Helminth Interactions with Bacteria in the Host Gut Are Essential for Its Immunomodulatory Effect
Abstract
1. Introduction
2. Material and Methods
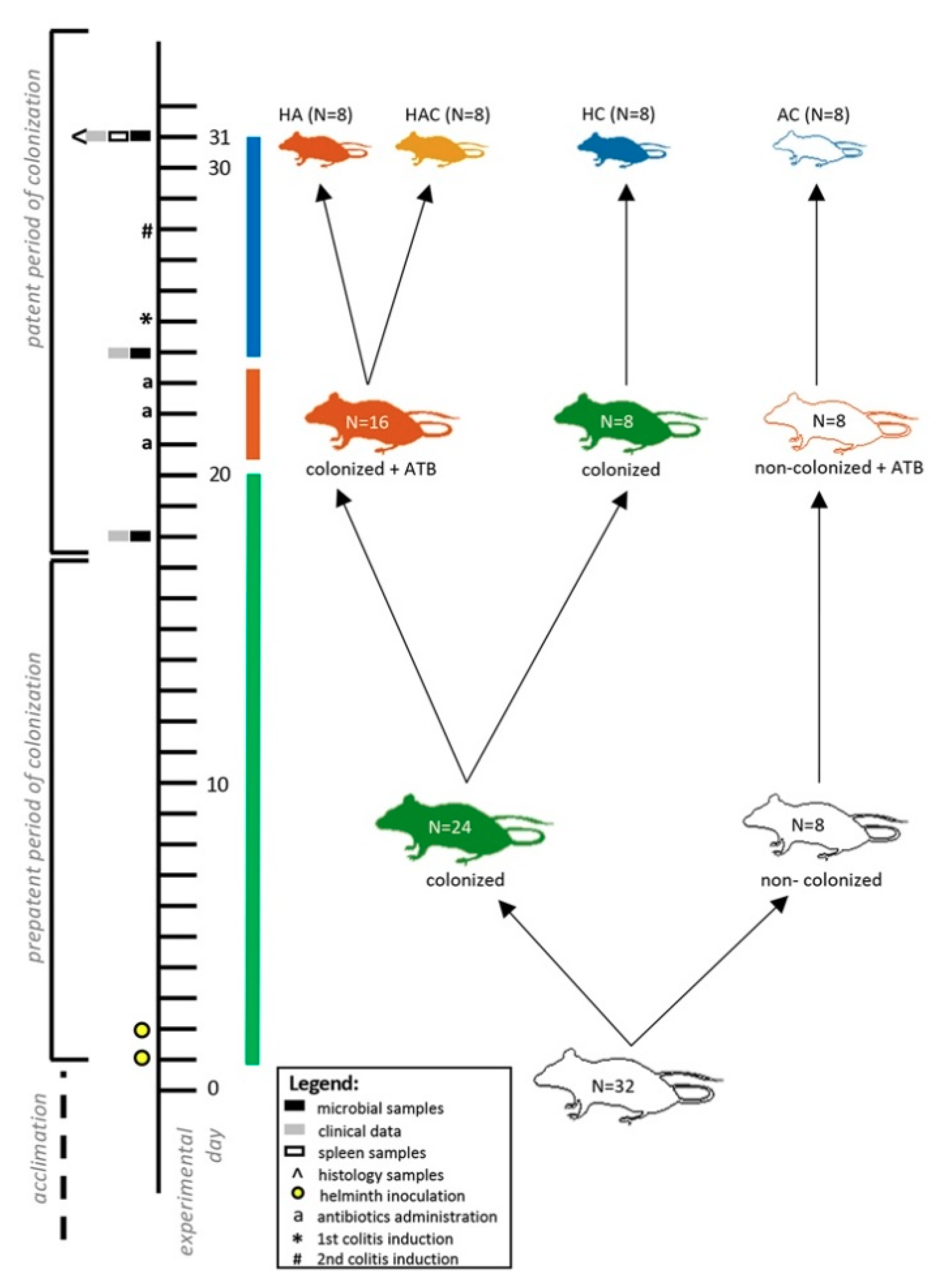
2.1. Ethics, Experimental Design, and Sampling
2.2. Antibiotics Administration and Colitis Induction
2.3. Analyses of Immune Markers
2.4. Histology
2.5. Microbial DNA Extraction, Amplification, and Sequence Processing
3. Results
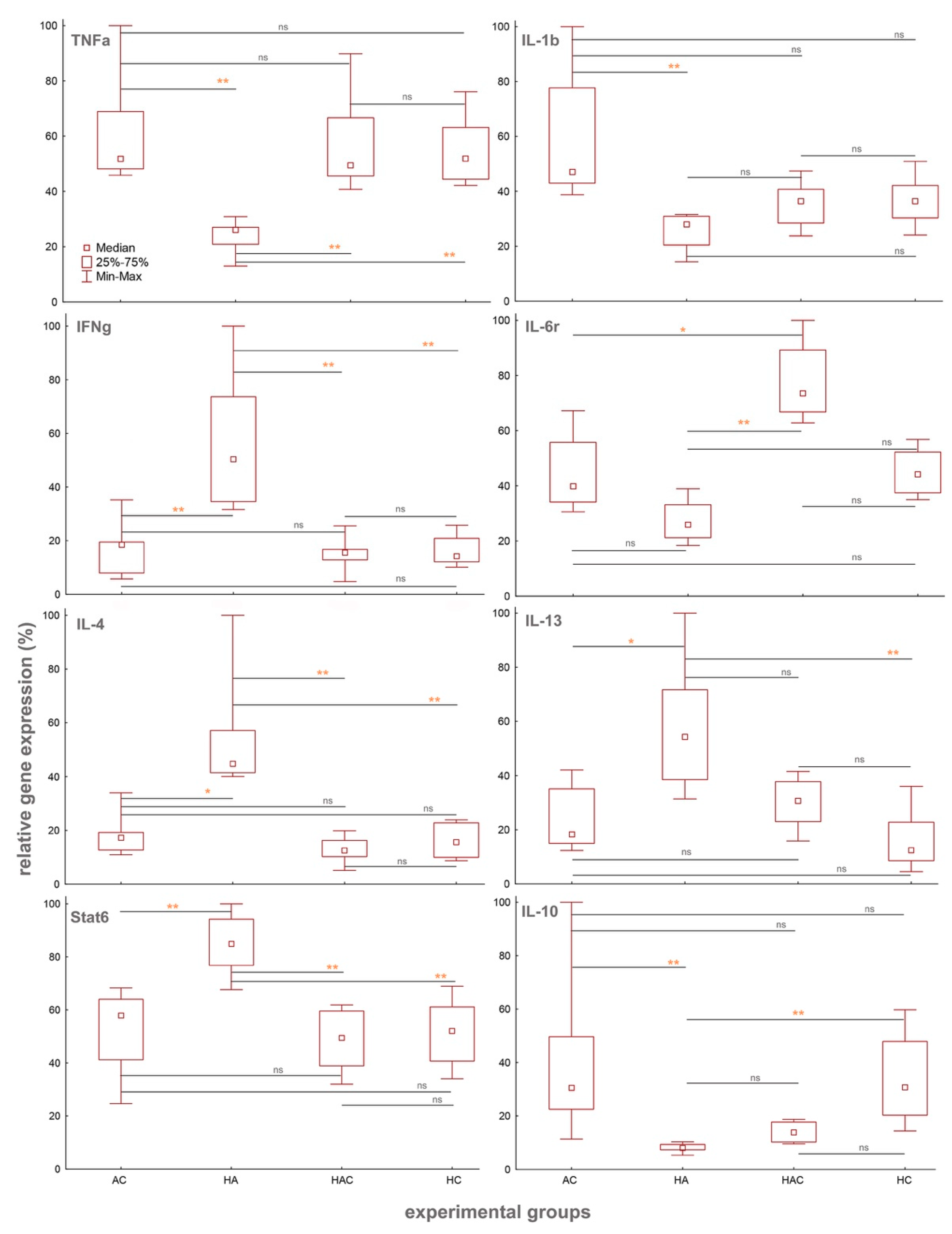
3.1. Cytokine Results
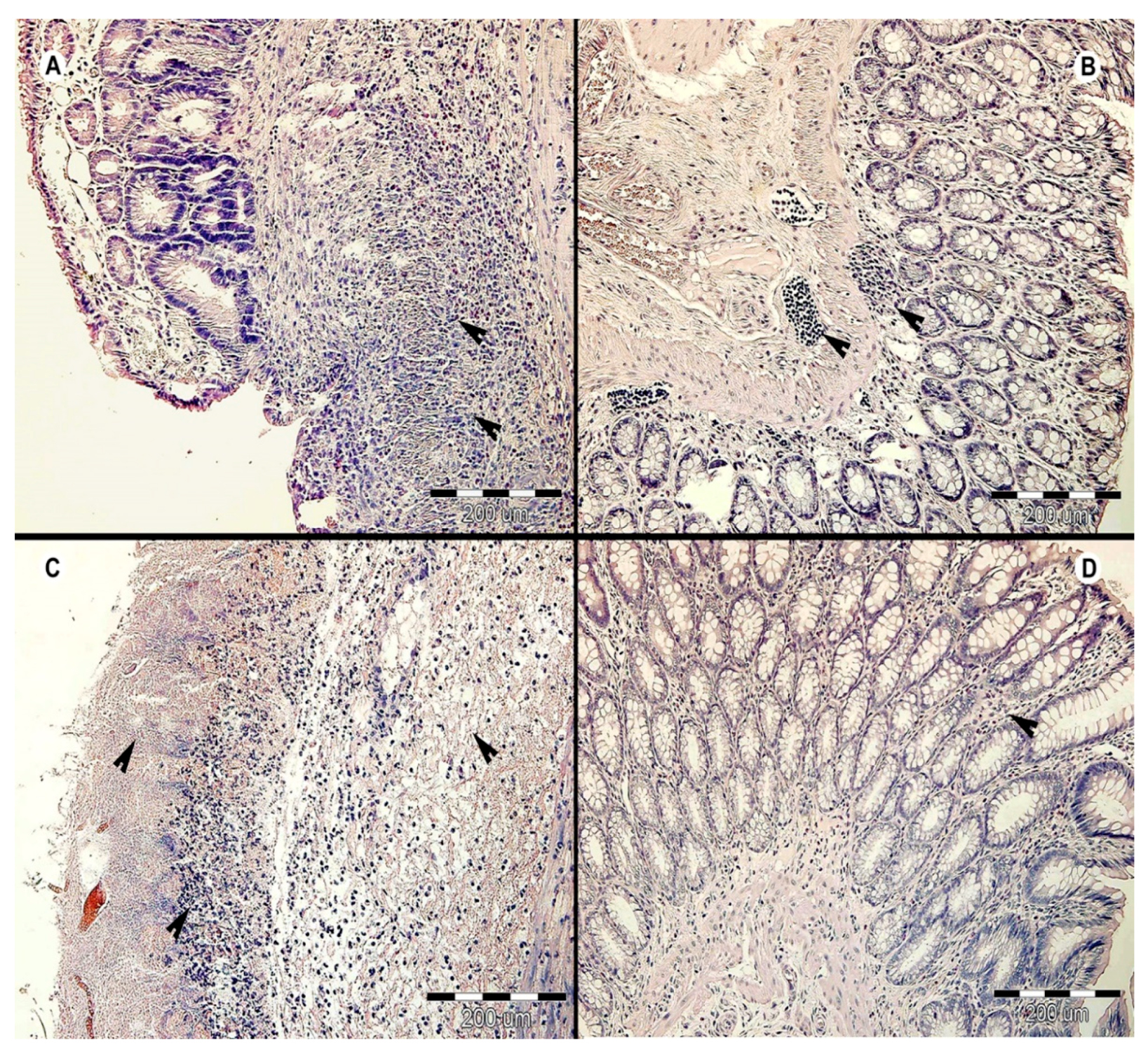
3.2. Histology Results
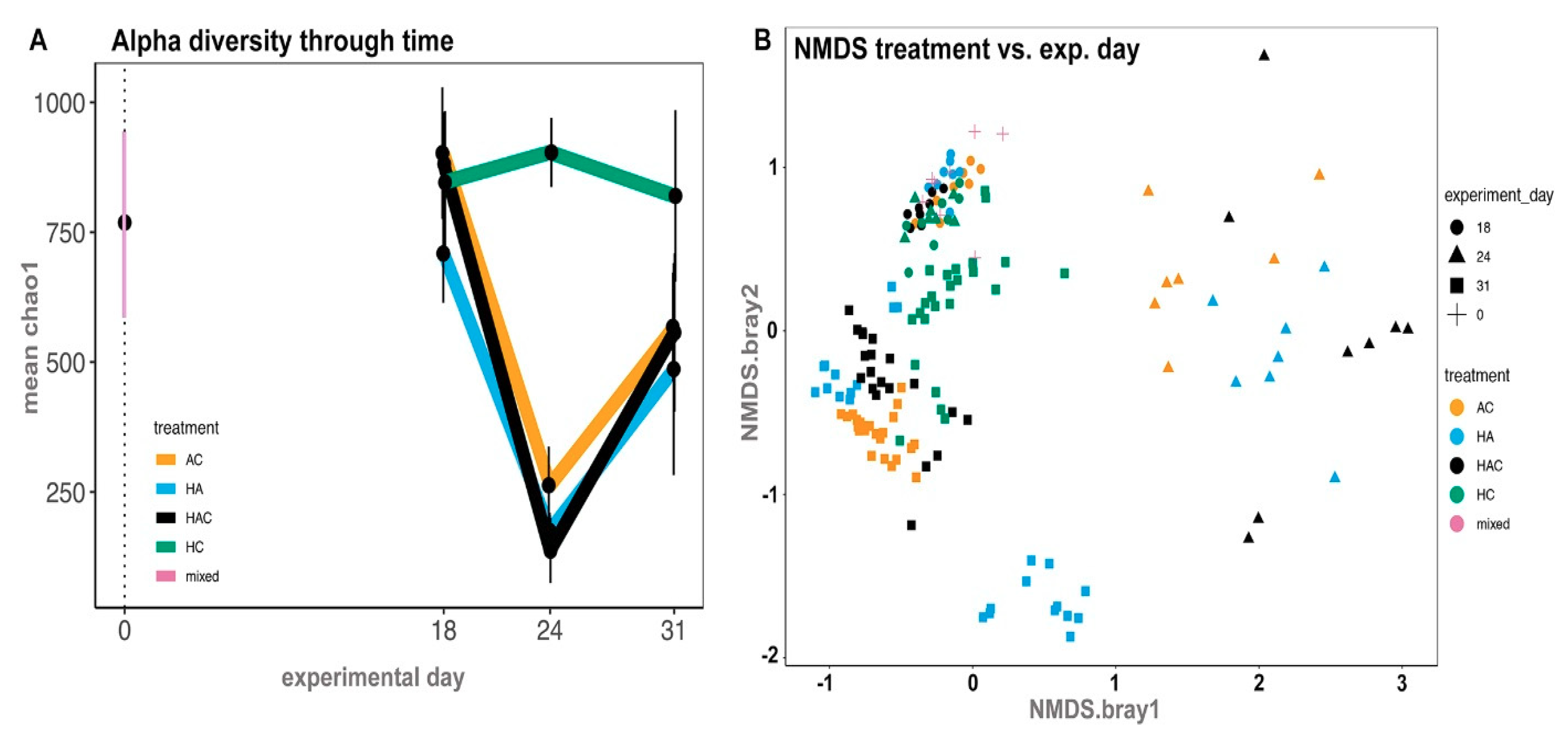
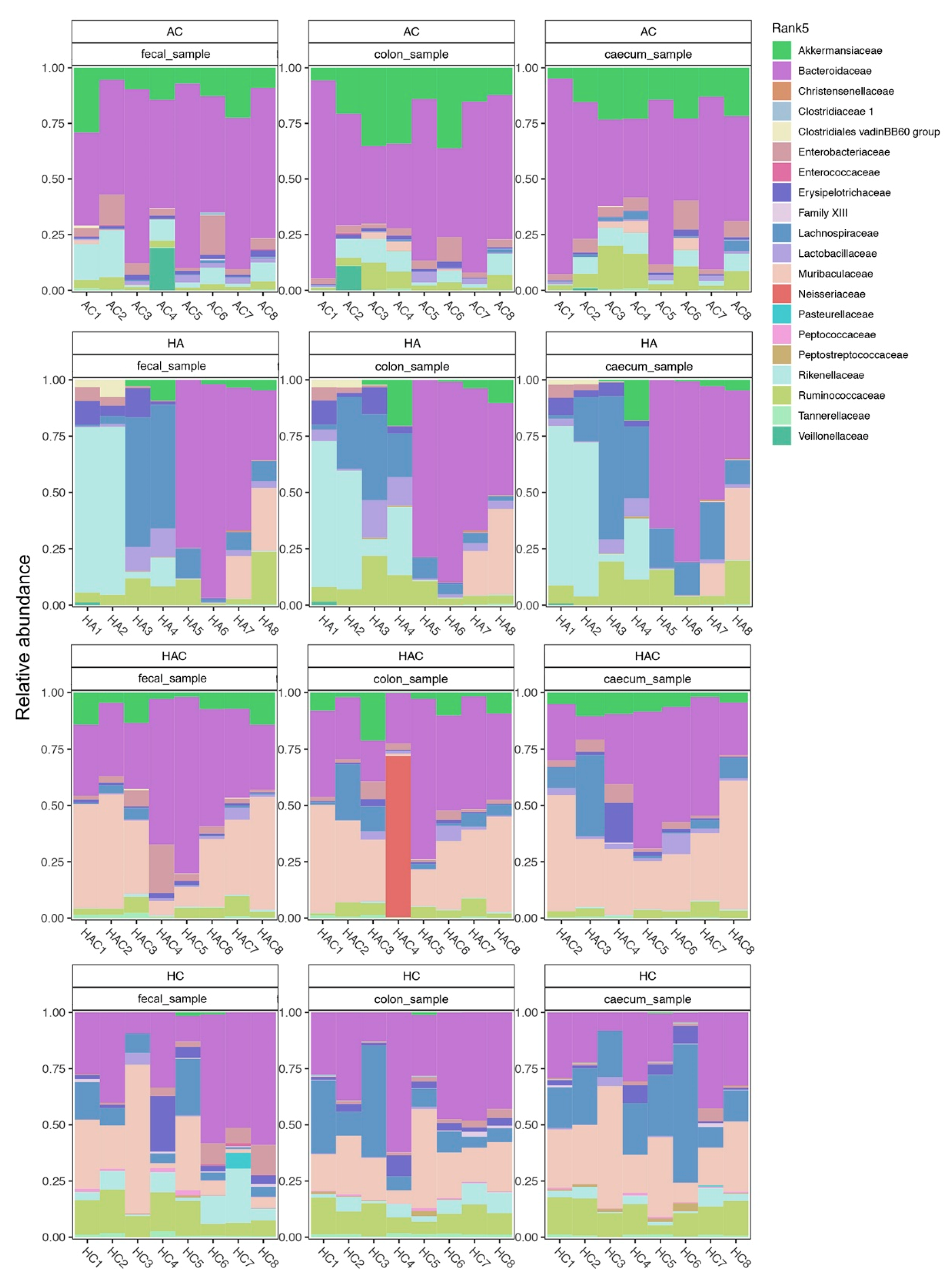
3.3. Microbial Results
3.4. Clinical Data
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Filyk, H.A.; Osborne, L.C. The Multibiome: The intestinal ecosystem’s influence on immune homeostasis, health, and disease. EbioMedicine 2016, 13, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Rook, G.A.W.; Bäckhed, F.; Levin, B.R.; McFall-Ngai, M.J.; McLean, A.R. Evolution, human-microbe interactions, and life history plasticity. Lancet 2017, 390, 521–530. [Google Scholar] [CrossRef]
- Gause, W.C.; Maizels, R.M. Macrobiota—Helminths as active participants and partners of the microbiota in host intestinal homeostasis. Curr. Opin. Microbiol. 2016, 32, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Maizels, R.M.; Smits, H.H.; McSorley, H.J. Modulation of host immunity by helminths: The expanding repertoire of parasite effector molecules. Immunity 2018, 49, 801–818. [Google Scholar] [CrossRef] [PubMed]
- Mubaraki, M.; Ahmad, M.; Hafiz, T.A.; Marie, M.A. The therapeutic prospect of crosstalk between prokaryotic and eukaryotic organisms in the human gut. FEMS Microbiol. Ecol. 2018, 94, 1–9. [Google Scholar] [CrossRef]
- Rapin, A.; Harris, N.L. Helminth—bacterial interactions: Cause and consequence. Trends Immunol. 2018, 39, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Parker, W.; Ollerton, J. Evolutionary biology and anthropology suggest biome reconstitution as a necessary approach toward dealing with immune disorders. Evol. Med. Public Health 2013, 2013, 89–103. [Google Scholar] [CrossRef]
- Rook, G.A.W.; Raison, C.L.; Lowry, C.A. Microbial ‘old friends’, immunoregulation and socioeconomic status. Clin. Exp. Immunol. 2014, 177, 1–12. [Google Scholar] [CrossRef]
- Sobotková, K.; Parker, W.; Levá, J.; Růžková, J.; Lukeš, J.; Jirků-Pomajbíková, K. Helminth therapy—From the parasite perspective. Trends Parasitol. 2019, 35, 501–515. [Google Scholar] [CrossRef]
- Grencis, R.K. Immunity to helminths: Resistance, regulation, and susceptibility to gastrointestinal nematodes. Annu. Rev. Immunol. 2015, 33, 201–225. [Google Scholar] [CrossRef]
- Harnett, M.M.; Harnett, W. Can parasitic worms cure the modern world’s ills? Trends Parasitol. 2017, 33, 694–705. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maizels, R.M.; McSorley, H.J. Regulation of the host immune system by helminth parasites. J. Allergy Clin. Immunol. 2016, 138, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, M.; Smeekens, S.P.; Vlamakis, H.; Jaeger, M.; Oosting, M.; Franzosa, E.A.; Jansen, T.; Jacobs, L.; Bonder, M.J.; Kurilshikov, A.; et al. Linking the human gut microbiome to inflammatory cytokine production capacity. Cell 2016, 167, 1125–1136.e8. [Google Scholar] [CrossRef]
- Lambring, C.B.; Siraj, S.; Patel, K.; Sankpal, U.T.; Mathew, S.; Basha, R. Impact of the microbiome on the immune system. Crit. Rev. Immunol. 2019, 39, 313–328. [Google Scholar] [CrossRef]
- Bernstein, C.N.; Forbes, J.D. Gut microbiome in inflammatory bowel disease and other chronic immune-mediated inflammatory diseases. Inflamm. Intest. Dis. 2017, 2, 116–123. [Google Scholar] [CrossRef]
- Bian, X.; Wu, W.; Yang, L.; Lv, L.; Wang, Q.; Li, Y.; Ye, J.; Fang, D.; Wu, J.; Jiang, X.; et al. Administration of Akkermansia muciniphila ameliorates Dextran Sulfate Sodium-induced ulcerative colitis in mice. Front. Microbiol. 2019, 10, 2259. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.J.; Kim, W.K.; Han, D.H.; Lee, K.; Ko, G. Lactobacillus fermentum species ameliorate dextran sulfate sodium-induced colitis by regulating the immune response and altering gut microbiota. Gut Microbes 2019, 10, 696–711. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.; Grinspan, A. Fecal microbiota transplantation for Inflammatory Bowel Diseases. Gastroenterol. Hepatol. 2016, 12, 374–379. [Google Scholar]
- Jenkins, T.P.; Brindley, P.J.; Gasser, R.B.; Cantacessi, C. Helminth microbiomes—A hidden treasure trove? Trends Parasitol. 2018, 35, 13–22. [Google Scholar] [CrossRef]
- White, E.C.; Houlden, A.; Bancroft, A.J.; Hayes, K.S.; Goldrick, M.; Grencis, R.K.; Roberts, I.A. Manipulation of host and parasite microbiotas: Survival strategies during chronic nematode infection. Sci. Adv. 2018, 4, 1–11. [Google Scholar] [CrossRef]
- Zaiss, M.M.; Rapin, A.; Lebon, L.; Dubey, L.K.; Mosconi, I.; Sarter, K.; Piersigilli, A.; Menin, L.; Walker, A.W.; Rougemont, J.; et al. The intestinal microbiota contributes to the ability of helminths to modulate allergic inflammation. Immunity 2015, 43, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Brosschot, T.P.; Reynolds, L.A. The impact of a helminth-modified microbiome on host immunity review-article. Mucosal Immunol. 2018, 11, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Walk, S.T.; Blum, A.M.; Ang-Sheng, S.; Weinstock, J.V.; Young, V.B. Alteration of the murine gut microbiota during infection with the parasitic helminth, Heligmosomoides polygyrus. Inflamm. Bowel Dis. 2010, 16, 1841–1849. [Google Scholar] [CrossRef] [PubMed]
- Rausch, S.; Held, J.; Fischer, A.; Heimesaat, M.M.; Kühl, A.A.; Bereswill, S.; Hartmann, S. Small intestinal nematode infection of mice is associated with increased enterobacterial loads alongside the intestinal tract. PLoS ONE 2013, 8, e74026. [Google Scholar] [CrossRef] [PubMed]
- Holm, J.B.; Sorobetea, D.; Kiilerich, P.; Ramayo-Caldas, Y.; Estellé, J.; Ma, T.; Madsen, L.; Kristiansen, K.; Svensson-Frej, M. Chronic Trichuris muris infection decreases diversity of the intestinal microbiota and concomitantly increases the abundance of lactobacilli. PLoS ONE 2015, 10, e0125495. [Google Scholar] [CrossRef] [PubMed]
- Houlden, A.; Hayes, K.S.; Bancroft, A.J.; Worthington, J.J.; Wang, P.; Grencis, R.K.; Roberts, I.S. Chronic Trichuris muris infection in C57BL/6 mice causes significant changes in host microbiota and metabolome: Effects reversed by pathogen clearance. PLoS ONE 2015, 10, e0125945. [Google Scholar] [CrossRef]
- Ramanan, D.; Bowcutt, R.; Lee, S.C.; Tang, M.S.; Kurtz, Z.D.; Ding, Y.; Honda, K.; Gause, W.C.; Blaser, M.J.; Bonneau, R.A.; et al. Helminth infection promotes colonization resistance via type 2 immunity. Science 2016, 352, 608–612. [Google Scholar] [CrossRef]
- Broadhurst, M.J.; Ardeshir, A.; Kanwar, B.; Mirpuri, J.; Gundra, U.M.; Leung, J.M.; Wiens, K.E.; Vujkovic-Cvijin, I.; Kim, C.C.; Yarovinsky, F.; et al. Therapeutic helminth infection of macaques with idiopathic chronic diarrhea alters the inflammatory signature and mucosal microbiota of the colon. PLoS Pathog. 2012, 8, e1003000. [Google Scholar] [CrossRef]
- Li, R.W.; Wu, S.; Li, W.; Navarro, K.; Couch, R.D.; Hill, D.; Urban, J.F., Jr. Alterations in the porcine colon microbiota induced by the gastrointestinal nematode Trichuris suis. Infect. Immun. 2012, 80, 2150–2157. [Google Scholar] [CrossRef]
- Stolzenbach, S.; Myhill, L.J.; Andersen, L.O.; Krych, L.; Mejer, H.; Williams, A.R.; Nejsum, P.; Stensvold, C.R.; Nielsen, D.S.; Thamsborg, S.M. Dietary inulin and Trichuris suis infection promote beneficial bacteria throughout the porcine gut. Front. Microbiol. 2020, 11, 312. [Google Scholar] [CrossRef]
- Cooper, P.; Walker, A.W.; Reyes, J.; Chico, M.; Salter, S.J.; Vaca, M.; Parkhill, J. Patent human infections with the whipworm, Trichuris trichiura, are not associated with alterations in the faecal microbiota. PLoS ONE 2013, 8, e76573. [Google Scholar] [CrossRef] [PubMed]
- Cantacessi, C.; Giacomin, P.; Croese, J.; Zakrzewski, M.; Sotillo, J.; McCann, L.; Nolan, M.J.; Mitreva, M.; Krause, L.; Loukas, A. Impact of experimental hookworm infection on the human gut microbiota. J. Infect. Dis. 2014, 210, 1431–1434. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Tang, M.S.; Lim, Y.A.L.; Choy, S.H.; Kurtz, Z.D.; Cox, L.M.; Gundra, U.M.; Cho, I.; Bonneau, R.; Blaser, M.J.; et al. Helminth colonization is associated with increased diversity of the gut microbiota. PLoS Negl. Trop. Dis. 2014, 8, e2880. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, T.P.; Rathnayaka, Y.; Perera, P.K.; Peachey, L.E.; Nolan, M.J.; Krause, L.; Rajakaruna, R.S.; Cantacessi, C. Infections by human gastrointestinal helminths are associated with changes in faecal microbiota diversity and composition. PLoS ONE 2017, 12, e0184719. [Google Scholar] [CrossRef]
- Lukeš, J.; Kuchta, R.; Scholz, T.; Pomajbíková, K. (Self-) infections with parasites: Re-interpretations for the present. Trends Parasitol. 2014, 30, 377–385. [Google Scholar] [CrossRef]
- Shi, M.; Wang, A.; Prescott, D.; Waterhouse, C.C.M.; Zhang, S.; McDougall, J.J.; Sharkey, K.A.; McKay, D.M. Infection with an intestinal helminth parasite reduces Freund’s complete adjuvant-induced monoarthritis in mice. Arthritis Rheum. 2011, 63, 434–444. [Google Scholar] [CrossRef]
- Matisz, C.E.; Leung, G.; Reyes, J.L.; Wang, A.; Sharkey, K.A.; McKay, D.M. Adoptive transfer of helminth antigen-pulsed dendritic cells protects against the development of experimental colitis in mice. Eur. J. Immunol. 2015, 45, 3126–3139. [Google Scholar] [CrossRef]
- Williamson, L.L.; McKenney, E.A.; Holzknecht, Z.E.; Belliveau, C.; Rawls, J.F.; Poulton, S.; Parker, W.; Bilbo, S.D. Got worms? Perinatal exposure to helminths prevents persistent immune sensitization and cognitive dysfunction induced by early-life infection. Brain. Behav. Immun. 2016, 51, 14–28. [Google Scholar] [CrossRef]
- Jirků-Pomajbíková, K.; Jirků, M.; Levá, J.; Sobotková, K.; Morien, E.; Parfrey, L.W. The benign helminth Hymenolepis diminuta ameliorates chemically induced colitis in a rat model system. Parasitology 2018, 145, 1324–1335. [Google Scholar] [CrossRef]
- Wang, A.; Fernando, M.; Leung, G.; Phan, V.; Smyth, D.; McKay, D.M. Exacerbation of oxazolone colitis by infection with the helminth Hymenolepis diminuta: Involvement of IL-5 and eosinophils. Am. J. Pathol. 2010, 177, 2850–2859. [Google Scholar] [CrossRef]
- Graepel, R.; Leung, G.; Wang, A.; Villemaire, M.; Jirik, F.R.; Sharkey, K.A.; McDougall, J.J.; McKay, D.M. Murine autoimmune arthritis is exaggerated by infection with the rat tapeworm, Hymenolepis diminuta. Int. J. Parasitol. 2013, 43, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.M.; Jaint, D.; Thomas, S.; Wilson, J.K.; Parker, W. Overcoming evolutionary mismatch by self-treatment with helminths: Current practices and experience. J. Evol. Med. 2015, 3, 1–22. [Google Scholar] [CrossRef]
- Liu, J.; Morey, R.A.; Wilson, J.K.; Parker, W. Practices and outcomes of self-treatment with helminths based on physicians’ observations. J. Helminthol. 2017, 91, 267–277. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.M. The immune response to and immunomodulation by Hymenolepis diminuta. Parasitology 2010, 137, 385–394. [Google Scholar] [CrossRef] [PubMed]
- McKenney, E.A.; Williamson, L.; Yoder, A.D.; Rawls, J.F.; Bilbo, S.D.; Parker, W. Alteration of the rat cecal microbiome during colonization with the helminth Hymenolepis diminuta. Gut Microbes 2015, 6, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Parfrey, L.W.; Jirků, M.; Šíma, R.; Jalovecká, M.; Sak, B.; Grigore, K.; Jirků-Pomajbíková, K. A benign helminth alters the host immune system and the gut microbiota in a rat model system. PLoS ONE 2017, 12, e0182205. [Google Scholar] [CrossRef]
- Shute, A.; Wang, A.; Jayme, T.S.; Strous, M.; McCoy, K.D.; Buret, A.G.; McKay, D.M. Worm expulsion is independent of alterations in composition of the colonic bacteria that occur during experimental Hymenolepis diminuta-infection in mice. Gut Microbes 2020, 11, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Knox, N.C.; Forbes, J.D.; van Domselaar, G.; Bernstein, C.N. The gut microbiome as a target for IBD treatment: Are we there yet? Curr. Treat. Options Gastroenterol. 2019, 17, 115–126. [Google Scholar] [CrossRef]
- Manichanh, C.; Reeder, J.; Gibert, P.; Varela, E.; Llopis, M.; Antolin, M.; Guigo, R.; Knight, R.; Guarner, F. Reshaping the gut microbiome with bacterial transplantation and antibiotic intake. Genome Res. 2010, 20, 1411–1419. [Google Scholar] [CrossRef]
- Hintze, K.J.; Cox, J.E.; Rrompato, G.; Benninghoff, A.D.; Ward, R.; Broadbent, J.; Lefevre, M. Broad scope method for creating humanized animal models for animal health and disease research through antibiotic treatment and human fecal transfer. Gut Microbes 2014, 5, 183–191. [Google Scholar] [CrossRef]
- Radwan, M.A.; AlQuadeib, B.T.; Šiller, L.; Wright, M.C.; Horrocks, B. Oral administration of amphotericin B nanoparticles: Antifungal activity, bioavailability and toxicity in rats. Drug Deliv. 2017, 24, 40–50. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Gonzales-Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Hannon, G.J. FASTX-Toolkit. 2010. Available online: http://hannonlab.cshl.edu/fastx_toolkit (accessed on 25 February 2020).
- Eren, A.M.; Morrison, H.G.; Lescault, P.J.; Reveillaud, J.; Vineis, J.H.; Sogin, M.L. Minimum entropy decomposition: Unsupervised oligotyping for sensitive partitioning of high-throughput marker gene sequences. ISME J. 2015, 9, 968–979. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, 590–596. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 25 February 2020).
- McMurdie, P.J.; Holmes, S. Phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Chao, A. Nonparametric estimation of the number of classes in a population. Scand. J. Stat. 1984, 11, 265–270. [Google Scholar]
- Bray, J.R.; Curtis, J.T. An ordination of the upland forest communities of Southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- Oksanen, F.J.; Guillaume Blanchet, F.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package Version 2.4-3. 2017. Available online: https://CRAN.R-project.org/package=vegan. (accessed on 25 February 2020).
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate—A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. Available online: https://ggplot2.tidyverse.org (accessed on 25 February 2020).
- Himmerich, H.; Fischer, J.; Bauer, K.; Kirkby, K.C.; Sack, U.; Krügel, U. Stress-induced cytokine changes in rats. Eur. Cytokine Netw. 2013, 24, 97–103. [Google Scholar] [CrossRef]
- Dea-Ayuela, M.A.; Rama-Iñiguez, S.; Bolás-Fernandez, F. Enhanced susceptibility to Trichuris muris infection of B10Br mice treated with the probiotic Lactobacillus casei. Int. Immunopharmacol. 2008, 8, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Hayes, K.S.; Bancroft, A.J.; Goldrick, M.; Portsmouth, C.; Roberts, I.S.; Grencis, R.K. Exploitation of the intestinal microflora by the parasitic nematode Trichuris muris. Science 2010, 328, 1391–1394. [Google Scholar] [CrossRef] [PubMed]
- Webb, R.A.; Hoque, T.; Dimas, S. Expulsion of the gastrointestinal cestode, Hymenolepis diminuta by tolerant rats: Evidence for mediation by a Th2 type immune enhanced goblet cell hyperplasia, increased mucin production and secretion. Parasite Immunol. 2007, 29, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Hunter, M.M.; Wang, A.; Hirota, C.L.; McKay, D.M. Neutralizing Anti-IL-10 Antibody blocks the protective effect of tapeworm infection in a murine model of chemically induced colitis. J. Immunol. 2005, 174, 7368–7375. [Google Scholar] [CrossRef]
- Persaud, R.; Wang, A.; Reardon, C.; McKay, D.M. Characterization of the immuno-regulatory response to the tapeworm Hymenolepis diminuta in the non-permissive mouse host. Int. J. Parasitol. 2007, 37, 393–403. [Google Scholar] [CrossRef]
- Melon, A.; Wang, A.; Phan, V.; McKay, D.M. Infection with Hymenolepis diminuta is more effective than daily corticosteroids in blocking chemically induced colitis in mice. J. Biomed. Biotechnol. 2010, 2010, 384523. [Google Scholar] [CrossRef]
- Reyes, J.L.; Lopes, F.; Leung, G.; Mancini, N.L.; Matisz, C.E.; Wang, A.; Thomson, E.A.; Graves, N.; Gilleard, J.; McKay, D.M. Treatment with cestode parasite antigens results in recruitment of CCR2+ myeloid cells, the adoptive transfer of which ameliorates colitis. Infect. Immun. 2016, 84, 3471–3483. [Google Scholar] [CrossRef]
- Ozkul, C.; Ruiz, V.E.; Battaglia, T.; Xu, J.; Roubaud-Baudron, C.; Cadwell, K.; Perer-Perez, G.I.; Blaser, M.J. A single early-in-life antibiotic course increases susceptibility to DSS-induced colitis. Genome Med. 2020, 12, 1–16. [Google Scholar] [CrossRef]
- Moges, R.; De Lamache, D.D.; Sajedy, S.; Renaus, B.S.; Hollenberg, M.D.; Muench, G.; Abbott, E.M.; Buret, A.G. Anti-inflammatory benefits of antibiotics: Tylvalosin induces apoptosis of porcine neutrophils and macrophages, promotes efferocytosis, an inhibits proinflammatoy CXCL-8, Il1a, and LTB4 production, while inducing the release of pro-resolving lipoxin A4 and resolvin D1. Front. Vet. Sci. 2018, 5, 57. [Google Scholar] [CrossRef]
- Giannoudaki, E.; Hernandez-Santana, Y.E.; Mulfaul, K.; Doyle, S.L.; Hams, E.; Fallon, P.G.; Mat, A.; O’Shea, D.; Kopf, M.; Hogan, A.E.; et al. Interleukin-36 cytokines alter the intestinal microbiome and can protect against obesity and metabolic dysfunction. Nat. Commun. 2019, 10, 1–14. [Google Scholar] [CrossRef]
- Huang, S.; Mao, J.; Zhou, L.; Xiong, X.; Deng, Y. The imbalance of gut microbiota and its correlation with plasma inflammatory cytokines in pemphigus vulgaris patients. Scand. J. Immunol. 2019, 90, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Saltykova, I.V.; Petrov, V.A.; Logacheva, M.D.; Ivanova, P.G.; Merzlikin, N.V.; Sazonov, A.E.; Ogorodova, L.M.; Brindley, P.J. Biliary microbiota, gallstone disease and infection with Opisthorchis felineus. PLoS Negl. Trop. Dis. 2016, 10, e0004809. [Google Scholar] [CrossRef] [PubMed]
- Fricke, W.F.; Song, Y.; Wang, A.J.; Smith, A.; Grinchuk, V.; Pei, C.; Ma, B.; Lu, N.; Urban, J.F., Jr.; Shea-Donohue, T.; et al. Type 2 immunity-dependent reduction of segmented filamentous bacteria in mice infected with the helminthic parasite Nippostrongylus brasiliensis. Microbiome 2015, 3, 40. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.A.; Hoffmann, C.; Abt, M.C.; Du, Y.; Kobuley, D.; Kirn, T.J.; Bushman, F.D.; Artis, D. Metagenomic analyses reveal antibiotic-induced temporal and spatial changes in intestinal microbiota with associated alterations in immune cell homeostasis. Mucosal Immunol. 2010, 3, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; El Khader, I.; Casellas, F.; López Vivancos, J.; García Cors, M.; Santiago, A.; Cuenca, S.; Guarner, F.; Manichanh, C. Short-term effect of antibiotics on human gut microbiota. PLoS ONE 2014, 9, e95476. [Google Scholar] [CrossRef]
- Zarrinpar, A.; Chaix, A.; Xu, Z.Z.; Chang, M.W.; Marotz, C.A.; Saghatelian, A.; Knight, R.; Panda, S. Antibiotic-induced microbiome depletion alters metabolic homeostasis by affecting gut signaling and colonic metabolism. Nat. Commun. 2018, 9, 2872. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jirků, M.; Lhotská, Z.; Frgelecová, L.; Kadlecová, O.; Petrželková, K.J.; Morien, E.; Jirků-Pomajbíková, K. Helminth Interactions with Bacteria in the Host Gut Are Essential for Its Immunomodulatory Effect. Microorganisms 2021, 9, 226. https://doi.org/10.3390/microorganisms9020226
Jirků M, Lhotská Z, Frgelecová L, Kadlecová O, Petrželková KJ, Morien E, Jirků-Pomajbíková K. Helminth Interactions with Bacteria in the Host Gut Are Essential for Its Immunomodulatory Effect. Microorganisms. 2021; 9(2):226. https://doi.org/10.3390/microorganisms9020226
Chicago/Turabian StyleJirků, Milan, Zuzana Lhotská, Lucia Frgelecová, Oldřiška Kadlecová, Klára Judita Petrželková, Evan Morien, and Kateřina Jirků-Pomajbíková. 2021. "Helminth Interactions with Bacteria in the Host Gut Are Essential for Its Immunomodulatory Effect" Microorganisms 9, no. 2: 226. https://doi.org/10.3390/microorganisms9020226
APA StyleJirků, M., Lhotská, Z., Frgelecová, L., Kadlecová, O., Petrželková, K. J., Morien, E., & Jirků-Pomajbíková, K. (2021). Helminth Interactions with Bacteria in the Host Gut Are Essential for Its Immunomodulatory Effect. Microorganisms, 9(2), 226. https://doi.org/10.3390/microorganisms9020226



