Pollution Gradients Altered the Bacterial Community Composition and Stochastic Process of Rural Polluted Ponds
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Sites and Water Quality
2.2. Sampling and Physical-Chemical Analysis
2.3. DNA Extraction, PCR Amplification and High-Throughput Sequencing
2.4. Statistical Analysis
2.5. Community Assembly Processes in Three Groups of Ponds
3. Results
3.1. Physiochemical Properties of Water and Sediment
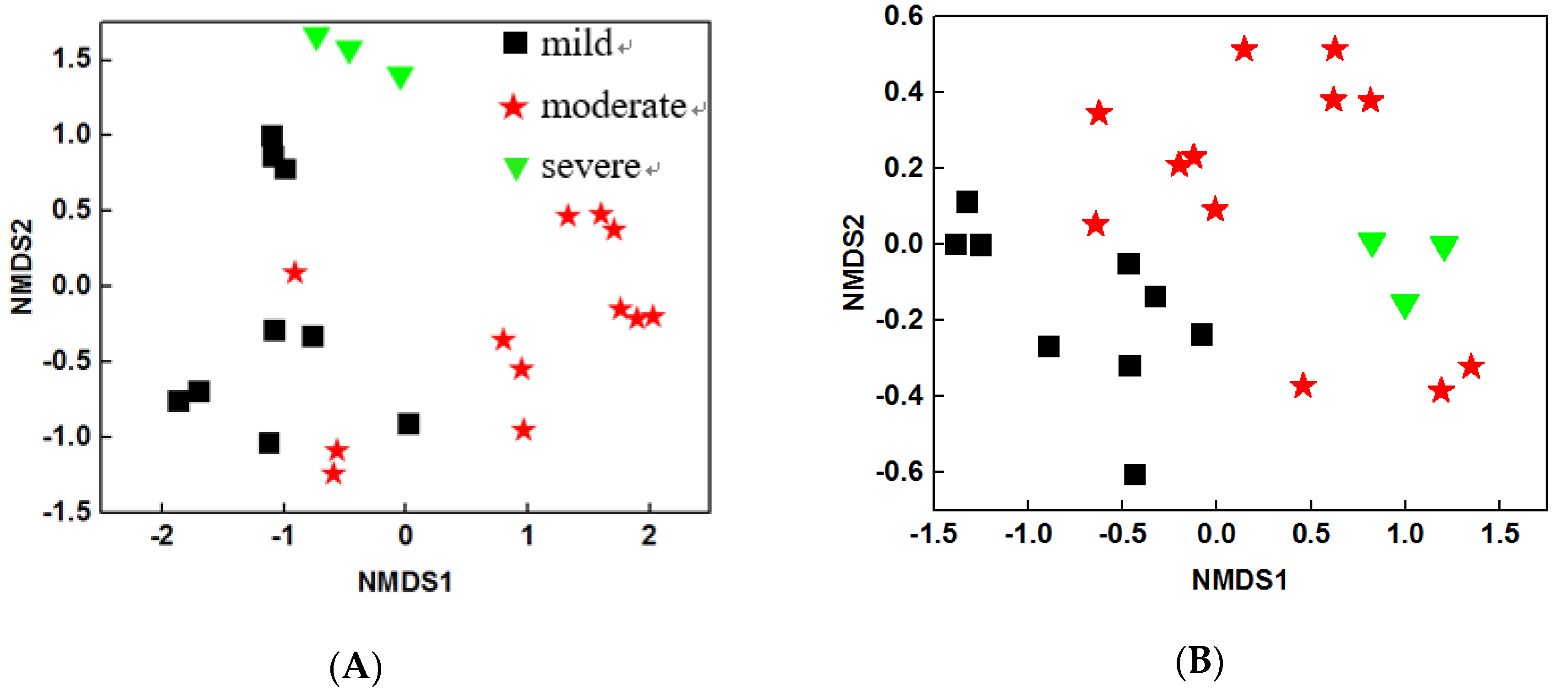
3.2. Bacterial Community Structure of Three Group Ponds
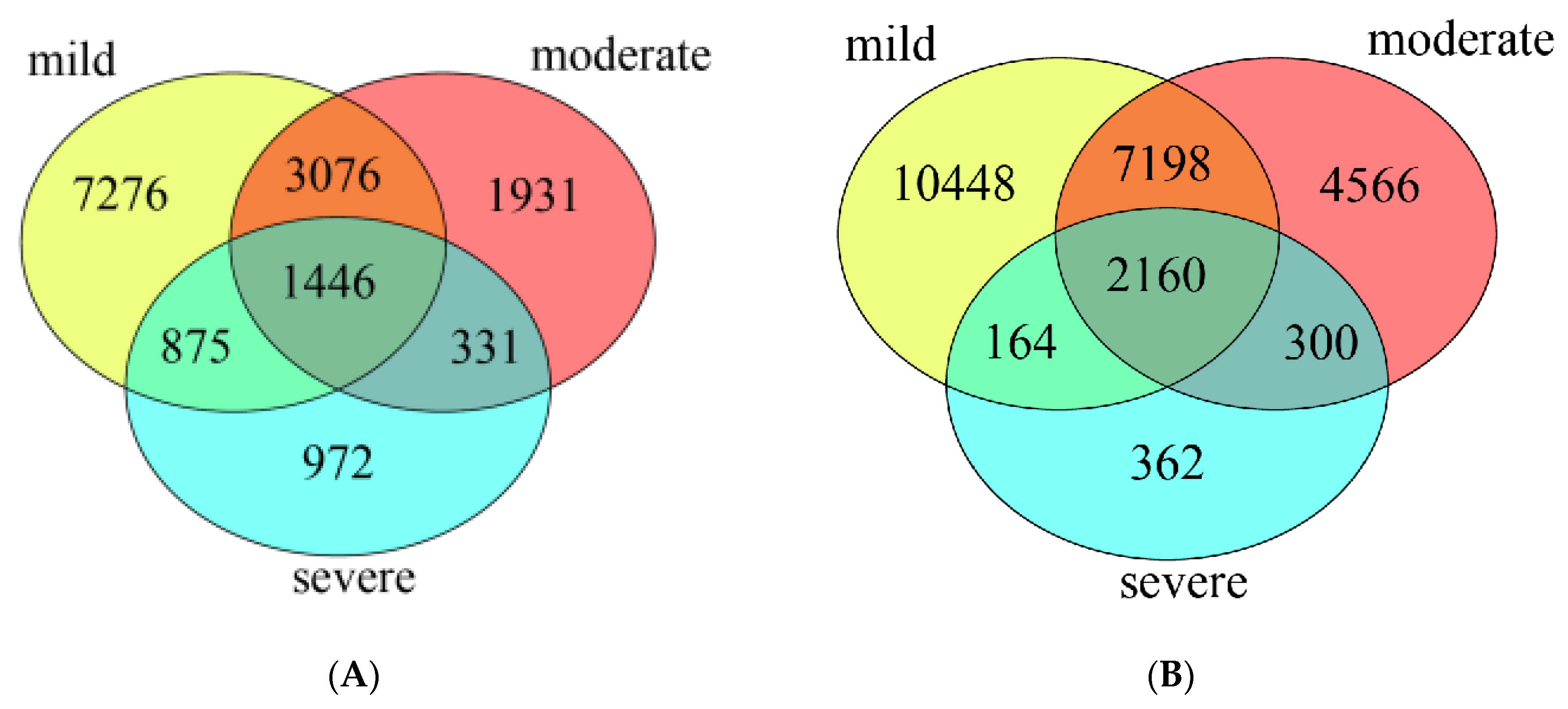
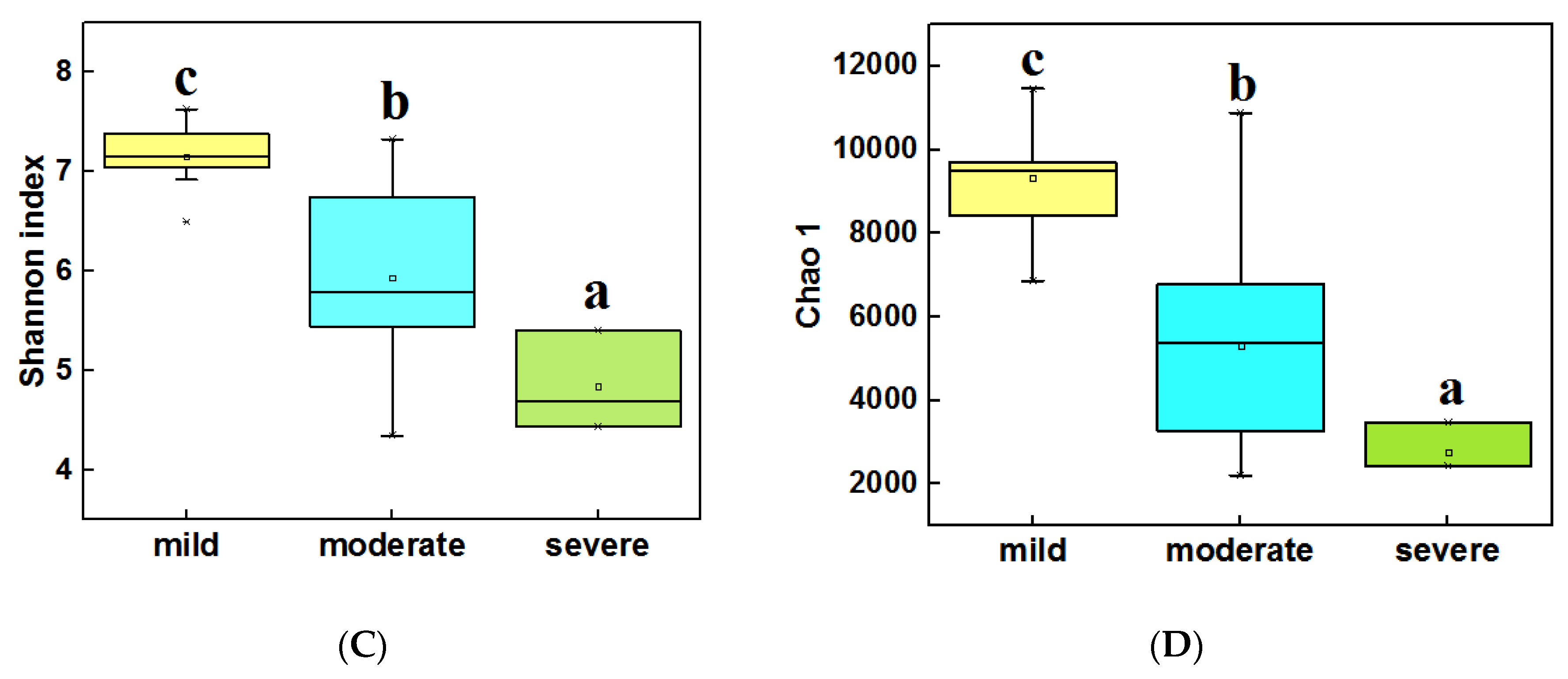
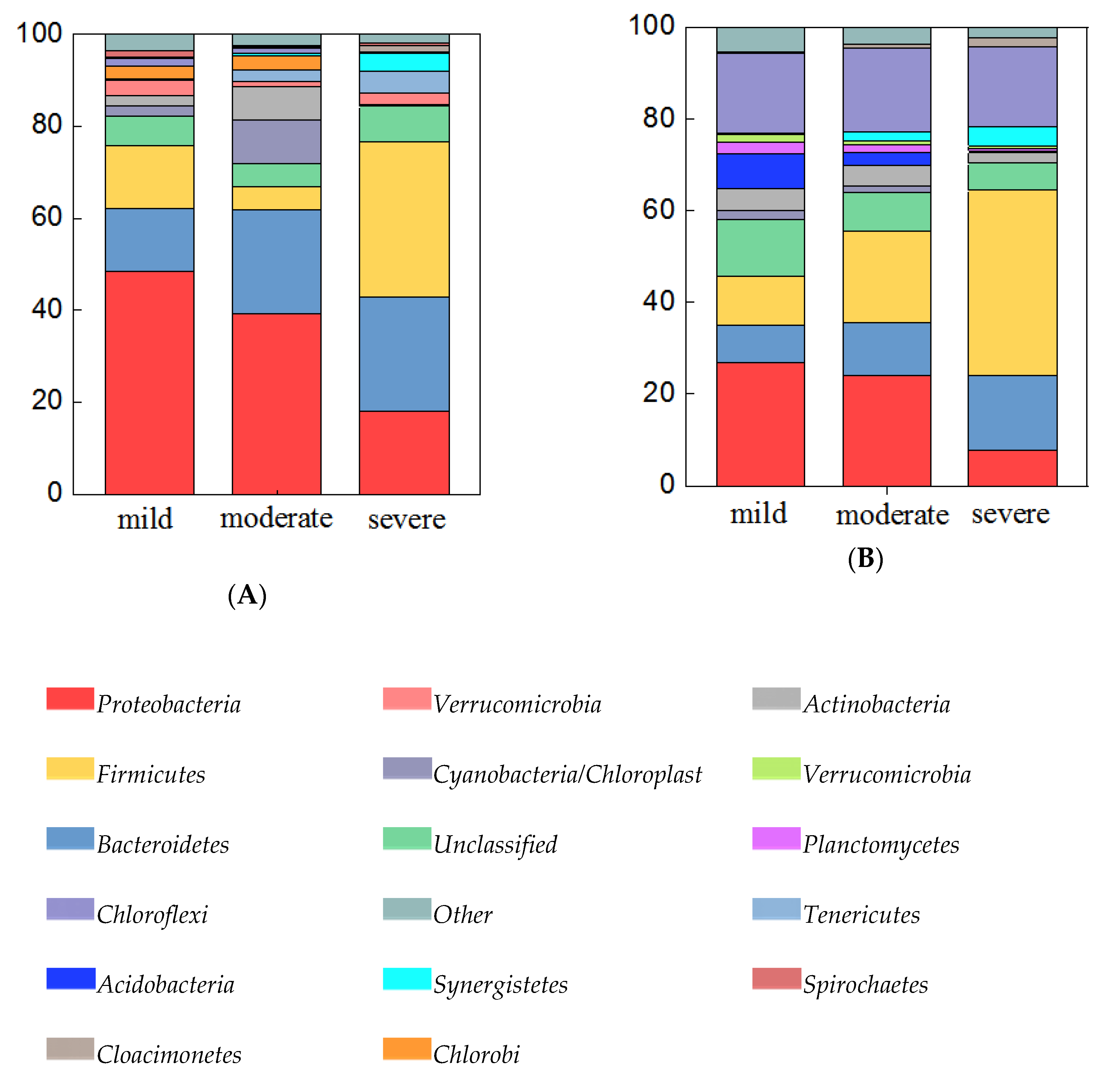
3.3. Diversity and Composition of Bacterial Community from Three Groups of Ponds
3.4. Effects of Environmental Variables on Bacterial Community
3.5. Ecological Processes in the Community Assembly
4. Discussion
4.1. The Alpha Diversity Was Significantly Different for Different Pollution Levels of Ponds
4.2. Beta Diversity of Bacterial Were Significantly Changed under Different Pollution Levels of Ponds
4.3. Environmental Variations Play a Crucial Role in Community Assembly Processes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sobek, S.; Algesten, G.; BERGSTRÖM, A.K.; Jansson, M.; Tranvik, L.J. The catchment and climate regulation of pCO2 in boreal lakes. Glob. Chang. Biol. 2003, 9, 630–641. [Google Scholar] [CrossRef]
- Downing, J.A.; Prairie, Y.; Cole, J.; Duarte, C.; Tranvik, L.; Striegl, R.G.; McDowell, W.; Kortelainen, P.; Caraco, N.; Melack, J. The global abundance and size distribution of lakes, ponds, and impoundments. Limnol. Oceanogr. 2006, 51, 2388–2397. [Google Scholar] [CrossRef]
- Williams, P.; Biggs, J.; Crowe, A.; Murphy, J.; Nicolet, P.; Weatherby, A.; Dunbar, M. Countryside survey: Ponds report from 2007. Available online: http://nora.nerc.ac.uk/id/eprint/9622/ (accessed on 27 January 2020).
- Céréghino, R.; Boix, D.; Cauchie, H.-M.; Martens, K.; Oertli, B. The ecological role of ponds in a changing world. Hydrobiologia 2014, 723, 1–6. [Google Scholar] [CrossRef]
- Hill, M.J.; Hassall, C.; Oertli, B.; Fahrig, L.; Robson, B.J.; Biggs, J.; Samways, M.J.; Usio, N.; Takamura, N.; Krishnaswamy, J. New policy directions for global pond conservation. Conserv. Lett. 2018, 11, e12447. [Google Scholar] [CrossRef]
- Takamura, N. Status of biodiversity loss in lakes and ponds in Japan. In The Biodiversity Observation Network in the Asia-Pacific Region; Springer: New York City, NY, USA, 2012; pp. 133–148. [Google Scholar]
- Cameron, K.A.; Hodson, A.J.; Osborn, A.M. Carbon and nitrogen biogeochemical cycling potentials of supraglacial cryoconite communities. Polar Biol. 2012, 35, 1375–1393. [Google Scholar] [CrossRef]
- Peterson, H.G. Rural drinking water and waterborne illness. Available online: https://pdfs.semanticscholar.org/2c8a/5eba02b91b2db00c193ad389951c3bd7eda5.pdf (accessed on 27 January 2020).
- Li, Y.; Wu, H.N.; Shen, Y.; Wang, C.; Wang, P.F.; Zhang, W.L.; Gao, Y.; Niu, L.H. Statistical determination of crucial taxa indicative of pollution gradients in sediments of Lake Taihu, China. Environ. Pollut. 2019, 246, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.B.; Liu, Y.Q.; Lin, X.G.; Zhang, H.Y.; Zeng, J.; Hou, J.Z.; Yang, Y.P.; Yao, T.D.; Knight, R.; Chu, H.Y. Geographic distance and pH drive bacterial distribution in alkaline lake sediments across Tibetan Plateau. Environ. Microbiol. 2012, 14, 2457–2466. [Google Scholar] [CrossRef]
- Sun, M.Y.; Dafforn, K.A.; Johnston, E.L.; Brown, M.V. Core sediment bacteria drive community response to anthropogenic contamination over multiple environmental gradients. Environ. Microbiol. 2013, 15, 2517–2531. [Google Scholar] [CrossRef]
- Gladyshev, M.I.; Kolmakova, O.V.; Tolomeev, A.P.; Anishchenko, O.V.; Makhutova, O.N.; Kolmakova, A.A.; Kravchuk, E.S.; Glushchenko, L.A.; Kolmakov, V.I.; Sushchik, N.N. Differences in organic matter and bacterioplankton between sections of the largest A rctic river: Mosaic or continuum? Limnol. Oceanogr. 2015, 60, 1314–1331. [Google Scholar] [CrossRef]
- Dai, J.Y.; Tang, X.M.; Gao, G.; Chen, D.; Shao, K.Q.; Cai, X.L.; Zhang, L. Effects of salinity and nutrients on sedimentary bacterial communities in oligosaline Lake Bosten, northwestern China. Aquat. Microb. Ecol. 2013, 69, 123–134. [Google Scholar] [CrossRef]
- Faris, K.P.; Ramsey, P.W.; Gibbons, S.M.; Frazar, C.; Rillig, M.C.; Moore, J.N.; Gannon, J.E.; Holben, W.E. Hyporheic Microbial Community Development Is a Sensitive Indicator of Metal Contamination. Environ. Sci. Technol. 2009, 43, 6158–6163. [Google Scholar] [CrossRef] [PubMed]
- Epelde, L.; Becerril, J.M.; Barrutia, O.; Gonzalez-Oreja, J.A.; Garbisu, C. Interactions between plant and rhizosphere microbial communities in a metalliferous soil. Environ. Pollut. 2010, 158, 1576–1583. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yao, J.; Si, Y.; Chen, H.; Russel, M.; Chen, K.; Qian, Y.; Zaray, G.; Bramanti, E. Short-time effect of heavy metals upon microbial community activity. J. Hazard. Mater. 2010, 173, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Wu, L.; Li, N.; Luo, Y.; Liu, L.; Zhao, Q.; Zhang, L.; Christie, P. Effects of multiple heavy metal contamination and repeated phytoextraction by Sedum plumbizincicola on soil microbial properties. Eur. J. Soil Biol. 2010, 46, 18–26. [Google Scholar] [CrossRef]
- Yin, H.; Niu, J.; Ren, Y.; Cong, J.; Zhang, X.; Fan, F.; Xiao, Y.; Zhang, X.; Deng, J.; Xie, M. An integrated insight into the response of sedimentary microbial communities to heavy metal contamination. Sci. Rep. 2015, 5, 14266. [Google Scholar] [CrossRef]
- Bier, R.L.; Voss, K.A.; Bernhardt, E.S. Bacterial community responses to a gradient of alkaline mountaintop mine drainage in Central Appalachian streams. ISME J. 2015, 9, 1378. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, X.; Zhou, J.; Li, G. Long-term oil contamination increases deterministic assembly processes in soil microbes. Ecol. Appl. 2015, 25, 1235–1243. [Google Scholar] [CrossRef]
- Kumari, P.; Choi, H.L.; Sudiarto, S.I.A. Assessment of Bacterial Community Assembly Patterns and Processes in Pig Manure Slurry. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Chave, J. Neutral theory and community ecology. Ecol. Lett. 2004, 7, 241–253. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Deng, Y.; Feng, K.; Cai, W.W.; Li, S.Z.; Yin, H.Q.; Xu, M.Y.; Ning, D.L.; Qu, Y.Y. Deterministic Assembly and Diversity Gradient Altered the Biofilm Community Performances of Bioreactors. Environ. Sci. Technol. 2019, 53, 1315–1324. [Google Scholar] [CrossRef]
- Vellend, M. Conceptual Synthesis in Community Ecology. Q. Rev. Biol. 2010, 85, 183–206. [Google Scholar] [CrossRef] [PubMed]
- Hubbell, S.P. The unified neutral theory of biodiversity and biogeography (MPB-32); Princeton University Press: Princeton, NJ, USA, 2001. [Google Scholar]
- Fargione, J.; Brown, C.S.; Tilman, D. Community assembly and invasion: An experimental test of neutral versus niche processes. Proc. Natl. Acad. Sci. USA 2003, 100, 8916–8920. [Google Scholar] [CrossRef] [PubMed]
- Adler, P.B.; HilleRisLambers, J.; Levine, J.M. A niche for neutrality. Ecol. Lett. 2007, 10, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Gravel, D.; Canham, C.D.; Beaudet, M.; Messier, C. Reconciling niche and neutrality: The continuum hypothesis. Ecol. Lett. 2006, 9, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Chase, J.M. Drought mediates the importance of stochastic community assembly. Proc. Natl. Acad. Sci. USA 2007, 104, 17430–17434. [Google Scholar] [CrossRef]
- Kardol, P.; Souza, L.; Classen, A.T. Resource availability mediates the importance of priority effects in plant community assembly and ecosystem function. Oikos 2013, 122, 84–94. [Google Scholar] [CrossRef]
- Chase, J.M. Stochastic community assembly causes higher biodiversity in more productive environments. Science 2010, 328, 1388–1391. [Google Scholar] [CrossRef]
- Zhou, J.Z.; Deng, Y.; Zhang, P.; Xue, K.; Liang, Y.T.; Van Nostrand, J.D.; Yang, Y.F.; He, Z.L.; Wu, L.Y.; Stahl, D.A.; et al. Stochasticity, succession, and environmental perturbations in a fluidic ecosystem. Proc. Natl. Acad. Sci. USA 2014, 111, E836–E845. [Google Scholar] [CrossRef]
- Langenheder, S.; Szekely, A.J. Species sorting and neutral processes are both important during the initial assembly of bacterial communities. ISME J. 2011, 5, 1086–1094. [Google Scholar] [CrossRef]
- Guo, X.; Feng, J.J.; Shi, Z.; Zhou, X.S.; Yuan, M.T.; Tao, X.Y.; Hale, L.; Yuan, T.; Wang, J.J.; Qin, Y.J.; et al. Climate warming leads to divergent succession of grassland microbial communities. Nat. Clim. Chang. 2018, 8. [Google Scholar] [CrossRef]
- Liu, L.; Chen, H.; Liu, M.; Yang, J.R.; Xiao, P.; Wilkinson, D.M.; Yang, J. Response of the eukaryotic plankton community to the cyanobacterial biomass cycle over 6 years in two subtropical reservoirs. ISME J. 2019, 13, 2196–2208. [Google Scholar] [CrossRef] [PubMed]
- Pei, Y.; Yu, Z.; Ji, J.; Khan, A.; Li, X. Microbial community structure and function indicate the severity of chromium contamination of the Yellow River. Front. Microbiol. 2018, 9, 38. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, P.; Wang, C.; Wang, X.; Miao, L.; Liu, S.; Yuan, Q. Bacterial communities in riparian sediments: A large-scale longitudinal distribution pattern and response to dam construction. Front. Microbiol. 2018, 9, 999. [Google Scholar] [CrossRef] [PubMed]
- TANG, T.; ZHAI, Y.; HUANG, K. Water quality analysis and Recommendations through comprehensive pollution index method. Manag. Sci. Eng. 2011, 5, 95–100. [Google Scholar]
- Kong, X.; Jin, D.; Tai, X.; Yu, H.; Duan, G.; Yan, X.; Pan, J.; Song, J.; Deng, Y. Bioremediation of dibutyl phthalate in a simulated agricultural ecosystem by Gordonia sp. strain QH-11 and the microbial ecological effects in soil. Sci. Total Environ. 2019, 667, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Feng, K.; Zhang, Z.J.; Cai, W.W.; Liu, W.Z.; Xu, M.Y.; Yin, H.Q.; Wang, A.J.; He, Z.L.; Deng, Y. Biodiversity and species competition regulate the resilience of microbial biofilm community. Mol. Ecol. 2017, 26, 6170–6182. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996. [Google Scholar] [CrossRef]
- Ning, D.; Deng, Y.; Tiedje, J.M.; Zhou, J. A general framework for quantitatively assessing ecological stochasticity. Proc. Natl. Acad. Sci. USA 2019, 116, 16892–16898. [Google Scholar] [CrossRef]
- Administration SEP. Environmental quality standards for surface water. Available online: http://down.foodmate.net/standard/sort/3/3528.html (accessed on 28 April 2002).
- C. SAotPR. Control standards of pollutants in sludge for agricultural use. Available online: http://openstd.samr.gov.cn/bzgk/gb/newGbInfo?hcno=B40A49F7734797DF61D90FE1F6BA5442 (accessed on 14 May 2018).
- Jia, S.Y.; Zhang, X.X.; Miao, Y.; Zhao, Y.T.; Ye, L.; Li, B.; Zhang, T. Fate of antibiotic resistance genes and their associations with bacterial community in livestock breeding wastewater and its receiving river water. Water Res. 2017, 124, 259–268. [Google Scholar] [CrossRef]
- Shade, A.; Peter, H.; Allison, S.D.; Baho, D.L.; Berga, M.; Burgmann, H.; Huber, D.H.; Langenheder, S.; Lennon, J.T.; Martiny, J.B.H.; et al. Fundamentals of microbial community resistance and resilience. Front. Microbiol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Mori, A.S.; Furukawa, T.; Sasaki, T. Response diversity determines the resilience of ecosystems to environmental change. Biol. Rev. 2013, 88, 349–364. [Google Scholar] [CrossRef]
- Beazley, M.J.; Martinez, R.J.; Rajan, S.; Powell, J.; Piceno, Y.M.; Tom, L.M.; Andersen, G.L.; Hazen, T.C.; Van Nostrand, J.D.; Zhou, J.Z.; et al. Microbial Community Analysis of a Coastal Salt Marsh Affected by the Deepwater Horizon Oil Spill. PLoS ONE 2012, 7, e41305. [Google Scholar] [CrossRef]
- Hazen, T.C.; Dubinsky, E.A.; DeSantis, T.Z.; Andersen, G.L.; Piceno, Y.M.; Singh, N.; Jansson, J.K.; Probst, A.; Borglin, S.E.; Fortney, J.L.; et al. Deep-Sea Oil Plume Enriches Indigenous Oil-Degrading Bacteria. Science 2010, 330, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Torsvik, V.; Sorheim, R.; Goksoyr, J. Total bacterial diversity in soil and sediment communities - A review. J. Ind. Microbiol. 1996, 17, 170–178. [Google Scholar] [CrossRef]
- Allison, S.D.; Martiny, J.B.H. Resistance, resilience, and redundancy in microbial communities. Proc. Natl. Acad. Sci. USA 2008, 105, 11512–11519. [Google Scholar] [CrossRef]
- Goni-Urriza, M.; Capdepuy, M.; Raymond, N.; Quentin, C.; Caumette, P. Impact of an urban effluent on the bacterial community structure in the Arga River (Spain), with special reference to culturable Gram-negative rods. Proc. Natl. Acad. Sci. USA 1999, 45, 826–832. [Google Scholar] [CrossRef]
- Da Silva, M.L.B.; Cantao, M.E.; Mezzari, M.P.; Ma, J.; Nossa, C.W. Assessment of Bacterial and Archaeal Community Structure in Swine Wastewater Treatment Processes. Proc. Natl. Acad. Sci. USA 2015, 70, 77–87. [Google Scholar] [CrossRef]
- Ducey, T.F.; Hunt, P.G. Microbial community analysis of swine wastewater anaerobic lagoons by next-generation DNA sequencing. Anaerobe 2013, 21, 50–57. [Google Scholar] [CrossRef]
- Isaacson, R.; Kim, H.B. The intestinal microbiome of the pig. Anim. Health Res. Rev. 2012, 13, 100–109. [Google Scholar] [CrossRef]
- Leung, K.; Topp, E. Bacterial community dynamics in liquid swine manure during storage: Molecular analysis using DGGE/PCR of 16S rDNA. Fems Microbiol. Ecol. 2001, 38, 169–177. [Google Scholar] [CrossRef][Green Version]
- Bai, Y.H.; Shi, Q.; Wen, D.H.; Li, Z.X.; Jefferson, W.A.; Feng, C.P.; Tang, X.Y. Bacterial Communities in the Sediments of Dianchi Lake, a Partitioned Eutrophic Waterbody in China. PLoS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.P.; Chen, H.Y.; Sun, J.; Zhang, X.; He, C.Q.; Liu, X.Y.; Huang, X.; Yang, M.; Wang, F.S.; Vaananen, K. Shifts in the structure and function of the microbial community in response to metal pollution of fresh water sediments in Finland. J. Soils Sediments 2018, 18, 3324–3333. [Google Scholar] [CrossRef]
- Shi, Y.-L.; Lu, X.-Z.; Yu, W.-G. A new β-agarase from marine bacterium Janthinobacterium sp. SY12. World J. Microbiol. Biotechnol. 2008, 24, 2659–2664. [Google Scholar] [CrossRef]
- Dini-Andreote, F.; Stegen, J.C.; van Elsas, J.D.; Salles, J.F. Disentangling mechanisms that mediate the balance between stochastic and deterministic processes in microbial succession. Proc. Natl. Acad. Sci. USA 2015, 112, E1326–E1332. [Google Scholar] [CrossRef]
- Martiny, A.C.; Jorgensen, T.M.; Albrechtsen, H.J.; Arvin, E.; Molin, S. Long-term succession of structure and diversity of a biofilm formed in a model drinking water distribution system. Appl. Environ. Microbiol. 2003, 69, 6899–6907. [Google Scholar] [CrossRef]


| Total Area (m2) | Water Area (m2) | Water Depth (m) | Water Capacity (m3) | Sediment Depth (m) | Sediment Volume (m3) | |
|---|---|---|---|---|---|---|
| 1# | 11,291.9 | 4848.3 | 1.74 | 8436.1 | 1.8 | 8727 |
| 2# | 6517 | 4784.3 | 1.272 | 6085.7 | 2.4 | 11,482.4 |
| 3# | 17,021.2 | 9213.2 | 1.08 | 9950.3 | 1.6 | 14,741.2 |
| 4# | 898.4 | 409.7 | 1.2 | 491.7 | 1.8 | 157.7 |
| 5# | 1197.6 | 857.1 | 2.64 | 2262.8 | 2.4 | 2057.4 |
| 6# | 3562.9 | 2358.2 | 0.95 | 2240.3 | 1.2 | 2829.9 |
| 7# | 1628.8 | 1044.4 | 1.8 | 1880 | 1.56 | 1629.3 |
| 8# | 2547.7 | 1539.4 | 6.28 | 9667.5 | 2.4 | 3694.6 |
| Mild | Moderate | Severe | Standard (mg/L)a | |
|---|---|---|---|---|
| pH | 7.10 ± 0.06A | 8.16 ± 0.18B | 7.69 ± 0.18AB | 6~9 |
| NH3-N | 3.39 ± 0.81A | 41.79 ± 8.09B | 451.33 ± 27.53C | 2.0 |
| TP | 9.64 ± 2.52A | 38.63 ± 7.47B | 72.23 ± 7.61C | 0.4 |
| COD | 634.78 ± 264.52A | 422.25 ± 72.57A | 1186.67 ± 455.81A | 40 |
| TN | 76.04 ± 34.00A | 97.11 ± 13.06A | 687.67 ± 142.54B | 2.0 |
| comprehensive pollution index range | 11.63–24.87 | 33.00–37.95 | 156.12 |
| Unit (mg/L) | Mild | Moderate | Severe |
|---|---|---|---|
| B (—)b | 35.84 ± 3.91A | 29.32 ± 2.06A | 34.87 ± 1.57A |
| Cr (500) | 27.35 ± 1.89AB | 21.00 ± 1.37A | 30.80 ± 0.60B |
| Ni (100) | 22.31 ± 1.70AB | 18.66 ± 1.25A | 25.93 ± 0.91B |
| Cu (500) | 29.47 ± 3.90A | 96.75 ± 25.14A | 70.82 ± 6.97A |
| Zn (1200) | 86.79 ± 7.49A | 204.81 ± 25.96B | 213.70 ± 16.63B |
| As (30) | 7.04 ± 0.65A | 6.65 ± 1.02A | 9.39 ± 1.01A |
| Cd (3) | 0.37 ± 0.06A | 0.31 ± 0.02A | 0.24 ± 0.02A |
| Pb (300) | 19.62 ± 1.02A | 27.91 ± 7.98A | 33.77 ± 12.22A |
| Hg (3) | 2.74 ± 1.35A | 0.83 ± 0.16A | 0.82 ± 0.1A |
| TP | 812.44 ± 25.63A | 956.67 ± 69.7A | 751.33 ± 72.84A |
| TN | 1876.22 ± 73AB | 2163.83 ± 192.08B | 1415.67 ± 85.93A |
| NH3-N | 93.52 ± 4.14A | 100.30 ± 7.27A | 80.59 ± 2.81A |
| pH (5.5–8.5) | 7.93 ± 0.13A | 8.24 ± 0.1A | 8.01 ± 0.33A |
| OM (≥20) | 7522.67 ± 599.74AB | 13950.25 ± 2084.48B | 6273.00 ± 403.35A |
| Groups | Bray Curtis | Jaccard | ||||||
|---|---|---|---|---|---|---|---|---|
| MRPP | PERMANOVA | MRPP | PERMANOVA | |||||
| water samples | δ | P | F | P | δ | P | F | P |
| mild vs. moderate | 0.7651 | 0.003** | 2.9581 | 0.005** | 0.8013 | 0.001*** | 3.2270 | 0.001*** |
| mild vs. severe | 0.6388 | 0.005** | 4.3320 | 0.006** | 0.7479 | 0.003** | 2.6988 | 0.004** |
| moderate vs. severe | 0.7176 | 0.011* | 3.6158 | 0.005** | 0.7625 | 0.001*** | 3.0440 | 0.005** |
| sediment samples | ||||||||
| mild vs. moderate | 0.6633 | 0.001*** | 2.9240 | 0.007** | 0.7718 | 0.001*** | 2.6000 | 0.001*** |
| mild vs. severe | 0.5908 | 0.005** | 4.2474 | 0.012** | 0.7302 | 0.003** | 3.2429 | 0.008** |
| moderate vs. severe | 0.6077 | 0.025* | 1.6452 | 0.092 | 0.7652 | 0.015* | 1.6279 | 0.035* |
| Groups | NST (Null Model Based on Taxonomic) | Null Model Based on Phylogenetic | |||||
|---|---|---|---|---|---|---|---|
| Group | Stochasticity Process (%) | Mean of Observed Similarity | Mean of Null Expected Similarity | Stochasticity Process (%) | F | Pc | |
| Water | mild | 0.634 | 0.147 | 0.057 | 0.391 | 25.567 | <0.001 |
| moderate | 0.403 | 0.125 | 0.032 | 0.257 | 2.846 | 0.106 | |
| severe | 0.214 | 0.352 | 0.057 | 0.163 | 0.469 | 0.531 | |
| Sediment | mild | 0.717 | 0.148 | 0.097 | 0.656 | 79.074 | <0.001 |
| moderate | 0.540 | 0.172 | 0.060 | 0.349 | 4.613 | 0.043 | |
| severe | 0.150 | 0.371 | 0.038 | 0.103 | 4.617 | 0.098 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tai, X.; Li, R.; Zhang, B.; Yu, H.; Kong, X.; Bai, Z.; Deng, Y.; Jia, L.; Jin, D. Pollution Gradients Altered the Bacterial Community Composition and Stochastic Process of Rural Polluted Ponds. Microorganisms 2020, 8, 311. https://doi.org/10.3390/microorganisms8020311
Tai X, Li R, Zhang B, Yu H, Kong X, Bai Z, Deng Y, Jia L, Jin D. Pollution Gradients Altered the Bacterial Community Composition and Stochastic Process of Rural Polluted Ponds. Microorganisms. 2020; 8(2):311. https://doi.org/10.3390/microorganisms8020311
Chicago/Turabian StyleTai, Xin, Rui Li, Bao Zhang, Hao Yu, Xiao Kong, Zhihui Bai, Ye Deng, Lan Jia, and Decai Jin. 2020. "Pollution Gradients Altered the Bacterial Community Composition and Stochastic Process of Rural Polluted Ponds" Microorganisms 8, no. 2: 311. https://doi.org/10.3390/microorganisms8020311
APA StyleTai, X., Li, R., Zhang, B., Yu, H., Kong, X., Bai, Z., Deng, Y., Jia, L., & Jin, D. (2020). Pollution Gradients Altered the Bacterial Community Composition and Stochastic Process of Rural Polluted Ponds. Microorganisms, 8(2), 311. https://doi.org/10.3390/microorganisms8020311






