Blastocystis sp. Prevalence and Subtypes Distribution amongst Syrian Refugee Communities Living in North Lebanon
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Approval
2.2. Questionnaire Survey
2.3. Sites Sampling and Collection of Samples
2.4. DNA Extraction and Molecular Subtyping of Blastocystis sp. Isolates
2.5. Statistical Analysis
3. Results
3.1. Analysis of the Cohort of Syrian Refugees and Prevalence of Blastocystis sp.
3.2. Screening for Blastocystis sp. Using qPCR
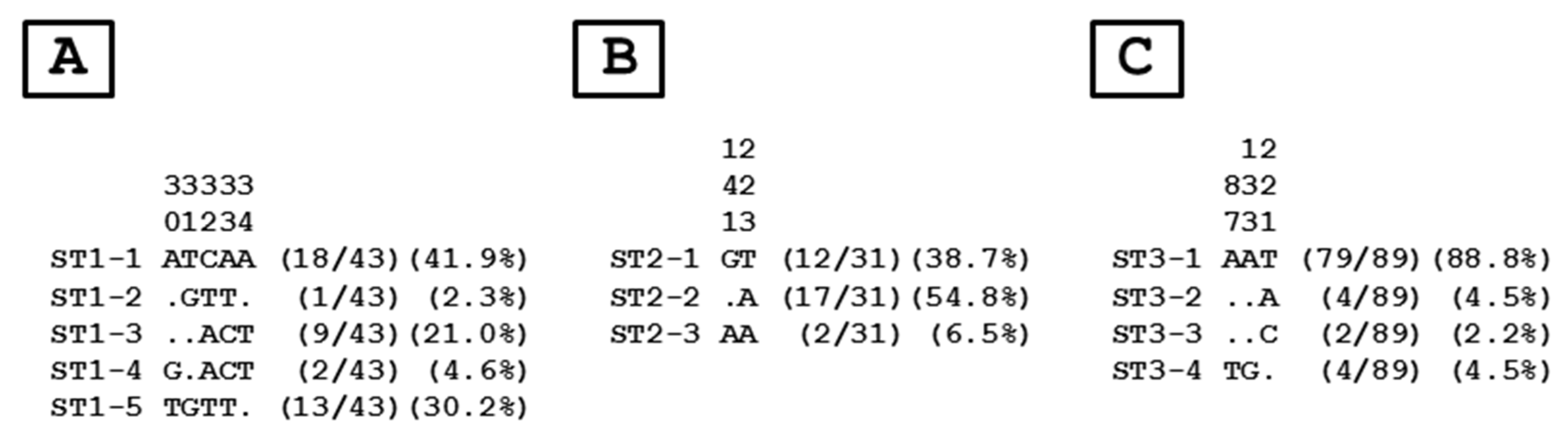
3.3. Distribution of Blastocystis sp. STs and Genotypes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stenzel, D.J.; Boreham, P.F.L. Blastocystis hominis revisited. Clin. Microbiol. Rev. 1996, 9, 563–584. [Google Scholar] [CrossRef]
- Tan, K.S.W. Blastocystis in humans and animals: New insights using modern methodologies. Vet. Parasitol. 2004, 126, 121–144. [Google Scholar] [CrossRef]
- Wawrzyniak, I.; Poirier, P.; Viscogliosi, E.; Meloni, D.; Texier, C.; Delbac, F.; El Alaoui, H. Blastocystis, an unrecognized parasite: An overview of pathogeneis and diagnosis. Ther. Adv. Infect. Dis. 2013, 1, 167–178. [Google Scholar] [CrossRef]
- Osman, M.; El Safadi, D.; Cian, A.; Benamrouz, S.; Nourrisson, C.; Poirier, P.; Pereira, B.; Razakandrainibe, R.; Pinon, A.; Lambert, C.; et al. Prevalence and risk factors for intestinal protozoan infections with Cryptosporidium, Giardia, Blastocystis and Dientamoeba among schoolchildren in Tripoli, Lebanon. PLoS Negl. Trop. Dis. 2016, 10, e0004496. [Google Scholar]
- Scanlan, P.D.; Stensvold, C.R. Blastocystis: Getting to grips with our guileful guest. Trends Parasitol. 2013, 29, 523–529. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Yoshida, K.; Nakajima, A.; Yamanari, K.; Iwatani, S.; Kimata, I. Fecal-oral transmission of the cyst form of Blastocystis hominis in rats. Parasitol. Res. 2004, 94, 391–396. [Google Scholar] [CrossRef]
- Tan, K.S.W. New insights on classification, identification, and clinical relevance of Blastocystis spp. Clin. Microbiol. Rev. 2008, 21, 639–665. [Google Scholar] [CrossRef] [PubMed]
- El Safadi, D.; Gaayeb, L.; Meloni, D.; Cian, A.; Poirier, P.; Wawrzyniak, I.; Delbac, F.; Dabboussi, F.; Delhaes, L.; Seck, M.; et al. Children of Senegal River Basin show the highest prevalence of Blastocystis sp. ever observed worldwide. BMC Infect. Dis. 2014, 14, 164. [Google Scholar] [CrossRef]
- Khaled, S.; Gantois, N.; Tidjani Ly, A.; Senghor, S.; Even, G.; Dautel, E.; Dejager, R.; Sawant, M.; Baydoun, M.; Benamrouz-Vanneste, S.; et al. Prevalence and subtype distribution of Blastocystis sp. in Senegalese school children. Microorganisms 2020, 8, 1408. [Google Scholar] [CrossRef] [PubMed]
- Lokmer, A.; Cian, A.; Froment, A.; Gantois, N.; Viscogliosi, E.; Chabé, M.; Ségurel, L. Use of shotgun metagenomics for the identification of protozoa in the gut microbiota of healthy individuals from worldwide populations with various industrialization levels. PLoS ONE 2019, 14, e0211139. [Google Scholar] [CrossRef] [PubMed]
- Noradilah, S.A.; Moktar, N.; Anuar, T.S.; Lee, I.L.; Salleh, F.M.; Manap, S.N.A.A.; Mohtar, N.S.H.M.; Azrul, S.M.; Abdullah, W.O.; Nordin, A.; et al. Molecular epidemiology of blastocystosis in Malaysia: Does seasonal variation play an important role in determining the distribution and risk factors of Blastocystis subtype infections in the Aboriginal community? Parasit. Vectors 2017, 10, 360. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Velazquez, L.; Maloney, J.G.; Molokin, A.; Moran, P.; Serrano-Vazquez, A.; Gonzalez, E.; Perez-Juarez, H.; Ximenez, C.; Santin, M. Use of next-generation amplicon sequencing to study Blastocystis genetic diversity in a rural human population from Mexico. Parasit. Vectors 2019, 12, 566. [Google Scholar] [CrossRef] [PubMed]
- Higuera, A.; Villamizar, X.; Herrera, G.; Giraldo, J.C.; Vasquez-A, L.R.; Urbano, P.; Villalobos, O.; Tovar, C.; Ramirez, J.D. Molecular detection and genotyping of intestinal protozoa from different biogeographical regions of Colombia. PeerJ 2020, 8, e8554. [Google Scholar] [CrossRef] [PubMed]
- Cian, A.; El Safadi, D.; Osman, M.; Moriniere, R.; Gantois, N.; Benamrouz-Vanneste, S.; Delgado-Viscogliosi, P.; Guyot, K.; Li, L.L.; Monchy, S.; et al. Molecular epidemiology of Blastocystis sp. in various animal groups from two French zoos and evaluation of potential zoonotic risk. PLoS ONE 2017, 12, e0169659. [Google Scholar] [CrossRef]
- Wang, W.; Owen, H.; Traub, R.J.; Cuttell, L.; Inpankaew, T.; Bielefeldt-Ohmann, H. Molecular epidemiology of Blastocystis in pigs and their in-contact humans in Southeast Queensland, Australia, and Cambodia. Vet. Parasitol. 2014, 203, 264–269. [Google Scholar] [CrossRef]
- Greige, S.; El Safadi, D.; Bécu, N.; Gantois, N.; Pereira, B.; Chabé, M.; Benamrouz-Vanneste, S.; Certad, G.; El Hage, R.; Chemaly, M.; et al. Prevalence and subtype distribution of Blastocystis sp. isolates from poultry in Lebanon and evidence of zoonotic potential. Parasit. Vectors 2018, 11, 389. [Google Scholar] [CrossRef]
- Clark, C.G.; van der Giezen, M.; Alfellani, M.A.; Stensvold, C.R. Recent developments in Blastocystis research. Adv. Parasitol. 2013, 82, 1–32. [Google Scholar]
- Stensvold, C.R.; Clark, C.G. Current status of Blastocystis: A personal view. Parasitol Int. 2016, 65, 763–771. [Google Scholar] [CrossRef]
- Andersen, L.O.; Stensvold, C.R. Blastocystis in health and disease: Are we moving from a clinical to a public health perspective? J. Clin. Microbiol. 2016, 54, 524–528. [Google Scholar] [CrossRef]
- Fréalle, E.; El Safadi, D.; Cian, A.; Aubry, E.; Certad, G.; Osman, M.; Wacrenier, A.; Dutoit, E.; Creusy, C.; Dubos, F.; et al. Acute Blastocystis-associated appendicular peritonitis in a child, Casablanca, Morocco. Emerg. Infect. Dis. 2015, 21, 91–94. [Google Scholar] [CrossRef]
- Salvador, F.; Sulleiro, E.; Sanchez-Montalva, A.; Alonso, C.; Santos, J.; Fuentes, I.; Molina, I. Epidemiological and clinical profile of adult patients with Blastocystis sp. infection in Barcelona, Spain. Parasit. Vectors 2016, 9, 548. [Google Scholar] [CrossRef] [PubMed]
- Ajjampur, S.S.; Tan, K.S.W. Pathogenic mechanisms in Blastocystis spp.—Interpreting results from in vitro and in vivo studies. Parasitol Int. 2016, 65, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Gentekaki, E.; Curtis, B.A.; Stairs, C.W.; Klimes, V.; Elias, M.; Salas-Leiva, D.E.; Herman, E.K.; Eme, L.; Arias, M.C.; Henrissat, B.; et al. Extreme genome diversity in the hyper-prevalent parasitic eukaryote Blastocystis. PLoS Biol. 2017, 15, e2003769. [Google Scholar] [CrossRef] [PubMed]
- Noël, C.; Dufernez, F.; Gerbod, D.; Edgcomb, V.P.; Delgado-Viscogliosi, P.; Ho, L.C.; Singh, M.; Wintjens, R.; Sogin, M.L.; Capron, M.; et al. Molecular phylogenies of Blastocystis isolates from different hosts: Implications for genetic diversity, identification of species, and zoonosis. J. Clin. Microbiol. 2005, 43, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Suresh, G.K.; Tan, K.S.; Thompson, R.C.; Traub, R.J.; Viscogliosi, E.; Yoshikawa, H.; Clark, C.G. Terminology for Blastocystis subtypes a consensus. Trends Parasitol. 2007, 23, 93–96. [Google Scholar] [CrossRef]
- Alfellani, M.A.; Taner-Mulla, D.; Jacob, A.S.; Imeede, C.A.; Yoshikawa, H.; Stensvold, C.R.; Clark, C.G. Genetic diversity of Blastocystis in livestock and zoo animals. Protist 2013, 164, 497–509. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Koyama, Y.; Tsuchiya, E.; Takami, K. Blastocystis phylogeny among various isolates from humans to insects. Parasitol. Int. 2016, 65, 750–759. [Google Scholar] [CrossRef]
- Zhao, G.H.; Hu, X.F.; Liu, T.L.; Hu, R.S.; Yu, Z.Q.; Yang, W.B.; Wu, Y.L.; Yu, S.K.; Song, J.K. Molecular characterization of Blastocystis sp. in captive wild animals in Qinling Mountains. Parasitol. Res. 2017, 116, 2327–2333. [Google Scholar] [CrossRef]
- Maloney, J.G.; Lombard, J.E.; Urie, N.J.; Shivley, C.B.; Santin, M. Zoonotic and genetically diverse subtypes of Blastocystis in US preweaned dairy heifer calves. Parasitol. Res. 2019, 118, 575–582. [Google Scholar] [CrossRef]
- Gantois, N.; Lamot, A.; Seesao, Y.; Creusy, C.; Li, L.L.; Monchy, S.; Benamrouz-Vanneste, S.; Karpouzopoulos, J.; Bourgain, J.L.; Rault, C.; et al. First report on the prevalence and subtype distribution of Blastocystis sp. in edible marine fish and marine mammals: A large-scale study conducted in Atlantic Northeast and on the coasts of Northern France. Microorganisms 2020, 8, 460. [Google Scholar] [CrossRef]
- Alfellani, M.A.; Stensvold, C.R.; Vidal-Lapiedra, A.; Onuoha, E.S.; Fagbenro-Beyioku, A.F.; Clark, C.G. Variable geographic distribution of Blastocystis subtypes and its potential implications. Acta Trop. 2013, 126, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, J.D.; Sánchez, A.; Hernández, C.; Florez, C.; Bernal, M.C.; Giraldo, J.C.; Reyes, P.; Lopez, M.C.; Garcia, L.; Cooper, P.J.; et al. Geographic distribution of human Blastocystis subtypes in South America. Infect. Genet. Evol. 2016, 41, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, P.A.; Jaimes, J.E.; Ramirez, J.D. A summary of Blastocystis subtypes in North and South America. Parasit. Vectors 2019, 12, 376. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Alfellani, M.A.; Norskov-Lauritsen, S.; Prip, K.; Victory, E.L.; Maddox, C.; Nielsen, H.V.; Clark, C.G. Subtype distribution of Blastocystis isolates from synanthropic and zoo animals and identification of a new subtype. Int. J. Parasitol. 2009, 39, 473–479. [Google Scholar] [CrossRef]
- El Safadi, D.; Meloni, D.; Poirier, P.; Osman, M.; Cian, A.; Gaayeb, L.; Wawrzyniak, I.; Delbac, F.; El Alaoui, H.; Delhaes, L.; et al. Molecular epidemiology of Blastocystis in Lebanon and correlation between subtype 1 and gastrointestinal symptoms. Am. J. Trop. Med. Hyg. 2013, 88, 1203–1206. [Google Scholar] [CrossRef]
- Greige, S.; El Safadi, D.; Khaled, S.; Gantois, N.; Baydoun, M.; Chemaly, M.; Benamrouz-Vanneste, S.; Chabé, M.; Osman, M.; Certad, G.; et al. First report on the prevalence and subtype distribution of Blastocystis sp. in dairy cattle in Lebanon and assessment of zoonotic transmission. Acta Trop. 2019, 194, 23–29. [Google Scholar] [CrossRef]
- Moosavi, A.; Haghighi, A.; Nazemalhosseini, E.; Zayeri, F.; Alebouyeh, M.; Khazan, H.; Kazemi, B.; Zali, M.R. Genetic variability of Blastocystis sp. isolated from symptomatic and asymptomatic individuals in Iran. Parasitol. Res. 2012, 111, 2311–2315. [Google Scholar] [CrossRef]
- Alinaghizade, A.; Mirjalali, H.; Mohebali, M.; Stensvold, C.R.; Rezaeian, M. Inter- and intra-subtype variation of Blastocystis subtypes isolated from diarrheic and non-diarrheic patients in Iran. Infect. Genet. Evol. 2017, 50, 77–82. [Google Scholar] [CrossRef]
- Mohamed, R.T.; El-Bali, M.A.; Mohamed, A.A.; Abdel-Fatah, M.A.; El-Malky, M.A.; Mowafy, N.M.; Zaghlool, D.A.; Bakri, R.A.; Al-Harthi, S.A. Subtyping of Blastocystis sp. isolated from symptomatic and asymptomatic individuals in Makkah, Saudi Arabia. Parasit. Vectors 2017, 10, 174. [Google Scholar] [CrossRef]
- Ghanem, N. Local Governance under Pressure: Research on Social Stability in T5 Area, North Lebanon. Menapolis and OXFAM Italia. 2016. Available online: https://reliefweb.int/report/lebanon/local-governance-under-pressure-research-social-stability-t5-area-north-lebanon (accessed on 7 March 2016).
- Poirier, P.; Wawrzyniak, I.; Albert, A.; El Alaoui, H.; Delbac, F.; Livrelli, V. Development and evaluation of a real-time PCR assay for detection and quantification of Blastocystis parasites in human stool samples: Prospective study of patients with hematological malignancies. J. Clin. Microbiol. 2011, 49, 975–983. [Google Scholar] [CrossRef]
- Badparva, E.; Sadraee, J.; Kheirandish, F.; Frouzandeh, M. Genetic diversity of human Blastocystis isolates in Khorramabad, central Iran. Iran J. Parasitol. 2014, 9, 44–49. [Google Scholar] [PubMed]
- Abu-Madi, M.; Aly, M.; Behnke, J.M.; Clark, C.G.; Balkhy, H. The distribution of Blastocystis subtypes in isolates from Qatar. Parasit. Vectors 2015, 8, 465. [Google Scholar] [CrossRef] [PubMed]
- AbuOdeh, R.; Ezzedine, S.; Samie, A.; Stensvold, C.R.; ElBakri, A. Prevalence and subtype distribution of Blastocystis in healthy individuals in Sharjah, United Arab Emirates. Infect. Genet. Evol. 2016, 37, 158–162. [Google Scholar] [CrossRef] [PubMed]
- El Safadi, D.; Cian, A.; Nourrisson, C.; Pereira, B.; Morelle, C.; Bastien, P.; Bellanger, A.P.; Botterel, F.; Candolfi, E.; Desoubeaux, G.; et al. Prevalence, risk factors for infection and subtype distribution of the intestinal parasite Blastocystis sp. from a large-scale multi-center study in France. BMC Infect. Dis. 2016, 16, 451. [Google Scholar] [CrossRef]
- Scanlan, P.D.; Stensvold, C.R.; Cotter, P.D. Development and application of a Blastocystis subtype-specific PCR assay reveals that mixed-subtype infections are common in a healthy human population. Appl. Environ. Microbiol. 2015, 81, 4071–4076. [Google Scholar] [CrossRef]
- Riabi, T.R.; Mirjalali, H.; Haghighi, A.; Nejad, M.R.; Pourhoseingholi, M.A.; Poirier, P.; Delbac, F.; Wawrzyniak, I.; Zali, M.R. Genetic diversity analysis of Blastocystis subtypes from both symptomatic and asymptomatic subjects using a barcoding region from the 18S rRNA gene. Infect. Genet. Evol. 2018, 61, 119–126. [Google Scholar] [CrossRef]
- Shirvani, G.; Fasihi-Harandi, M.; Raiesi, O.; Bazargan, N.; Zahedi, M.J.; Sharifi, I.; Kalantari-Khandani, B.; Nooshadokht, M.; Shabandoust, H.; Mohammadi, M.A.; et al. Prevalence and molecular subtyping of Blastocystis from patients with irritable bowel syndrome, inflammatory bowel disease and chronic urticaria in Iran. Acta Parasitol. 2020, 65, 90–96. [Google Scholar] [CrossRef]
- Forsell, J.; Granlund, M.; Stensvold, C.R.; Clark, C.G.; Evengard, B. Subtype analysis of Blastocystis isolates in Swedish patients. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1689–1696. [Google Scholar] [CrossRef][Green Version]
- Khoshnood, S.; Rafiei, A.; Saki, J.; Alizadeh, K. Prevalence and genotype characterization of Blastocystis hominis among the Baghmalek people in Southwestern Iran in 2013–2014. Jundishapur J. Microbiol. 2015, 8, e23930. [Google Scholar] [CrossRef]
- Khademvatan, S.; Masjedizadeh, R.; Rahim, F.; Mahbodfar, H.; Salehi, R.; Yousefi-Razin, E.; Foroutan, M. Blastocystis and irritable bowel syndrome: Frequency and subtypes from Iranian patients. Parasitol. Int. 2017, 66, 142–145. [Google Scholar] [CrossRef]
- Piranshahi, A.R.; Tavalla, M.; Khademvatan, S. Genomic analysis of Blastocystis hominis isolates in patients with HIV-positive using locus SSU-rDNA. J. Parasit. Dis. 2018, 42, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Audebert, C.; Even, G.; Cian, A.; Blastocystis Investigation Group; Loywick, A.; Merlin, S.; Viscogliosi, E.; Chabé,, M. Colonization with the enteric protozoa Blastocystis is associated with increased diversity of human gut bacterial microbiota. Sci. Rep. 2016, 6, 25255. [Google Scholar] [CrossRef] [PubMed]
- Beghini, F.; Pasolli, E.; Truong, T.D.; Putignani, L.; Caccio, S.M.; Segata, N. Large-scale comparative metagenomics of Blastocystis, a common member of the human gut microbiome. ISME J. 2017, 11, 2848–2863. [Google Scholar] [CrossRef] [PubMed]
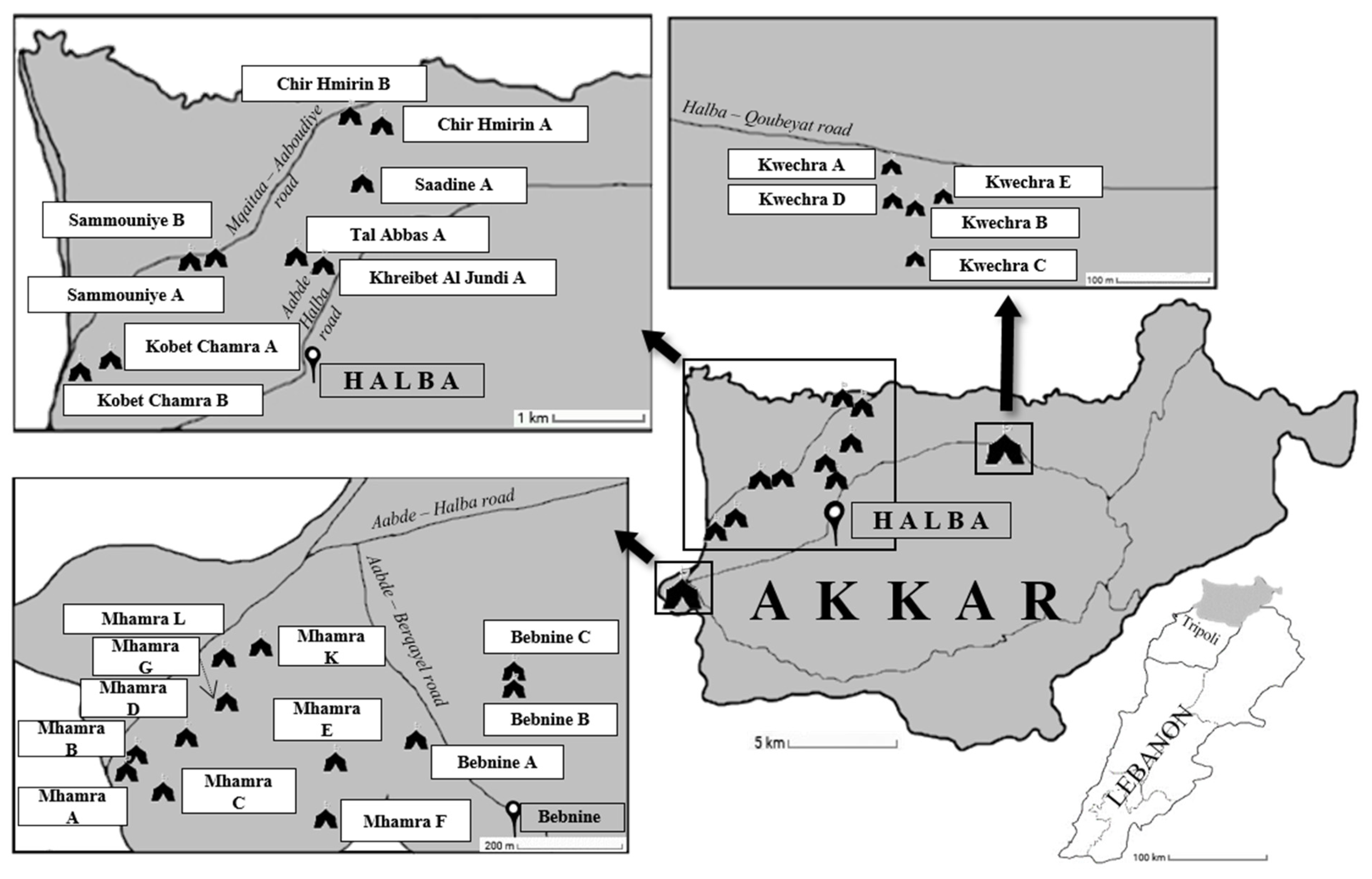
| ITS a | Samples (n) | Positive Samples (n) | Prevalence (%) | Blastocystis sp. STs | ||||
|---|---|---|---|---|---|---|---|---|
| ST1 | ST2 | ST3 | ST10 | MI b | ||||
| With WI c | ||||||||
| Kwechra A | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Kwechra B | 6 | 6 | 100 | 2 | 1 | 2 | 0 | 1 |
| Kwechra C | 7 | 4 | 57.1 | 0 | 0 | 2 | 0 | 2 |
| Kwechra D | 13 | 12 | 92.3 | 4 | 2 | 4 | 0 | 2 |
| Kwechra E | 7 | 5 | 71.4 | 1 | 0 | 4 | 0 | 0 |
| Mhamra A | 16 | 8 | 50 | 2 | 0 | 6 | 0 | 0 |
| Mhamra B | 21 | 15 | 71.4 | 3 | 0 | 11 | 0 | 1 |
| Mhamra C | 39 | 21 | 53.8 | 2 | 8 | 5 | 0 | 6 |
| Mhamra D | 14 | 7 | 50 | 2 | 3 | 1 | 0 | 1 |
| Mhamra E | 17 | 10 | 58.8 | 1 | 0 | 7 | 0 | 2 |
| Chir Hmirin A | 3 | 3 | 100 | 2 | 0 | 1 | 0 | 0 |
| Chir Hmirin B | 2 | 2 | 100 | 0 | 0 | 2 | 0 | 0 |
| Khraybet Al Jundi A | 16 | 9 | 56.3 | 1 | 3 | 3 | 0 | 2 |
| Sammouniye A | 3 | 2 | 66.7 | 0 | 0 | 1 | 0 | 1 |
| Saadine A | 17 | 5 | 29.4 | 0 | 2 | 3 | 0 | 0 |
| Tal Abbas A | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total | 184 | 109 | 59.2 | 20 | 19 | 52 | 0 | 18 |
| Without WI | ||||||||
| Bebnine A | 8 | 4 | 50 | 2 | 0 | 1 | 0 | 1 |
| Bebnine B | 10 | 5 | 50 | 2 | 1 | 2 | 0 | 0 |
| Bebnine C | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Mhamra F | 4 | 4 | 100 | 1 | 2 | 0 | 0 | 1 |
| Mhamra G | 6 | 4 | 66.7 | 0 | 1 | 2 | 1 | 0 |
| Mhamra K | 2 | 2 | 100 | 0 | 0 | 2 | 0 | 0 |
| Mhamra L | 6 | 5 | 83.3 | 2 | 1 | 2 | 0 | 0 |
| Kobet Chamra A | 16 | 11 | 68.8 | 4 | 1 | 6 | 0 | 0 |
| Kobet Chamra B | 58 | 43 | 74.1 | 9 | 3 | 20 | 0 | 11 |
| Sammouniye B | 11 | 8 | 72.7 | 3 | 3 | 2 | 0 | 0 |
| Total | 122 | 86 | 70.5 | 23 | 12 | 37 | 1 | 13 |
| Grand total | 306 | 195 | 63.7 | 43 | 31 | 89 | 1 | 31 |
| Cohort | Blastocystis sp. STs | Reference | ||
|---|---|---|---|---|
| ST1 | ST2 | ST3 | ||
| Lebanon | 12 (34.3%) | 12 (34.3%) | 11 (31.4%) | [35] |
| Lebanon | 35 (25.4%) | 39 (28.2%) | 64 (46.4%) | [4] |
| Lebanon | 8 (14.8%) | 11 (20.4%) | 35 (64.8%) | [16] |
| Lebanon | 13 (26.0%) | 12 (24.0%) | 25 (50.0%) | [36] |
| Total Lebanon | 68 (24.5%) | 74 (26.7%) | 135 (48.8%) | |
| Syrian refugees | 43 (26.4%) | 31 (19.0%) | 89 (54.6%) | Present study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khaled, S.; Gantois, N.; Ayoubi, A.; Even, G.; Sawant, M.; El Houmayraa, J.; Nabot, M.; Benamrouz-Vanneste, S.; Chabé, M.; Certad, G.; et al. Blastocystis sp. Prevalence and Subtypes Distribution amongst Syrian Refugee Communities Living in North Lebanon. Microorganisms 2021, 9, 184. https://doi.org/10.3390/microorganisms9010184
Khaled S, Gantois N, Ayoubi A, Even G, Sawant M, El Houmayraa J, Nabot M, Benamrouz-Vanneste S, Chabé M, Certad G, et al. Blastocystis sp. Prevalence and Subtypes Distribution amongst Syrian Refugee Communities Living in North Lebanon. Microorganisms. 2021; 9(1):184. https://doi.org/10.3390/microorganisms9010184
Chicago/Turabian StyleKhaled, Salma, Nausicaa Gantois, Aisha Ayoubi, Gaël Even, Manasi Sawant, Jinane El Houmayraa, Mathieu Nabot, Sadia Benamrouz-Vanneste, Magali Chabé, Gabriela Certad, and et al. 2021. "Blastocystis sp. Prevalence and Subtypes Distribution amongst Syrian Refugee Communities Living in North Lebanon" Microorganisms 9, no. 1: 184. https://doi.org/10.3390/microorganisms9010184
APA StyleKhaled, S., Gantois, N., Ayoubi, A., Even, G., Sawant, M., El Houmayraa, J., Nabot, M., Benamrouz-Vanneste, S., Chabé, M., Certad, G., El Safadi, D., Dabboussi, F., Hamze, M., & Viscogliosi, E. (2021). Blastocystis sp. Prevalence and Subtypes Distribution amongst Syrian Refugee Communities Living in North Lebanon. Microorganisms, 9(1), 184. https://doi.org/10.3390/microorganisms9010184







