Borrelia miyamotoi—An Emerging Human Tick-Borne Pathogen in Europe
Abstract
1. Introduction
2. Review
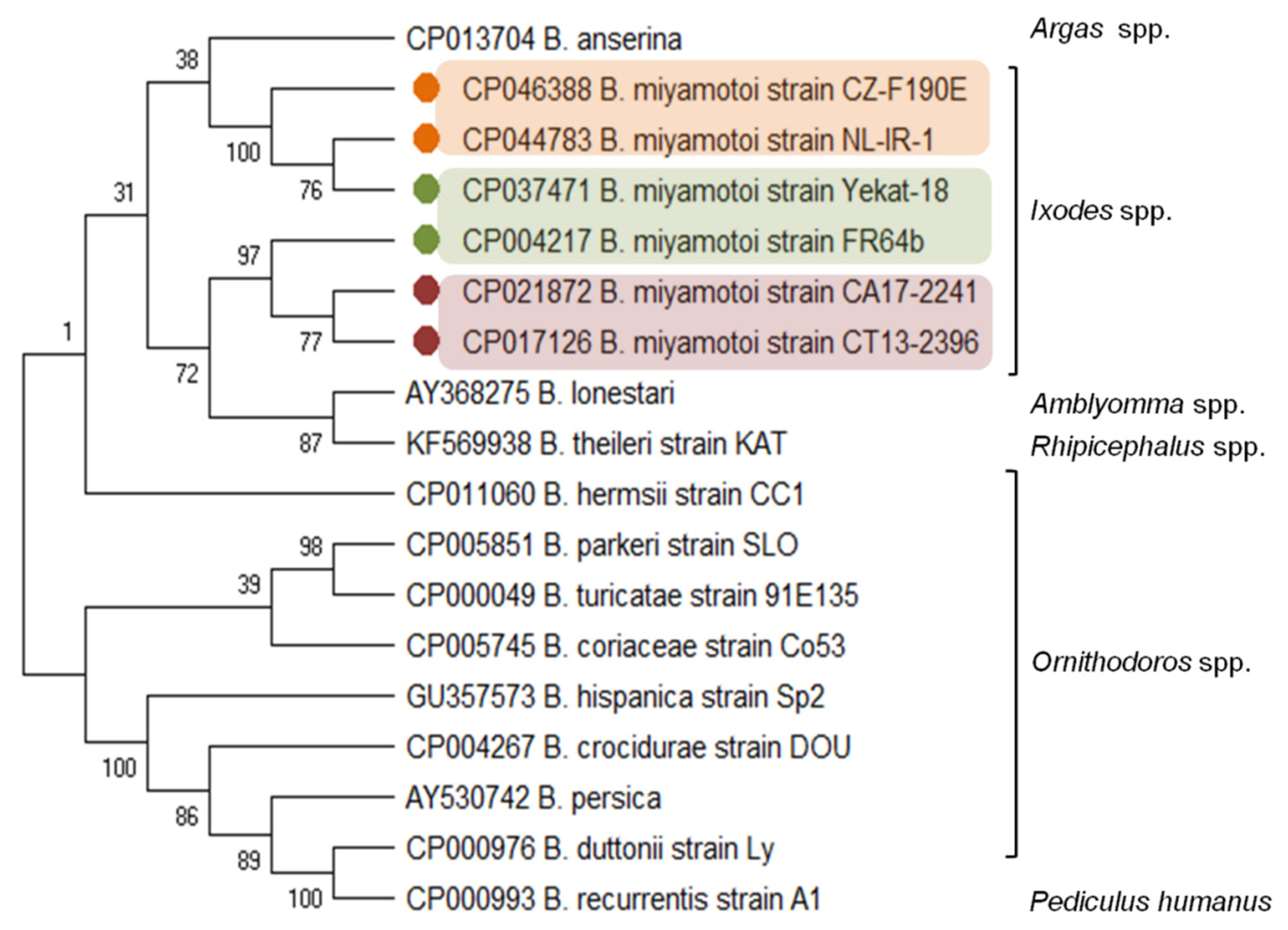
2.1. Taxonomic Position
2.2. Genome Organization and Genetic Diversity
2.3. Vectors and Reservoirs
2.4. Borrelia miyamotoi Disease (BMD) in Europe
2.5. Clinical Manifestation
2.6. Diagnosis
2.7. Treatment and Prevention
3. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mysterud, A.; Jore, S.; Østerås, O.; Viljugrein, H. Emergence of tick-borne diseases at northern latitudes in Europe: A comparative approach. Sci. Rep. 2017, 7, 16316. [Google Scholar] [CrossRef] [PubMed]
- Dantas-Torres, F.; Chomel, B.; Otranto, D. Ticks and tick-borne diseases: A One Health perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, A.; Silaghi, C.; Obiegala, A.; Rudolf, I.; Hubálek, Z.; Földvári, G.; Plantard, O.; Vayssier-Taussat, M.; Bonnet, S.; Špitalská, E.; et al. Ixodes ricinus and its transmitted pathogens in urban and peri-urban areas in Europe: New hazards and relevance for public health. Front. Public Health 2014, 2, 251. [Google Scholar] [CrossRef] [PubMed]
- Azagi, T.; Hoornstra, D.; Kremer, K.; Hovius, J.W.R.; Sprong, H. Evaluation of disease causality of rare Ixodes ricinus-borne infections in Europe. Pathogens 2020, 9, 150. [Google Scholar] [CrossRef]
- Telford, S.R.; Goethert, H.K.; Molloy, P.J.; Berardi, V.P.; Chowdri, H.R.; Gugliotta, J.L.; Lepore, T.J. Borrelia miyamotoi disease: Neither Lyme disease nor relapsing fever. Clin. Lab. Med. 2015, 35, 867–882. [Google Scholar] [CrossRef]
- Cutler, S.J.; Vayssier-Taussat, M.; Estrada-Peña, A.; Potkonjak, A.; Mihalca, A.D.; Zeller, H. A new Borrelia on the block: Borrelia miyamotoi—A human health risk? Eurosurveillance 2019, 24, 1800170. [Google Scholar] [CrossRef]
- Fukunaga, M.; Takahashi, Y.; Tsuruta, Y.; Matsushita, O.; Ralph, D.; McClelland, M.; Nakao, M. Genetic and phenotypic analysis of Borrelia miyamotoi sp. nov., isolated from the ixodid tick Ixodes persulcatus, the vector for Lyme disease in Japan. Int. J. Syst. Bacteriol. 1995, 45, 804–810. [Google Scholar] [CrossRef]
- Platonov, A.E.; Karan, L.S.; Kolyasnikova, N.M.; Makhneva, N.A.; Toporkova, M.G.; Maleev, V.V.; Fish, D.; Krause, P.J. Humans infected with relapsing fever spirochete Borrelia miyamotoi, Russia. Emerg. Infect. Dis. 2011, 17, 1816–1823. [Google Scholar] [CrossRef]
- Sato, K.; Takano, A.; Konnai, S.; Nakao, M.; Ito, T.; Koyama, K.; Kaneko, M.; Ohnishi, M.; Kawabata, H. Human infections with Borrelia miyamotoi, Japan. Emerg. Infect. Dis. 2014, 20, 1391–1393. [Google Scholar] [CrossRef]
- Krause, P.J.; Narasimhan, S.; Wormser, G.P.; Rollend, L.; Fikrig, E.; Lepore, T.; Barbour, A.; Fish, D. Human Borrelia miyamotoi infection in the United States. N. Engl. J. Med. 2013, 368, 291–293. [Google Scholar] [CrossRef]
- Molloy, P.J.; Telford, S.R., III; Chowdri, H.R.; Lepore, T.J.; Gugliotta, J.L.; Weeks, K.E.; Hewins, M.E.; Goethert, H.K.; Berardi, V.P. Borrelia miyamotoi disease in the Northeastern United States: A case series. Ann. Intern. Med. 2015, 163, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Hansford, K.M.; Fonville, M.; Jahfari, S.; Sprong, H.; Medlock, J.M. Borrelia miyamotoi in host-seeking Ixodes ricinus ticks in England. Epidemiol. Infect. 2015, 143, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Hovius, J.W.R.; De Wever, B.; Sohne, M.; Brouwer, M.C.; Coumou, J.; Wagemakers, A.; Oei, A.; Knol, H.; Narasimhan, S.; Hodiamont, C.J.; et al. A case of meningoencephalitis by the relapsing fever spirochaete Borrelia miyamotoi in Europe. Lancet 2013, 382, 658. [Google Scholar] [CrossRef]
- Jahfari, S.; Hofhuis, A.; Fonville, M.; van der Giessen, J.; van Pelt, W.; Sprong, H. Molecular detection of tick-borne pathogens in humans with tick bites and erythema migrans, in the Netherlands. PLoS Negl. Trop. Dis. 2016, 10, e0005042. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Schwartz, I. Genus Borrelia. In Bergey’s Manual of Systematic Bacteriology Vol. 4: The Bacteroidetes, Spirochaetes, Tenericutes (Mollicutes), Acidobacteria, Fibrobacteres, Fusobacteria, Dictyoglomi, Gemmatimonadetes, Lentisphaerae, Verrucomicrobia, Chlamydiae, and Planctomycetes; Krieg, N., Staley, J., Brown, D., Hedlund, B., Paster, B., Ward, N., Ludwig, W., Whitman, W., Eds.; Springer: New York, NY, USA, 2011; pp. 484–531. ISBN 978-0-387-68572-4. [Google Scholar]
- Krause, P.J.; Fish, D.; Narasimhan, S.; Barbour, A.G. Borrelia miyamotoi infection in nature and in humans. Clin. Microbiol. Infect. 2015, 21, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Wang, G. Borrelia burgdorferi and other Borrelia species. In Molecular Medical Microbiology, 2nd ed.; Tang, Y.-W., Sussman, M., Liu, D., Poxton, I., Schwartzman, J., Eds.; Academic Press: Cambridge, MA, USA, 2015; pp. 1867–1909. ISBN 9780123971692. [Google Scholar]
- Wagemakers, A.; Oei, A.; Fikrig, M.M.; Miellet, W.R.; Hovius, J.W. The relapsing fever spirochete Borrelia miyamotoi is cultivable in a modified Kelly-Pettenkofer medium, and is resistant to human complement. Parasites Vectors 2014, 7, 418. [Google Scholar] [CrossRef]
- Stanek, G.; Reiter, M. The expanding Lyme Borrelia complex-clinical significance of genomic species? Clin. Microbiol. Infect. 2011, 17, 487–493. [Google Scholar] [CrossRef]
- Margos, G.; Marosevic, D.; Cutler, S.; Derdakova, M.; Diuk-Wasser, M.; Emler, S.; Fish, D.; Gray, J.; Hunfeldt, K.P.; Jaulhac, B.; et al. There is inadequate evidence to support the division of the genus Borrelia. Int. J. Syst. Evol. Microbiol. 2017, 67, 1081–1084. [Google Scholar] [CrossRef]
- Adeolu, M.; Gupta, R.S. A phylogenomic and molecular marker based proposal for the division of the genus Borrelia into two genera: The emended genus Borrelia containing only the members of the relapsing fever Borrelia, and the genus Borreliella gen. nov. containing the members of the Lyme disease Borrelia (Borrelia burgdorferi sensu lato complex). Antonie Leeuwenhoek 2014, 105, 1049–1072. [Google Scholar] [CrossRef]
- Margos, G.; Fingerle, V.; Oskam, C.; Stevenson, B.; Gofton, A. Comment on: Gupta, 2019, Distinction between Borrelia and Borreliella is more robustly supported by molecular and phenotypic characteristics than all other neighbouring prokaryotic genera: Response to Margos’ et al. “The genus Borrelia reloaded” PLoS ONE. Ticks Tick Borne Dis. 2020, 11, 101320. [Google Scholar] [CrossRef]
- Stevenson, B.; Fingerle, V.; Wormser, G.P.; Margos, G. Public health and patient safety concerns merit retention of Lyme borreliosis-associated spirochetes within the genus Borrelia, and rejection of the genus novum Borreliella. Ticks Tick Borne Dis. 2019, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Barbour, A.G. Phylogeny of a relapsing fever Borrelia species transmitted by the hard tick Ixodes scapularis. Infect. Genet. Evol. 2014, 27, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Hue, F.; Langeroudi, A.G.; Barbour, A.G. Borne agent of human infection. Genome Announc. 2013, 1, 5–6. [Google Scholar] [CrossRef]
- Kuleshov, K.V.; Koetsveld, J.; Goptar, I.A.; Markelov, M.L.; Kolyasnikova, N.M. Whole-genome sequencing of six Borrelia miyamotoi clinical strains isolated in Russia. Genome Announc. 2018, 6, e01424-17. [Google Scholar] [CrossRef] [PubMed]
- Kingry, L.C.; Replogle, A.; Dolan, M.; Sexton, C.; Padgett, K.A.; Schriefer, M.E. Chromosome and large linear plasmid sequences of a Borrelia miyamotoi strain isolated from Ixodes pacificus ticks from California. Genome Announc. 2017, 5, e00960-17. [Google Scholar] [CrossRef]
- Kingry, L.C.; Replogle, A.; Batra, D.; Rowe, L.A.; Sexton, C.; Dolan, M.; Connally, N.; Petersen, J.M.; Schriefer, M.E. Toward a complete north American Borrelia miyamotoi genome. Genome Announc. 2017, 5, e01557-16. [Google Scholar] [CrossRef] [PubMed]
- Kuleshov, K.V.; Hoornstra, D.; Sprong, H.; Platonov, A.E.; Hovius, J.W. Draft whole-genome sequences of two western European Borrelia miyamotoi isolates. Microbiol. Resour. Announc. 2019, 8, e01314-19. [Google Scholar] [CrossRef]
- Kuleshov, K.V.; Margos, G.; Fingerle, V.; Koetsveld, J.; Goptar, I.A.; Markelov, M.L.; Kolyasnikova, N.M.; Sarksyan, D.S.; Kirdyashkina, N.P.; Shipulin, G.A.; et al. Whole genome sequencing of Borrelia miyamotoi isolate Izh-4: Reference for a complex bacterial genome. BMC Genom. 2020, 21, 16. [Google Scholar] [CrossRef]
- Barbour, A.G. Multiple and diverse vsp and vlp sequences in Borrelia miyamotoi, a hard tick-borne zoonotic pathogen. PLoS ONE 2016, 11, e0146283. [Google Scholar] [CrossRef]
- Bergström, S.; Normark, J. Microbiological features distinguishing Lyme disease and relapsing fever spirochetes. Wien. Klin. Wochenschr. 2018, 130, 484–490. [Google Scholar] [CrossRef]
- Schwan, T.G.; Battisti, J.M.; Porcella, S.F.; Raffel, S.J.; Schrumpf, M.E.; Fischer, E.R.; Carroll, J.A.; Stewart, P.E.; Rosa, P.; Somerville, G.A. Glycerol-3-phosphate acquisition in spirochetes: Distribution and biological activity of glycerophosphodiester phosphodiesterase (GlpQ) among Borrelia species. J. Bacteriol. 2003, 185, 1346–1356. [Google Scholar] [CrossRef] [PubMed]
- Bacon, R.M.; Pilgard, M.A.; Johnson, B.J.B.; Raffel, S.J.; Schwan, T.G. Glycerophosphodiester phosphodiesterase gene (glpQ) of Borrelia lonestari identified as a target for differentiating Borrelia species associated with hard ticks (Acari:Ixodidae). J. Clin. Microbiol. 2004, 42, 2326–2328. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.J.; Carroll, M.; Fedorova, N.; Brancato, J.; Dumouchel, C.; Akosa, F.; Narasimhan, S.; Fikrig, E.; Lane, R.S. Human Borrelia miyamotoi infection in California: Serodiagnosis is complicated by multiple endemic Borrelia species. PLoS ONE 2018, 13, e0191725. [Google Scholar] [CrossRef] [PubMed]
- Wagemakers, A.; Jahfari, S.; de Wever, B.; Spanjaard, L.; Starink, M.V.; de Vries, H.J.C.; Sprong, H.; Hovius, J.W. Borrelia miyamotoi in vectors and hosts in The Netherlands. Ticks Tick Borne Dis. 2017, 8, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Hoornstra, D.; Koetsveld, J.; Sprong, H.; Platonov, A.E.; Hovius, J.W. Borrelia miyamotoi disease in an immunocompetent patient, Western Europe. Emerg. Infect. Dis. 2018, 24, 1770–1772. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Iwabu-Itoh, Y.; Bazartseren, B.; Naranbaatar, O.; Yondonjamts, E.; Furuno, K.; Lee, K.; Sato, K.; Kawabata, H.; Takada, N.; Andoh, M.; et al. Tick surveillance for Borrelia miyamotoi and phylogenetic analysis of isolates in Mongolia and Japan. Ticks Tick Borne Dis. 2017, 8, 850–857. [Google Scholar] [CrossRef]
- Jiang, B.G.; Jia, N.; Jiang, J.F.; Zheng, Y.C.; Chu, Y.L.; Jiang, R.R.; Wang, Y.W.; Liu, H.B.; Wei, R.; Zhang, W.H.; et al. Borrelia miyamotoi infections in humans and ticks, Northeastern China. Emerg. Infect. Dis. 2018, 24, 236–241. [Google Scholar] [CrossRef]
- Takano, A.; Toyomane, K.; Konnai, S.; Ohashi, K.; Nakao, M.; Ito, T.; Andoh, M.; Maeda, K.; Watarai, M.; Sato, K.; et al. Tick surveillance for relapsing fever spirochete Borrelia miyamotoi in Hokkaido, Japan. PLoS ONE 2014, 9, e104532. [Google Scholar] [CrossRef]
- Rar, V.; Livanova, N.; Tkachev, S.; Kaverina, G.; Tikunov, A.; Sabitova, Y.; Igolkina, Y.; Panov, V.; Livanov, S.; Fomenko, N.; et al. Detection and genetic characterization of a wide range of infectious agents in Ixodes pavlovskyi ticks in Western Siberia, Russia. Parasites Vectors 2017, 10, 258. [Google Scholar] [CrossRef]
- Scoles, G.; Papero, M.; Beati, L.; Fish, D. A relapsing fever group spirochete transmitted by Ixodes scapularis ticks. Vector Borne Zoonotic Dis. 2001, 1, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Barbour, A.G.; Bunikis, J.; Travinsky, B.; Hoen, A.G.; Diuk-Wasser, M.A.; Fish, D.; Tsao, J.I. Niche partitioning of Borrelia burgdorferi and Borrelia miyamotoi in the same tick vector and mammalian reservoir species. Am. J. Trop. Med. Hyg. 2009, 81, 1120–1131. [Google Scholar] [CrossRef] [PubMed]
- Crowder, C.D.; Carolan, H.E.; Rounds, M.A.; Honig, V.; Mothes, B.; Haag, H.; Nolte, O.; Luft, B.J.; Grubhoffer, L.; Ecker, D.J.; et al. Prevalence of Borrelia miyamotoi in Ixodes ticks in Europe and the United States. Emerg. Infect. Dis. 2014, 20, 1678–1682. [Google Scholar] [CrossRef] [PubMed]
- Lynn, G.E.; Graham, C.B.; Horiuchi, K.; Eisen, L.; Johnson, T.L.; Lane, R.S.; Eisen, R.J. Prevalence and geographic distribution of Borrelia miyamotoi in host-seeking Ixodes pacificus (Acari: Ixodidae) nymphs in Mendocino County, California Geoffrey. J. Med. Entomol. 2018, 55, 711–716. [Google Scholar] [CrossRef]
- Ogden, N.H.; Margos, G.; Aanensen, D.M.; Drebot, M.A.; Feil, E.J.; Hanincová, K.; Schwartz, I.; Tyler, S.; Lindsay, L.R. Investigation of genotypes of Borrelia burgdorferi in Ixodes scapularis ticks collected during surveillance in Canada. Appl. Environ. Microbiol. 2011, 77, 3244–3254. [Google Scholar] [CrossRef]
- Sormunen, J.J.; Penttinen, R.; Klemola, T.; Hänninen, J.; Vuorinen, I.; Laaksonen, M.; Sääksjärvi, I.E.; Ruohomäki, K.; Vesterinen, E.J. Tick-borne bacterial pathogens in southwestern Finland. Parasites Vectors 2016, 9, 168. [Google Scholar] [CrossRef]
- Kjelland, V.; Rollum, R.; Korslund, L.; Slettan, A.; Tveitnes, D. Borrelia miyamotoi is widespread in Ixodes ricinus ticks in southern Norway. Ticks Tick Borne Dis. 2015, 6, 516–521. [Google Scholar] [CrossRef]
- Kjelland, V.; Paulsen, K.M.; Rollum, R.; Jenkins, A.; Stuen, S.; Soleng, A.; Edgar, K.S.; Lindstedt, H.H.; Vaino, K.; Gibory, M.; et al. Tick-borne encephalitis virus, Borrelia burgdorferi sensu lato, Borrelia miyamotoi, Anaplasma phagocytophilum and Candidatus Neoehrlichia mikurensis in Ixodes ricinus ticks collected from recreational islands in southern Norway. Ticks Tick Borne Dis. 2018, 9, 1098–1102. [Google Scholar] [CrossRef]
- Fraenkel, C.J.; Garpmo, U.; Berglund, J. Determination of novel Borrelia genospecies in Swedish Ixodes ricinus ticks. J. Clin. Microbiol. 2002, 40, 3308–3312. [Google Scholar] [CrossRef]
- Szekeres, S.; Coipan, E.C.; Rigó, K.; Majoros, G.; Jahfari, S.; Sprong, H.; Földvári, G. Eco-epidemiology of Borrelia miyamotoi and Lyme borreliosis spirochetes in a popular hunting and recreational forest area in Hungary. Parasites Vectors 2015, 8, 309. [Google Scholar] [CrossRef]
- Wodecka, B.; Leońska, A.; Skotarczak, B. A comparative analysis of molecular markers for the detection and identification of Borrelia spirochaetes in Ixodes ricinus. J. Med. Microbiol. 2010, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Kiewra, D.; Stańczak, J.; Richter, M. Ixodes ricinus ticks (Acari, Ixodidae) as a vector of Borrelia burgdorferi sensu lato and Borrelia miyamotoi in Lower Silesia, Poland—Preliminary study. Ticks Tick Borne Dis. 2014, 5, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Kowalec, M.; Szewczyk, T.; Welc-Falȩciak, R.; Siński, E.; Karbowiak, G.; Bajer, A. Ticks and the city—Are there any differences between city parks and natural forests in terms of tick abundance and prevalence of spirochaetes? Parasites Vectors 2017, 10, 573. [Google Scholar] [CrossRef] [PubMed]
- Kubiak, K.; Dziekońska-Rynko, J.; Szymańska, H.; Kubiak, D.; Dmitryjuk, M.; Dzika, E. Questing Ixodes ricinus ticks (Acari, Ixodidae) as a vector of Borrelia burgdorferi sensu lato and Borrelia miyamotoi in an urban area of north-eastern Poland. Exp. Appl. Acarol. 2019, 78, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Kalmár, Z.; Sprong, H.; Mihalca, A.D.; Gherman, C.M.; Dumitrache, M.O.; Coipan, E.C.; Fonville, M.; Cozma, V. Borrelia miyamotoi and Candidatus Neoehrlichia mikurensis in Ixodes ricinus ticks, Romania. Emerg. Infect. Dis. 2016, 22, 550–551. [Google Scholar] [CrossRef] [PubMed]
- Hamšíková, Z.; Coipan, C.; Mahríková, L.; Minichová, L.; Sprong, H.; Kazimírová, M. Borrelia miyamotoi and co-infection with Borrelia afzelii in Ixodes ricinus ticks and rodents from Slovakia. Microb. Ecol. 2017, 73, 1000–1008. [Google Scholar] [CrossRef]
- Vaculová, T.; Derdáková, M.; Špitalská, E.; Václav, R.; Chvostáč, M.; Rusňáková Tarageľová, V. Simultaneous occurrence of Borrelia miyamotoi, Borrelia burgdorferi sensu lato, Anaplasma phagocytophilum and Rickettsia helvetica in Ixodes ricinus ticks in urban foci in Bratislava, Slovakia. Acta Parasitol. 2019, 64, 19–30. [Google Scholar] [CrossRef]
- Oechslin, C.P.; Heutschi, D.; Lenz, N.; Tischhauser, W.; Péter, O.; Rais, O.; Beuret, C.M.; Leib, S.L.; Bankoul, S.; Ackermann-Gäumann, R. Prevalence of tick-borne pathogens in questing Ixodes ricinus ticks in urban and suburban areas of Switzerland. Parasites Vectors 2017, 10, 558. [Google Scholar] [CrossRef]
- Cochez, C.; Heyman, P.; Heylen, D.; Fonville, M.; Hengeveld, P.; Takken, W.; Simons, L.; Sprong, H. The Presence of Borrelia miyamotoi, a relapsing fever spirochaete, in questing Ixodes ricinus in Belgium and in The Netherlands. Zoonoses Public Health 2015, 62, 331–333. [Google Scholar] [CrossRef]
- Lernout, T.; De Regge, N.; Tersago, K.; Fonville, M.; Suin, V.; Sprong, H. Prevalence of pathogens in ticks collected from humans through citizen science in Belgium. Parasites Vectors 2019, 12, 550. [Google Scholar] [CrossRef]
- Lejal, E.; Marsot, M.; Chalvet-Monfray, K.; Cosson, J.F.; Moutailler, S.; Vayssier-Taussat, M.; Pollet, T. A three-years assessment of Ixodes ricinus-borne pathogens in a French peri-urban forest. Parasites Vectors 2019, 12, 551. [Google Scholar] [CrossRef]
- Nebbak, A.; Dahmana, H.; Almeras, L.; Raoult, D.; Boulanger, N.; Jaulhac, B.; Mediannikov, O.; Parola, P. Co-infection of bacteria and protozoan parasites in Ixodes ricinus nymphs collected in the Alsace region, France. Ticks Tick Borne Dis. 2019, 10, 101241. [Google Scholar] [CrossRef] [PubMed]
- Boyer, P.H.; Koetsveld, J.; Zilliox, L.; Sprong, H.; Talagrand-Reboul, É.; Hansmann, Y.; De Martino, S.J.; Boulanger, N.; Hovius, J.W.; Jaulhac, B. Assessment of Borrelia miyamotoi in febrile patients and ticks in Alsace, an endemic area for Lyme borreliosis in France. Parasites Vectors 2020, 13, 199. [Google Scholar] [CrossRef] [PubMed]
- Blazejak, K.; Raulf, M.K.; Janecek, E.; Jordan, D.; Fingerle, V.; Strube, C. Shifts in Borrelia burgdorferi (s.l.) geno-species infections in Ixodes ricinus over a 10-year surveillance period in the city of Hanover (Germany) and Borrelia miyamotoi-specific reverse line blot detection. Parasites Vectors 2018, 11, 304. [Google Scholar] [CrossRef] [PubMed]
- Page, S.; Daschkin, C.; Anniko, S.; Krey, V.; Nicolaus, C.; Maxeiner, H.G. First report of Borrelia miyamotoi in an Ixodes ricinus tick in Augsburg, Germany. Exp. Appl. Acarol. 2018, 74, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Geller, J.; Nazarova, L.; Katargina, O.; Järvekülg, L.; Fomenko, N.; Golovljova, I. Detection and Genetic Characterization of Relapsing fever spirochete Borrelia miyamotoi in Estonian ticks. PLoS ONE 2012, 7, e51914. [Google Scholar] [CrossRef] [PubMed]
- Ravagnan, S.; Tomassone, L.; Montarsi, F.; Krawczyk, A.I.; Mastrorilli, E.; Sprong, H.; Milani, A.; Rossi, L.; Capelli, G. First detection of Borrelia miyamotoi in Ixodes ricinus ticks from northern Italy. Parasites Vectors 2018, 11, 130. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.; Parreira, R.; Lopes, N.; Maia, C.; Carreira, T.; Sousa, C.; Faria, S.; Campino, L.; Vieira, M. Molecular identification of Borrelia miyamotoi in Ixodes ricinus from Portugal. Vector Borne Zoonotic Dis. 2015, 15, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Potkonjak, A.; Kleinerman, G.; Gutiérrez, R.; Savić, S.; Vračar, V.; Nachum-Biala, Y.; Jurišić, A.; Rojas, A.; Petrović, A.; Ivanović, I.; et al. Occurrence of Borrelia burgdorferi sensu lato in Ixodes ricinus ticks with first identification of Borrelia miyamotoi in Vojvodina, Serbia. Vector Borne Zoonotic Dis. 2016, 16, 631–635. [Google Scholar] [CrossRef]
- Díaz, P.; Arnal, J.L.; Remesar, S.; Pérez-Creo, A.; Venzal, J.M.; Vázquez-López, M.E.; Prieto, A.; Fernández, G.; López, C.M.; Panadero, R.; et al. Molecular identification of Borrelia spirochetes in questing Ixodes ricinus from northwestern Spain. Parasites Vectors 2017, 10, 615. [Google Scholar] [CrossRef]
- Palomar, A.M.; Portillo, A.; Santibáñez, P.; Santibáñez, S.; Oteo, J.A. Borrelia miyamotoi: Should this pathogen be considered for the diagnosis of tick-borne infectious diseases in Spain? Enferm. Infecc. Microbiol. Clin. 2018, 36, 568–571. [Google Scholar] [CrossRef] [PubMed]
- Remesar, S.; Díaz, P.; Venzal, J.M.; Prieto, A.; Estrada-Peña, A.; López, C.M.; Panadero, R.; Fernández, G.; Díez-Baños, P.; Morrondo, P. Longitudinal study of infection with Borrelia spp. in questing ticks from north-western Spain. Vector Borne Zoonotic Dis. 2019, 19, 785–792. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, K.; Şen, E.; Sato, K.; Kawabata, H.; Ohashi, N.; Masuzawa, T. Characterization of the emerging relapsing fever pathogen, Borrelia miyamotoi, from the Ixodes ricinus tick in the rural Trakya (Thrace) Region of Northwestern Turkey. Vector Borne Zoonotic Dis. 2016, 16, 797–799. [Google Scholar] [CrossRef] [PubMed]
- Capligina, V.; Seleznova, M.; Akopjana, S.; Freimane, L.; Lazovska, M.; Krumins, R.; Kivrane, A.; Namina, A.; Aleinikova, D.; Kimsis, J.; et al. Large-scale countrywide screening for tick-borne pathogens in field-collected ticks in Latvia during 2017–2019. Parasites Vectors 2020, 13, 351. [Google Scholar] [CrossRef]
- Lambert, J.S.; Cook, M.J.; Healy, J.E.; Murtagh, R.; Avramovic, G.; Lee, S.H. Metagenomic 16S rRNA gene sequencing survey of Borrelia species in Irish samples of Ixodes ricinus ticks. bioRxiv 2018. [Google Scholar] [CrossRef]
- Szekeres, S.; Lügner, J.; Fingerle, V.; Margos, G.; Földvári, G. Prevalence of Borrelia miyamotoi and Borrelia burgdorferi sensu lato in questing ticks from a recreational coniferous forest of East Saxony, Germany. Ticks Tick Borne Dis. 2017, 8, 922–927. [Google Scholar] [CrossRef]
- Rǎileanu, C.; Tauchmann, O.; Vasić, A.; Wöhnke, E.; Silaghi, C. Borrelia miyamotoi and Borrelia burgdorferi (sensu lato) identification and survey of tick-borne encephalitis virus in ticks from north-eastern Germany. Parasites Vectors 2020, 13, 106. [Google Scholar] [CrossRef]
- Michelet, L.; Delannoy, S.; Devillers, E.; Umhang, G.; Aspan, A.; Juremalm, M.; Chirico, J.; van der Wal, F.J.; Sprong, H.; Boye Pihl, T.P.; et al. High-throughput screening of tick-borne pathogens in Europe. Front. Cell. Infect. Microbiol. 2014, 4, 103. [Google Scholar] [CrossRef]
- Layzell, S.J.; Bailey, D.; Peacey, M.; Nuttall, P.A. Prevalence of Borrelia burgdorferi and Borrelia miyamotoi in questing Ixodes ricinus ticks from four sites in the UK. Ticks Tick Borne Dis. 2018, 9, 217–224. [Google Scholar] [CrossRef]
- Siński, E.; Welc-Falȩciak, R.; Zajkowska, J. Borrelia miyamotoi: A human tick-borne relapsing fever spirochete in Europe and its potential impact on public health. Adv. Med. Sci. 2016, 61, 255–260. [Google Scholar] [CrossRef]
- Richter, D.; Debski, A.; Hubalek, Z.; Matuschka, F.-R. Absence of Lyme disease spirochetes in larval Ixodes ricinus ticks. Vector Borne Zoonotic Dis. 2012, 12, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Richter, D.; Schlee, D.B.; Matuschka, F.R. Relapsing fever-like spirochetes infecting European vector tick of Lyme disease agent. Emerg. Infect. Dis. 2003, 9, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Van Duijvendijk, G.; Coipan, C.; Wagemakers, A.; Fonville, M.; Ersöz, J.; Oei, A.; Földvári, G.; Hovius, J.; Takken, W.; Sprong, H. Larvae of Ixodes ricinus transmit Borrelia afzelii and B. miyamotoi to vertebrate hosts. Parasites Vectors 2016, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Burri, C.; Schumann, O.; Schumann, C.; Gern, L. Are Apodemus spp. mice and Myodes glareolus reservoirs for Borrelia miyamotoi, Candidatus Neoehrlichia mikurensis, Rickettsia helvetica, R. monacensis and Anaplasma phagocytophilum? Ticks Tick Borne Dis. 2014, 5, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Talagrand-Reboul, E.; Boyer, P.H.; Bergström, S.; Vial, L.; Boulanger, N. Relapsing fevers: Neglected tick-borne diseases. Front. Cell. Infect. Microbiol. 2018, 8, 98. [Google Scholar] [CrossRef] [PubMed]
- Majerová, K.; Hönig, V.; Houda, M.; Papežík, P.; Fonville, M.; Sprong, H.; Rudenko, N.; Golovchenko, M.; Černá Bolfíková, B.; Hulva, P.; et al. Hedgehogs, squirrels, and blackbirds as sentinel hosts for active surveillance of Borrelia miyamotoi and Borrelia burgdorferi complex in urban and rural environments. Microorganisms 2020, 8, 1908. [Google Scholar] [CrossRef]
- Wagemakers, A.; Staarink, P.J.; Sprong, H.; Hovius, J.W.R. Borrelia miyamotoi: A widespread tick-borne relapsing fever spirochete. Trends Parasitol. 2015, 31, 260–269. [Google Scholar] [CrossRef]
- Taylor, K.R.; Takano, A.; Konnai, S.; Shimozuru, M.; Kawabata, H.; Tsubota, T. Borrelia miyamotoi infections among wild rodents show age and month independence and correlation with Ixodes persulcatus larval attachment in Hokkaido, Japan. Vector Borne Zoonotic Dis. 2013, 13, 92–97. [Google Scholar] [CrossRef]
- Chowdri, H.R.; Gugliotta, J.L.; Berardi, V.P.; Goethert, H.K.; Molloy, P.J.; Sterling, S.L.; Telford, S.R. Borrelia miyamotoi infection presenting as human granulocytic anaplasmosis: A case report. Ann. Intern. Med. 2013, 159, 21–27. [Google Scholar] [CrossRef]
- Gugliotta, J.L.; Goethert, H.K.; Berardi, V.P.; Telford, S.R. Meningoencephalitis from Borrelia miyamotoi in an immunocompromised patient. N. Engl. J. Med. 2013, 368, 240–245. [Google Scholar] [CrossRef]
- Boden, K.; Lobenstein, S.; Hermann, B.; Margos, G.; Fingerle, V. Borrelia miyamotoi-associated neuroborreliosis in immunocompromised person. Emerg. Infect. Dis. 2016, 22, 1617–1620. [Google Scholar] [CrossRef] [PubMed]
- Henningsson, A.J.; Asgeirsson, H.; Hammas, B.; Karlsson, E.; Parke, Å.; Hoornstra, D.; Wilhelmsson, P.; Hovius, J.W. Two cases of Borrelia miyamotoi meningitis, Sweden, 2018. Emerg. Infect. Dis. 2019, 25, 1965–1968. [Google Scholar] [CrossRef] [PubMed]
- Tobudic, S.; Burgmann, H.; Stanek, G.; Winkler, S.; Schötta, A.; Obermüller, M.; Markowicz, M.; Lagler, H. Human Human Borrelia miyamotoi Infection, AustriaBorrelia miyamotoi Infection, Austria. Emerg. Infect. Dis. 2020, 26, 2201–2204. [Google Scholar] [CrossRef] [PubMed]
- Fiecek, B.; Lewandowska, G.; Roguska, U.; Rozej-Bielicka, W.; Tylewska-Wierzbanowska, S.; Chmielewski, T. Borrelia miyamotoi DNA in a Patient Suspected of Lyme Borreliosis. 2019, pp. 1–14. Available online: https://assets.researchsquare.com/files/rs-5981/v2/manuscript.pdf (accessed on 11 January 2021). [CrossRef]
- Franck, M.; Ghozzi, R.; Pajaud, J.; Lawson-Hogban, N.E.; Mas, M.; Lacout, A.; Perronne, C. Borrelia miyamotoi: 43 cases diagnosed in France by Real-Time PCR in patients with persistent polymorphic signs and symptoms. Front. Med. 2020, 7, 55. [Google Scholar] [CrossRef]
- Jahfari, S.; Herremans, T.; Platonov, A.E.; Kuiper, H.; Karan, L.S.; Vasilieva, O.; Koopmans, M.P.G.; Hovius, J.W.R.; Sprong, H. High seroprevalence of Borrelia miyamotoi antibodies in forestry workers and individuals suspected of human granulocytic anaplasmosis in The Netherlands. New Microbes New Infect. 2014, 2, 144–149. [Google Scholar] [CrossRef][Green Version]
- Reiter, M.; Stelzer, T.; Schötta, A.M.; Markowicz, M.; Leschnik, M.; Harsch, A.; Reiß, E.; Kneusel, R.E.; Stockinger, H.; Stanek, G. Glycerophosphodiester phosphodiesterase identified as non-reliable serological marker for Borrelia miyamotoi disease. Microorganisms 2020, 8, 1846. [Google Scholar] [CrossRef]
- Sudhindra, P.; Wang, G.; Schriefer, M.E.; McKenna, D.; Zhuge, J.; Krause, P.J.; Marques, A.R.; Wormser, G.P. Insights into Borrelia miyamotoi infection from an untreated case demonstrating relapsing fever, monocytosis and a positive C6 Lyme serology. Diagn. Microbiol. Infect. Dis. 2016, 86, 93–96. [Google Scholar] [CrossRef]
- Yamano, K.; Ito, T.; Kiyanagi, K.; Yamazaki, H.; Sugawara, M.; Saito, T.; Ohashi, N.; Zamoto-Niikura, A.; Sato, K.; Kawabata, H. Case report: Clinical features of a case of suspected Borrelia miyamotoi disease in Hokkaido, Japan. Am. J. Trop. Med. Hyg. 2017, 97, 84–87. [Google Scholar] [CrossRef][Green Version]
- Karan, L.; Makenov, M.; Kolyasnikova, N.; Stukolova, O.; Toporkova, M.; Olenkova, O. Dynamics of spirochetemia and early PCR detection of Borrelia miyamotoi. Emerg. Infect. Dis. 2018, 24, 860–867. [Google Scholar] [CrossRef]
- Wagemakers, A.; Koetsveld, J.; Narasimhan, S.; Wickel, M.; Deponte, K.; Bleijlevens, B.; Jahfari, S.; Sprong, H.; Karan, L.S.; Sarksyan, D.S.; et al. Variable major proteins as targets for specific antibodies against Borrelia miyamotoi. J. Immunol. 2016, 196, 4185–4195. [Google Scholar] [CrossRef]
- Wormser, G.P. Doxycycline for prevention of spirochetal infections: Status report. Clin. Infect. Dis. 2020, 71, 2014–2017. [Google Scholar] [CrossRef] [PubMed]

| Part of Europe | Country | B. miyamotoi Prevalence (%) | Reference |
|---|---|---|---|
| North | Denmark | 0.2–1.3 | [80] |
| Estonia | 0.4 (I.r.) 1; 2.7 (I.p.) 2 | [68] | |
| Finland | 0.56 | [48] | |
| Latvia | 1.1 (I.r.) 1; 1.27 (I.p.) 2 | [76] | |
| Norway | 0.9–1.3 | [49,50] | |
| Sweden | 0.7 (B.m.-like) 3 | [51] | |
| Central | Czech Republic | 3.2 | [45] |
| Hungary | 4.8 | [52] | |
| Poland | 0.5–3.9 | [53,54,55,56] | |
| Romania | 1.5 | [57] | |
| Slovakia | 0.75–1.0 | [58,59] | |
| Switzerland | 2.5 | [60] | |
| West | Ireland | 10 | [77] |
| England | 0.4 (N) 4–0.73 | [12,81] | |
| France | 1.2–2.2 | [63,64,65] | |
| Belgium | 1.1–2.4 | [61,62] | |
| Germany | 0.8–8.9 | [66,67,78,79] | |
| The Netherlands | 2.5 (N) 4–3.8 | [36,61] | |
| South | Italy | 0.74 (N) 4 | [69] |
| Portugal | 0.16 | [70] | |
| Serbia | 1.4 (N) 4 | [71] | |
| Spain | 0.6–1.0 | [72,73,74] | |
| Turkey (European part) | 0.4 | [75] |
| Country | Patient | Number of Cases, Percentage of Cases among Persons Studied | Case | Assay | Reference |
|---|---|---|---|---|---|
| The Netherlands | 70-year-old man | 1 single case | Meningoencephalitis | qPCR | [13] |
| The Netherlands | 42-year-old man | 1/626, 0.16% | EM 1, asymptomatic | qPCR | [14] |
| Germany | 74-year-old woman | 1 single case | Neuroborreliosis, Immunocompromised | qPCR | [93] |
| Sweden | 53-year-old woman 66-year-old woman | 2 single cases | Meningitis, Immunocompetent Meningitis, Immunocompromised | PCR | [94] |
| Poland | 47-year-old man | 1/133, 0.75% | Neuroborreliosis, patient with alcohol abuse | Nested PCR | [96] |
| Austria | 51-year-old woman | 1 single case | Symptomatic | qPCR | [95] |
| France | NA 2 | 43/824, 5.22% | Symptomatic | qPCR | [97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubiak, K.; Szczotko, M.; Dmitryjuk, M. Borrelia miyamotoi—An Emerging Human Tick-Borne Pathogen in Europe. Microorganisms 2021, 9, 154. https://doi.org/10.3390/microorganisms9010154
Kubiak K, Szczotko M, Dmitryjuk M. Borrelia miyamotoi—An Emerging Human Tick-Borne Pathogen in Europe. Microorganisms. 2021; 9(1):154. https://doi.org/10.3390/microorganisms9010154
Chicago/Turabian StyleKubiak, Katarzyna, Magdalena Szczotko, and Małgorzata Dmitryjuk. 2021. "Borrelia miyamotoi—An Emerging Human Tick-Borne Pathogen in Europe" Microorganisms 9, no. 1: 154. https://doi.org/10.3390/microorganisms9010154
APA StyleKubiak, K., Szczotko, M., & Dmitryjuk, M. (2021). Borrelia miyamotoi—An Emerging Human Tick-Borne Pathogen in Europe. Microorganisms, 9(1), 154. https://doi.org/10.3390/microorganisms9010154






