Secretion of Iron(III)-Reducing Metabolites during Protein Acquisition by the Ectomycorrhizal Fungus Paxillus involutus
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Strain and Culture Conditions
2.2. Iron(III)-Reducing Activities and Fe2+ Concentrations
2.3. Determination of •OH Production
2.4. X-ray Absorption Spectroscopy (XAS)
2.5. Identification of Extracellular Secondary Metabolites
2.6. Production of Extracellular Iron(III)-Reducing Metabolites
2.7. NMR Spectroscopy
3. Results
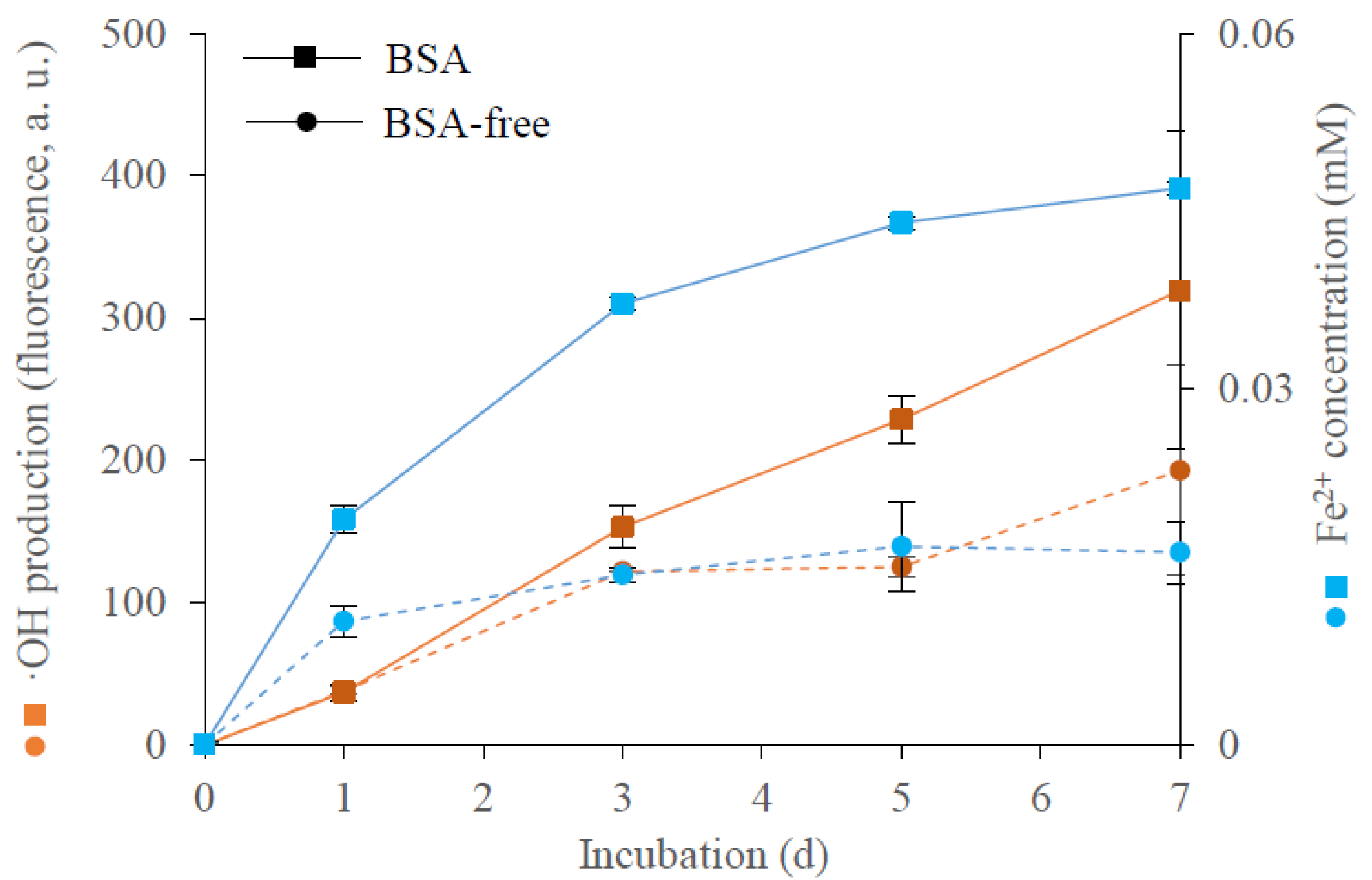
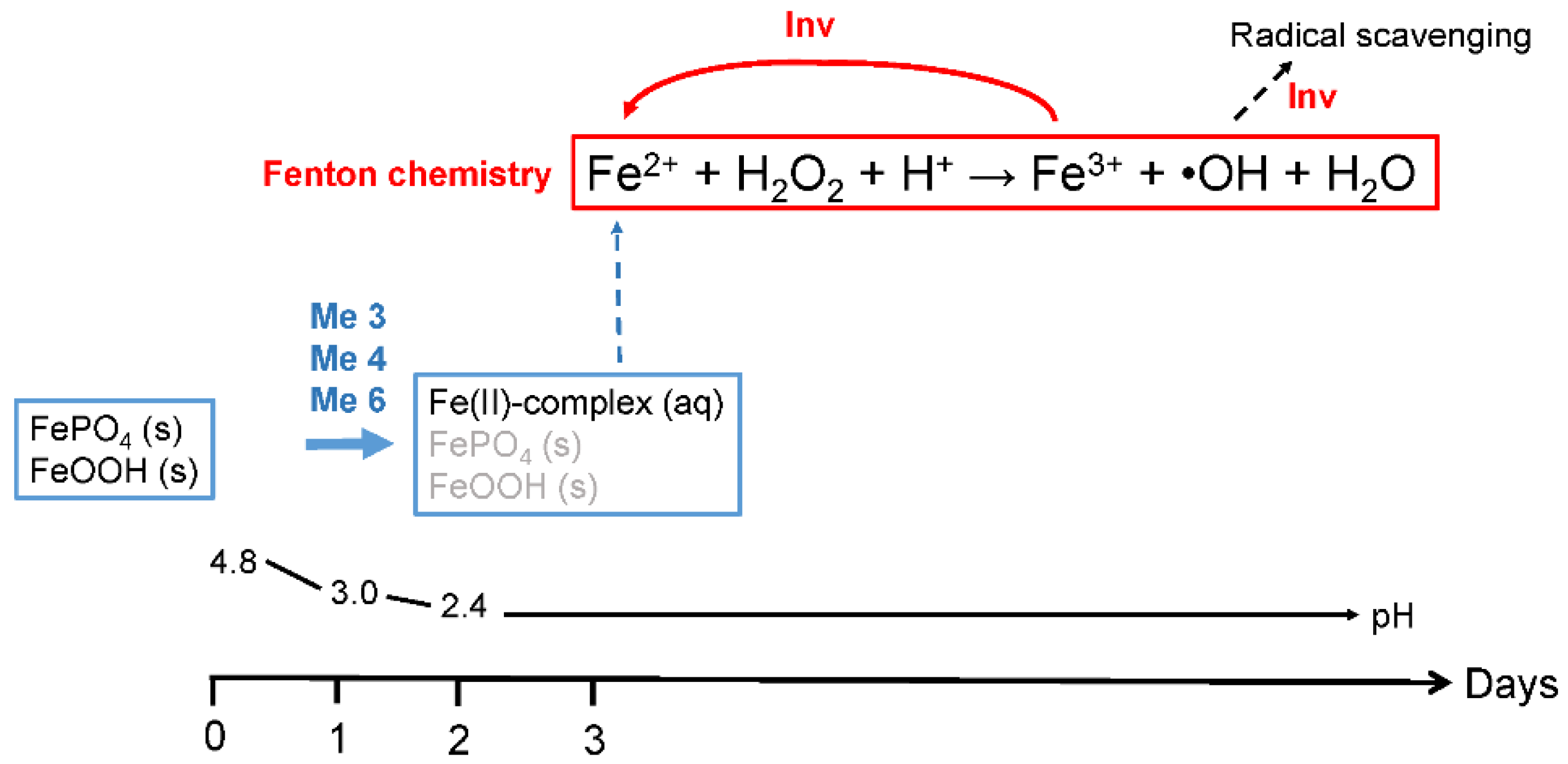
3.1. OH Production and Iron(III) Reduction
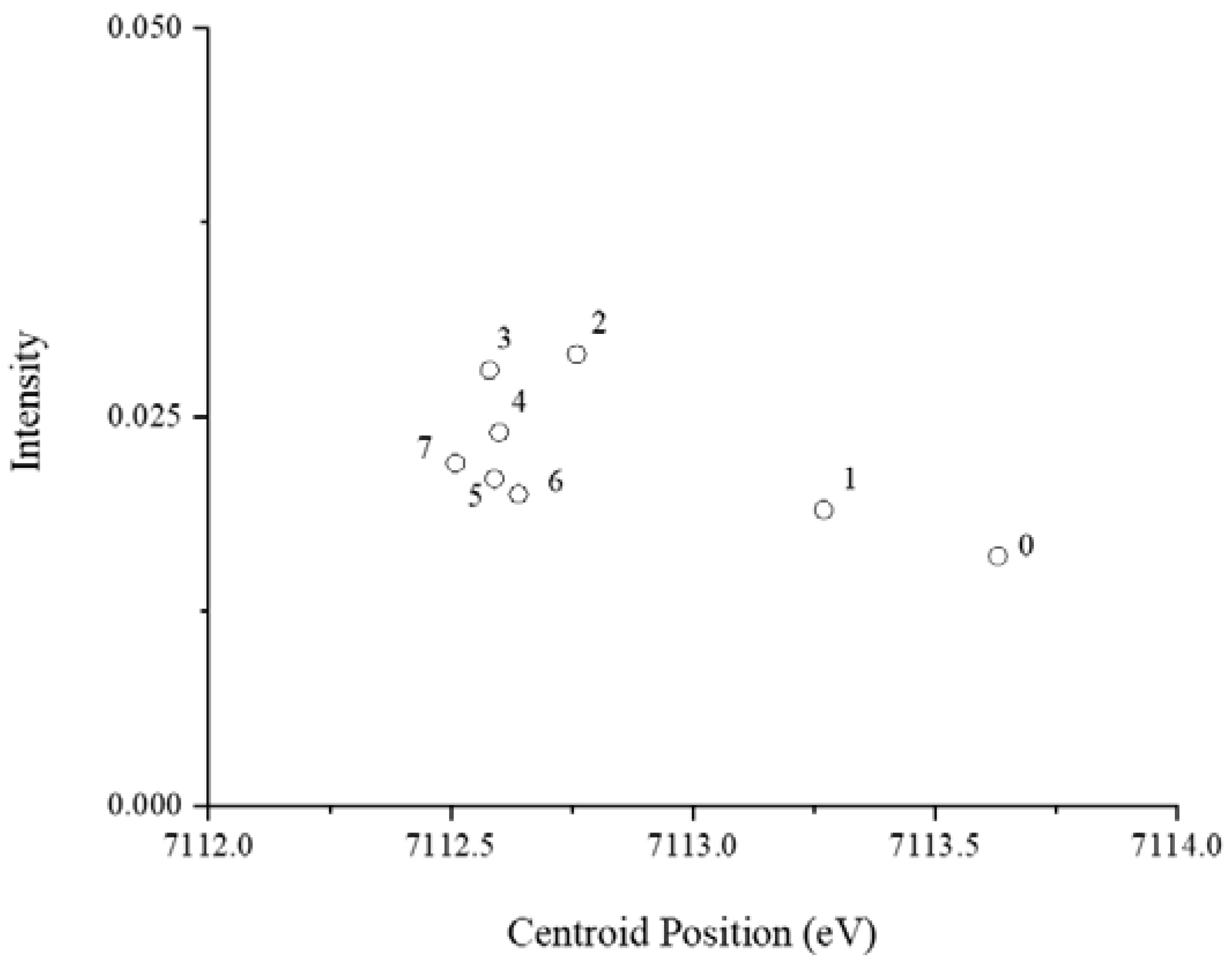
3.2. Fe Speciation in the Protein Growth Medium
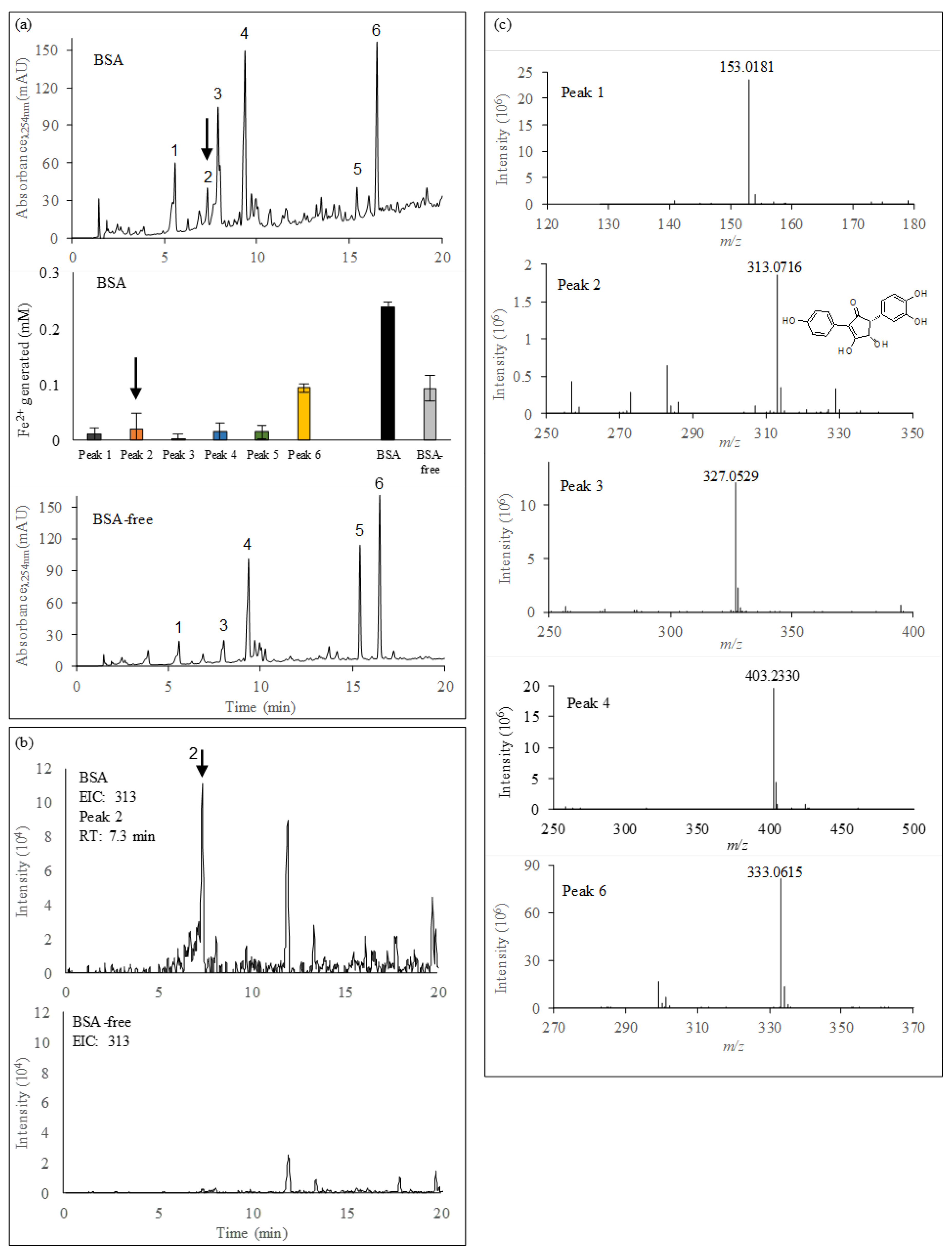
3.3. Characterization of Iron(III)-Reducing Metabolites
3.4. Identification of Involutin
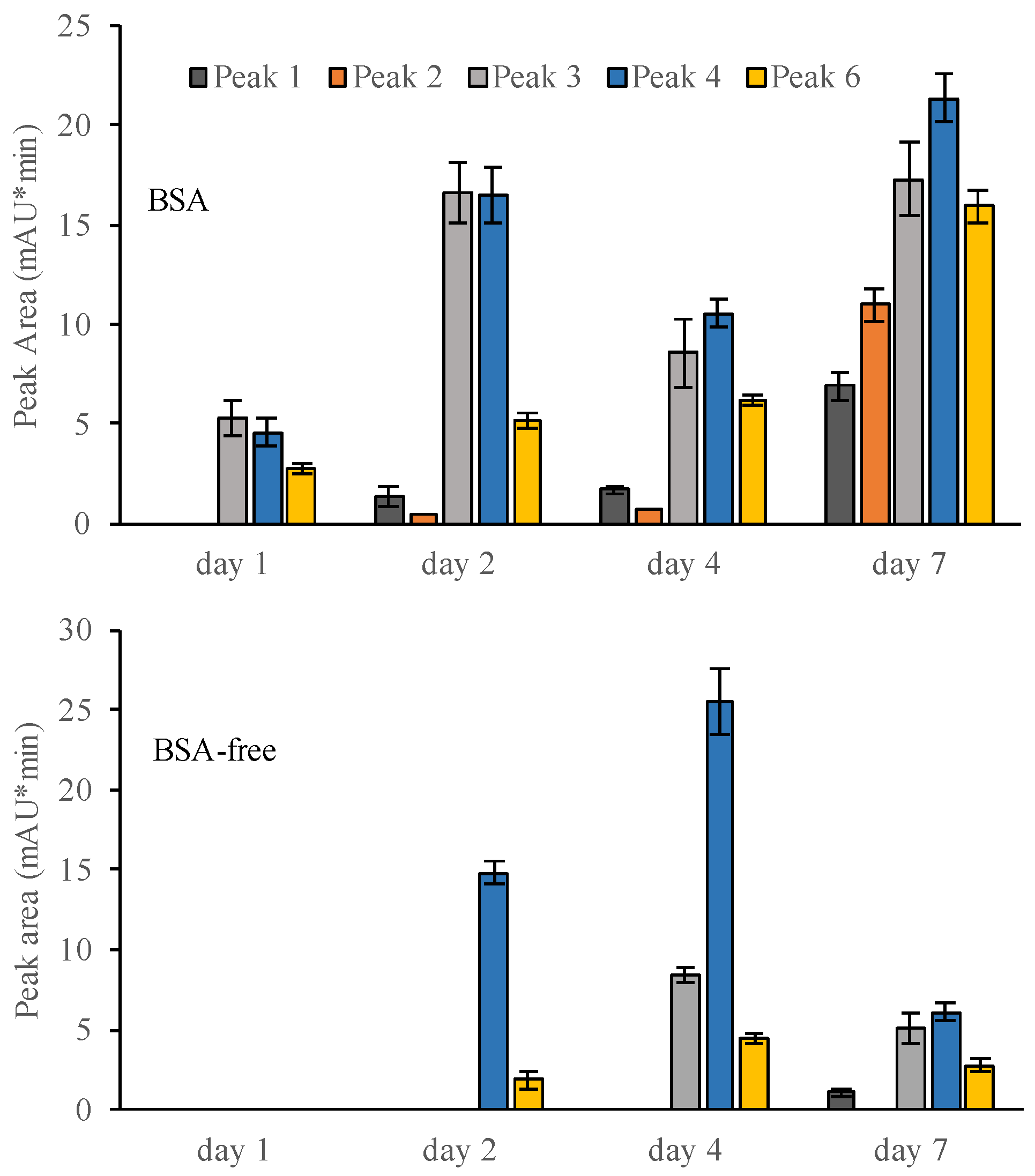
3.5. Production of Iron(III)-Reducing Metabolites over Time
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nannipieri, N.; Eldor, P. The chemical and functional characterization of soil N and its biotic components. Soil Biol. Biochem. 2009, 41, 2357–2369. [Google Scholar] [CrossRef]
- Piccolo, A. The supramolecular structure of humic substances. Soil Sci. 2001, 166, 810–832. [Google Scholar] [CrossRef]
- Jilling, A.; Keiluweit, M.; Contosta, A.R.; Frey, S.; Schimel, J.; Schnecker, J.; Smith, R.G.; Tiemann, L.; Grandy, A.S. Minerals in the rhizosphere: Overlooked mediators of soil nitrogen availability to plants and microbes. Biogeochemistry 2018, 139, 103–122. [Google Scholar] [CrossRef]
- Näsholm, T.; Kielland, K.; Ganeteg, U. Uptake of organic nitrogen by plants. New Phytol. 2009, 182, 31–48. [Google Scholar] [CrossRef]
- Schimel, J.P.; Bennet, J. Nitrogen mineralization: Challenges of a changing paradigm. Ecology 2004, 85, 591–602. [Google Scholar] [CrossRef]
- Read, D.J.; Leake, J.R.; Perez-Moreno, J. Mycorrhizal fungi as drivers of ecosystem processes in heathland and boreal forest biomes. Can. J. Bot. 2004, 82, 1243–1263. [Google Scholar] [CrossRef]
- Kohler, A.; Kuo, A.; Nagy, L.G.; Morin, E.; Barry, K.W.; Buscot, F.; Canbäck, B.; Choi, C.; Cichocki, N.; Clum, A.; et al. Convergent losses of decay mechanisms and rapid turnover of symbiosis genes in mycorrhizal mutualists. Nat. Genet. 2015, 47, 410–415. [Google Scholar] [CrossRef]
- Zak, D.R.; Pellitier, P.T.; Argiroff, W.A.; Castillo, B.; James, T.Y.; Nave, L.E.; Averill, C.; Beidler, K.V.; Bhatnagar, J.; Blesh, J.; et al. Exploring the role of ectomycorrhizal fungi in soil carbon dynamics. New Phytol. 2019, 223, 33–39. [Google Scholar] [CrossRef]
- Pellitier, P.M.; Zak, D.R. Ectomycorrhizal fungi and the enzymatic liberation of nitrogen from soil organic matter: Why evolutionary history matters. New Phytol. 2018, 217, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.; Nicolás, C.; Bentzer, J.; Ellström, M.; Smits, M.; Rineau, F.; Canbäck, B.; Floudas, D.; Carleer, R.; Lackner, G.; et al. Ectomycorrhizal fungi decompose soil organic matter using oxidative mechanisms adapted from saprotrophic ancestors. New Phytol. 2016, 209, 1705–1719. [Google Scholar] [CrossRef] [PubMed]
- Nicolás, C.; Martin-Bertelsen, T.; Floudas, D.; Bentzer, J.; Smits, M.; Johansson, T.; Troein, C.; Persson, P.; Tunlid, A. The soil organic matter decomposition mechanisms in ectomycorrhizal fungi are tuned for liberating soil organic nitrogen. ISME J. 2019, 13, 977–988. [Google Scholar] [PubMed]
- Rineau, F.; Roth, D.; Shah, F.; Smits, M.; Johansson, T.; Canbäck, B.; Olsen, P.B.; Persson, P.; Grell, M.N.; Lindquist, E.; et al. The ectomycorrhizal fungus Paxillus involutus converts organic matter in plant litter using a trimmed brown-rot mechanism involving Fenton chemistry. Environ. Microbiol. 2012, 14, 1477–1478. [Google Scholar] [PubMed]
- Jensen, K.A., Jr.; Houtman, C.J.; Ryan, Z.C.; Hammel, K.E. Pathways for extracellular Fenton chemistry in the brown rot basidiomycete Gloeophyllum trabeum. Appl. Environ. Microbiol. 2001, 67, 2705–2711. [Google Scholar]
- Newcombe, D.; Paszczynski, A.; Gajewska, W.; Kröger, M.; Feis, G.; Crawford, R. Production of small molecular weight catalysts and the mechanism of trinitrotoluene degradation by several Gloeophyllum species. Enzym. Microb. Technol. 2002, 30, 506–517. [Google Scholar] [CrossRef]
- Shah, F.; Schwenk, D.; Nicolás, C.; Persson, P.; Hoffmeister, D.; Tunlid, A. Involutin is an Fe3+ reductant secreted by the ectomycorrhizal fungus Paxillus involutus during Fenton-based decomposition of organic matter. Appl. Environ. Microbiol. 2015, 81, 8427–8433. [Google Scholar] [CrossRef] [PubMed]
- Braesel, J.; Götze, S.; Shah, F.; Heine, D.; Tauber, J.; Hertweck, C.; Tunlid, A.; Stallforth, P.; Hoffmeister, D. Three redundant synthetases secure redox-active pigment production in the Basidiomycete Paxillus involutus. Chem. Biol. 2015, 22, 1325–1334. [Google Scholar] [CrossRef] [PubMed]
- Schneider, P.; Bouhired, S.; Hoffmeister, D. Characterization of the atromentin biosynthesis genes and enzymes in the homobasidiomycete Tapinella panuoides. Fungal Genet. Biol. 2008, 45, 1487–1496. [Google Scholar] [CrossRef]
- Op de Beeck, M.; Troein, C.; Peterson, C.; Persson, P.; Tunlid, A. Fenton reaction facilitates organic nitrogen acquisition by an ectomycorrhizal fungus. New Phytol. 2018, 218, 335–343. [Google Scholar] [CrossRef]
- Shah, F.; Rineau, F.; Canbäck, B.; Johansson, T.; Tunlid, A. The molecular components of the extracellular protein-degradation pathways of the ectomycorrhizal fungus Paxillus involutus. New Phytol. 2013, 200, 875–887. [Google Scholar] [CrossRef]
- Klementiev, K.V. VIPER for windows, freeware. J. Phys. D Appl. Phys. 2001, 34, 209–217. [Google Scholar]
- Sundman, A.; Karlsson, T.; Sjöberg, S.; Persson, P. Impact of iron-organic matter complexes on aqueous phosphate concentrations. Chem. Geol. 2015, 426, 109–117. [Google Scholar] [CrossRef]
- Szytuła, A.; Burewicz, A.; Dimitrijević, Ž.; Kraśnicki, S.; Rżany, H.; Todorović, J.; Wanic, A.; Wolski, W. Neutron diffraction studies of α-FeOOH. Phys. Status Solidi 1968, 26, 429–434. [Google Scholar] [CrossRef]
- Zabinsk, S.J.; Rehr, J.; Ankudinov, A.; Albers, R.; Eller, M. FEFF code for ab initio calculations of XAFS. Phys. Rev. B 1995, 52, 2995–3009. [Google Scholar]
- Sundman, A.; Karlsson, T.; Laudon, H.; Persson, P. XAS study of iron speciation in soils and waters from a boreal catchment. Chem. Geol. 2014, 364, 93–102. [Google Scholar] [CrossRef]
- Wilke, M.; Farges, F.; Petit, P.-E.; Brown, G.E., Jr.; Martin, F. Oxidation state and coordination of Fe in minerals: An Fe K-XANES spectroscopic study. Am. Mineral. 2001, 86, 714–730. [Google Scholar] [CrossRef]
- Gill, M.; Steglich, W. Pigments of fungi (Macromycetes). Prog. Chem. Org. Nat. Prod. 1987, 51, 1–317. [Google Scholar]
- Feling, R.; Polborn, K.; Steglich, W.; Mühlbacher, J.; Bringmann, G. The absolute configuration of the mushroom metabolites involutin and chamonixin. Tetrahedron 2001, 57, 7857–7863. [Google Scholar] [CrossRef]
- Kälvö, D.; Menkis, A.; Broberg, A. Secondary metabolites from the root rot biocontrol fungus Phlebiopsis gigantea. Molecules 2018, 23, 1417. [Google Scholar] [CrossRef] [PubMed]
- Arai, Y.; Sparks, D.L. Phosphate reaction dynamics in soils and soil components: A multiscale approach. Adv. Agron. 2007, 94, 135–179. [Google Scholar]
- Wallander, H.; Wickman, T.; Jacks, G. Apatite as a P source in mycorrhizal and non-mycorrhizal Pinus sylvestris seedlings. Plant Soil 1997, 196, 123–131. [Google Scholar] [CrossRef]
- Rosling, A. Trees, mycorrhiza and minerals—field relevance of in vitro experiments. Geomicrobiol. J. 2009, 26, 389–401. [Google Scholar] [CrossRef]
- Peretyazhko, T.; Sposito, G. Fe(III) reduction and phosphorous solubilization in humid tropical forest soils. Geochim. Cosmochim. Acta 2005, 69, 3643–3652. [Google Scholar] [CrossRef]
- Morgan, B.; Lahav, O. The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution—basic principles and a simple heuristic description. Chemosphere 2007, 68, 2080–2084. [Google Scholar] [CrossRef]
- Jones, A.M.; Griffin, P.J.; Collins, R.N.; Waite, T.D. Ferrous iron oxidation under acidic conditions—The effect of ferric oxide surfaces. Geochim. Cosmochim. Acta 2014, 145, 1–12. [Google Scholar] [CrossRef]
- Kerem, Z.; Jensen, K.A.; Hammel, K.E. Biodegradative mechanism of the brown rot basidiomycete Gloeophyllum trabeum: Evidence for an extracellular hydroquinone-driven mechanism. FEBS Lett. 1999, 446, 49–54. [Google Scholar] [CrossRef]
- Wei, D.; Houtman, C.J.; Kapich, A.N.; Hunt, C.G.; Cullen, D.; Hammel, K.E. Laccase and its role in the production of extracellular reactive oxygen species during wood decay by the brown rot basidiomycete Postia placenta. Appl. Environ. Microbiol. 2010, 76, 2091–2097. [Google Scholar] [CrossRef]
- Levasseur, A.; Drula, E.; Lombard, V.; Coutinho, P.M.; Henrissat, B. Expansion of the enzymatic repertoire of the CAZy database to integrate auxiliary redox enzymes. Biotechnol. Biofuels 2013, 6, 41. [Google Scholar] [CrossRef]
- Korripally, P.; Timokhin, V.I.; Houtman, C.J.; Mozuch, M.D.; Hammel, K.E. Evidence from Serpula lacrymans that 2,5-dimethoxyhydroquinone is a lignocellulolytic agent of divergent brown rot basidiomycetes. Appl. Environ. Microb. 2013, 79, 2377–2383. [Google Scholar] [CrossRef]
- Jensen, K.A., Jr.; Ryan, Z.R.; Vanden Wymelenberg, A.; Cullen, D.; Hammel, K.E. An NADH:quinone oxidoreductase active during biodegradation by the brown-rot basidiomycete Gloeophyllum trabeum. Appl. Environ. Microbiol. 2002, 68, 2699–2703. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Mahaney, J.; Jellison, J.; Cao, J.; Gressler, J.; Hoffmeister, D.; Goodell, B. Fungal variegatic acid and extracellular polysaccharides promote the site-specific generation of reactive oxygen species. J. Ind. Microbiol. Biotechnol. 2017, 44, 329–338. [Google Scholar] [CrossRef]
- Aranates, V.; Jellison, J.; Goodell, B. Peculiarities of brown-rot fungi and biochemical Fenton reaction with regard to their potential as a model for bioprocessing biomass. Appl. Microbiol. Biotechnol. 2012, 94, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Lapeyrie, F.; Chilvers, G.A.; Bhem, C.A. Oxalic acid synthesis by the mycorrhizal fungus Paxillus involutus (Batsch. ex Fr.) Fr. New Phytol. 1987, 106, 139–146. [Google Scholar] [CrossRef]
- Schmalenberger, A.; Duran, A.L.; Bray, A.W.; Bridge, J.; Bonneville, S.; Benning, L.G.; Romero-Gonzalez, M.E.; Leake, J.R.; Banwart, S.A. Oxalate secretion by ectomycorrhizal Paxillus involutus is mineral-specific and controls calcium weathering from minerals. Sci. Rep. 2015, 5, 12187. [Google Scholar] [CrossRef] [PubMed]
- Arantes, V.; Qian, Y.; Milagres, A.M.F.; Jellison, J.; Goodell, B. Effect of pH and oxalic acid on the reduction of Fe3+ by a biomimetic chelator and on Fe3+ desorption/adsorption onto wood: Implications for brown-rot decay. Int. Biodeterior. Biodegrad. 2009, 63, 478–483. [Google Scholar] [CrossRef]
- Eastwood, D.C.; Floudas, D.; Binder, M.; Majcherczyk, A.; Schneider, P.; Aerts, A.; Asiegbu, F.O.; Baker, S.E.; Barry, K.; Bendiksby, M.; et al. The plant cell wall-decomposing machinery underlies the functional diversity of forest fungi. Science 2011, 333, 762–765. [Google Scholar] [CrossRef] [PubMed]
- Tauber, J.P.; Gallegos-Monterrosa, R.; Kovács, A.T.; Shelest, E.; Hoffmeister, D. Dissimilar pigment regulation in Serpula lacrymans and Paxillus involutus during inter-kingdom interactions. Microbiology 2018, 164, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Tauber, J.P.; Schroeckh, V.; Shelest, E.; Brakhage, A.A.; Hoffmeister, D. Bacteria induce pigment formation in the basidiomycete Serpula lacrymans. Environ. Microbiol. 2016, 18, 5218–5227. [Google Scholar] [CrossRef]
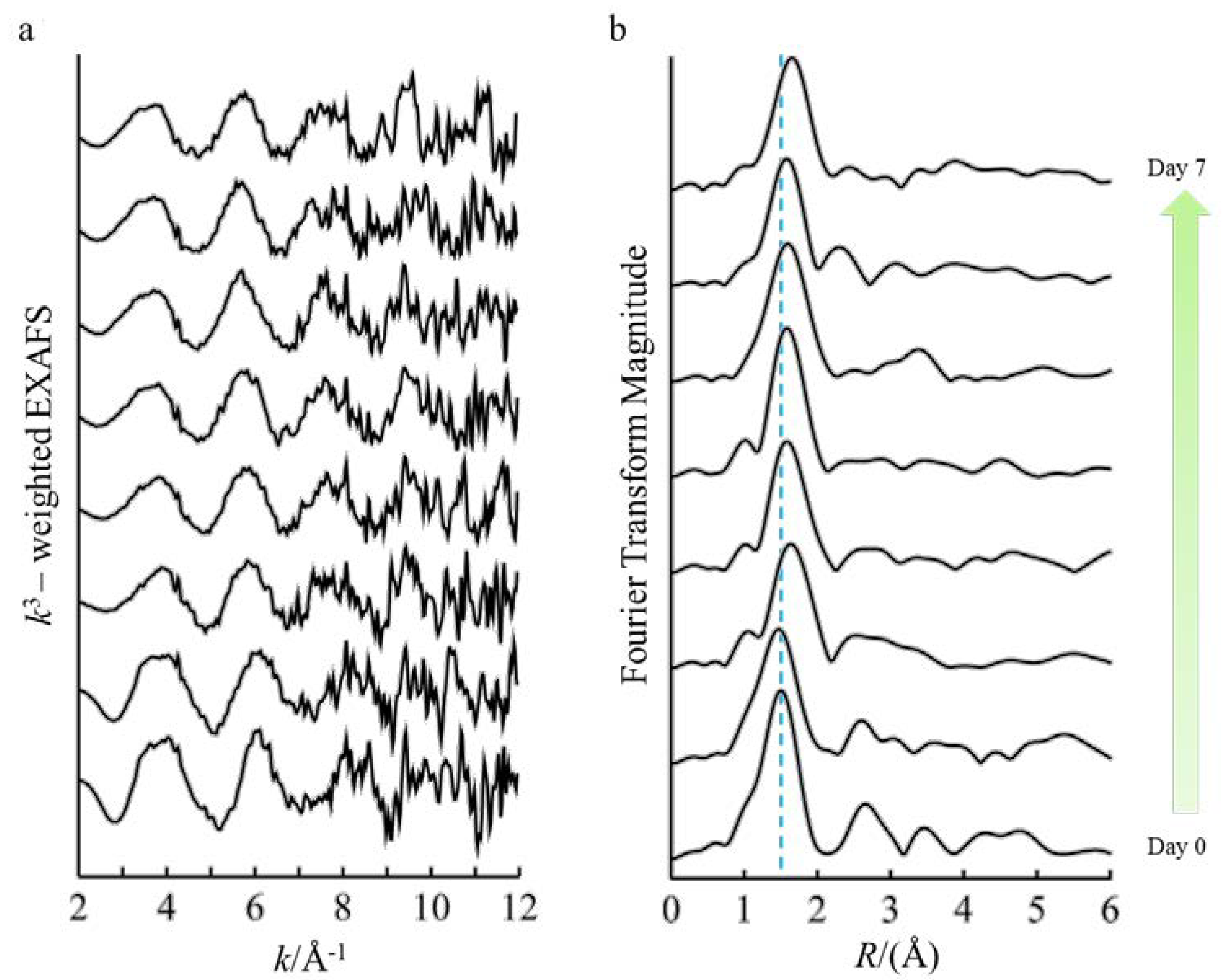
| Path | R (Å) | CN | σ2 (Å2) | ΔE0 (eV) | |
|---|---|---|---|---|---|
| Day 7 | Fe–O | 2.12 | 3.2 | 0.0031 | −9.1 |
| Day 6 | Fe–O | 2.09 | 4.1 | 0.0069 | −11.6 |
| Day 5 | Fe–O | 2.10 | 4.6 | 0.0071 | −10.7 |
| Day 4 | Fe–O | 2.08 | 3.8 | 0.0050 | −11.8 |
| Day 3 | Fe–O | 2.08 | 3.9 | 0.0068 | −9.5 |
| Day 2 | Fe–O | 2.09 | 3.8 | 0.0067 | −5.8 |
| Day 1 | Fe–O | 1.99 | 5.6 | 0.0110 | −11.8 |
| Fe–P | 3.15 | 3.9 | 0.0085 | ||
| Fe–Fe | 3.10 | 3.9 | 0.0100 | ||
| Day 0 | Fe–O | 1.99 | 5.9 | 0.0088 | −10.6 |
| Fe–P | 3.19 | 3.5 | 0.0085 | ||
| Fe-Fe | 3.11 | 2.8 | 0.0100 |
| RT (min) | MolecularIons (m/z) | Daughter Ions MS/MS (m/z) | RDB | Delta MMA | Predicted Formulas Molecular Ions | |
|---|---|---|---|---|---|---|
| Peak 1 | 5.6 | 153.0181 | 109.0278, 153.0181 | 5.5 | −0.365 | C7H6O4 |
| Peak 2 | 7.3 | 313.0716 | 135.0435, 149.0226, 163.0389, 295.2282, 313.0716 | 11.5 | 0.865 | C17H13O6 (involutin) |
| Peak 3 | 7.8 | 327.0529 | 140.9846, 188.0342, 235.9256, 285.1706, 327.0529 | 12.5 | 0.881 | C17H12O7 |
| Peak 4 | 9.0 | 403.2330 | 201.1121, 403.2330 | 3.5 | 0.416 | C20H36O8 |
| Peak 6 | 16.2 | 333.0615 | 267.1600, 299.0557, 333.0615 | 10.5 | 1.276 | C16H14O8 |
| Chemical Shift (ppm) | Peak Multiplicity | Peak Integration (Number of Protons) | C. No |
|---|---|---|---|
| Involutin | |||
| 6.58 | d | 0.85 | 3′ |
| 6.68 | s | 0.84 | 6′ |
| 6.71 | d | 1.12 | 2′ |
| 6.76 | d | 2.1 | 8,10 |
| 7.74 | d | 2 | 7,11 |
| Involutin enol form | |||
| 6.82 | d | 2.07 | 8,10 |
| 6.99 | d | 0.96 | 3′ |
| 7.26 | d * | 2.01 | 7,11 |
| 7.32 | d | 1.17 | 2′ |
| 7.89 | d | 1.01 | 6′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, F.; Gressler, M.; Nehzati, S.; Op De Beeck, M.; Gentile, L.; Hoffmeister, D.; Persson, P.; Tunlid, A. Secretion of Iron(III)-Reducing Metabolites during Protein Acquisition by the Ectomycorrhizal Fungus Paxillus involutus. Microorganisms 2021, 9, 35. https://doi.org/10.3390/microorganisms9010035
Shah F, Gressler M, Nehzati S, Op De Beeck M, Gentile L, Hoffmeister D, Persson P, Tunlid A. Secretion of Iron(III)-Reducing Metabolites during Protein Acquisition by the Ectomycorrhizal Fungus Paxillus involutus. Microorganisms. 2021; 9(1):35. https://doi.org/10.3390/microorganisms9010035
Chicago/Turabian StyleShah, Firoz, Markus Gressler, Susan Nehzati, Michiel Op De Beeck, Luigi Gentile, Dirk Hoffmeister, Per Persson, and Anders Tunlid. 2021. "Secretion of Iron(III)-Reducing Metabolites during Protein Acquisition by the Ectomycorrhizal Fungus Paxillus involutus" Microorganisms 9, no. 1: 35. https://doi.org/10.3390/microorganisms9010035
APA StyleShah, F., Gressler, M., Nehzati, S., Op De Beeck, M., Gentile, L., Hoffmeister, D., Persson, P., & Tunlid, A. (2021). Secretion of Iron(III)-Reducing Metabolites during Protein Acquisition by the Ectomycorrhizal Fungus Paxillus involutus. Microorganisms, 9(1), 35. https://doi.org/10.3390/microorganisms9010035





