Biodiversity of Algae and Cyanobacteria in Biological Soil Crusts Collected Along a Climatic Gradient in Chile Using an Integrative Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Cultivation of Strains and Morphological Identification
2.3. DNA Extraction, PCR
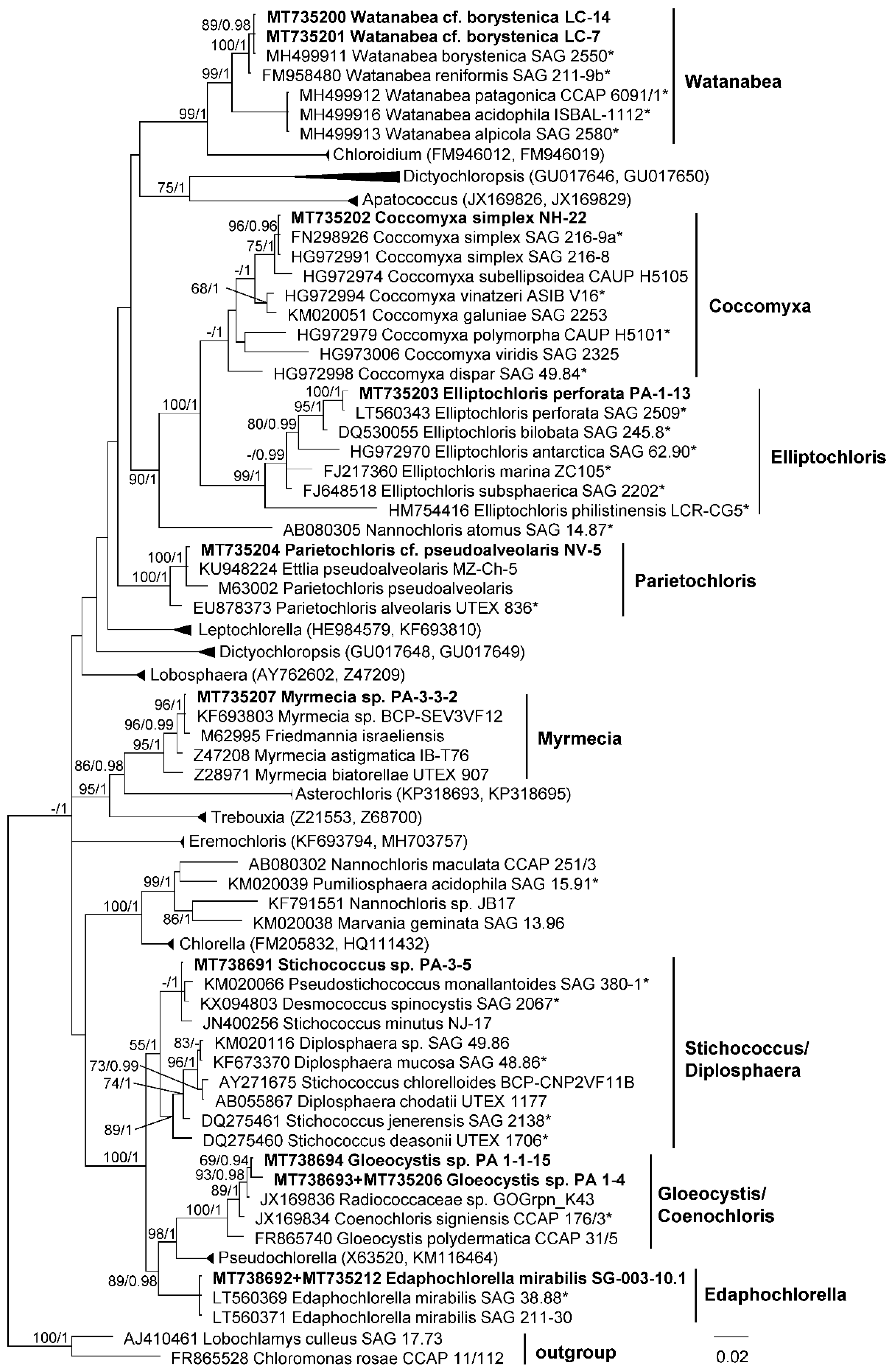
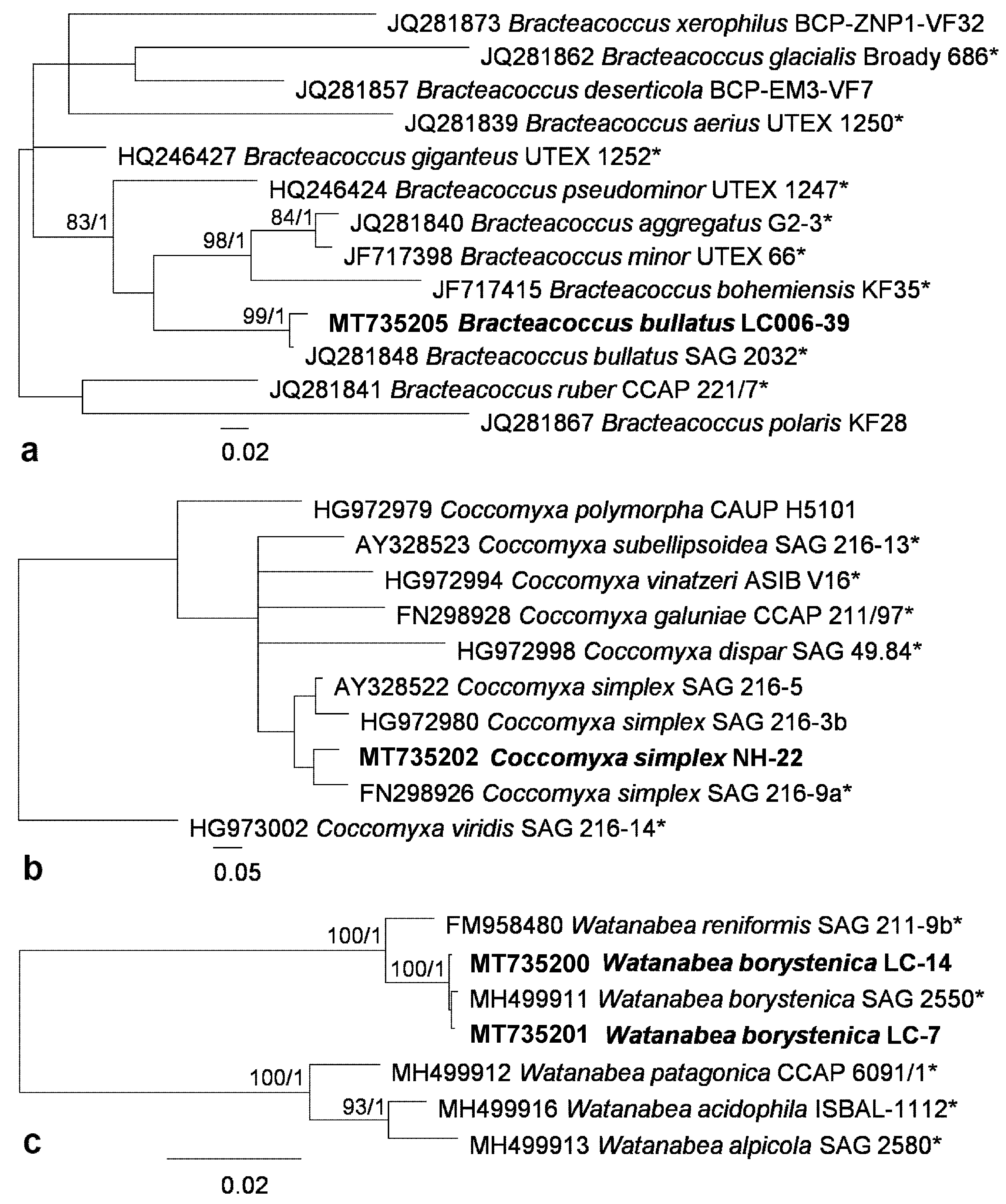
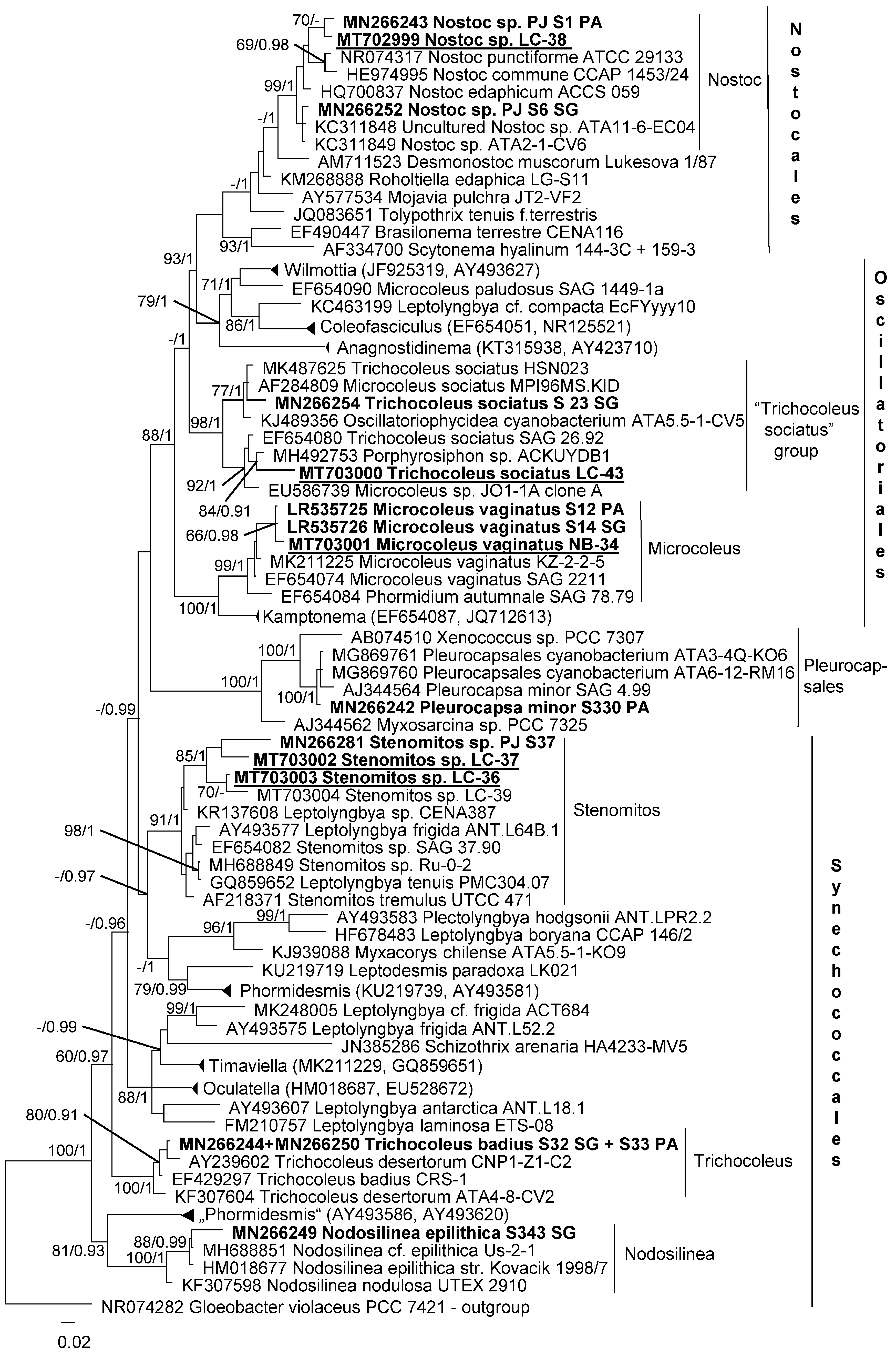
2.4. Phylogenetic Analyses
3. Results
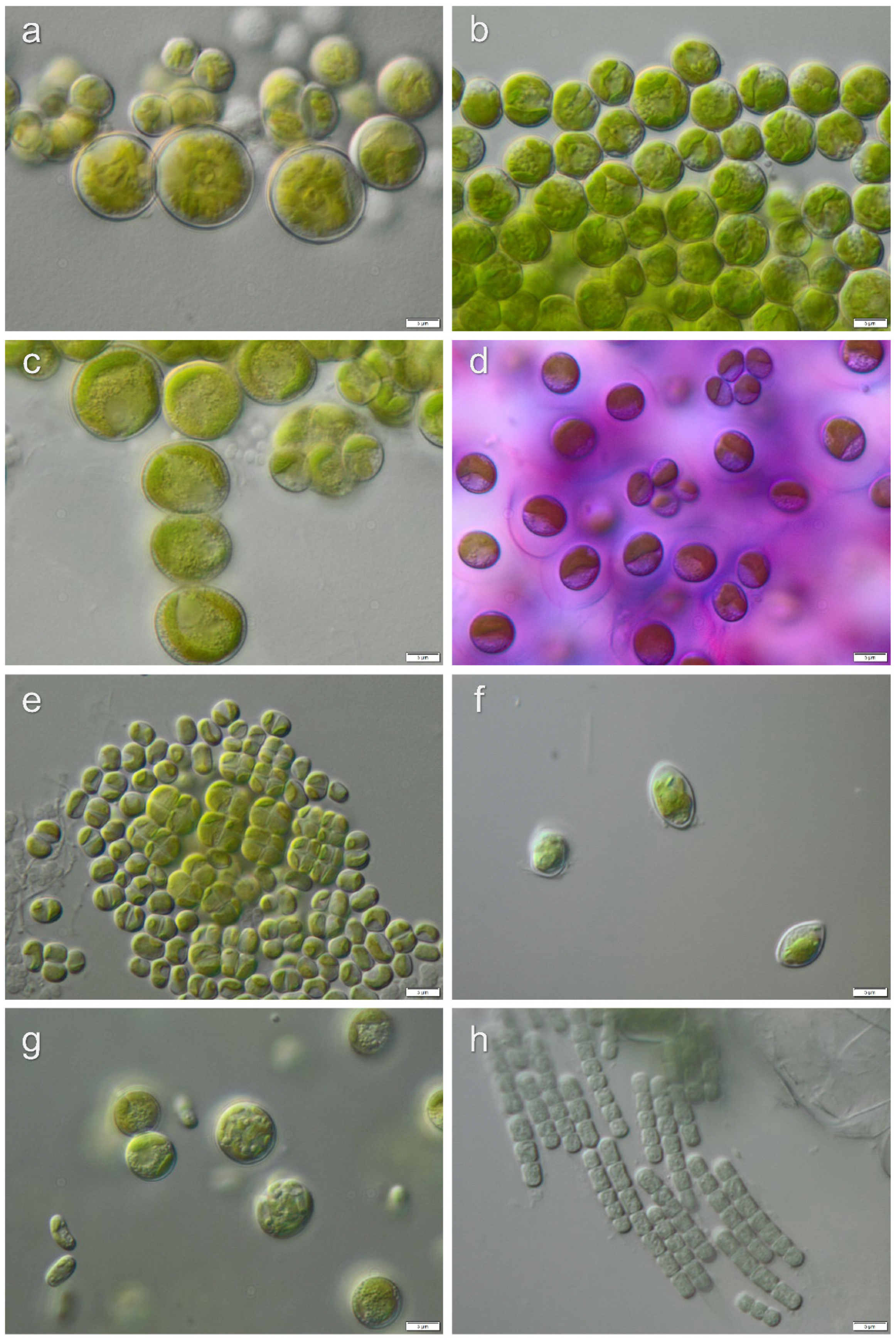
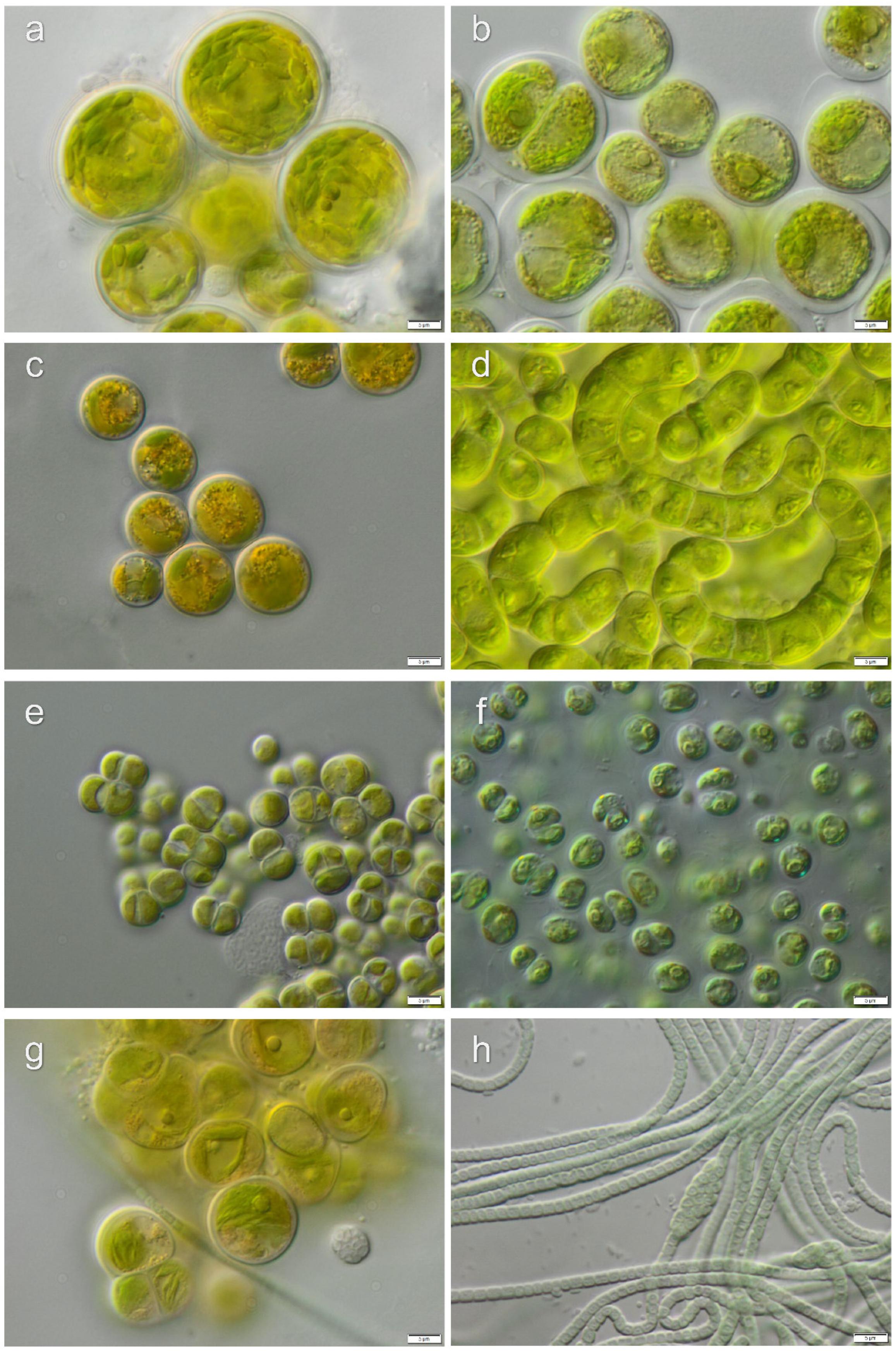
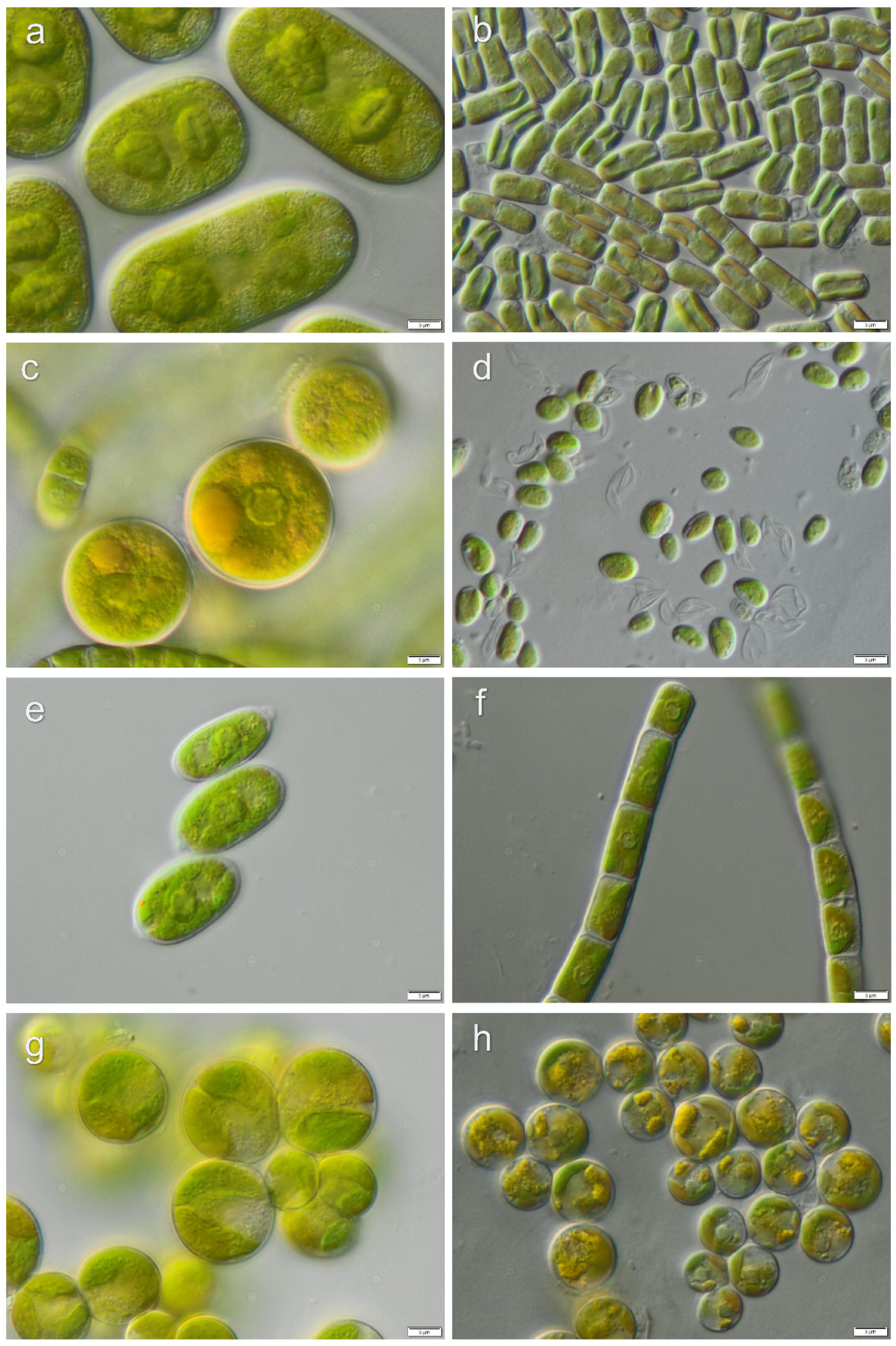
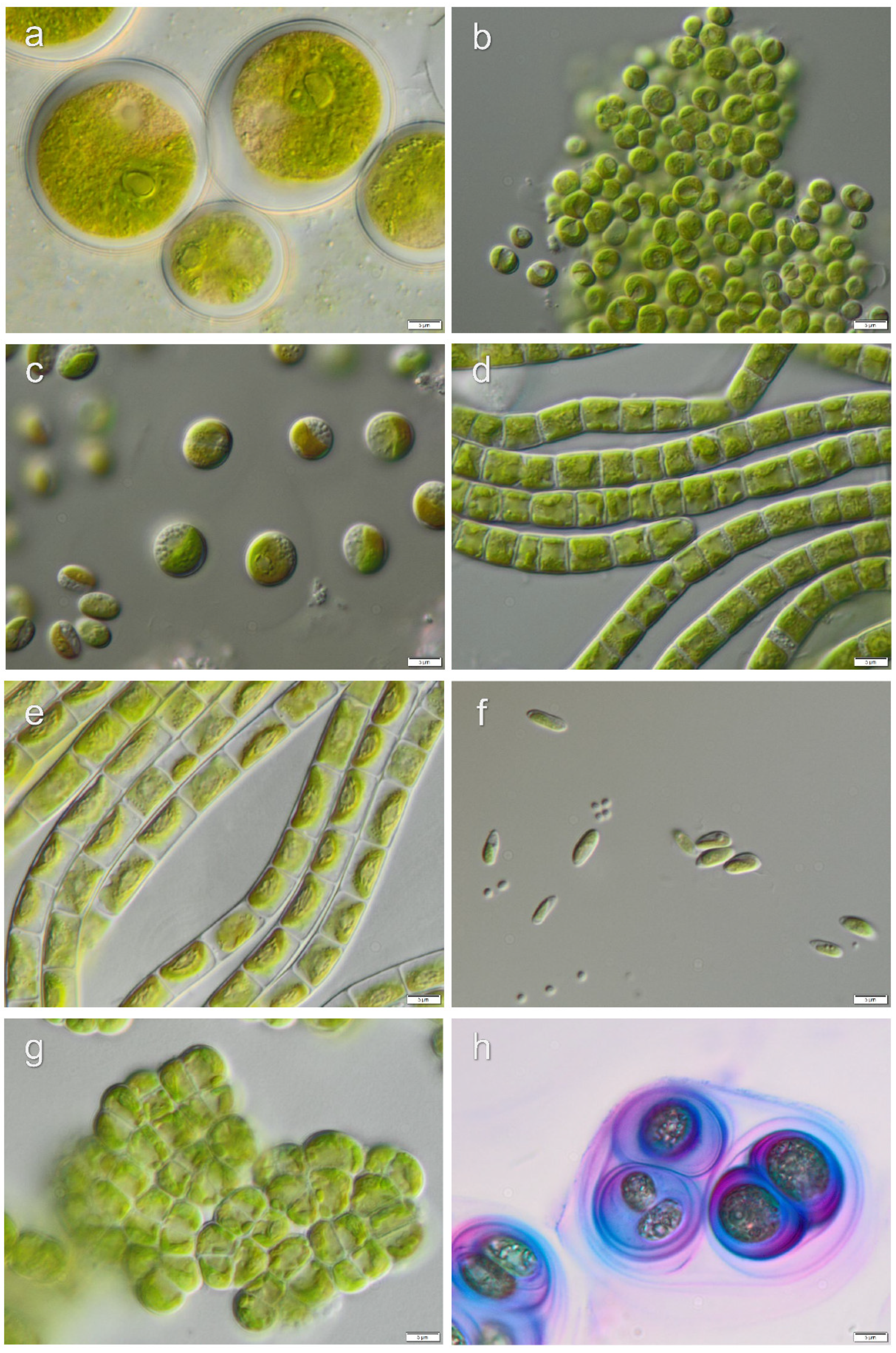
3.1. Diversity of Algae—Identification Based on Morphology and Molecular Phylogeny
3.2. Diversity of Cyanobacteria—Identification Based on Morphology and Molecular Phylogeny
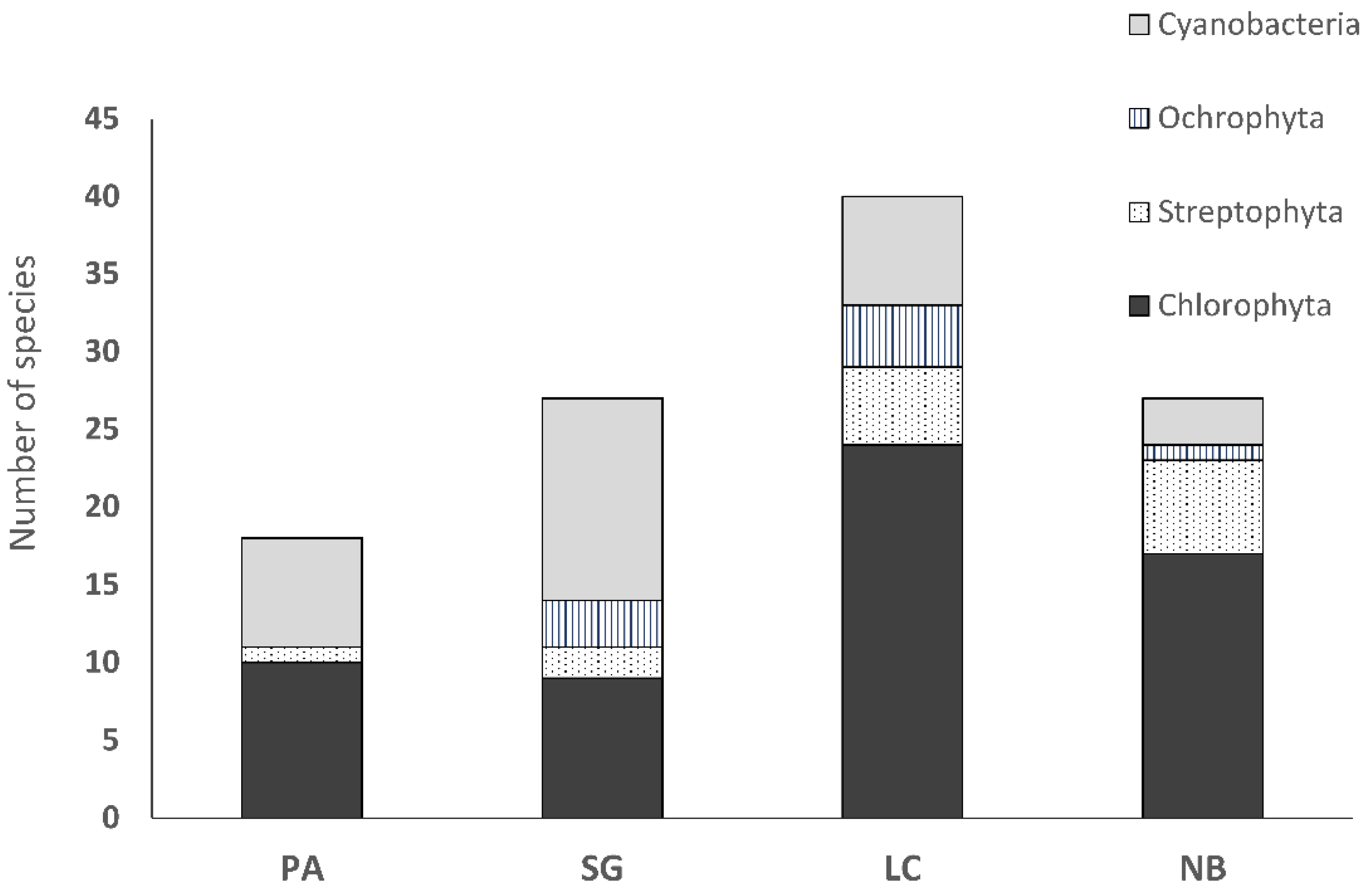
3.3. Biocrust Community Composition and Species Richness
4. Discussion
4.1. Differences in Community Composition of Biocrust Algae and Cyanobacteria along the Precipitation Gradient
4.2. Pan de Azúcar Biocrusts as an Example for an Extreme Biocrust Habitat
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Belnap, J.; Eldridge, D. Disturbance and Recovery of Biological Soil Crusts; Springer International Publishing: Berlin, Germany, 2001; Volume 150, pp. 363–383. [Google Scholar]
- Chamizo, S.; Cantón, Y.; Rodríguez-Caballero, E.; Domingo, F. Biocrusts positively affect the soil water balance in semiarid ecosystems. Ecohydrology 2016, 9, 1208–1221. [Google Scholar] [CrossRef]
- Rodríguez-Caballero, E.; Belnap, J.; Büdel, B.; Crutzen, P.J.; Andreae, M.O.; Poschl, U.; Weber, B. Dryland photoautotrophic soil surface communities endangered by global change. Nat. Geosci. 2018, 11, 185–189. [Google Scholar] [CrossRef]
- Baumann, K.; Jung, P.; Samolov, E.; Lehnert, L.W.; Büdel, B.; Karsten, U.; Bendix, J.; Achilles, S.; Schermer, M.; Matus, F.; et al. Biological soil crusts along a climatic gradient in Chile: Richness and imprints of phototrophic microorganisms in phosphorus biogeochemical cycling. Soil Biol. Biochem. 2018, 127, 286–300. [Google Scholar] [CrossRef]
- Tucker, C.L.; Antoninka, A.; Day, N.K.; Poff, B.; Reed, S. Biological soil crust salvage for dryland restoration: An opportunity for natural resource restoration. Restor. Ecol. 2020, 13115. [Google Scholar] [CrossRef]
- Maestre, F.T.; Bowker, M.A.; Castillo-Monroy, A.P.; Ochoa-Hueso, R. Species richness effects on ecosystem multifunctionality depend on evenness, composition and spatial pattern. J. Ecol. 2011, 100, 317–330. [Google Scholar] [CrossRef]
- Bowker, M.A.; Maestre, F.T.; Mau, R.L. Diversity and patch-size distributions of biological soil crusts regulate dryland ecosystem multifunctionality. Ecosystems 2013, 16, 923–933. [Google Scholar] [CrossRef]
- Hu, C.; Liu, Y.; Song, L.; Zhang, D. Effect of desert soil algae on the stabilization of fine sands. Environ. Biol. Fishes 2002, 14, 281–292. [Google Scholar] [CrossRef]
- Miller, M.E.; Belote, R.T.; Bowker, M.A.; Garman, S.L. Alternative states of a semiarid grassland ecosystem: Implications for ecosystem services. Ecosphere 2011, 2. [Google Scholar] [CrossRef]
- Kristiansen, J. 16. Dispersal of freshwater algae—a review. Hydrobiol. 1996, 336, 151–157. [Google Scholar] [CrossRef]
- Sharma, N.K.; Rai, A.; Singh, S.; Brown, R.M. Airborne algae: Their present status and relevance1. J. Phycol. 2007, 43, 615–627. [Google Scholar] [CrossRef]
- Moore, K.A.; Elmendorf, S.C. Propagule vs. niche limitation: Untangling the mechanisms behind plant species’ distributions. Ecol. Lett. 2006, 9, 797–804. [Google Scholar] [CrossRef]
- Pedrós-Alió, C. Marine microbial diversity: Can it be determined? Trends Microbiol. 2006, 14, 257–263. [Google Scholar] [CrossRef]
- Colesie, C.; Felde, V.J.M.N.L.; Büdel, B. Composition and macrostructure of biological soil crusts. In Photosynthetic Adaptation; Weber, B., Büdel, B., Belnap, J., Eds.; Springer International Publishing: Cham, Germany, 2016; Volume 226, pp. 159–172. [Google Scholar]
- Safriel, U.; Adeel, Z.; Niemeijer, D.; Puigdefabregas, J.; White, R.; Lal, R.; Winslow, M.; Ziedler, J.; Prince, S.; Archer, E.; et al. Dryland Systems. In Ecosystems and Human Well-being: Current State and Trends: Findings of the Condition and Trends Working Group; Hassan, R., Scholes, R., Ash, N., Eds.; Island Press: Washington, DC, USA, 2005; pp. 623–662. [Google Scholar]
- Fischer, T.; Gypser, S.; Subbotina, M.; Veste, M. Synergic hydraulic and nutritional feedback mechanisms control surface patchiness of biological soil crusts on tertiary sands at a post-mining site. J. Hydrol. Hydromechanics 2014, 62, 293–302. [Google Scholar] [CrossRef]
- Baumann, K.; Glaser, K.; Mutz, J.-E.; Karsten, U.; MacLennan, A.; Hu, Y.; Michalik, D.; Kruse, J.; Eckhardt, K.-U.; Schall, P.; et al. Biological soil crusts of temperate forests: Their role in P cycling. Soil Biol. Biochem. 2017, 109, 156–166. [Google Scholar] [CrossRef]
- Glaser, K.; Baumann, K.; Leinweber, P.; Mikhailyuk, T.; Karsten, U. Algal richness in BSCs in forests under different management intensity with some implications for P cycling. Biogeosciences 2018, 15, 4181–4192. [Google Scholar] [CrossRef]
- Büdel, B.; Dulić, T.; Darienko, T.; Rybalka, N.; Friedl, T. Cyanobacteria and Algae of Biological Soil Crusts. In Photosynthetic Adaptation; Weber, B., Büdel, B., Belnap, J., Eds.; Springer International Publishing: Cham, Germany, 2016; Volume 226, pp. 55–80. [Google Scholar]
- Schwabe, G.H. Zur Autotrophen Vegetation in Ariden Böden. Blaualgen Und Lebensraum IV. Österreichische Bot. Z. 1960, 107, 281–309. [Google Scholar] [CrossRef]
- Forest, H.S.; Weston, C.R. Blue-green algae from the Atacama Desert of Northern Chile. J. Phycol. 1966, 2, 163–164. [Google Scholar] [CrossRef]
- Patzelt, D.J.; Hodač, L.; Friedl, T.; Pietrasiak, N.; Johansen, J.R. Biodiversity of soil cyanobacteria in the hyper-arid Atacama Desert, Chile. J. Phycol. 2014, 50, 698–710. [Google Scholar] [CrossRef]
- Mühlsteinová, R.; Johansen, J.R.; Pietrasiak, N.; Martin, M.P. Polyphasic characterization of Kastovskya adunca gen. nov. et comb. nov. (Cyanobacteria: Oscillatoriales), from desert soils of the Atacama Desert, Chile. Phytotaxa 2014, 163, 216. [Google Scholar] [CrossRef]
- Mühlsteinová, R.; Johansen, J.R.; Pietrasiak, N.; Martin, M.P.; Osorio-Santos, K.; Warren, S.D. Polyphasic characterization of Trichocoleus desertorum sp. nov. (Pseudanabaenales, Cyanobacteria) from desert soils and phylogenetic placement of the genus Trichocoleus. Phytotaxa 2014, 163, 241. [Google Scholar] [CrossRef]
- Osorio-Santos, K.; Pietrasiak, N.; Bohunická, M.; Miscoe, L.H.; Kováčik, L.; Martin, M.P.; Johansen, J.R. Seven new species of Oculatella (Pseudanabaenales, Cyanobacteria): Taxonomically recognizing cryptic diversification. Eur. J. Phycol. 2014, 49, 450–470. [Google Scholar] [CrossRef]
- Pietrasiak, N.; Mühlsteinová, R.; Siegesmund, M.A.; Johansen, J.R. Phylogenetic placement of Symplocastrum (Phormidiaceae, Cyanophyceae) with a new combination S. californicum and two new species: S. flechtnerae and S. torsivum. Phycol. 2014, 53, 529–541. [Google Scholar] [CrossRef]
- Pietrasiak, N.; Osorio-Santos, K.; Shalygin, S.; Martin, M.P.; Johansen, J.R. When is a lineage a species? A case study in Myxacorys gen. nov. (Synechococcales: Cyanobacteria) with the description of two new species from the Americas. J. Phycol. 2019, 55, 976–996. [Google Scholar] [CrossRef] [PubMed]
- Jung, P.; Baumann, K.; Lehnert, L.W.; Samolov, E.; Achilles, S.; Schermer, M.; Wraase, L.M.; Eckhardt, K.; Bader, M.; Leinweber, P.; et al. Desert breath-How fog promotes a novel type of soil biocenosis, forming the coastal Atacama Desert’s living skin. Geobiology 2019, 18, 113–124. [Google Scholar] [CrossRef]
- Jung, P.; Mikhailyuk, T.; Emrich, D.; Baumann, K.; Dultz, S.; Büdel, B. Shifting boundaries: Ecological and geographical range extension based on three new species in the cyanobacterial genera Cyanocohniella, Oculatella, and, Aliterella. J. Phycol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Darienko, T.; Kang, W.; Orzechowski, A.K.; Pröschold, T. Pleurastrosarcina terriformae, a new species of a rare desert trebouxiophycean alga discovered by an integrative approach. Extremophiles 2019, 23, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Samolov, E.; Mikhailyuk, T.; Lukešová, A.; Glaser, K.; Büdel, B.; Karsten, U. Usual alga from unusual habitats: Biodiversity of Klebsormidium (Klebsormidiophyceae, Streptophyta) from the phylogenetic superclade G isolated from biological soil crusts. Mol. Phylogenetics Evol. 2019, 133, 236–255. [Google Scholar] [CrossRef]
- Rindi, F.; Mikhailyuk, T.; Sluiman, H.J.; Friedl, T.; Lopez-Bautista, J.M. Phylogenetic relationships in Interfilum and Klebsormidium (Klebsormidiophyceae, Streptophyta). Mol. Phylogenetics Evol. 2011, 58, 218–231. [Google Scholar] [CrossRef]
- Lehnert, L.; Thies, B.; Trachte, K.; Achilles, S.; Osses, P.; Baumann, K.; Bendix, J.; Schmidt, J.; Samolov, E.; Jung, P.; et al. A Case Study on Fog/Low Stratus Occurrence at Las Lomitas, Atacama Desert (Chile) as a Water Source for Biological Soil Crusts. Aerosol Air Qual. Res. 2018, 18, 254–269. [Google Scholar] [CrossRef]
- Larrain, H.; Velásquez, F.; Cereceda, P.; Espejo, R.; Pinto, R.; Osses, P.; Schemenauer, R. Fog measurements at the site “Falda Verde” north of Chañaral compared with other fog stations of Chile. Atmospheric Res. 2002, 64, 273–284. [Google Scholar] [CrossRef]
- Bernhard, N.; Moskwa, L.-M.; Schmidt, K.; Oeser, R.; Aburto, F.; Bader, M.; Baumann, K.; Von Blanckenburg, F.; Boy, J.; Brink, L.V.D.; et al. Pedogenic and microbial interrelations to regional climate and local topography: New insights from a climate gradient (arid to humid) along the Coastal Cordillera of Chile. Catena 2018, 170, 335–355. [Google Scholar] [CrossRef]
- Schulz, K.; Mikhailyuk, T.; Dreßler, M.; Leinweber, P.; Karsten, U. Biological Soil Crusts from Coastal Dunes at the Baltic Sea: Cyanobacterial and Algal Biodiversity and Related Soil Properties. Microb. Ecol. 2015, 71, 178–193. [Google Scholar] [CrossRef] [PubMed]
- Starr, R.C.; Zeikus, J.A. UTEX—The culture collevtion of algae at the university of Texas ataustin 1 2 3. J. Phycol. 1993, 29, 1–106. [Google Scholar] [CrossRef]
- Rippka, R. Pasteur culture collection of cyanobacterial strains in axenic culture. Cat. Taxon. Handb. Cat. Strains 1992/1993 1992, 1, 1–103. [Google Scholar]
- Ettl, H.; Gartner, G. Syllabus der Boden-, Luft- und Flechtenalgen, 2nd ed.; Springer: Berlin, Germany, 2013. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Süßwasserflora von Mitteleuropa, Bd. 19/2: Cyanoprokaryota: Bd. 2/Part 2: Oscillatoriales, German ed.; Springer Spektrum: Berlin, Germany, 2007. [Google Scholar]
- Komárek, J. Bd. 19/3: Cyanoprokaryota: 3. Teil/3rd Part: Heterocytous Genera; Springer Spektrum: Berlin, Germany, 2013. [Google Scholar]
- Marin, B.; Klingberg, M.; Melkonian, M. Phylogenetic relationships among the Cryptophyta: Analyses of nuclear-encoded SSU rRNA sequences support the monophyly of extant plastid-containing lineages. Protist 1998, 149, 265–276. [Google Scholar] [CrossRef]
- Marin, B.; Palm, A.; Klingberg, M.; Melkonian, M. Phylogeny and Taxonomic Revision of Plastid-Containing Euglenophytes based on SSU rDNA Sequence Comparisons and Synapomorphic Signatures in the SSU rRNA Secondary Structure. Protist 2003, 154, 99–145. [Google Scholar] [CrossRef]
- Mikhailyuk, T.; Lukešová, A.; Glaser, K.; Holzinger, A.; Obwegeser, S.; Nyporko, S.; Friedl, T.; Karsten, U. New taxa of streptophyte algae (Streptophyta) from terrestrial habitats revealed using an integrative approach. Protist 2018, 169, 406–431. [Google Scholar] [CrossRef]
- Lane, D.J. 16S/23S rRNA Sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E., Goodfellow, M., Eds.; John Wiley and Sons: New York, NY, USA, 1991. [Google Scholar]
- Goff, L.J.; Moon, D.A. PCR amplification of nuclear and plastid genes from algal herbarium specimens and algal spores1. J. Phycol. 1993, 29, 381–384. [Google Scholar] [CrossRef]
- Hoef-Emden, K.; Melkonian, M. Revision of the genus Cryptomonas (Cryptophyceae): A combination of molecular phylogeny and morphology provides insights into a long-hidden dimorphism. Protist 2003, 154, 371–409. [Google Scholar] [CrossRef]
- Williams, L.; Jung, P.; Zheng, L.-J.; Maier, S.; Peer, T.; Grube, M.; Weber, B.; Büdel, B. Assessing recovery of biological soil crusts across a latitudinal gradient in Western Europe. Restor. Ecol. 2017, 26, 543–554. [Google Scholar] [CrossRef]
- Mühling, M.; Woolven-Allen, J.; Murrell, J.C.; Joint, I. Improved group-specific PCR primers for denaturing gradient gel electrophoresis analysis of the genetic diversity of complex microbial communities. ISME J. 2008, 2, 379–392. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Jung, P.; Schermer, M.; Briegel-Williams, L.; Baumann, K.; Leinweber, P.; Karsten, U.; Lehnert, L.; Achilles, S.; Bendix, J.; Büdel, B. Water availability shapes edaphic and lithic cyanobacterial communities in the Atacama Desert. J. Phycol. 2019, 55, 1306–1318. [Google Scholar] [CrossRef]
- Akaike, H. A new look at the statistical model identification. IEEE Trans. Autom. Control. 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Boil. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinform. 2003, 19, 1572–1574. [Google Scholar] [CrossRef]
- Zwickl, D.J. Genetic Algorithm Approaches for the Phylogenetic Analysis of Large Biological Sequence Datasets Under the Maximum Likelihood Criterion. Ph.D. Thesis, The University of Texas at Austin, Austin, TX, USA, May 2006. [Google Scholar]
- Darienko, T.; Gustavs, L.; Pröschold, T.; Verbruggen, H. Species concept and nomenclatural changes within the genera Elliptochloris and Pseudochlorella (Trebouxiophyceae) based on an integrative approach. J. Phycol. 2016, 52, 1125–1145. [Google Scholar] [CrossRef]
- Rindi, F.; Guiry, M.D.; López-Bautista, J.M. Distribution, Morphology, and Phylogeny of Klebsormidium (Klebsormidiales, Charophyceae) in Urban Environments in Europe 1. J. Phycol. 2008, 44, 1529–1540. [Google Scholar] [CrossRef]
- Nakada, T.; Tomita, M.; Wu, J.; Nozaki, H. Taxonomic revision of Chlamydomonas subg. Amphichloris (Volvocales, Chlorophyceae), with resurrection of the genus Dangeardinia and descriptions of Ixipapillifera gen. nov. and Rhysamphichloris gen. nov. J. Phycol. 2016, 52, 283–304. [Google Scholar] [CrossRef]
- Mikhailyuk, T.; Holzinger, A.; Tsarenko, P.; Glaser, K.; Demchenko, E.; Karsten, U. Dictyosphaerium-like morphotype in terrestrial algae: What is Xerochlorella (Trebouxiophyceae, Chlorophyta)? J. Phycol. 2020, 56, 671–686. [Google Scholar] [CrossRef]
- Darienko, T.; Pröschold, T. Reevaluation and discovery of new species of the rare genus Watanabea and establishment of Massjukichlorella gen. nov. (Trebouxiophyceae, Chlorophyta) using an integrative approach. J. Phycol. 2019, 55, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, L. Algae of terrestrial habitats. Bot. Rev. 1989, 55, 77–105. [Google Scholar] [CrossRef]
- Bowker, M.A.; Belnap, J.; Büdel, B.; Sannier, C.; Pietrasiak, N.; Eldridge, D.J.; Rivera-Aguilar, V. Controls on Distribution Patterns of Biological Soil Crusts at Micro- to Global Scales. In Photosynthetic Adaptation; Weber, B., Büdel, B., Belnap, J., Eds.; Springer International Publishing: Cham, Germany, 2016; Volume 226, pp. 173–197. [Google Scholar]
- Ullmann, I.; Büdel, B. Biological Soil Crusts of Africa. In Photosynthetic Adaptation; Weber, B., Büdel, B., Belnap, J., Eds.; Springer International Publishing: Cham, Germany, 2001; Volume 150, pp. 107–118. [Google Scholar]
- Warren-Rhodes, K.A.; Rhodes, K.L.; Pointing, S.B.; Ewing, S.A.; Lacap, D.C.; Gómez-Silva, B.; Amundson, R.; Friedmann, E.I.; McKay, C.P. Hypolithic cyanobacteria, dry limit of photosynthesis, and microbial ecology in the hyperarid Atacama Desert. Microb. Ecol. 2006, 52, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Lange, O.L.; Kilian, E.; Ziegler, H. Water vapor uptake and photosynthesis of lichens: Performance differences in species with green and blue-green algae as phycobionts. Oecologia 1986, 71, 104–110. [Google Scholar] [CrossRef]
- Nienow, J.A. Ecology of Subaerial Algae. Nova Hedwig. Beih. 1996, 112, 537–552. [Google Scholar]
- Mikhailyuk, T.I.; Sluiman, H.J.; Massalski, A.; Mudimu, O.; Demchenko, E.M.; Kondratyuk, S.Y.; Friedl, T. New Streptophyte Green Algae from Terrestrial Habitats and an Assessment of the Genus Interfilum (Klebsormidiophyceae, Streptophyta) 1. J. Phycol. 2008, 44, 1586–1603. [Google Scholar] [CrossRef] [PubMed]
- Voytsekhovich, A.; Dymytrova, L.; Nadyeina, O. Photobiont composition of some taxa of the genera Micarea and Placynthiella (Lecanoromycetes, lichenized Ascomycota) from Ukraine. Folia Cryptogam. Est. 2011, 48, 135–148. [Google Scholar]
- Karsten, U.; Herburger, K.; Holzinger, A. Dehydration, temperature, and light tolerance in members of the aeroterrestrial green algal genus Interfilum (Streptophyta) from biogeographically different temperate soils. J. Phycol. 2014, 50, 804–816. [Google Scholar] [CrossRef]
- Dong, H.; Rech, J.A.; Jiang, H.; Sun, H.; Buck, B.J. Endolithic cyanobacteria in soil gypsum: Occurrences in Atacama (Chile), Mojave (United States), and Al-Jafr Basin (Jordan) Deserts. J. Geophys. Res. Space Phys. 2007, 112, 112. [Google Scholar] [CrossRef]
- Gallego, B.C.; Suzuki, S.; Nealson, K.H.; Wierzchos, J.; Ascaso, C.; Artieda, O.; Ríos, A.D.L. Ignimbrite textural properties as determinants of endolithic colonization patterns from hyper-arid Atacama Desert. Int. Microbiol. 2014, 17. [Google Scholar]
- Wierzchos, J.; Casero, M.C.; Artieda, O.; Ascaso, C. Endolithic microbial habitats as refuges for life in polyextreme environment of the Atacama Desert. Curr. Opin. Microbiol. 2018, 43, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Dillon, J.G.; Tatsumi, C.M.; Tandingan, P.G.; Castenholz, R.W. Effect of environmental factors on the synthesis of scytonemin, a UV-screening pigment, in a cyanobacterium (Chroococcidiopsis sp.). Arch. Microbiol. 2002, 177, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, R.; Molina, A.; Falvey, M. The Atacama surface solar maximum. Bull. Am. Meteorol. Soc. 2015, 96, 405–418. [Google Scholar] [CrossRef]
- Dojani, S.; Kauff, F.; Weber, B.; Büdel, B. Genotypic and phenotypic diversity of cyanobacteria in biological soil crusts of the Succulent Karoo and Nama Karoo of southern Africa. Microb. Ecol. 2013, 67, 286–301. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Martín, M.Á.; Absalón, I.B.; Perona, E.; Fernández-Valbuena, L.; Garcia-Pichel, F.; Mateo, P. Cyanobacterial biocrust diversity in Mediterranean ecosystems along a latitudinal and climatic gradient. New Phytol. 2018, 221, 123–141. [Google Scholar] [CrossRef] [PubMed]



| Species | Localities | |||
|---|---|---|---|---|
| NB | LC | SG | PA | |
| Chlorophyta—45 species | ||||
| Chlorophyceae—23 species | ||||
| Bracteacoccus bullatus | − | 1 | − | − |
| Bracteacoccus medionucleatus | − | 1 | − | − |
| Bracteacoccus sp. | 1 | 1 | − | − |
| Chlamydomonas cf. pseudoelegans | − | 1 | − | − |
| Chlamydomonas sp.1 | 1 | − | − | − |
| Chlamydomonas sp.2 | − | 1 | − | − |
| Chlorococcum cf. minimum | − | 1 | − | − |
| Chlorococcum cf. minutum | − | 1 | − | − |
| Chlorococcum cf. oleofaciens | − | 1 | − | − |
| Chlorococcum echinozygotum | − | 1 | − | − |
| Chlorococcum sp.1 | − | − | 1 | 1 |
| Chlorococcum sp.2 | 1 | − | − | − |
| Chlorosarcinopsis cf. gelatinosa | − | − | 1 | − |
| Fasciculochloris sp. | − | 1 | 1 | − |
| Heterochlamydomonas cf. inaequalis | − | 1 | − | − |
| Heterochlamydomonas sp. | − | − | 1 | − |
| Ixipapillifera sp. | − | 1 | − | − |
| Lobochlamys segnis | 1 | 1 | − | − |
| Macrochloris sp. | 1 | − | − | − |
| Neospongiococcum cf. excentricum | 1 | − | − | − |
| Oogamochlamydinia gen. sp. | − | 1 | − | − |
| Pseudomuriella aurantiaca | 1 | − | − | − |
| Tetracystis intermedia | − | − | 1 | − |
| 7 | 14 | 5 | 1 | |
| Trebouxiophyceae—22 species | ||||
| Chlorella sp. | 1 | 1 | − | − |
| Chloroidium sp. | − | − | − | 1 |
| Coccomyxa simplex | 1 | − | − | − |
| Desmococcus sp. | − | − | 1 | − |
| Diplosphaera chodatii | 1 | 1 | 1 | 1 |
| Edaphochlorella mirabilis | − | − | 1 | − |
| Elliptochloris perforata | − | − | − | 1 |
| Elliptochloris subsphaerica | 1 | 1 | 1 | − |
| Gloeocystis cf. vesiculosa | 1 | 1 | − | − |
| Gloeocystis sp. | − | − | − | 1 |
| Keratococcus raphidioides | − | 1 | − | − |
| Leptosira cf. erumpens | − | 1 | − | − |
| Lobosphaera incisa | 1 | 1 | − | 1 |
| Myrmecia cf. astigmatica | − | − | − | 1 |
| Myrmecia cf. bisecta | 1 | 1 | − | − |
| Myrmecia sp. | 1 | − | − | 1 |
| Neocystis cf. brevis | 1 | − | − | − |
| Parietochloris cf. pseudoalveolaris | 1 | − | − | − |
| Stichococcus sp. | − | − | − | 1 |
| Trebouxia sp. | − | − | − | 1 |
| Watanabea borystenica | − | 1 | − | − |
| Xerochlorella minuta | − | 1 | − | − |
| 10 | 10 | 4 | 9 | |
| Streptophyta—12 species | ||||
| Klebsormidiophyceae—10 species | ||||
| Interfilum massjukiae | 1 | − | − | 1 |
| Interfilum sp. 1 | 1 | − | − | − |
| Interfilum sp. 2 | − | 1 | − | − |
| Klebsormidium chilense | − | 1 | − | − |
| Klebsormidium delicatum var. americanum | 1 | − | − | − |
| Klebsormidium delicatum var. deserticum | − | − | 1 | − |
| Klebsormidium deserticola | − | − | 1 | − |
| Klebsormidium fluitans | 1 | − | − | − |
| Klebsormidium nitens | − | 1 | − | − |
| Klebsormidium sp. | 1 | − | − | − |
| Klebsormidium sylvaticum | 1 | − | − | − |
| 6 | 3 | 2 | 1 | |
| Zygnematophyceae—2 species | ||||
| Cylindrocystis brebissonii | − | 1 | − | − |
| Cylindrocystis crassa | − | 1 | − | − |
| − | 2 | − | − | |
| Ochrophyta—5 species | ||||
| Xanthophyceae—3 species | ||||
| Botrydiopsis cf. constricta | − | 1 | − | − |
| Botrydiopsis cf. intercedens | − | 1 | 1 | − |
| Xantonema exile | − | 1 | − | − |
| − | 3 | 1 | − | |
| Eustigmatophyceae—2 species | ||||
| Vischeria magna | 1 | − | 1 | − |
| Vischeria vischeri | − | 1 | 1 | − |
| 1 | 1 | 2 | − | |
| Species | Localities | |||
|---|---|---|---|---|
| NB | LC | SG | PA | |
| Chroococcidiopsidales—1 species | ||||
| Chroococcidiopsis sp. | − | − | − | 1 |
| – | – | – | 1 | |
| Nostocales—5 species | ||||
| Nostoc cf. edaphicum | − | − | 1 | − |
| Nostoc cf. punctiforme | − | 1 | 1 | − |
| Nostoc sp. 1 | 1 | − | − | − |
| Nostoc sp. 2 | − | 1 | − | − |
| Nostoc sp. 3 | − | − | 1 | 1 |
| 1 | 2 | 3 | 1 | |
| Oscillatoriales—8 species | ||||
| Microcoleus sp. | − | − | 1 | − |
| Microcoleus vaginatus | 1 | − | 1 | 1 |
| Oscillatoria cf. tenuis | − | − | 1 | − |
| Phormidium sp. | − | − | 1 | − |
| Pseudophormidium cf. hollerbachianum | − | − | − | 1 |
| Myxacorys sp.1 | 1 | − | − | − |
| Myxacorys sp.2 | − | − | 1 | − |
| “Trichocoleus” sociatus | − | 1 | 1 | − |
| 2 | 1 | 6 | 2 | |
| Pleurocapsales—1 species | ||||
| Pleurocapsa minor | − | − | − | 1 |
| – | – | – | 1 | |
| Synechococcales—9 species | ||||
| Leptolyngbya henningsii | − | 1 | − | − |
| Leptolyngbya sp. | − | − | 1 | − |
| Leptolyngbya tenuis | − | 1 | − | − |
| Nodosilinea epilithica | − | − | 1 | − |
| Phormidesmis sp. | − | − | − | 1 |
| Stenomitos sp. 1 | − | 1 | − | − |
| Stenomitos sp. 2 | − | 1 | − | − |
| Stenomitos sp. 3 | − | − | 1 | − |
| Trichocoleus cf. badius | − | − | 1 | 1 |
| – | 4 | 4 | 2 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samolov, E.; Baumann, K.; Büdel, B.; Jung, P.; Leinweber, P.; Mikhailyuk, T.; Karsten, U.; Glaser, K. Biodiversity of Algae and Cyanobacteria in Biological Soil Crusts Collected Along a Climatic Gradient in Chile Using an Integrative Approach. Microorganisms 2020, 8, 1047. https://doi.org/10.3390/microorganisms8071047
Samolov E, Baumann K, Büdel B, Jung P, Leinweber P, Mikhailyuk T, Karsten U, Glaser K. Biodiversity of Algae and Cyanobacteria in Biological Soil Crusts Collected Along a Climatic Gradient in Chile Using an Integrative Approach. Microorganisms. 2020; 8(7):1047. https://doi.org/10.3390/microorganisms8071047
Chicago/Turabian StyleSamolov, Elena, Karen Baumann, Burkhard Büdel, Patrick Jung, Peter Leinweber, Tatiana Mikhailyuk, Ulf Karsten, and Karin Glaser. 2020. "Biodiversity of Algae and Cyanobacteria in Biological Soil Crusts Collected Along a Climatic Gradient in Chile Using an Integrative Approach" Microorganisms 8, no. 7: 1047. https://doi.org/10.3390/microorganisms8071047
APA StyleSamolov, E., Baumann, K., Büdel, B., Jung, P., Leinweber, P., Mikhailyuk, T., Karsten, U., & Glaser, K. (2020). Biodiversity of Algae and Cyanobacteria in Biological Soil Crusts Collected Along a Climatic Gradient in Chile Using an Integrative Approach. Microorganisms, 8(7), 1047. https://doi.org/10.3390/microorganisms8071047






