Animal Fat as a Substrate for Production of n-6 Fatty Acids by Fungal Solid-State Fermentation
Abstract
1. Introduction
2. Materials and Methods
2.1. Production Microorganism
2.2. Inoculation of Solid-State Fermentation Substrate and Fermentation Conditions
2.3. Preparation of AF Emulsion
2.4. Humidity and Substrate Utilization Analysis
2.5. Estimation of Fungal Biomass and Glucosamine Amount in the Fermented Bioproduct
2.6. Complex Lipid and Carotenoid Pigments Extraction
2.7. Analysis of Carotenoid Pigments by High-Performance Liquid Chromatography (HPLC)
2.8. Analysis of Fatty Acid Profile and Content in Fermented Bioproduct
2.9. Analysis of Lipid Classes by Thin-Layer Chromatography (TLC)
2.10. Antioxidant Activity Assay
2.11. ATR-FTIR Spectroscopy Analysis
2.12. Data Analysis
3. Results
3.1. Wheat Bran Utilization and U.isabellina Growth Characteristics During SSF
3.2. Fungal Lipids Accumulation and Lipid Profile
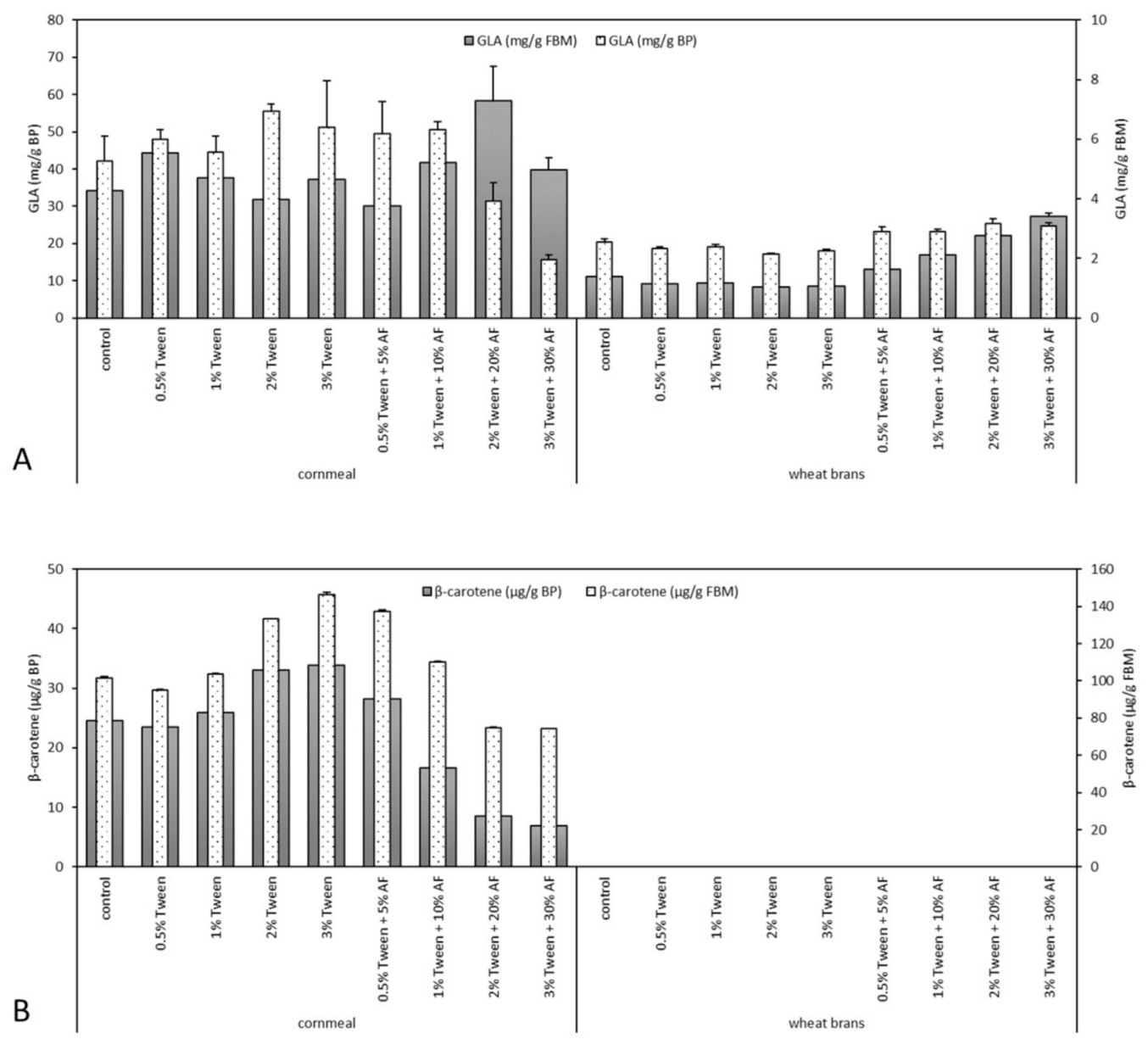
3.3. γ-Linolenic Acid Yield
3.4. β-Carotene Accumulation
3.5. Glucosamine Accumulation
3.6. Antioxidant Activity
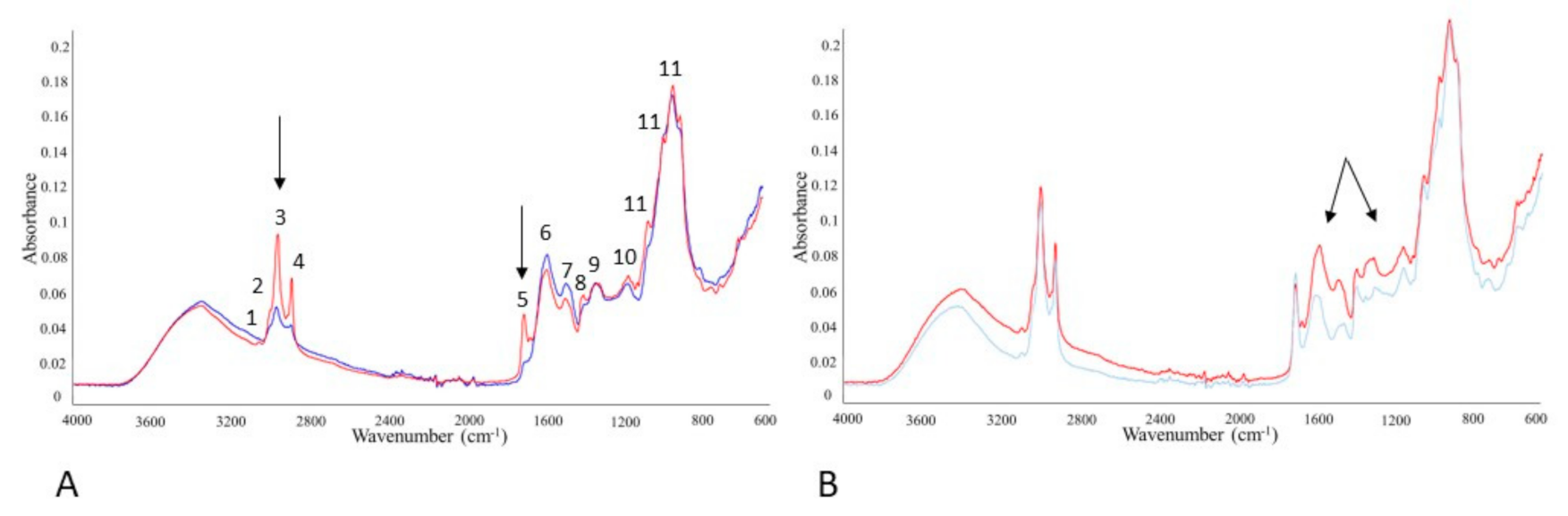
3.7. Monitoring SFF Bioproducts by FTIR-ATR Spectroscopy
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zalouk, S.; Barbati, S.; Sergent, M.; Ambrosio, M. Disposal of Animal By-Products by Wet Air Oxidation: Performance Optimization and Kinetics. Chemosphere 2009, 74, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Toldrá-Reig, F.; Mora, L.; Toldrá, F. Trends in Biodiesel Production from Animal Fat Waste. Appl. Sci. 2020, 10, 3644. [Google Scholar] [CrossRef]
- Encinar, J.M.; Sánchez, N.; Martínez, G.; García, L. Study of Biodiesel Production from Animal Fats with High Free Fatty Acid Content. Bioresour. Technol. 2011, 102, 10907–10914. [Google Scholar] [CrossRef] [PubMed]
- Mata, T.; Mendes, A.; Caetano, N.; Martins, A. Properties and Sustainability of Biodiesel from Animal Fats and Fish Oil. Chem. Eng. Trans. 2014, 38, 175–180. [Google Scholar] [CrossRef]
- Asadi, S.Z.; Khosravi-Darani, K.; Nikoopour, H.; Bakhoda, H. Evaluation of the Effect of Process Variables on the Fatty Acid Profile of Single Cell Oil Produced by Mortierella Using Solid-State Fermentation. Crit. Rev. Biotechnol. 2015, 35, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Klempová, T.; Slaný, O.; Šišmiš, M.; Marcinčák, S.; Čertík, M. Dual Production of Polyunsaturated Fatty Acids and Beta-Carotene with Mucor wosnessenskii by the Process of Solid-State Fermentation Using Agro-Industrial Waste. J. Biotechnol. 2020, 311, 1–11. [Google Scholar] [CrossRef]
- Daskalaki, A.; Vasiliadou, I.A.; Bellou, S.; Tomaszewska-Hetman, L.; Chatzikotoula, C.; Kompoti, B.; Papanikolaou, S.; Vayenas, D.; Pavlou, S.; Aggelis, G. Data on Cellular Lipids of Yarrowia lipolytica Grown on Fatty Substrates. Data Brief 2018, 21, 1037–1044. [Google Scholar] [CrossRef]
- Čertík, M.; Klempová, T.; Guothová, L.; Mihálik, D.; Kraic, J. Biotechnology for the Functional Improvement of Cereal-Based Materials Enriched with PUFA and Pigments: Biotechnologically Enriched Cereals with PUFAs and Pigments. Eur. J. Lipid Sci. Technol. 2013, 115, 1247–1256. [Google Scholar] [CrossRef]
- Čertík, M.; Shimizu, S. Biosynthesis and Regulation of Microbial Polyunsaturated Fatty Acid Production. J. Biosci. Bioeng. 1999, 87, 1–14. [Google Scholar] [CrossRef]
- Szczęsna-Antczak, M.; Antczak, T.; Piotrowicz-Wasiak, M.; Rzyska, M.; Binkowska, N.; Bielecki, S. Relationships between Lipases and Lipids in Mycelia of Two Mucor Strains. Enzym. Microb. Technol. 2006, 39, 1214–1222. [Google Scholar] [CrossRef]
- Soccol, C.R.; Da Costa, E.S.F.; Letti, L.A.J.; Karp, S.G.; Woiciechowski, A.L.; Vandenberghe, L.P.d.S. Recent Developments and Innovations in Solid State Fermentation. Biotechnol. Res. Innov. 2017, 1, 52–71. [Google Scholar] [CrossRef]
- Lizardi-Jiménez, M.A.; Hernández-Martínez, R. Solid State Fermentation (SSF): Diversity of Applications to Valorize Waste and Biomass. 3 Biotech 2017, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Slaný, O.; Klempová, T.; Shapaval, V.; Zimmermann, B.; Kohler, A.; Čertík, M. Biotransformation of Animal Fat-By Products into ARA-Enriched Fermented Bioproducts by Solid-State Fermentation of Mortierella alpina. J. Fungi 2020, 6, 236. [Google Scholar] [CrossRef] [PubMed]
- Stressler, T.; Eisele, T.; Rost, J.; Haunschild, E.-M.; Kuhn, A.; Fischer, L. Production of Polyunsaturated Fatty Acids by Mortierella alpina Using Submerse and Solid State Fermentation. Chem. Ing. Tech. 2013, 85, 318–322. [Google Scholar] [CrossRef]
- Peng, J.W.; Wu, H.S. Kinetic Study of Glucosamine Production Using Aspergillus sydowii BCRC 31742 under Solid-State Fermentation. Molecules 2020, 25, 4832. [Google Scholar] [CrossRef]
- Klempová, T.; Basil, E.; Kubatova, A.; Čertík, M. Biosynthesis of Gamma-Linolenic Acid and Beta-Carotene by Zygomycetes Fungi. Biotechnol. J. 2013, 8, 794–800. [Google Scholar] [CrossRef]
- Slaný, O.; Klempová, T.; Marcinčák, S.; Čertík, M. Production of High-Value Bioproducts Enriched with γ-Linolenic Acid and β-Carotene by Filamentous Fungi Umbelopsis isabellina Using Solid-State Fermentations. Ann. Microbiol. 2020, 70, 5. [Google Scholar] [CrossRef]
- Hernández-Almanza, A.; Montañez-Sáenz, J.; Martínez-Ávila, C.; Rodríguez-Herrera, R.; Aguilar, C.N. Carotenoid Production by Rhodotorula glutinis YB-252 in Solid-State Fermentation. Food Biosci. 2014, 7, 31–36. [Google Scholar] [CrossRef]
- Pyo, Y.-H.; Seo, S.-Y. Simultaneous Production of Natural Statins and Coenzyme Q10 by Monascus pilosus Fermentation Using Different Solid Substrates. Food Sci. Biotechnol. 2010, 19, 1635–1641. [Google Scholar] [CrossRef]
- Dos Santos Costa, R.; de Almeida, S.S.; Cavalcanti, E.; d’Avila, C.; Freire, D.M.G.; Moura-Nunes, N.; Monteiro, M.; Perrone, D. Enzymes Produced by Solid State Fermentation of Agro-Industrial by-Products Release Ferulic Acid in Bioprocessed Whole-Wheat Breads. Food Res. Int. 2020, 109843. [Google Scholar] [CrossRef]
- Aguieiras, E.C.G.; Cavalcanti-Oliveira, E.D.; Cammarota, M.C.; Freire, D.M.G. Solid-State Fermentation for the Production of Lipases for Environmental and Biodiesel Applications. In Current Developments in Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2018; pp. 123–168. ISBN 9780444639905. [Google Scholar]
- Ertan, F.; Balkan, B.; Balkan, S.; Aktac, T. Solid State Fermentation for the Production of α-Amylase from Penicillium chrysogenum Using Mixed Agricultural by-Products as Substrate. Biologia 2006, 61, 657–661. [Google Scholar] [CrossRef]
- Vasiliadou, I.A.; Bellou, S.; Daskalaki, A.; Tomaszewska- Hetman, L.; Chatzikotoula, C.; Kompoti, B.; Papanikolaou, S.; Vayenas, D.; Pavlou, S.; Aggelis, G. Biomodification of Fats and Oils and Scenarios of Adding Value on Renewable Fatty Materials through Microbial Fermentations: Modelling and Trials with Yarrowia lipolytica. J. Clean. Prod. 2018, 200, 1111–1129. [Google Scholar] [CrossRef]
- Lopes, M.; Gomes, A.S.; Silva, C.M.; Belo, I. Microbial Lipids and Added Value Metabolites Production by Yarrowia lipolytica from Pork Lard. J. Biotechnol. 2018, 265, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Xiong, D.; Zhang, H.; Xie, Y.; Tang, N.; Berenjian, A.; Song, Y. Conversion of Mutton Fat to Cocoa Butter Equivalent by Increasing the Unsaturated Fatty Acids at the Sn-2 Position of Triacylglycerol Through Fermentation by Yarrowia lipolytica. Am. J. Biochem. Biotechnol. 2015, 11, 57–65. [Google Scholar] [CrossRef]
- Radha, P.; Narayanan, S.; Chaudhuri, A.; Anjum, S.; Thomas, D.L.; Pandey, R.; Ramani, K. Synthesis of Single-Cell Oil by Yarrowia lipolytica MTCC 9520 Utilizing Slaughterhouse Lipid Waste for Biodiesel Production. Biomass Conv. Bioref. 2020, 1–12. [Google Scholar] [CrossRef]
- Papanikolaou, I.; Chevalot, M.; Komai, S. Single Cell Oil Production by Yarrowia lipolytica Growing on an Industrial Derivative of Animal Fat in Batch Cultures. Appl. Microbiol. Biotechnol. 2002, 58, 308–312. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Aggelis, G. Selective Uptake of Fatty Acids by the Yeast Yarrowia lipolytica. Eur. J. Lipid Sci. Technol. 2003, 105, 651–655. [Google Scholar] [CrossRef]
- Nykiel-Szymańska, J.; Stolarek, P.; Bernat, P. Elimination and Detoxification of 2,4-D by Umbelopsis isabellina with the Involvement of Cytochrome P450. Env. Sci. Pollut. Res. 2018, 25, 2738–2743. [Google Scholar] [CrossRef]
- Janicki, T.; Długoński, J.; Krupiński, M. Detoxification and Simultaneous Removal of Phenolic Xenobiotics and Heavy Metals with Endocrine-Disrupting Activity by the Non-Ligninolytic Fungus Umbelopsis isabellina. J. Hazard. Mater. 2018, 360, 661–669. [Google Scholar] [CrossRef]
- Gardeli, C.; Athenaki, M.; Xenopoulos, E.; Mallouchos, A.; Koutinas, A.A.; Aggelis, G.; Papanikolaou, S. Lipid Production and Characterization by Mortierella (Umbelopsis) isabellina Cultivated on Lignocellulosic Sugars. J. Appl. Microbiol. 2017, 123, 1461–1477. [Google Scholar] [CrossRef]
- Somacal, S.; Pinto, V.S.; Vendruscolo, R.G.; Somacal, S.; Wagner, R.; Ballus, C.A.; Kuhn, R.C.; Mazutti, M.A.; Menezes, C.R. Maximization of Microbial Oil Containing Polyunsaturated Fatty Acid Production by Umbelopsis (Mortierella) isabellina. Biocatal. Agric. Biotechnol. 2020, 30, 101831. [Google Scholar] [CrossRef]
- Krishna, C. Solid-State Fermentation Systems—An Overview. Crit. Rev. Biotechnol. 2005, 25, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Dulf, F.V.; Vodnar, D.C.; Toşa, M.I.; Dulf, E.-H. Simultaneous Enrichment of Grape Pomace with γ-Linolenic Acid and Carotenoids by Solid-State Fermentation with Zygomycetes Fungi and Antioxidant Potential of the Bioprocessed Substrates. Food Chem. 2020, 310, 125927. [Google Scholar] [CrossRef] [PubMed]
- Marcinčák, S.; Klempová, T.; Bartkovský, M.; Marcinčáková, D.; Zdolec, N.; Popelka, P.; Mačanga, J.; Čertík, M. Effect of Fungal Solid-State Fermented Product in Broiler Chicken Nutrition on Quality and Safety of Produced Breast Meat. Biomed Res. Int. 2018, 2018, 1–8. [Google Scholar] [CrossRef]
- Zamani, A.; Jeihanipour, A.; Edebo, L.; Niklasson, C.; Taherzadeh, M.J. Determination of Glucosamine and N -Acetyl Glucosamine in Fungal Cell Walls. J. Agric. Food Chem. 2008, 56, 8314–8318. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley Sloane, G.H. A Simple Method for the Isolation and Purification of Total Lipides from Animal Tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Čertík, M.; Shimizu, S. Kinetic Analysis of Oil Biosynthesis by an Arachidonic Acid-Producing Fungus, Mortierella alpina 1S-4. Appl. Microbiol. Biotechnol. 2000, 54, 224–230. [Google Scholar] [CrossRef]
- Gajdoš, P.; Nicaud, J.-M.; Rossignol, T.; Čertík, M. Single Cell Oil Production on Molasses by Yarrowia lipolytica Strains Overexpressing DGA2 in Multicopy. Appl. Microbiol. Biotechnol. 2015, 99, 8065–8074. [Google Scholar] [CrossRef]
- Kosa, G.; Kohler, A.; Tafintseva, V.; Zimmermann, B.; Forfang, K.; Afseth, N.K.; Tzimorotas, D.; Vuoristo, K.S.; Horn, S.J.; Mounier, J.; et al. Microtiter Plate Cultivation of Oleaginous Fungi and Monitoring of Lipogenesis by High-Throughput FTIR Spectroscopy. Microb. Cell Fact 2017, 16, 101. [Google Scholar] [CrossRef]
- Forfang, K.; Zimmermann, B.; Kosa, G.; Kohler, A.; Shapaval, V. FTIR Spectroscopy for Evaluation and Monitoring of Lipid Extraction Efficiency for Oleaginous Fungi. PLoS ONE 2017, 12, e0170611. [Google Scholar] [CrossRef]
- Kosa, G.; Zimmermann, B.; Kohler, A.; Ekeberg, D.; Afseth, N.K.; Mounier, J.; Shapaval, V. High-Throughput Screening of Mucoromycota Fungi for Production of Low- and High-Value Lipids. Biotechnol. Biofuels 2018, 11, 66. [Google Scholar] [CrossRef] [PubMed]
- Dzurendova, S.; Zimmermann, B.; Kohler, A.; Tafintseva, V.; Slany, O.; Certik, M.; Shapaval, V. Microcultivation and FTIR Spectroscopy-Based Screening Revealed a Nutrient-Induced Co-Production of High-Value Metabolites in Oleaginous Mucoromycota Fungi. PLoS ONE 2020, 15, e0234870. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, S.; Dimou, A.; Fakas, S.; Diamantopoulou, P.; Philippoussis, A.; Galiotou-Panayotou, M.; Aggelis, G. Biotechnological Conversion of Waste Cooking Olive Oil into Lipid-Rich Biomass Using Aspergillus and Penicillium Strains: Waste Oil Valourization by Higher Fungi. J. Appl. Microbiol. 2011, 110, 1138–1150. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, P.K.; Bajpai, P.; Ward, O.P. Optimisation of Culture Conditions for Production of Eicosapentaenoic Acid by Mortierella elongata NRRL 5513. J. Ind. Microbiol. 1992, 9, 11–17. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Chevalot, I.; Komaitis, M.; Aggelis, G.; Marc, I. Kinetic Profile of the Cellular Lipid Composition in an Oleaginous Yarrowia lipolytica Capable of Producing a Cocoa-Butter Substitute from Industrial Fats. Antonie van Leeuwenhoek 2001, 80, 215–224. [Google Scholar] [CrossRef]
- Márová, I.; Szotkowski, M.; Vanek, M.; Rapta, M.; Byrtusova, D.; Mikheichyk, N.; Haronikova, A.; Čertík, M.; Shapaval, V. Utilization of Animal Fat Waste as Carbon Source by Carotenogenic Yeasts — A Screening Study. Eurobiotech J. 2017, 1, 310–318. [Google Scholar] [CrossRef]
- Szczęsna-Antczak, M.; Struszczyk-Świta, K.; Rzyska, M.; Szeląg, J.; Stańczyk, Ł.; Antczak, T. Oil Accumulation and in Situ Trans/Esterification by Lipolytic Fungal Biomass. Bioresour. Technol. 2018, 265, 110–118. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Aggelis, G. Yarrowia lipolytica: A Model Microorganism Used for the Production of Tailor-Made Lipids. Eur. J. Lipid Sci. Technol. 2010, 112, 639–654. [Google Scholar] [CrossRef]
- Ng, T.-K.; Yu, A.-Q.; Ling, H.; Pratomo Juwono, N.K.; Choi, W.J.; Leong, S.S.J.; Chang, M.W. Engineering Yarrowia lipolytica towards Food Waste Bioremediation: Production of Fatty Acid Ethyl Esters from Vegetable Cooking Oil. J. Biosci. Bioeng. 2020, 129, 31–40. [Google Scholar] [CrossRef]
- Najjar, A.; Robert, S.; Guérin, C.; Violet-Asther, M.; Carrière, F. Quantitative Study of Lipase Secretion, Extracellular Lipolysis, and Lipid Storage in the Yeast Yarrowia lipolytica Grown in the Presence of Olive Oil: Analogies with Lipolysis in Humans. Appl. Microbiol. Biotechnol. 2011, 89, 1947–1962. [Google Scholar] [CrossRef]
- Stredansky, M.; Conti, E.; Stredanska, S.; Zanetti, F. γ-Linolenic Acid Production with Thamnidium elegans by Solid-State Fermentation on Apple Pomace. Bioresour. Technol. 2000, 73, 41–45. [Google Scholar] [CrossRef]
- Čertík, M.; Adamechová, Z.; Laoteng, K. Microbial Production of γ-Linolenic Acid: Submerged versus Solid-State Fermentations. Food Sci. Biotechnol. 2012, 21, 921–926. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Aggelis, G. Sources of Microbial Oils with Emphasis to Mortierella (Umbelopsis) isabellina Fungus. World J. Microbiol. Biotechnol. 2019, 35, 63. [Google Scholar] [CrossRef] [PubMed]
- Čertík, M.; Adamechová, Z.; Guothová, L. Simultaneous Enrichment of Cereals with Polyunsaturated Fatty Acids and Pigments by Fungal Solid State Fermentations. J. Biotechnol. 2013, 168, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Jangbua, P.; Laoteng, K.; Kitsubun, P.; Nopharatana, M.; Tongta, A. Gamma-Linolenic Acid Production of Mucor rouxii by Solid-State Fermentation Using Agricultural by-Products. Lett. Appl. Microbiol. 2009, 49, 91–97. [Google Scholar] [CrossRef]
- Merali, Z.; Collins, S.R.A.; Elliston, A.; Wilson, D.R.; Käsper, A.; Waldron, K.W. Characterization of Cell Wall Components of Wheat Bran Following Hydrothermal Pretreatment and Fractionation. Biotechnol. Biofuels 2015, 8, 23. [Google Scholar] [CrossRef]
- Artmann, D.J.; Amrain, W.; Murauer, A.; Ganzera, M.; Vrabl, P.; Schinagl, C.W.; Burgstaller, W. Critical Evaluation of a Putative Glucosamine Excretion by Aspergillus niger CBS120.49 and Penicillium ochrochloron CBS123.824 under Citric Acid Producing Conditions. Sci. Rep. 2019, 9, 7496. [Google Scholar] [CrossRef]
- Liu, L.; Liu, Y.; Shin, H.; Chen, R.; Li, J.; Du, G.; Chen, J. Microbial Production of Glucosamine and N-Acetylglucosamine: Advances and Perspectives. Appl. Microbiol. Biotechnol. 2013, 97, 6149–6158. [Google Scholar] [CrossRef]
- Hur, S.J.; Lee, S.Y.; Kim, Y.-C.; Choi, I.; Kim, G.-B. Effect of Fermentation on the Antioxidant Activity in Plant-Based Foods. Food Chem. 2014, 160, 346–356. [Google Scholar] [CrossRef]
- Torino, M.I.; Limón, R.I.; Martínez-Villaluenga, C.; Mäkinen, S.; Pihlanto, A.; Vidal-Valverde, C.; Frias, J. Antioxidant and Antihypertensive Properties of Liquid and Solid State Fermented Lentils. Food Chem. 2013, 136, 1030–1037. [Google Scholar] [CrossRef]
- Fernandez-Orozco, R.; Frias, J.; Muñoz, R.; Zielinski, H.; Piskula, M.K.; Kozlowska, H.; Vidal-Valverde, C. Fermentation as a Bio-Process to Obtain Functional Soybean Flours. J. Agric. Food Chem. 2007, 55, 8972–8979. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.; Ding, H.; Lin, H.; Chen, W. AOD: The Antioxidant Protein Database. Sci. Rep. 2017, 7, 7449. [Google Scholar] [CrossRef] [PubMed]
- Elias, R.J.; Kellerby, S.S.; Decker, E.A. Antioxidant Activity of Proteins and Peptides. Crit. Rev. Food Sci. Nutr. 2008, 48, 430–441. [Google Scholar] [CrossRef] [PubMed]

| FA | [%] | Lipid Structure | [%] |
|---|---|---|---|
| C14:0 | 2.23 | ||
| C16:0 | 25.66 | Polar lipids | 0.14 |
| C16:1 n-7 | 2.18 | Monoacylglycerols | 0.28 |
| C18:0 | 21.33 | Diacylglycerols | 2.08 |
| C18:1 n-9 | 38.34 | Sterol structures | 9.38 |
| C18:1 n-7 | 1.94 | Free fatty acids | 7.20 |
| C18:2 n-6 | 5.47 | Triacylglycerols | 66.54 |
| C18:3 n-3 | 0.81 | Esterified sterols | 11.23 |
| C20:0 | 0.23 | Other lipid structures | 3.15 |
| Other FAs | 1.81 |
| Cereal Matrix | Tween® 40 [%(w/w)] | Animal Fat [%(w/w)] |
|---|---|---|
| wheat bran/cornmeal (20 g) | 0 | 0 |
| 0.5 | 0 | |
| 1 | 0 | |
| 2 | 0 | |
| 3 | 0 | |
| 0.5 | 5 | |
| 1 | 10 | |
| 2 | 20 | |
| 3 | 30 |
| Cereal Matrix Utilization [%] | Fermented Bioproduct Humidity [%] | aw [Non-Fermented Substrate] | aw [Fermented Bioproduct] | pH [Fermented Bioproduct] | FBM [mg/g BP] | |
|---|---|---|---|---|---|---|
| Cornmeal (CM) | 22.0 ± 1.5 | 62.0 ± 1.9 | 0.959 ± 0.014 | 0.957 ± 0.004 | 5.1 ± 0.1 | 242.7 ± 12.5 |
| CM + 0.5% Tween 40 | 21.2 ± 0.8 | 60.5 ± 0.3 | 0.958 ± 0.008 | 0.959 ± 0.007 | 4.9 ± 0.0 | 247.2 ± 19.8 |
| CM + 1% Tween 40 | 21.9 ± 1.4 | 62.6 ± 1.2 | 0.957 ± 0.020 | 0.962 ± 0.005 | 4.5 ± 0.3 | 250.6 ± 13.6 |
| CM + 2% Tween 40 | 24.7 ± 1.6 | 60.0 ± 1.9 | 0.958 ± 0.009 | 0.964 ± 0.011 | 4.2 ± 0.2 | 248.3 ± 7.9 |
| CM + 3% Tween 40 | 28.5 ± 1.6 | 62.3 ± 1.3 | 0.960 ± 0.010 | 0.965 ± 0.001 | 4.1 ± 0.1 | 231.8 ± 15.7 |
| CM + 0.5% Tween 40 + 5% AF | 25.4 ± 1.9 | 60.6 ± 1.5 | 0.958 ± 0.017 | 0.964 ± 0.004 | 4.6 ± 0.1 | 206.2 ± 19.4 |
| CM + 1% Tween 40 + 10% AF | 23.8 ± 1.0 | 54.8 ± 0.6 | 0.957 ± 0.007 | 0.966 ± 0.014 | 4.2 ± 0.1 | 151.4 ± 16.0 |
| CM + 2% Tween 40 + 20% AF | 12.9 ± 1.1 | 47.7 ± 1.8 | 0.951 ± 0.005 | 0.967 ± 0.011 | 4.3 ± 0.1 | 113.4 ± 9.7 |
| CM + 3% Tween 40 + 30% AF | 6.6 ± 0.1 | 38.5 ± 1.6 | 0.943 ± 0.005 | 0.964 ± 0.014 | 3.9 ± 0.1 | 93.0 ± 1.0 |
| Wheat Bran (WB) | 17.3 ± 0.3 | 59.7 ± 0.4 | 0.933 ± 0.027 | 0.938 ± 0.025 | 4.6 ± 0.5 | 229.9 ± 10.4 |
| WB + 0.5% Tween 40 | 17.4 ± 0.3 | 59.4 ± 1.0 | 0.933 ± 0.015 | 0.940 ± 0.007 | 5.1 ± 0.6 | 255.1 ± 8.9 |
| WB + 1% Tween 40 | 17.4 ± 0.3 | 59.3 ± 0.4 | 0.932 ± 0.037 | 0.941 ± 0.015 | 5.3 ± 0.3 | 253.3 ± 7.4 |
| WB + 2% Tween 40 | 19.5 ± 0.9 | 59.1 ± 0.6 | 0.934 ± 0.016 | 0.941 ± 0.008 | 5.3 ± 0.2 | 257.2 ± 2.9 |
| WB + 3% Tween 40 | 21.8 ± 0.8 | 59.0 ± 0.1 | 0.933 ± 0.004 | 0.943 ± 0.014 | 5.4 ± 0.3 | 266.9 ± 2.4 |
| WB + 0.5% Tween 40 + 5% AF | 15.8 ± 1.0 | 56.4 ± 1.3 | 0.939 ± 0.025 | 0.939 ± 0.005 | 5.1 ± 0.1 | 222.1 ± 10.0 |
| WB + 1% Tween 40 + 10% AF | 13.8 ± 0.6 | 52.4 ± 1.0 | 0.937 ± 0.017 | 0.934 ± 0.018 | 5.0 ± 0.1 | 171.9 ± 18.2 |
| WB + 2% Tween 40 + 20% AF | 10.3 ± 0.3 | 46.4 ± 0.6 | 0.928 ± 0.015 | 0.925 ± 0.011 | 4.6 ± 0.4 | 143.3 ± 10.0 |
| WB + 3% Tween 40 + 30% AF | 8.9 ± 0.6 | 39.7 ± 0.2 | 0.924 ± 0.009 | 0.921 ± 0.015 | 4.5 ± 0.2 | 113.3 ± 1.7 |
| IU | TFA | Fatty Acids (%) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| (%/BP) | C16:0 | C16:1, n-7 | C18:0 | C18:1, n-9 | C18:2, n-6 | C18:3, n-6 | Other FAs | ||
| Cornmeal | |||||||||
| CM nf | 1.19 | 3.5 | 13.2 | 0.0 | 2.4 | 27.1 | 55.2 | 0.0 | 2.1 |
| CM f | 0.69 | 9.9 | 18.6 | 1.6 | 3.3 | 44.0 | 22.8 | 5.3 | 4.4 |
| + 0.5% Tween 40 nf | 1.15 | 3.5 | 15.3 | 0.0 | 2.5 | 26.3 | 52.8 | 0.0 | 3.1 |
| + 0.5% Tween 40 f | 0.68 | 11.5 | 19.5 | 1.6 | 3.3 | 43.8 | 22.3 | 5.2 | 4.3 |
| + 1% Tween 40 nf | 1.15 | 4.2 | 15.8 | 0.0 | 2.4 | 26.2 | 51.9 | 0.0 | 3.7 |
| + 1% Tween 40 f | 0.74 | 11.4 | 19.7 | 1.5 | 3.4 | 42.9 | 22.7 | 5.6 | 4.2 |
| + 2% Tween 40 nf | 1.09 | 4.1 | 21.5 | 0.0 | 2.6 | 23.9 | 48.2 | 0.0 | 3.7 |
| + 2% Tween 40 f | 0.71 | 13.5 | 21.1 | 1.5 | 3.5 | 43.3 | 21.3 | 5.1 | 4.1 |
| + 3% Tween 40 nf | 1.13 | 4.5 | 24.3 | 0.0 | 2.6 | 21.9 | 47.9 | 0.0 | 3.3 |
| + 3% Tween 40 f | 0.76 | 13.9 | 22.4 | 1.5 | 3.6 | 41.3 | 21.8 | 5.4 | 4.0 |
| + 0.5% T + 5%AF nf | 0.78 | 6.8 | 20.7 | 1.2 | 10.7 | 32.6 | 30.3 | 0.0 | 4.4 |
| + 0.5% T + 5%AF f | 0.71 | 15.2 | 20.1 | 1.8 | 7.1 | 43.3 | 18.3 | 4.7 | 4.8 |
| + 1% T + 10%AF nf | 0.46 | 10.7 | 24.3 | 1.6 | 14.8 | 34.5 | 19.5 | 0.0 | 5.3 |
| + 1% T + 10%AF f | 0.49 | 17.6 | 21.2 | 1.9 | 8.5 | 43.7 | 16.1 | 3.6 | 5.0 |
| + 2% T + 20%AF nf | 0.40 | 15.4 | 25.1 | 1.8 | 15.9 | 35.6 | 16.1 | 0.0 | 5.6 |
| + 2% T + 20%AF f | 0.40 | 19.9 | 22.8 | 2.0 | 12.1 | 42.2 | 13.7 | 1.9 | 5.4 |
| + 3% T + 30%AF nf | 0.31 | 21.7 | 27.2 | 2.0 | 17.8 | 35.5 | 12.0 | 0.0 | 5.5 |
| + 3% T + 30%AF f | 0.35 | 22.4 | 24.4 | 2.1 | 14.9 | 39.7 | 12.5 | 0.9 | 5.5 |
| Wheat Bran | |||||||||
| WB nf | 1.46 | 3.6 | 17.1 | 0.0 | 1.0 | 17.3 | 56.1 | 0.0 | 8.5 |
| WB f | 1.41 | 3.4 | 14.4 | 0.5 | 2.1 | 29.0 | 40.3 | 7.5 | 6.3 |
| + 0.5% Tween 40 nf | 1.42 | 3.6 | 20.7 | 0.0 | 1.1 | 16.6 | 54.7 | 0.0 | 6.8 |
| + 0.5% Tween 40 f | 1.40 | 2.8 | 14.5 | 0.5 | 2.3 | 29.1 | 38.8 | 8.3 | 6.5 |
| + 1% Tween 40 nf | 1.39 | 3.8 | 21.8 | 0.2 | 1.2 | 16.7 | 53.4 | 0.0 | 6.8 |
| + 1% Tween 40 f | 1.40 | 2.8 | 14.6 | 0.5 | 2.3 | 29.6 | 37.8 | 8.5 | 6.8 |
| + 2% Tween 40 nf | 1.31 | 3.8 | 26.7 | 0.0 | 1.5 | 15.8 | 50.1 | 0.0 | 6.0 |
| + 2% Tween 40 f | 1.39 | 2.6 | 15.4 | 0.5 | 2.4 | 28.7 | 38.1 | 8.3 | 6.6 |
| + 3% Tween 40 nf | 1.17 | 4.6 | 33.6 | 0.1 | 1.8 | 13.7 | 44.9 | 0.0 | 5.9 |
| + 3% Tween 40 f | 1.35 | 3.1 | 17.2 | 0.6 | 2.5 | 28.5 | 37.6 | 7.3 | 6.3 |
| + 0.5% T + 5%AF nf | 1.08 | 6.6 | 22.1 | 1.1 | 9.5 | 26.5 | 34.6 | 0.0 | 6.3 |
| + 0.5% T + 5%AF f | 1.21 | 4.6 | 17.2 | 1.1 | 5.0 | 36.1 | 28.1 | 6.3 | 6.1 |
| + 1% T + 10%AF nf | 0.99 | 9.9 | 23.7 | 1.2 | 10.6 | 27.2 | 30.8 | 0.0 | 6.6 |
| + 1% T + 10%AF f | 1.02 | 6.8 | 19.1 | 1.5 | 8.9 | 37.9 | 22.3 | 4.3 | 6.1 |
| + 2% T + 20%AF nf | 0.83 | 14.4 | 25.8 | 1.7 | 12.8 | 31.1 | 22.2 | 0.0 | 6.4 |
| + 2% T + 20%AF f | 0.86 | 11.7 | 22.3 | 1.7 | 11.4 | 40.6 | 15.4 | 2.7 | 5.9 |
| + 3% T + 30%AF nf | 0.76 | 23.4 | 26.1 | 1.6 | 16.0 | 33.3 | 17.1 | 0.0 | 6.0 |
| + 3% T + 30%AF f | 0.83 | 20.0 | 23.5 | 1.7 | 11.2 | 40.6 | 15.8 | 1.6 | 5.6 |
| Lipid Class (%) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Polar Lipids | 1,2-Diacylglycerols | Sterols | Free Fatty Acids | Coenzyme Q | Triacylglycerols | Esterified Sterols | Others | |
| Cornmeal | ||||||||
| CM nf | 6.0 | 5.6 | 5.1 | 3.9 | 2.1 | 66.2 | 9.2 | 1.9 |
| CM f | 4.7 | 8.8 | 11.8 | 12.1 | 0.8 | 50.0 | 11.1 | 0.7 |
| + 0.5% Tween 40 nf | 4.7 | 3.4 | 4.0 | 4.2 | 2.0 | 71.6 | 8.0 | 2.0 |
| + 0.5% Tween 40 f | 4.3 | 7.1 | 9.7 | 11.5 | 1.2 | 53.0 | 12.1 | 1.1 |
| + 1% Tween 40 nf | 4.6 | 2.8 | 3.7 | 4.1 | 1.2 | 72.9 | 8.5 | 2.4 |
| + 1% Tween 40 f | 4.3 | 5.6 | 7.6 | 13.8 | 1.1 | 56.4 | 10.5 | 0.7 |
| + 2% Tween 40 nf | 3.8 | 3.1 | 5.2 | 5.3 | 1.8 | 71.2 | 7.7 | 1.9 |
| + 2% Tween 40 f | 5.3 | 6.3 | 10.9 | 11.9 | 1.2 | 51.5 | 11.8 | 1.1 |
| + 3% Tween 40 nf | 3.7 | 2.7 | 4.1 | 7.2 | 2.6 | 69.9 | 8.7 | 1.2 |
| + 3% Tween 40 f | 5.5 | 6.2 | 7.7 | 14.2 | 0.7 | 53.7 | 11.1 | 0.9 |
| + 0.5% T + 5%AF nf | 4.1 | 2.8 | 2.5 | 4.8 | 1.1 | 74.2 | 5.7 | 5.0 |
| + 0.5% T + 5%AF f | 4.1 | 7.0 | 8.2 | 13.2 | 0.8 | 55.7 | 8.5 | 2.5 |
| + 1% T + 10%AFnf | 4.2 | 1.0 | 2.8 | 6.8 | 0.9 | 69.8 | 4.4 | 10.2 |
| + 1% T + 10%AF f | 3.8 | 5.3 | 6.9 | 13.5 | 0.7 | 59.9 | 7.7 | 2.3 |
| + 2% T + 20%AF nf | 4.1 | 5.1 | 2.7 | 6.6 | 0.3 | 60.1 | 4.1 | 16.2 |
| + 2% T + 20%AF f | 3.9 | 5.4 | 9.3 | 12.5 | 0.3 | 56.8 | 5.1 | 6.9 |
| + 3% T + 30%AF nf | 6.1 | 2.4 | 2.3 | 7.5 | 0.3 | 63.7 | 3.7 | 14.1 |
| + 3% T + 30%AF f | 3.4 | 5.2 | 3.9 | 12.0 | 0.0 | 66.9 | 3.5 | 5.1 |
| Wheat Bran | ||||||||
| WB nf | 8.8 | 9.1 | 3.2 | 6.6 | 0.6 | 45.5 | 13.3 | 13.1 |
| WB f | 8.4 | 7.4 | 10.4 | 16.6 | 1.9 | 24.2 | 19.5 | 11.6 |
| + 0.5% Tween 40 nf | 9.6 | 10.2 | 3.3 | 8.8 | 0.2 | 45.9 | 7.9 | 14.1 |
| + 0.5% Tween 40 f | 11.0 | 7.8 | 11.4 | 14.5 | 1.8 | 27.0 | 13.1 | 13.5 |
| + 1% Tween 40 nf | 8.8 | 8.4 | 3.5 | 8.1 | 0.3 | 45.9 | 13.2 | 11.9 |
| + 1% Tween 40 f | 12.2 | 7.1 | 11.9 | 14.0 | 1.8 | 27.7 | 17.8 | 12.5 |
| + 2% Tween 40 nf | 9.8 | 14.2 | 5.5 | 6.1 | 0.0 | 47.3 | 8.2 | 8.9 |
| + 2% Tween 40 f | 11.5 | 7.4 | 15.1 | 13.5 | 1.6 | 22.9 | 16.7 | 13.2 |
| + 3% Tween 40 nf | 10.1 | 14.2 | 5.9 | 6.0 | 0.2 | 43.6 | 11.6 | 8.5 |
| + 3% Tween 40 f | 10.9 | 7.4 | 15.2 | 10.3 | 1.6 | 30.7 | 13.0 | 10.9 |
| + 0.5% T + 5%AF nf | 7.3 | 10.8 | 9.4 | 7.6 | 0.0 | 50.3 | 7.7 | 7.0 |
| + 0.5% T + 5%AF f | 8.7 | 7.2 | 10.9 | 15.9 | 1.1 | 31.7 | 16.1 | 8.5 |
| + 1% T + 10%AF nf | 8.7 | 10.1 | 9.5 | 7.5 | 0.0 | 52.0 | 6.2 | 6.1 |
| + 1% T + 10%AF f | 8.5 | 7.1 | 11.6 | 16.8 | 0.7 | 38.3 | 11.8 | 5.2 |
| + 2% T + 20%AF nf | 6.0 | 7.2 | 10.0 | 9.1 | 0.0 | 61.9 | 3.4 | 2.5 |
| + 2% T + 20%AF f | 7.4 | 6.3 | 10.5 | 17.6 | 0.3 | 47.7 | 7.0 | 3.3 |
| + 3% T + 30%AF nf | 5.8 | 6.9 | 8.2 | 9.2 | 0.0 | 61.3 | 5.3 | 3.3 |
| + 3% T + 30%AF f | 7.5 | 6.3 | 10.8 | 15.9 | 0.0 | 49.7 | 6.9 | 3.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slaný, O.; Klempová, T.; Shapaval, V.; Zimmermann, B.; Kohler, A.; Čertík, M. Animal Fat as a Substrate for Production of n-6 Fatty Acids by Fungal Solid-State Fermentation. Microorganisms 2021, 9, 170. https://doi.org/10.3390/microorganisms9010170
Slaný O, Klempová T, Shapaval V, Zimmermann B, Kohler A, Čertík M. Animal Fat as a Substrate for Production of n-6 Fatty Acids by Fungal Solid-State Fermentation. Microorganisms. 2021; 9(1):170. https://doi.org/10.3390/microorganisms9010170
Chicago/Turabian StyleSlaný, Ondrej, Tatiana Klempová, Volha Shapaval, Boris Zimmermann, Achim Kohler, and Milan Čertík. 2021. "Animal Fat as a Substrate for Production of n-6 Fatty Acids by Fungal Solid-State Fermentation" Microorganisms 9, no. 1: 170. https://doi.org/10.3390/microorganisms9010170
APA StyleSlaný, O., Klempová, T., Shapaval, V., Zimmermann, B., Kohler, A., & Čertík, M. (2021). Animal Fat as a Substrate for Production of n-6 Fatty Acids by Fungal Solid-State Fermentation. Microorganisms, 9(1), 170. https://doi.org/10.3390/microorganisms9010170






