A Novel Lactic Acid Bacteria Mixture: Macrophage-Targeted Prophylactic Intervention in Colorectal Cancer Management
Abstract
1. Introduction
2. Materials and Methods
2.1. Macrophage Culture
2.2. Bacterial Culture
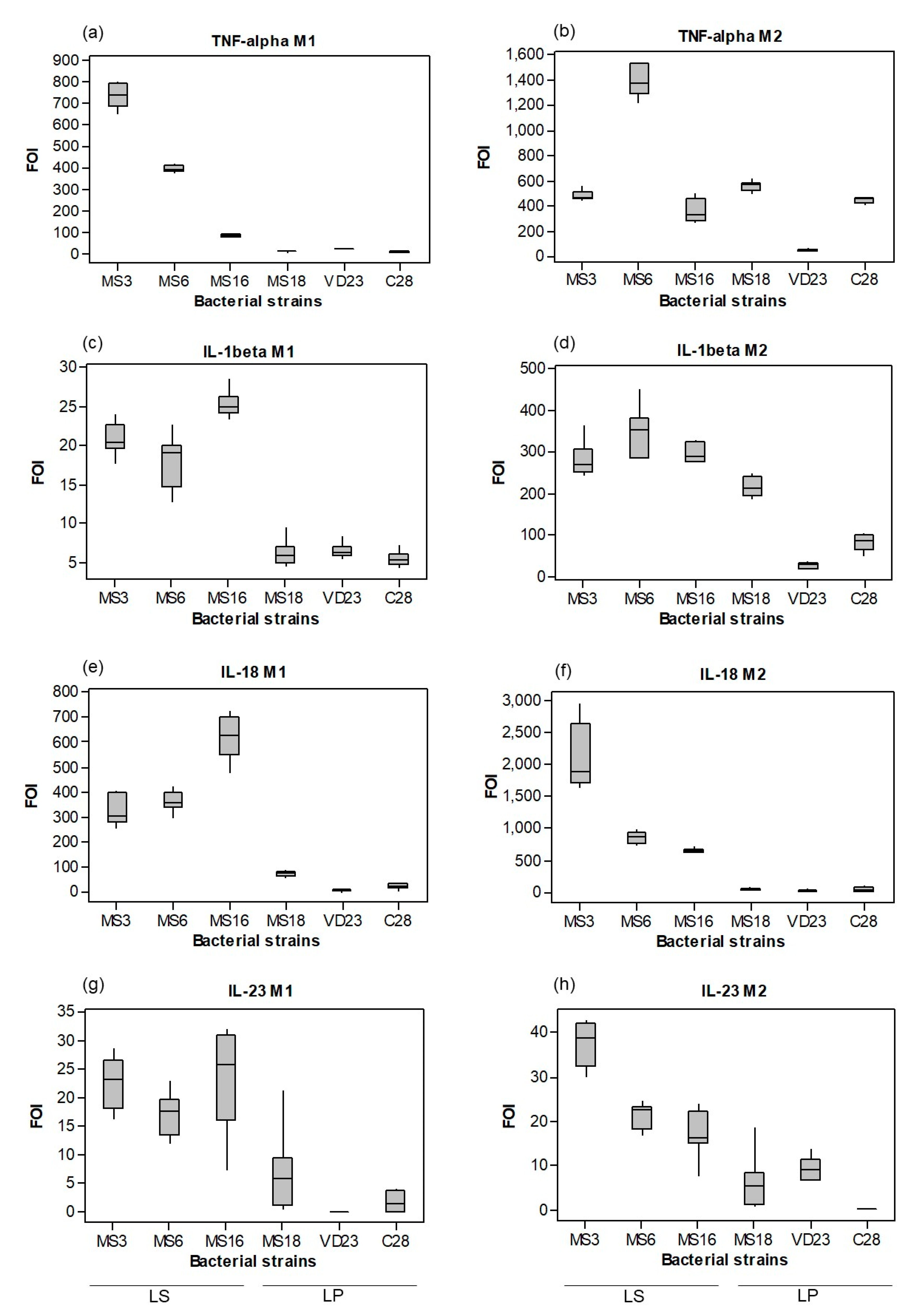
2.3. Modulatory Effect of Live Probiotic Bacteria on Cytokine Production
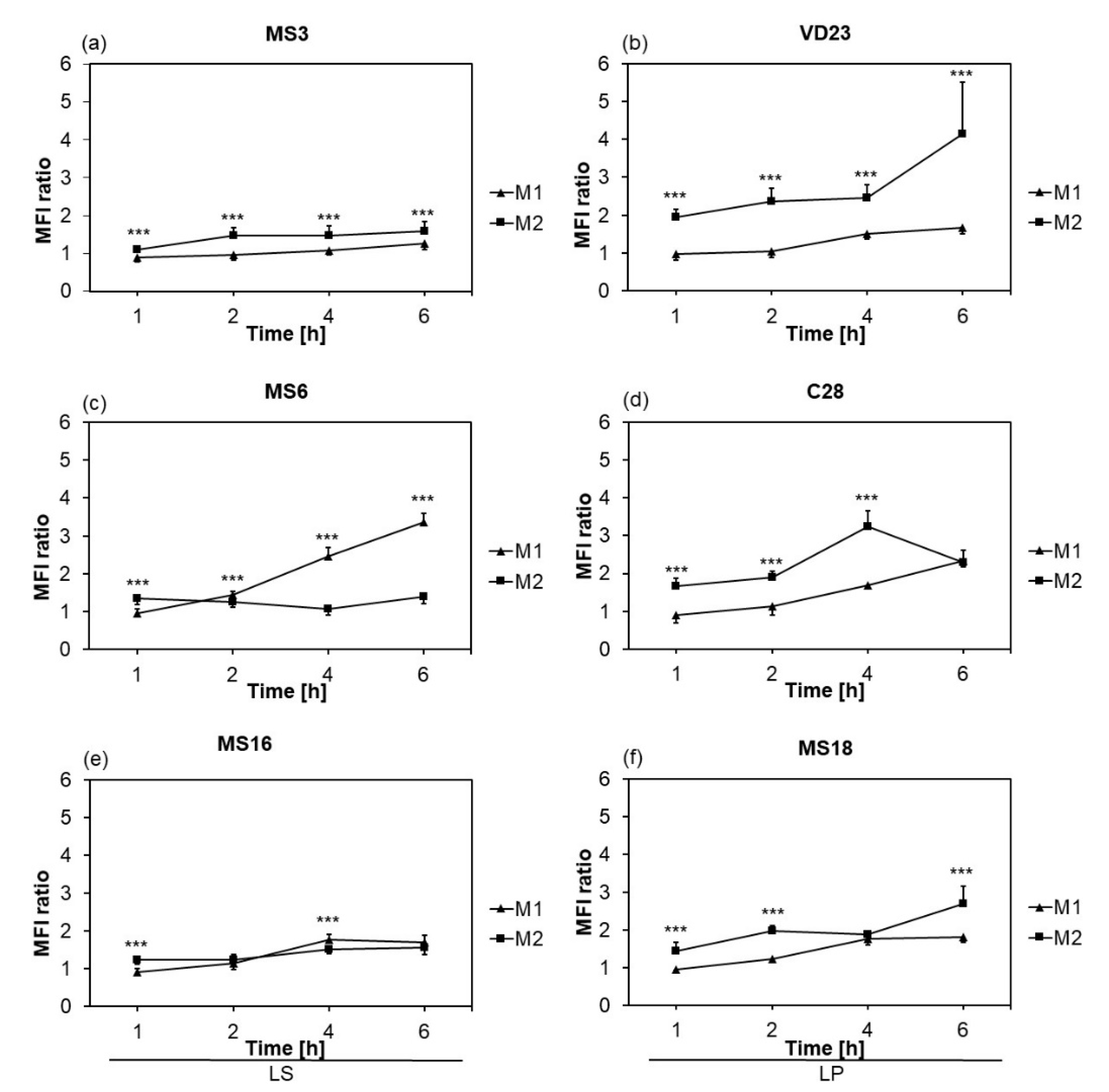
2.4. Phagocytosis Assay of Live Probiotic Bacteria
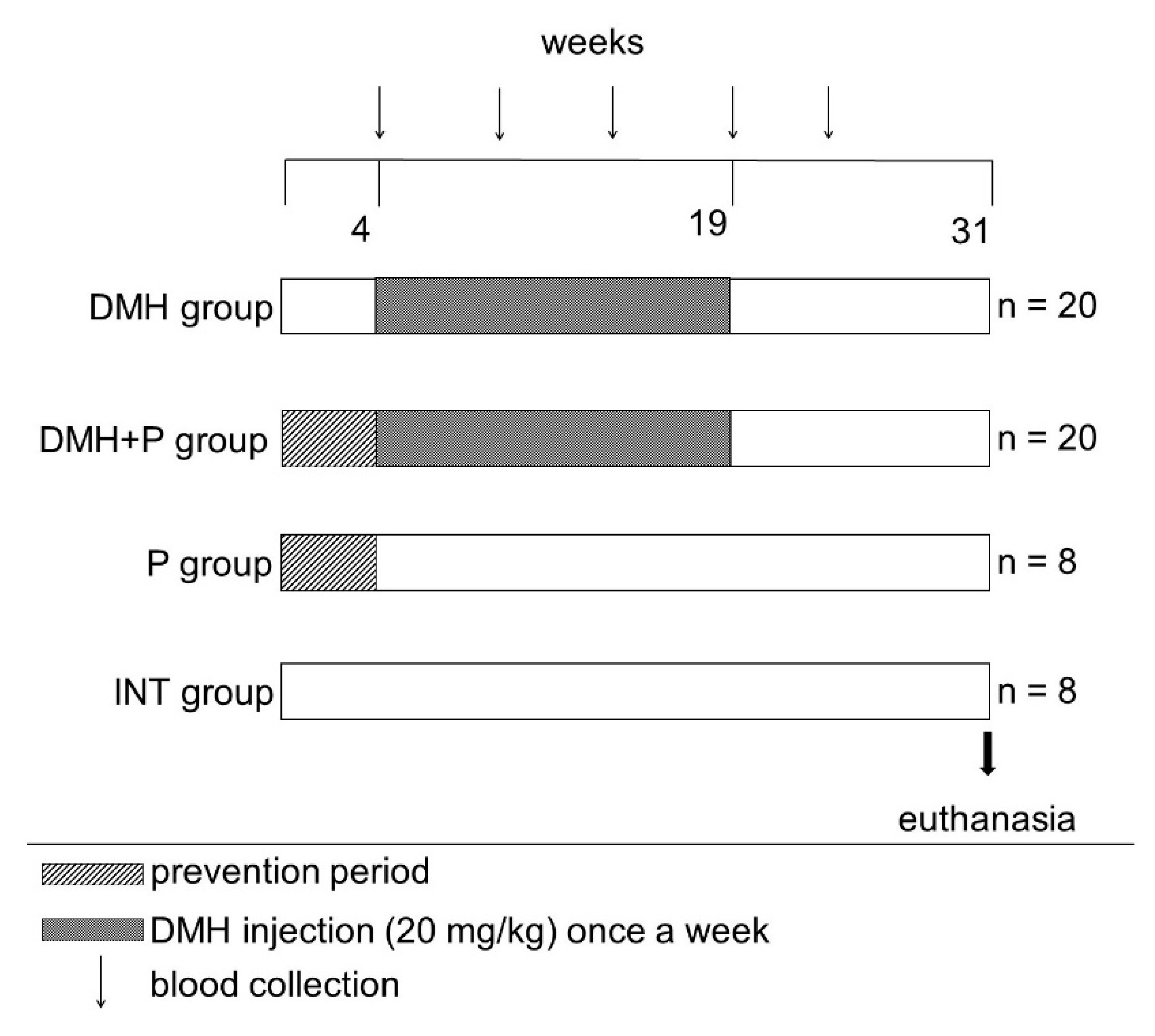
2.5. Animals and Diet
2.6. In Vivo Investigation of Probiotic Effects on Colorectal Cancer
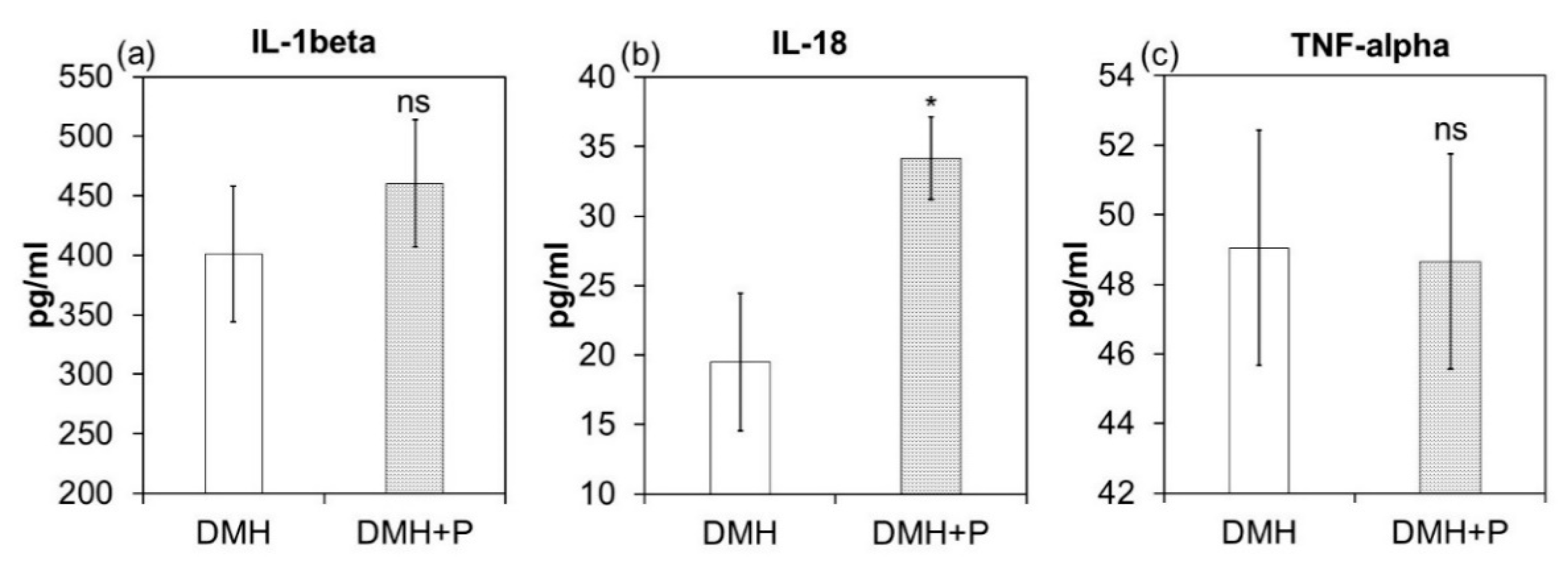
2.7. Measurement of Probiotic Effects on Tumour Incidence, Colon Length and Tumour Cytokine Production
2.8. Statistical Analysis
3. Results
3.1. In Vitro Responses of Macrophages
3.1.1. Probiotic Induction of Macrophage Cytokine Profiles was Both Strain- and Subset-Dependent
3.1.2. Phagocytic Activity of Macrophages was Strain- and Subset-Dependent
3.2. In Vivo Anti-Tumour Effect of LAB Mixture
3.2.1. Animal Health Status
3.2.2. LAB Mixture Treatment had a Selective Effect on Tumour Tissue Secretion of Cytokines.
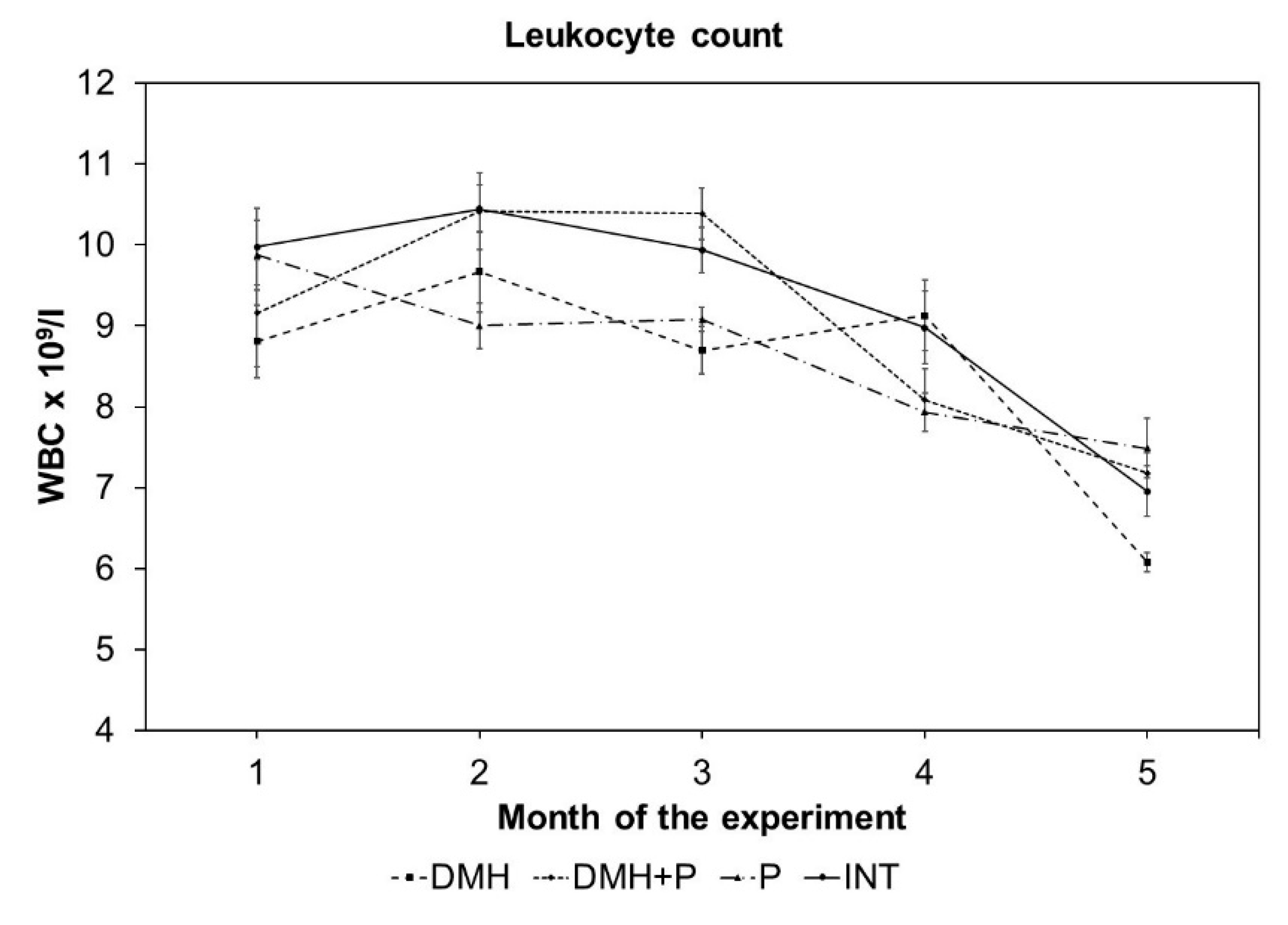
3.2.3. LAB Mixture Treatment had a Positive Effect on Leukocyte Count
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The human microbiome project. Nature 2007, 449, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Pamp, S.J.; Hill, J.A.; Surana, N.K.; Edelman, S.M.; Troy, E.B.; Reading, N.C.; Villablanca, E.J.; Wang, S.; Mora, J.R.; et al. Gut immune maturation depends on colonization with a host-specific microbiota. Cell 2012, 149, 1578–1593. [Google Scholar] [CrossRef] [PubMed]
- Schaubeck, M.; Clavel, T.; Calasan, J.; Lagkouvardos, I.; Haange, S.B.; Jehmlich, N.; Basic, M.; Dupont, A.; Hornef, M.; von Bergen, M.; et al. Dysbiotic gut microbiota causes transmissible Crohn’s disease-like ileitis independent of failure in antimicrobial defence. Gut 2016, 65, 225–237. [Google Scholar] [CrossRef]
- Sobhani, I.; Amiot, A.; Le Baleur, Y.; Levy, M.; Auriault, M.L.; Van Nhieu, J.T.; Delchier, J.C. Microbial dysbiosis and colon carcinogenesis: Could colon cancer be considered a bacteria-related disease? Ther. Adv. Gastroenterol. 2013, 6, 215–229. [Google Scholar] [CrossRef]
- Butto, L.; Haller, D. Dysbiosis in intestinal inflammation: Cause or consequence. Int. J. Med. Microbiol. 2016, 306, 302–309. [Google Scholar] [CrossRef]
- Cenit, M.C.; Olivares, M.; Codoñer-Franch, P.; Sanz, Y. Intestinal Microbiota and Celiac Disease: Cause, Consequence or Co-Evolution? Nutrients 2015, 7, 6900–6923. [Google Scholar] [CrossRef]
- Allen-Vercoe, E. Fusobacterium varium in ulcerative colitis: Is it population-based? Dig. Dis. Sci. 2015, 60, 7–8. [Google Scholar] [CrossRef][Green Version]
- Takaishi, H.; Matsuki, T.; Nakazawa, A.; Takada, T.; Kado, S.; Asahara, T.; Kamada, N.; Sakuraba, A.; Yajima, T.; Higuchi, H.; et al. Imbalance in intestinal microflora constitution could be involved in the pathogenesis of inflammatory bowel disease. Int. J. Med. Microbiol. 2008, 298, 463–472. [Google Scholar] [CrossRef]
- McFarland, L. Use of probiotics to correct dysbiosis of normal microbiota following disease or disruptive events: A systematic review. BMJ Open 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Fijan, S. Microorganisms with claimed probiotic properties: An overview of recent literature. Int. J. Environ. Res. Public Health 2014, 11, 4745–4767. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Nybom, S.; Meriluoto, J.; Collado, M.C.; Vesterlund, S.; El-Nezami, H. Interaction of probiotics and pathogens—Benefits to human health? Curr. Opin. Biotechnol. 2010, 21, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Foligne, B.; Nutten, S.; Grangette, C.; Dennin, V.; Goudercourt, D.; Poiret, S.; Dewulf, J.; Brassart, D.; Mercenier, A.; Pot, B. Correlation between in vitro and in vivo immunomodulatory properties of lactic acid bacteria. World J. Gastroenterol. 2007, 13, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Paolillo, R.; Romano Carratelli, C.; Sorrentino, S.; Mazzola, N.; Rizzo, A. Immunomodulatory effects of Lactobacillus plantarum on human colon cancer cells. Int. Immunopharmacol. 2009, 9, 1265–1271. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Kumar, A.; Nagpal, R.; Mohania, D.; Behare, P.; Verma, V.; Kumar, P.; Poddar, D.; Aggarwal, P.K.; Henry, C.J.; et al. Cancer-preventing attributes of probiotics: An update. Int. J. Food Sci. Nutr. 2010, 61, 473–496. [Google Scholar] [CrossRef]
- Hagemann, T.; Balkwill, F.; Lawrence, T. Inflammation and cancer: A double-edged sword. Cancer Cell 2007, 12, 300–301. [Google Scholar] [CrossRef]
- Lee, S.H.; Starkey, P.M.; Gordon, S. Quantitative analysis of total macrophage content in adult mouse tissues. Immunochemical studies with monoclonal antibody F4/80. J. Exp. Med. 1985, 161, 475–489. [Google Scholar] [CrossRef]
- Biswas, S.K.; Chittezhath, M.; Shalova, I.N.; Lim, J.Y. Macrophage polarization and plasticity in health and disease. Immunol. Res. 2012, 53, 11–24. [Google Scholar] [CrossRef]
- Mills, C.D. M1 and M2 Macrophages: Oracles of Health and Disease. Crit. Rev. Immunol. 2012, 32, 463–488. [Google Scholar] [CrossRef]
- Jetten, N.; Verbruggen, S.; Gijbels, M.; Post, M.; De Winther, M.; Donners, M. Anti-inflammatory M2, but not pro-inflammatory M1 macrophages promote angiogenesis in vivo. Angiogenesis 2014, 17, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.; Louise Jones, J. Jekyll and Hyde: The role of the microenvironment on the progression of cancer. J. Pathol. 2011, 223, 162–176. [Google Scholar] [CrossRef] [PubMed]
- Sica, A.; Mantovani, A. Macrophage plasticity and polarization: In vivo veritas. J. Clin. Investig. 2012, 122, 787–795. [Google Scholar] [CrossRef]
- Ciszek-Lenda, M.; Nowak, B.; Sróttek, M.; Gamian, A.; Marcinkiewicz, J. Immunoregulatory potential of exopolysaccharide from Lactobacillus rhamnosus KL37: Effects on the production of inflammatory mediators by mouse macrophages. Int. J. Exp. Pathol. 2011, 92, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Habil, N.; Beal, J.; Foey, A.D. Lactobacillus casei strain Shirota selectively modulates macrophage subset cytokine production. Int. J. Probiotics Prebiotics 2012, 7, 1–12. [Google Scholar]
- Isidro, R.A.; Appleyard, C.B. Colonic macrophage polarization in homeostasis, inflammation, and cancer. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 311, G59–G73. [Google Scholar] [CrossRef]
- Shida, K.; Nanno, M.; Nagata, S. Flexible cytokine production by macrophages and T cells in response to probiotic bacteria: A possible mechanism by which probiotics exert multifunctional immune regulatory activities. Gut Microbes 2011, 2, 109–114. [Google Scholar] [CrossRef]
- Poh, A.R.; Ernst, M. Targeting Macrophages in Cancer: From Bench to Bedside. Front. Oncol. 2018, 8, 49. [Google Scholar] [CrossRef]
- Demeckova, V. Benefits of Fermented Liquid Diets for Sows and Their Piglets. Ph.D. Dissertation, University of Plymouth, Plymouth, UK, 2003. [Google Scholar]
- Savvidou, S. Selection of a Chicken Lactobacillus Strain with Probiotic Properties and Its Application in Poultry Production. Ph.D. Dissertation, University of Plymouth, Plymouth, UK, 2009. [Google Scholar]
- Brooks, P.; Beal, J.; Savvidou, S.; Demeckova, V. Components of Animal Feed and Use Thereof. UK Patent GB2485476, 4 September 2013. [Google Scholar]
- Chapman, C.M.; Gibson, G.R.; Rowland, I. Health benefits of probiotics: Are mixtures more effective than single strains? Eur. J. Nutr. 2011, 50. [Google Scholar] [CrossRef]
- Daigneault, M.; Preston, J.A.; Marriott, H.M.; Whyte, M.K.; Dockrell, D.H. The identification of markers of macrophage differentiation in PMA-stimulated THP-1 cells and monocyte-derived macrophages. PLoS ONE 2010, 5, e8668. [Google Scholar] [CrossRef]
- Al-Shaghdali, K.; Durante, B.; Hayward, C.; Beal, J.; Foey, A. Macrophage subsets exhibit distinct E. coli-LPS tolerisable cytokines associated with the negative regulators, IRAK-M and Tollip. PLoS ONE 2019, 14, e0214681. [Google Scholar] [CrossRef]
- Maskens, A.P. Histogenesis and growth pattern of 1,2-dimethylhydrazine-induced rat colon adenocarcinoma. Cancer Res. 1976, 36, 1585–1592. [Google Scholar] [PubMed]
- Tomayko, M.M.; Reynolds, C.P. Determination of subcutaneous tumor size in athymic (nude) mice. Cancer Chemother. Pharmacol. 1989, 24, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Christoffersen, T.E.; Hult, L.T.; Kuczkowska, K.; Moe, K.M.; Skeie, S.; Lea, T.; Kleiveland, C.R. In vitro comparison of the effects of probiotic, commensal and pathogenic strains on macrophage polarization. Probiotics Antimicrob. Proteins 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Habil, N.; Al-Murrani, W.; Beal, J.; Foey, A.D. Probiotic bacterial strains differentially modulate macrophage cytokine production in a strain-dependent and cell subset-specific manner. Benef. Microbes 2011, 2, 283–293. [Google Scholar] [CrossRef]
- Carswell, E.A.; Old, L.J.; Kassel, R.L.; Green, S.; Fiore, N.; Williamson, B. An endotoxin-induced serum factor that causes necrosis of tumors. Proc. Natl. Acad. Sci. USA 1975, 72, 3666–3670. [Google Scholar] [CrossRef]
- Ikemoto, S.; Sugimura, K.; Yoshida, N.; Wada, S.; Yamamoto, K.; Kishimoto, T. TNF alpha, IL-1 beta and IL-6 production by peripheral blood monocytes in patients with renal cell carcinoma. Anticancer Res. 2000, 20, 317–321. [Google Scholar]
- Choo, M.K.; Sakurai, H.; Koizumi, K.; Saiki, I. Stimulation of cultured colon 26 cells with TNF-alpha promotes lung metastasis through the extracellular signal-regulated kinase pathway. Cancer Lett. 2005, 230, 47–56. [Google Scholar] [CrossRef]
- Al Obeed, O.A.; Alkhayal, K.A.; Al Sheikh, A.; Zubaidi, A.M.; Vaali-Mohammed, M.A.; Boushey, R.; Mckerrow, J.H.; Abdulla, M.H. Increased expression of tumor necrosis factor-α is associated with advanced colorectal cancer stages. World J. Gastroenterol. 2014, 20, 18390–18396. [Google Scholar] [CrossRef]
- Zhao, P.; Zhang, Z. TNF-α promotes colon cancer cell migration and invasion by upregulating TROP-2. Oncol. Lett. 2018, 15, 3820–3827. [Google Scholar] [CrossRef]
- Elkabets, M.; Ribeiro, V.S.; Dinarello, C.A.; Ostrand-Rosenberg, S.; Di Santo, J.P.; Apte, R.N.; Vosshenrich, C.A. IL-1β regulates a novel myeloid-derived suppressor cell subset that impairs NK cell development and function. Eur. J. Immunol. 2010, 40, 3347–3357. [Google Scholar] [CrossRef] [PubMed]
- Voronov, E.; Reich, E.; Dotan, S.; Dransh, P.; Cohen, I.; Huszar, M.; Fogel, M.; Kleinman, H.; White, R.; Apte, R. Effects of IL-1 molecules on growth patterns of 3-MCA-induced cell lines: An interplay between immunogenicity and invasive potential. J. Immunotoxicol. 2010, 7, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Chun, E.; Lavoie, S.; Michaud, M.; Gallini, C.A.; Kim, J.; Soucy, G.; Odze, R.; Glickman, J.N.; Garrett, W.S. CCL2 Promotes Colorectal Carcinogenesis by Enhancing Polymorphonuclear Myeloid-Derived Suppressor Cell Population and Function. Cell Rep. 2015, 12, 244–257. [Google Scholar] [CrossRef] [PubMed]
- Hyodo, Y.; Matsui, K.; Hayashi, N.; Tsutsui, H.; Kashiwamura, S.; Yamauchi, H.; Hiroishi, K.; Takeda, K.; Tagawa, Y.; Iwakura, Y.; et al. IL-18 up-regulates perforin-mediated NK activity without increasing perforin messenger RNA expression by binding to constitutively expressed IL-18 receptor. J. Immunol. 1999, 162, 1662–1668. [Google Scholar]
- Pages, F.; Berger, A.; Lebel-Binay, S.; Zinzindohoue, F.; Danel, C.; Piqueras, B.; Carriere, O.; Thiounn, N.; Cugnenc, P.H.; Fridman, W.H. Proinflammatory and antitumor properties of interleukin-18 in the gastrointestinal tract. Immunol. Lett. 2000, 75, 9–14. [Google Scholar] [CrossRef]
- Kumar, R.S.; Kanmani, P.; Yuvaraj, N.; Paari, K.A.; Pattukumar, V.; Thirunavukkarasu, C.; Arul, V. Lactobacillus plantarum AS1 isolated from south Indian fermented food Kallappam suppress 1,2-dimethyl hydrazine (DMH)-induced colorectal cancer in male Wistar rats. Appl. Biochem. Biotechnol. 2012, 166, 620–631. [Google Scholar] [CrossRef]
- Mohania, D.; Kansal, V.K.; Sagwal, R.; Shah, D. Anticarcinogenic effect of probiotic Dahi and Piroxicam on DMH-induced colorectal carcinogenesis in Wistar rats. Am. J. Cancer Ther. Pharmacol. 2013, 1, 8–24. [Google Scholar]
- Salcedo, R.; Worschech, A.; Cardone, M.; Jones, Y.; Gyulai, Z.; Dai, R.M.; Wang, E.; Ma, W.; Haines, D.; O’hUigin, C.; et al. MyD88-mediated signaling prevents development of adenocarcinomas of the colon: Role of interleukin 18. J. Exp. Med. 2010, 207, 1625–1636. [Google Scholar] [CrossRef]
- Hirota, S.A.; Ng, J.; Lueng, A.; Khajah, M.; Parhar, K.; Li, Y.; Lam, V.; Potentier, M.S.; Ng, K.; Bawa, M.; et al. NLRP3 inflammasome plays a key role in the regulation of intestinal homeostasis. Inflamm. Bowel Dis. 2011, 17, 1359–1372. [Google Scholar] [CrossRef]
- Mager, L.F.; Wasmer, M.H.; Rau, T.T.; Krebs, P. Cytokine-Induced Modulation of Colorectal Cancer. Front. Oncol. 2016, 6, 96. [Google Scholar] [CrossRef]
- Neurath, M.F. IL-23 in inflammatory bowel diseases and colon cancer. Cytokine Growth Factor Rev. 2019, 45. [Google Scholar] [CrossRef] [PubMed]
- Izcue, A.; Hue, S.; Buonocore, S.; Arancibia-Cárcamo, C.V.; Ahern, P.P.; Iwakura, Y.; Maloy, K.J.; Powrie, F. Interleukin-23 restrains regulatory T cell activity to drive T cell-dependent colitis. Immunity 2008, 28, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Ljujic, B.; Radosavljevic, G.; Jovanovic, I.; Pavlovic, S.; Zdravkovic, N.; Milovanovic, M.; Acimovic, L.; Knezevic, M.; Bankovic, D.; Zdravkovic, D.; et al. Elevated serum level of IL-23 correlates with expression of VEGF in human colorectal carcinoma. Arch. Med. Res. 2010, 41, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, J.; Li, L.; Zhang, J.; Wang, X.; Yang, C.; Li, Y.; Lan, F.; Lin, P. IL-23 selectively promotes the metastasis of colorectal carcinoma cells with impaired Socs3 expression via the STAT5 pathway. Carcinogenesis 2014, 35, 1330–1340. [Google Scholar] [CrossRef]
- Gratchev, A.; Kzhyshkowska, J.; Utikal, J.; Goerdt, S. Interleukin-4 and dexamethasone counterregulate extracellular matrix remodelling and phagocytosis in type-2 macrophages. Scand. J. Immunol. 2005, 61, 10–17. [Google Scholar] [CrossRef]
- Lingnau, M.; Höflich, C.; Volk, H.D.; Sabat, R.; Döcke, W.D. Interleukin-10 enhances the CD14-dependent phagocytosis of bacteria and apoptotic cells by human monocytes. Hum. Immunol. 2007, 68, 730–738. [Google Scholar] [CrossRef]
- Smythies, L.E.; Sellers, M.; Clements, R.H.; Mosteller-Barnum, M.; Meng, G.; Benjamin, W.H.; Orenstein, J.M.; Smith, P.D. Human intestinal macrophages display profound inflammatory anergy despite avid phagocytic and bacteriocidal activity. J. Clin. Investig. 2005, 115, 66–75. [Google Scholar] [CrossRef]
- Fadok, V.A.; Bratton, D.L.; Frasch, S.C.; Warner, M.L.; Henson, P.M. The role of phosphatidylserine in recognition of apoptotic cells by phagocytes. Cell Death Differ. 1998, 5, 551–562. [Google Scholar] [CrossRef]
- McDonald, P.P.; Fadok, V.A.; Bratton, D.; Henson, P.M. Transcriptional and translational regulation of inflammatory mediator production by endogenous TGF-beta in macrophages that have ingested apoptotic cells. J. Immunol. 1999, 163, 6164–6172. [Google Scholar]
- Zhang, M.; Hutter, G.; Kahn, S.A.; Azad, T.D.; Gholamin, S.; Xu, C.Y.; Liu, J.; Achrol, A.S.; Richard, C.; Sommerkamp, P.; et al. Anti-CD47 Treatment Stimulates Phagocytosis of Glioblastoma by M1 and M2 Polarized Macrophages and Promotes M1 Polarized Macrophages In Vivo. PLoS ONE 2016, 11, e0153550. [Google Scholar] [CrossRef]
- Moslehi-Jenabian, S.; Pedersen, L.L.; Jespersen, L. Beneficial effects of probiotic and food borne yeasts on human health. Nutrients 2010, 2, 449–473. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Singh, A.; Kumar, P.; Chaudhari, A.; Nareshkumar, G. Probiotic Escherichia coli CFR 16 producing pyrroloquinoline quinone (PQQ) ameliorates 1,2-dimethylhydrazine-induced oxidative damage in colon and liver of rats. Appl. Biochem. Biotechnol. 2014, 173, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Cui, G.; Goll, R.; Olsen, T.; Steigen, S.E.; Husebekk, A.; Vonen, B.; Florholmen, J. Reduced expression of microenvironmental Th1 cytokines accompanies adenomas-carcinomas sequence of colorectum. Cancer Immunol. Immunother. 2007, 56, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.H.; Vogel, P.; Body-Malapel, M.; Lamkanfi, M.; Kanneganti, T.D. IL-18 production downstream of the Nlrp3 inflammasome confers protection against colorectal tumor formation. J. Immunol. 2010, 185, 4912–4920. [Google Scholar] [CrossRef] [PubMed]
- Senju, H.; Kumagai, A.; Nakamura, Y.; Yamaguchi, H.; Nakatomi, K.; Fukami, S.; Shiraishi, K.; Harada, Y.; Nakamura, M.; Okamura, H.; et al. Effect of IL-18 on the Expansion and Phenotype of Human Natural Killer Cells: Application to Cancer Immunotherapy. Int. J. Biol. Sci. 2018, 14, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Harrison, O.J.; Srinivasan, N.; Pott, J.; Schiering, C.; Krausgruber, T.; Ilott, N.E.; Maloy, K.J. Epithelial-derived IL-18 regulates Th17 cell differentiation and Foxp3⁺ Treg cell function in the intestine. Mucosal Immunol. 2015, 8, 1226–1236. [Google Scholar] [CrossRef]
- Bailly, Y.; Duprat, P. Normal Blood Cell Values, Rat. In Hemapoietic System; Jones, T.C., Ward, J.M., Mohr, U., Hunt, R.D., Eds.; Springer: Berlin/Heidelberg, Germany, 1990; pp. 27–38. [Google Scholar]
- Valiathan, R.; Ashman, M.; Asthana, D. Effects of Ageing on the Immune System: Infants to Elderly. Scand. J. Immunol. 2016, 83, 255–266. [Google Scholar] [CrossRef]
- Evans, C.F.; Galustian, C.; Bodman-Smith, M.; Dalgleish, A.G.; Kumar, D. The effect of colorectal cancer upon host peripheral immune cell function. Colorectal Dis. 2010, 12, 561–569. [Google Scholar] [CrossRef]
- Osterlund, P.; Ruotsalainen, T.; Korpela, R.; Saxelin, M.; Ollus, A.; Valta, P.; Kouri, M.; Elomaa, I.; Joensuu, H. Lactobacillus supplementation for diarrhoea related to chemotherapy of colorectal cancer: A randomised study. Br. J. Cancer 2007, 97, 1028–1034. [Google Scholar] [CrossRef]
- Jacouton, E.; Chain, F.; Sokol, H.; Langella, P.; Bermúdez-Humarán, L.G. Probiotic Strain. Front. Immunol. 2017, 8, 1553. [Google Scholar] [CrossRef]
| Name. | Accession Number | Code | Origin |
|---|---|---|---|
| Lactobacillus plantarum VD23 | NCIMB 41608 | VD23 | isolated from the faeces of pigs [29] |
| Lactobacillus plantarum C28 | NCIMB 41605 | C28 | isolated from the faeces of pigs [29] |
| Lactobacillus plantarum MS18 | NCIMB 41607 | MS18 | isolated from a healthy chicken small intestine [30] |
| Lactobacillus salivarius MS3 | NCIMB 41606 | MS3 | isolated from a healthy chicken small intestine [30] |
| Lactobacillus salivarius MS6 | NCIMB 41609 | MS6 | isolated from a healthy chicken small intestine [30] |
| Lactobacillus salivarius MS16 | NCIMB 41610 | MS16 | isolated from a healthy chicken small intestine [30] |
| Groups | Body-Weight Gain (g) | Food Intake (g/rat/day) | FER (%) 1 | Gut Length (cm) |
|---|---|---|---|---|
| DMH | 378.6 ± 13.7 | 22.5 ± 0.08 A | 7.67 ± 0.23 a | 18.58 ± 1.04 a |
| DMH+P | 350.3 ± 11.1 | 21.5 ± 0.08 B | 8.09 ± 0.25 ab | 21.3 ± 0.6 b |
| P | 400.5 ± 16 | 23.3 ± 0.07 C | 8.68 ± 0.31 b | 21.5 ± 0.52 b |
| INT | 404.6 ± 17.7 | 24.2 ± 0.09 D | 7.78 ± 0.21 a | 21.2 ± 0.52 b |
| Groups | Tumour Incidence | Tumour Multiplicity 1 | Total Number of Tumours | Numbers of Tumours in Proximal Colon | Numbers of Tumours in Distal Colon | Average Tumour Volume (cm3) 1 |
|---|---|---|---|---|---|---|
| DMH | 4/8 (50%) | 2.25 ± 1.25 | 9 | 7 | 2 | 2.42 ± 0.56 |
| DMH+P | 4/8 (50%) | 1.5 ± 0.29 | 6 | 5 | 1 | 1.66 ± 0.64 |
| P | 0/8 (0%) | 0 | 0 | 0 | 0 | 0 |
| INT | 0/8 (0%) | 0 | 0 | 0 | 0 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hradicka, P.; Beal, J.; Kassayova, M.; Foey, A.; Demeckova, V. A Novel Lactic Acid Bacteria Mixture: Macrophage-Targeted Prophylactic Intervention in Colorectal Cancer Management. Microorganisms 2020, 8, 387. https://doi.org/10.3390/microorganisms8030387
Hradicka P, Beal J, Kassayova M, Foey A, Demeckova V. A Novel Lactic Acid Bacteria Mixture: Macrophage-Targeted Prophylactic Intervention in Colorectal Cancer Management. Microorganisms. 2020; 8(3):387. https://doi.org/10.3390/microorganisms8030387
Chicago/Turabian StyleHradicka, Petra, Jane Beal, Monika Kassayova, Andrew Foey, and Vlasta Demeckova. 2020. "A Novel Lactic Acid Bacteria Mixture: Macrophage-Targeted Prophylactic Intervention in Colorectal Cancer Management" Microorganisms 8, no. 3: 387. https://doi.org/10.3390/microorganisms8030387
APA StyleHradicka, P., Beal, J., Kassayova, M., Foey, A., & Demeckova, V. (2020). A Novel Lactic Acid Bacteria Mixture: Macrophage-Targeted Prophylactic Intervention in Colorectal Cancer Management. Microorganisms, 8(3), 387. https://doi.org/10.3390/microorganisms8030387







