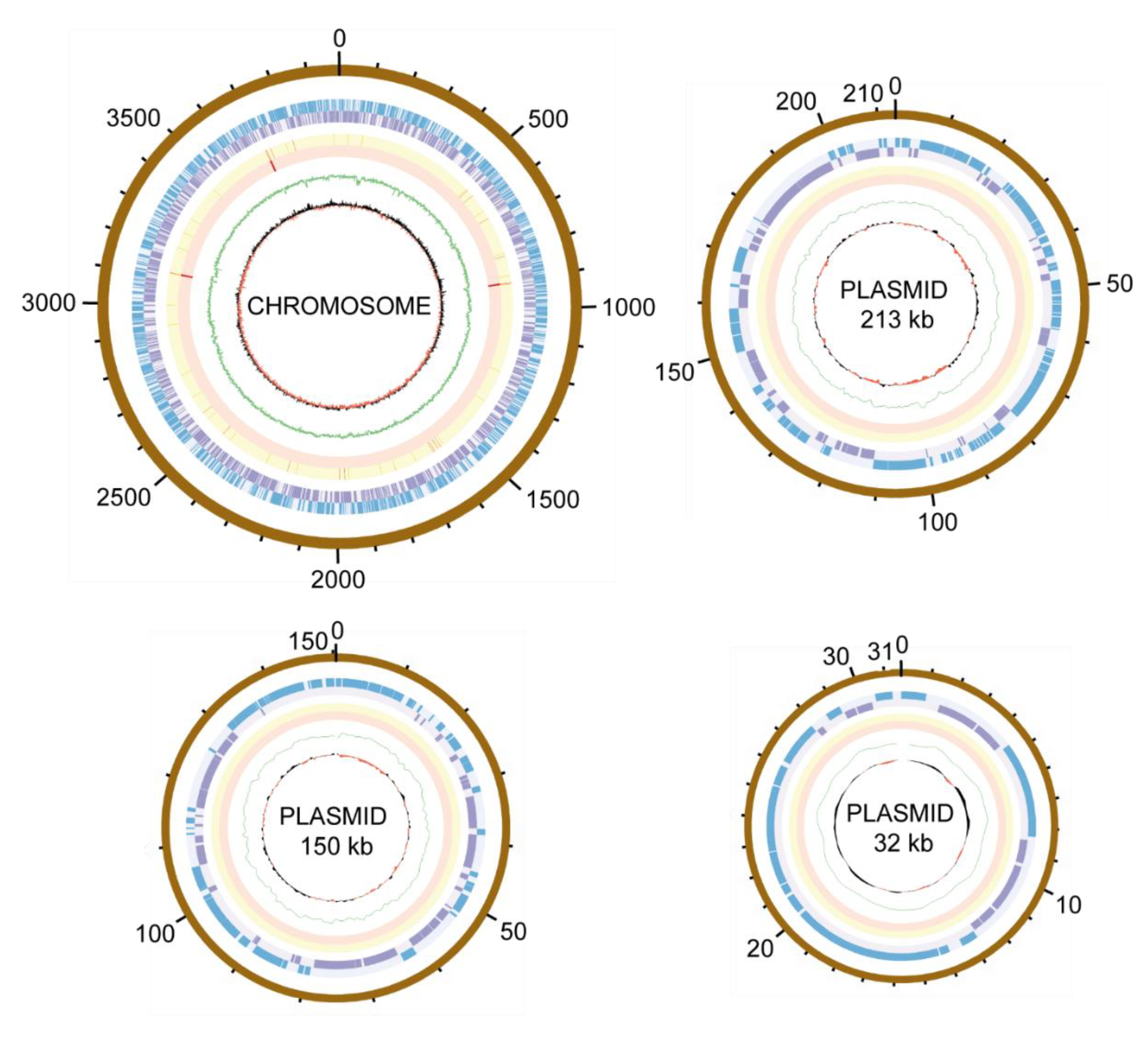
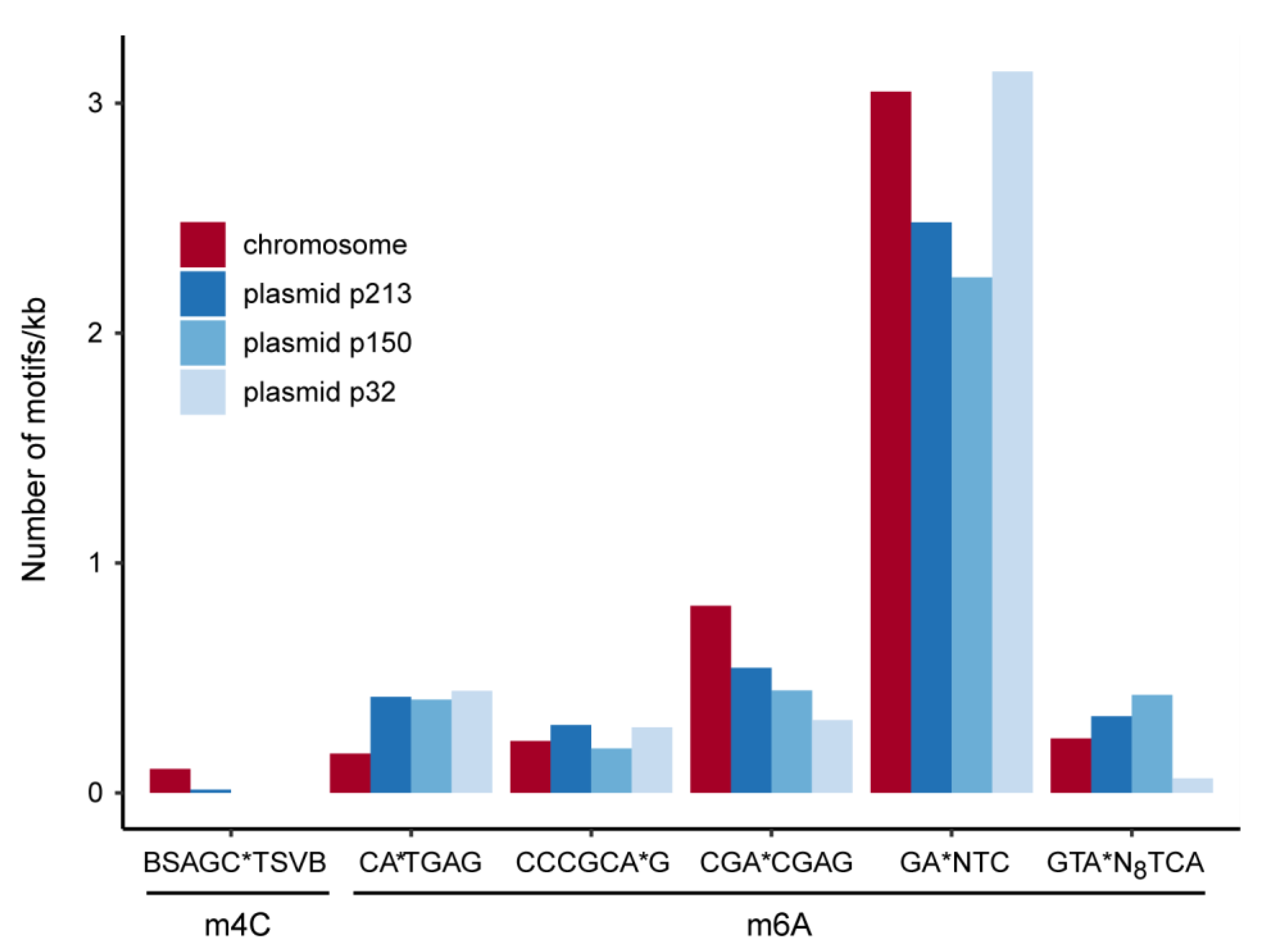
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Yabuuchi, E.; Yano, I.; Oyaizu, H.; Hashimoto, Y.; Ezaki, T.; Yamamoto, H. Proposals of Sphingomonas paucimobilis gen. nov. and comb. nov., Sphingomonas parapaucimobilis sp. nov., Sphingomonas yanoikuyae sp. nov., Sphingomonas adhaesiva sp. nov., Sphingomonas capsulata comb, nov., and Two Genospecies of the Genus. Sphingomonas. Microbiol. Immunol. 1990, 34, 99–119. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, M.; Sawada, H.; Oyaizu, H.; Yokota, A. Phylogenetic evidence for Sphingomonas and Rhizomonas as nonphotosynthetic members of the alpha-4 subclass of the Proteobacteria. Int. J. Syst. Evol. Microbiol. 1994, 44, 308–314. [Google Scholar] [CrossRef]
- White, D.C.; Sutton, S.D.; Ringelberg, D.B. The genus Sphingomonas: Physiology and ecology. Curr. Opin. Biotechnol. 1996, 7, 301–306. [Google Scholar] [CrossRef]
- Takeuchi, M.; Hamana, K.; Hiraishi, A. Proposal of the genus Sphingomonas sensus stricto and three new genera, Sphingobium, Novosphingobium Sphingopyxis, on the basis of phylogenetic and chemotaxonomic analyses. Int. J. Syst. Evol. Microbiol. 2001, 51, 1405–1417. [Google Scholar] [CrossRef]
- Hiraishi, A.; Kuraishi, H.; Kawahara, K. Emendation of the description of Blastomonas natatoria (Sly 1985) Sly and Cahill 1997 as an aerobic photosynthetic bacterium and reclassification of Erythromonas ursincola Yurkov et al. 1997 as Blastomonas ursincola comb. nov. Int. J. Syst. Evol. Microbiol. 2000, 50, 1113–1118. [Google Scholar] [CrossRef]
- Kim, M.K.; Schubert, K.; Im, W.T.; Kim, K.H.; Lee, S.T.; Overmann, J.; Affiliations, V. Sphingomonas kaistensis sp. nov., a novel alphaproteobacterium containing pufLM genes. Int. J. Syst. Evol. Microbiol. 2007, 57, 1527–1534. [Google Scholar] [CrossRef]
- Salka, I.; Srivastava, A.; Allgaier, M.; Grossart, H.P. The draft genome sequence of Sphingomonas sp. strain FukuSWIS1, obtained from acidic Lake Grosse Fuchskuhle, indicates photoheterotrophy and a potential for humic matter degradation. Genome Announc. 2014, 2, e01183-14. [Google Scholar] [CrossRef]
- Tahon, G.; Willems, A. Isolation and characterization of aerobic anoxygenic phototrophs from exposed soils from the Sør Rondane Mountains, East Antarctica. Syst. Appl. Microbiol. 2017, 40, 357–369. [Google Scholar] [CrossRef]
- Huang, H.D.; Wang, W.; Ma, T.; Li, G.Q.; Liang, F.L.; Liu, R.-L. Sphingomonas sanxanigenens sp. nov., isolated from soil. Int. J. Syst. Evol. Microbiol. 2009, 59, 719–723. [Google Scholar] [CrossRef]
- Manandhar, P.; Zhang, G.; Lama, A.; Liu, F.; Hu, Y. Sphingomonas montana sp. nov., isolated from a soil sample from the Tanggula Mountain in the Qinghai Tibetan Plateau. Antonie Leeuwenhoek 2017, 110, 1659–1668. [Google Scholar] [PubMed]
- Asker, D.; Beppu, T.; Ueda, K. Sphingomonas jaspsi sp. nov., a novel carotenoid-producing bacterium isolated from Misasa, Tottori, Japan. Int. J. Syst. Evol. Microbiol. 2007, 57, 1435–1441. [Google Scholar] [CrossRef]
- Čuperová, Z.; Holzer, E.; Salka, I.; Sommaruga, R.; Koblížek, M. Temporal changes and altitudinal distribution of aerobic anoxygenic phototrophs in mountain lakes. Appl. Environ. Microbiol. 2013, 79, 6439–6446. [Google Scholar]
- Marizcurrena, J.J.; Morales, D.; Smircich, P.; Castro-Sowinski, S. Draft Genome Sequence of the UV-Resistant Antarctic Bacterium Sphingomonas sp. Strain UV9. Microbiol. Resour. Announc. 2019, 8, e01651-18. [Google Scholar] [CrossRef] [PubMed]
- Buonaurio, R.; Stravato, V.M.; Kosako, Y.; Fujiwara, N.; Naka, T.; Kobayashi, K.; Cappelli, C.; Yabuuchi, E. Sphingomonas melonis sp. nov., a novel pathogen that causes brown spots on yellow Spanish melon fruits. Int. J. Syst. Evol. Microbiol. 2002, 52, 2081–2087. [Google Scholar]
- Shin, S.C.; Ahn, D.H.; Lee, J.K.; Kim, S.J.; Hong, S.G.; Kim, E.H.; Park, H. Genome sequence of Sphingomonas sp. strain PAMC 26605, isolated from arctic lichen (Ochrolechia sp.). J. Bacteriol. 2012, 194, 1607. [Google Scholar] [CrossRef]
- Busse, H.J.; Denner, E.B.; Buczolits, S.; Salkinoja-Salonen, M.; Bennasar, A.; Kämpfer, P. Sphingomonas aurantiaca sp. nov., Sphingomonas aerolata sp. nov. and Sphingomonas faeni sp. nov., air-and dustborne and Antarctic, orange-pigmented, psychrotolerant bacteria, and emended description of the genus Sphingomonas. Int. J. Syst. Evol. Microbiol. 2003, 53, 1253–1260. [Google Scholar] [CrossRef]
- Amato, P.; Parazols, M.; Sancelme, M.; Laj, P.; Mailhot, G.; Delort, A.-M. Microorganisms isolated from the water phase of tropospheric clouds at the Puy de Dôme: Major groups and growth abilities at low temperatures. FEMS Microbiol. Ecol. 2007, 59, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Angelakis, E.; Roux, V.; Raoult, D. Sphingomonas mucosissima bacteremia in patient with sickle cell disease. Emerg. Infect. Dis. 2009, 15, 133. [Google Scholar] [PubMed]
- Ryan, M.P.; Adley, C.C. Sphingomonas paucimobilis: A persistent Gram-negative nosocomial infectious organism. J. Hosp. Infect. 2010, 75, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Denner, E.B.; Paukner, S.; Kämpfer, P.; Moore, E.R.; Abraham, W.R.; Busse, H.J.; Wanner, G.; Lubitz, W. Sphingomonas pituitosa sp. nov., an exopolysaccharide-producing bacterium that secretes an unusual type of sphingan. Int. J. Syst. Evol. Microbiol. 2001, 51, 827–841. [Google Scholar] [CrossRef] [PubMed]
- Gai, Z.; Wang, X.; Zhang, X.; Su, F.; Tang, H.; Tai, C.; Tao, F.; Ma, C.; Xu, P. Genome sequence of Sphingomonas elodea ATCC 31461, a highly productive industrial strain of gellan gum. J. Bacteriol. 2011, 193, 7015–7016. [Google Scholar] [CrossRef]
- Kera, Y.; Abe, K.; Kasai, D.; Fukuda, M.; Takahashi, S. Draft genome sequences of Sphingobium sp. strain TCM1 and Sphingomonas sp. strain TDK1, haloalkyl phosphate flame retardant-and plasticizer-degrading bacteria. Genome Announc. 2016, 4, e00668-16. [Google Scholar] [CrossRef]
- Kopejtka, K.; Tomasch, J.; Zeng, Y.; Selyanin, V.; Dachev, M.; Piwosz, K.; Tichý, M.; Bína, D.; Gardian, Z.; Bunk, B.; et al. Simultaneous presence of bacteriochlorophyll and xanthorhodopsin genes in a freshwater bacterium. mSystems 2020, 5, e01044-20. [Google Scholar] [CrossRef]
- Shiba, T.; Simidu, U. Erythrobacter longus gen. nov., sp. nov., an aerobic bacterium which contains bacteriochlorophyll a. Int. J. Syst. Bacteriol. 1982, 32, 211–217. [Google Scholar] [CrossRef]
- Yurkov, V.V.; Csotonyi, J.T. New light on aerobic anoxygenic phototrophs. In The Purple Phototrophic Bacteria. Advances in Photosynthesis and Respiration; Hunter, C.N., Daldal, F., Thurnauer, M.C., Beatty, J.T., Eds.; Springer Verlag: Dordrecht, The Netherlands, 2009; Volume 28, pp. 31–35. [Google Scholar]
- Zeng, Y.; Feng, F.; Medová, H.; Dean, J.; Koblížek, M. Functional type 2 photosynthetic reaction centers found in the rare bacterial phylum Gemmatimonadetes. Proc. Natl. Acad. Sci. USA 2014, 111, 7795–7800. [Google Scholar] [CrossRef]
- Coleman, A.W. Enhanced detection of bacteria in natural environments by fluorochrome staining of DNA. Limnol. Oceanogr. 1980, 25, 948–951. [Google Scholar] [CrossRef]
- Fecskeová, L.K.; Piwosz, K.; Hanusová, M.; Nedoma, J.; Znachor, P.; Koblížek, M. Diel changes and diversity of pufM expression in freshwater communities of anoxygenic phototrophic bacteria. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Sader, J.E.; Chon, J.W.; Mulvaney, P. Calibration of rectangular atomic force microscope cantilevers. Rev. Sci. Instrum. 1999, 70, 3967–3969. [Google Scholar] [CrossRef]
- Hertz, H. Über die Berührung fester elastischer Körper. J. Reine Angew. Math. 1882, 92, 156–171. [Google Scholar]
- Sneddon, I.N. The relation between load and penetration in the axisymmetric Boussinesq problem for a punch of arbitrary profile. Int. J. Eng. Sci. 1965, 3, 47–57. [Google Scholar] [CrossRef]
- Rico, F.; Roca-Cusachs, P.; Gavara, N.; Farré, R.; Rotger, M.; Navajas, D. Probing mechanical properties of living cells by atomic force microscopy with blunted pyramidal cantilever tips. Phys. Rev. E 2005, 72, 021914. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Felsenstein, J. Evolutionary trees from DNA sequences: A maximum likelihood approach. J. Mol. Evol. 1981, 17, 368–376. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Nei, M. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol. 1993, 10, 512–526. [Google Scholar]
- Tavaré, S. Some probabilistic and statistical problems in the analysis of DNA sequences. Some Mathematical Questions in Biology: DNA Sequence Analysis. In Lectures on Mathematics in the Life Sciences, 2nd ed.; Miura, R.M., Ed.; The American Mathematical Society: Providence, RI, USA,, 1986; Volume 17, pp. 57–86. [Google Scholar]
- Le, S.Q.; Gascuel, O. An improved general amino acid replacement matrix. Mol. Biol. Evol. 2008, 25, 1307–1320. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Göker, M.; Spröer, C.; Klenk, H.P. When should a DDH experiment be mandatory in microbial taxonomy? Arch. Microbiol. 2013, 195, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.H.; Ha, S.M.; Lim, J.; Kwon, S.; Chun, J. A large-scale evaluation of algorithms to calculate average nucleotide identity. Antonie Leeuwenhoek 2017, 110, 1281–1286. [Google Scholar]
- Xu, L.; Dong, Z.; Fang, L.; Luo, Y.; Wei, Z.; Guo, H.; Zhang, G.; Gu, Y.Q.; Coleman-Derr, D.; Xia, Q.; et al. OrthoVenn2: A web server for whole-genome comparison and annotation of orthologous clusters across multiple species. Nucleic Acids Res. 2019, 47, W52–W58. [Google Scholar] [CrossRef]
- Bartling, P.; Brinkmann, H.; Bunk, B.; Overmann, J.; Göker, M.; Petersen, J. The Composite 259-kb Plasmid of Martelella mediterranea DSM 17316T–A Natural Replicon with Functional RepABC Modules from Rhodobacteraceae and Rhizobiaceae. Front. Microbiol. 2017, 8, 1787. [Google Scholar] [CrossRef]
- Bartling, P.; Vollmers, J.; Petersen, J. The first world swimming championships of roseobacters—Phylogenomic insights into an exceptional motility phenotype. Syst. Appl. Microbiol. 2018, 41, 544–554. [Google Scholar] [CrossRef]
- Zeng, Y.; Nupur Wu, N.; Madsen, A.M.; Chen, X.; Gardiner, A.T.; Koblížek, M. Gemmatimonas groenlandica sp. nov. is an aerobic anoxygenic phototroph in the phylum Gemmatimonadetes. Front. Microbiol. 2021, 11, 606612. [Google Scholar] [CrossRef]
- Zhang, D.C.; Busse, H.J.; Liu, H.C.; Zhou, Y.G.; Schinner, F.; Margesin, R. Sphingomonas glacialis sp. nov., a psychrophilic bacterium isolated from alpine glacier cryoconite. Int. J. Syst. Evol. Microbiol. 2011, 61, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Liu, H.C.; Zhang, J.L.; Zhou, Y.G.; Xin, Y.H. Sphingomonas psychrolutea sp. nov., a psychrotolerant bacterium isolated from glacier ice. Int. J. Syst. Evol. Microbiol. 2015, 65, 2955–2959. [Google Scholar] [CrossRef] [PubMed]
- Holmes, B.; Owen, R.J.; Evans, A.; Malnick, H.; Willcox, W.R. Pseudomonas paucimobilis, a new species isolated from human clinical specimens, the hospital environment, and other sources. Int. J. Syst. Evol. Microbiol. 1977, 27, 133–146. [Google Scholar] [CrossRef]
- Goris, J.; Konstantinidis, K.T.; Klappenbach, J.A.; Coenye, T.; Vandamme, P.; Tiedje, J.M. DNA–DNA hybridization values and their relationship to whole-genome sequence similarities. Int. J. Sys. Evol. Microbiol. 2007, 57, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Lang, A.S.; Beatty, J.T. Genetic analysis of a bacterial genetic exchange element: The gene transfer agent of Rhodobacter capsulatus. Proc. Nat. Acad. Sci. USA 2000, 97, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Imhoff, J.F.; Rahn, T.; Künzel, S.; Neulinger, S.C. Photosynthesis is widely distributed among Proteobacteria as demonstrated by the phylogeny of PufLM reaction center proteins. Front. Microbiol. 2018, 8, 2679. [Google Scholar] [CrossRef]
- Imhoff, J.F.; Rahn, T.; Künzel, S.; Neulinger, S.C. Phylogeny of anoxygenic photosynthesis based on sequences of photosynthetic reaction center proteins and a key enzyme in bacteriochlorophyll biosynthesis, the chlorophyllide reductase. Microorganisms 2019, 7, 576. [Google Scholar] [CrossRef] [PubMed]
- Flusberg, B.; Webster, D.; Lee, J.; Travers, K.J.; Olivares, E.C.; Clark, T.A.; Korlach, J.; Turner, S.W. Direct detection of DNA methylation during single-molecule, real-time sequencing. Nat. Methods 2010, 7, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Blow, M.J.; Clark, T.A.; Daum, C.G.; Deutschbauer, A.M.; Fomenkov, A.; Fries, R.; Froula, J.; Kang, D.D.; Malmstrom, R.R.; Morgan, R.D.; et al. The epigenomic landscape of prokaryotes. PLoS Genet. 2016, 12, e1005854. [Google Scholar] [CrossRef] [PubMed]
- Zweiger, G.; Marczynski, G.; Shapiro, L. A Caulobacter DNA methyltransferase that functions only in the predivisional cell. J. Mol. Biol. 1994, 235, 472–485. [Google Scholar] [CrossRef]
- Gonzalez, D.; Kozdon, J.B.; McAdams, H.H.; Shapiro, L.; Collier, J. The functions of DNA methylation by CcrM in Caulobacter crescentus: A global approach. Nucleic Acids Res. 2014, 42, 3720–3735. [Google Scholar] [CrossRef]
- Domian, I.J.; Reisenauer, A.; Shapiro, L. Feedback control of a master bacterial cell-cycle regulator. Proc. Nat. Acad. Sci. 1999, 96, 6648–6653. [Google Scholar] [CrossRef]
- Gonzalez, D.; Collier, J. DNA methylation by CcrM activates the transcription of two genes required for the division of Caulobacter crescentus. Mol. Microbiol. 2013, 88, 203–218. [Google Scholar] [CrossRef]
- Loenen, W.A.; Dryden, D.T.; Raleigh, E.A.; Wilson, G.G.; Murray, N.E. Highlights of the DNA cutters: A short history of the restriction enzymes. Nucleic Acids Res. 2014, 42, 3–19. [Google Scholar] [CrossRef]

| Parameter | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| Colony color | Yellow | Yellow | Orange-yellow | Deep yellow | Yellow |
| Cell width [μm] | 0.7–0.8 | 0.5 | 0.5–0.6 | 0.68–0.85 | 0.7 |
| Cell length [μm] | 1.1–2.3 | 0.8 | 1.8–2.2 | 1.2–1.9 | 1.4 |
| Motility | No | No | No | No | Yes |
| Genome characteristics ‡ | |||||
| G + C content [%] | 65.9 | 65.7 | 64.2 | 67.1 | 65.7 |
| PGC | Yes | Yes | Yes | No | No |
| Xanthorhodopsin | Yes | Yes | Yes | No | No |
| Utilization of | |||||
| D-mannose | Yes | No | Yes | Yes | No |
| D-melibiose | Yes | No | No | No | Yes |
| L-rhamnose | Yes | No | No | No | No |
| Phenylacetate | No | No | No | Yes | No |
| Antibiotics resistance | |||||
| Penicillin G 100 μg ml−1 | No | Yes | n.d. | Yes | Yes |
| Tetracycline 30 μg ml−1 | Yes | No | n.d. | No | No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopejtka, K.; Zeng, Y.; Kaftan, D.; Selyanin, V.; Gardian, Z.; Tomasch, J.; Sommaruga, R.; Koblížek, M. Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas sp. AAP5. Microorganisms 2021, 9, 768. https://doi.org/10.3390/microorganisms9040768
Kopejtka K, Zeng Y, Kaftan D, Selyanin V, Gardian Z, Tomasch J, Sommaruga R, Koblížek M. Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas sp. AAP5. Microorganisms. 2021; 9(4):768. https://doi.org/10.3390/microorganisms9040768
Chicago/Turabian StyleKopejtka, Karel, Yonghui Zeng, David Kaftan, Vadim Selyanin, Zdenko Gardian, Jürgen Tomasch, Ruben Sommaruga, and Michal Koblížek. 2021. "Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas sp. AAP5" Microorganisms 9, no. 4: 768. https://doi.org/10.3390/microorganisms9040768
APA StyleKopejtka, K., Zeng, Y., Kaftan, D., Selyanin, V., Gardian, Z., Tomasch, J., Sommaruga, R., & Koblížek, M. (2021). Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas sp. AAP5. Microorganisms, 9(4), 768. https://doi.org/10.3390/microorganisms9040768






