Use of Lactic Acid Bacteria During Pikeperch (Sander lucioperca) Larval Rearing
Abstract
1. Introduction
Objective
2. Materials and Methods
2.1. Salinity Stress Challenge
2.2. Statistical Analysis
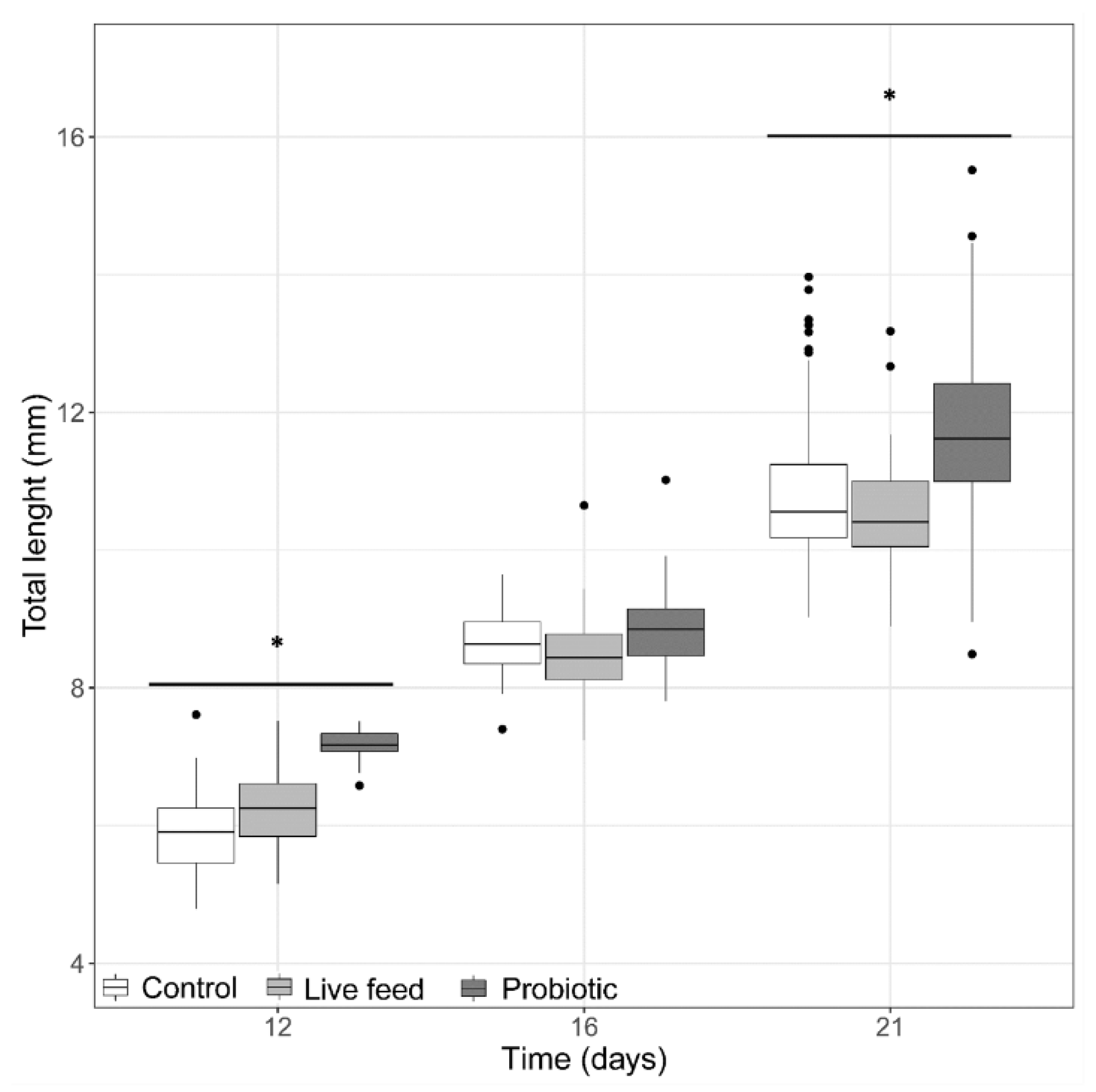
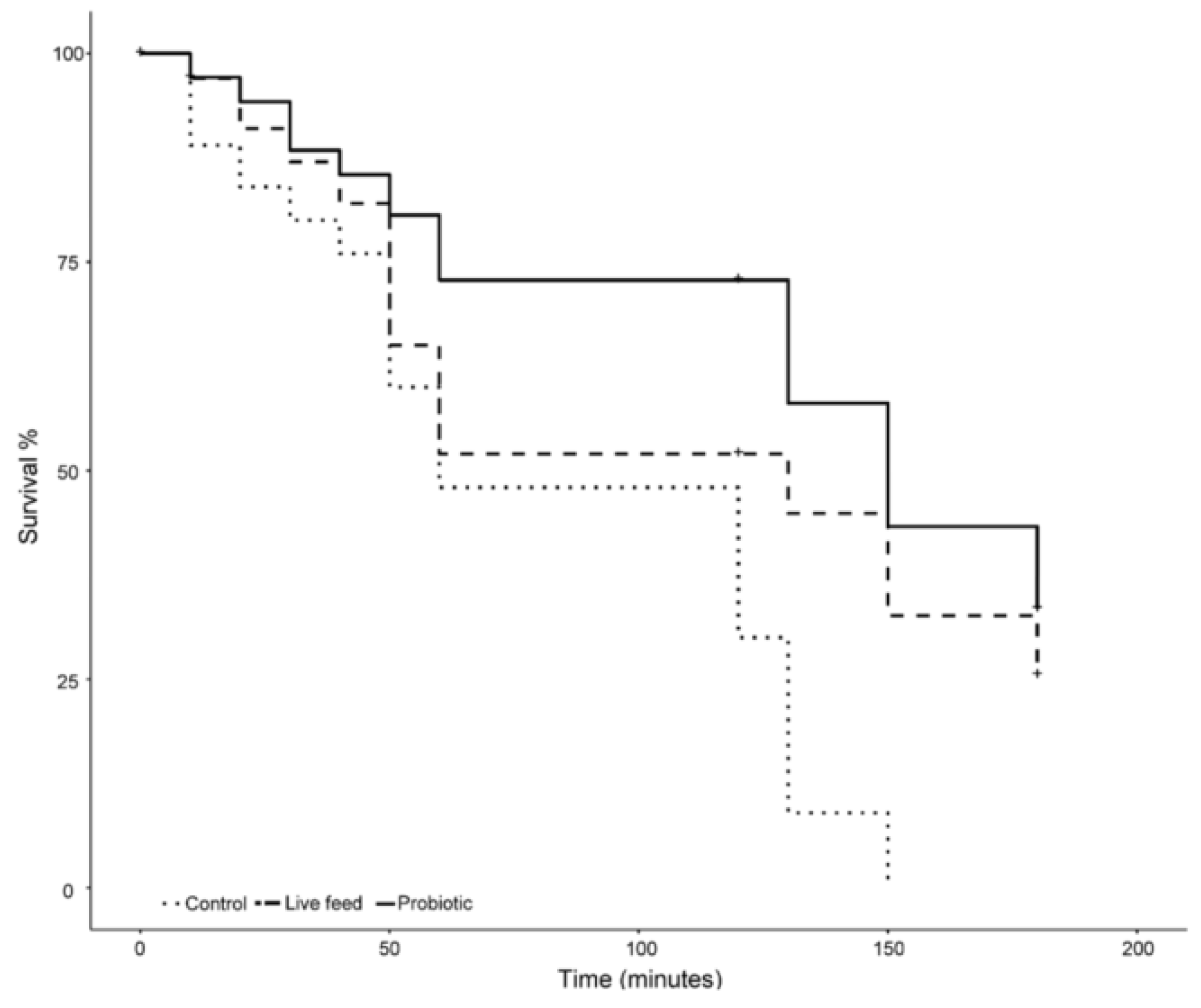
3. Results
3.1. Larval Growth
3.2. Survival
3.3. Salinity Stress Tolerance
4. Discussion
5. Conclusions
Funding
Acknowledgements
Conflicts of Interest
Abbreviations
| dph | days post hatch |
| RAS | Recirculation aquaculture systems |
| TL | Total length |
| BW | Body weight |
| MH | Myomere height |
| ED | Eye diameter |
| SF | Stomach fullness |
| FFPW | Faculty of Fisheries and Protection of Waters |
| USB | University of South Bohemia |
| LMM | Linear mixed model |
| GLMM | Generalized linear mixed models |
| LAB | Lactic acid bacteria |
References
- Kestemont, P.; Dabrowski, K.; Summerfelt, R.C. Biology and Culture of Percid Fishes: Principles and Practices; Springer: Dordrecht, The Netherlands, 2015. [Google Scholar]
- Policar, T.; Schaefer, F.J.; Panana, E.; Meyer, S.; Teerlinck, S.; Toner, D.; Żarski, D. Recent progress in European percid fish culture production technology—Tackling bottlenecks. In Aquaculture International; Springer International Publishing: New York, NY, USA, 2019; Volume 27, pp. 1151–1174. [Google Scholar]
- Steenfeldt, S.; Fontaine, P.; Overton, J.L.; Policar, T.; Toner, D.; Falahatkar, B.; Horváth, Á.; Khemis, I.B.; Hamza, N. Mohammed Mhetli Current Status of Eurasian Percid Fishes Aquaculture. In Biology and Culture of Percid Fishes; Springer: Dordrecht, The Netherlands, 2015; pp. 817–841. [Google Scholar]
- Yanes-Roca, C.; Mráz, J.; Born-Torrijos, A.; Holzer, A.S.; Imentai, A.; Policar, T. Introduction of rotifers (Brachionus plicatilis) during pikeperch first feeding. Aquaculture 2018, 497, 260–268. [Google Scholar] [CrossRef]
- Imentai, A.; Yanes-Roca, C.; Malinovskyi, O.; Policar, T. Effect of Brachionus plicatilis density on pikeperch (Sander lucioperca L.) larva performance at first feeding. J. Appl. Ichthyol. 2019, 35, 1292–1294. [Google Scholar] [CrossRef]
- Imentai, A.; Yanes-Roca, C.; Steinbach, C.; Policar, T. Optimized application of rotifers Brachionus plicatilis for rearing pikeperch Sander lucioperca L. larvae. Aquac. Int. 2019, 27, 1137–1149. [Google Scholar] [CrossRef]
- Lubzens, E.; Tandler, A.; Minkoff, G. Rotifers as food in aquaculture. Hydrobiologia 1989, 186–187, 387–400. [Google Scholar] [CrossRef]
- Kestemont, P.; Henrotte, E. Nutritional Requirements and Feeding of Broodstock and Early Life Stages of Eurasian Perch and Pikeperch. In Biology and Culture of Percid Fishes; Springer: Dordrecht, The Netherlands, 2015; pp. 539–564. [Google Scholar]
- Lund, I.; el Kertaoui, N.; Izquierdo, M.S.; Dominguez, D.; Hansen, B.W.; Kestemont, P. The importance of phospholipids combined with long-chain PUFA in formulated diets for pikeperch (Sander lucioperca) larvae. Br. J. Nutr. 2018, 120, 628–644. [Google Scholar] [CrossRef]
- Lund, I.; Vilhelm, P.; Winding, B. Comparative Biochemistry and Physiology, Part A Dietary supplementation of essential fatty acids in larval pikeperch (Sander lucioperca); short and long term effects on stress tolerance and metabolic physiology. Comp. Biochem. Physiol. Part A 2012, 162, 340–348. [Google Scholar] [CrossRef]
- Villamil, L.; Figueras, A.; Planas, M.; Novoa, B. Control of Vibrio alginolyticus in Artemia culture by treatment with bacterial probiotics. Aquaculture 2003, 219, 43–56. [Google Scholar] [CrossRef]
- Fuller, R. Probiotics in man and animals. J. Appl. Bacteriol. 1989, 66, 365–378. [Google Scholar] [PubMed]
- Gatesoupe, F. The use of probiotics in aquaculture. Aquaculture 1999, 180, 147–165. [Google Scholar] [CrossRef]
- Vanbelle, M.; Teller, E.; Focant, M. Probiotics in animal nutrition: A review. Arch. für Tierernährung 1990, 40, 543–567. [Google Scholar] [CrossRef] [PubMed]
- Hai, N.V. The use of probiotics in aquaculture. J. Appl. Microbiol. 2015, 119, 917–935. [Google Scholar] [CrossRef] [PubMed]
- Balcázar, J.L.; de Blas, I.; Ruiz-Zarzuela, I.; Cunningham, D.; Vendrell, D.; Múzquiz, J.L. The role of probiotics in aquaculture. Vet. Microbiol. 2006, 114, 173–186. [Google Scholar] [CrossRef] [PubMed]
- Ljubobratovic, U.; Kosanovic, D.; Vukotic, G.; Molnar, Z.; Stanisavljevic, N.; Ristovic, T.; Peter, G.; Lukic, J.; Jeney, G. Supplementation of lactobacilli improves growth, regulates microbiota composition and suppresses skeletal anomalies in juvenile pike-perch (Sander lucioperca) reared in recirculating aquaculture system (RAS): A pilot study. Res. Vet. Sci. 2017, 115, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Ljubobratovic, U.; Kosanovic, D.; Demény, F.Z.; Krajcsovics, A.; Vukotic, G.; Stanisavljevic, N.; Golic, N.; Jeney, G.; Lukic, J. The effect of live and inert feed treatment with lactobacilli on weaning success in intensively reared pike-perch larvae. Aquaculture 2019, 516, 734608. [Google Scholar] [CrossRef]
- Klein, G.; Pack, A.; Bonaparte, C.; Reuter, G. Taxonomy and physiology of probiotic lactic acid bacteria. Int. J. Food Microbiol. 1998, 41, 103–125. [Google Scholar] [CrossRef]
- Munro, P.O.; Barbour, A.; Blrkbeck, T.H. Comparison of the gut bacterial flora of start-feeding larval turbot reared under different conditions. J. Appl. Bacteriol. 1994, 77, 560–566. [Google Scholar] [CrossRef]
- Ringø, E.; Birkbeck, T.H.; Munro, P.O.; Vadstein, O.; Hjelmeland, K. The effect of early exposure to Vibrio pelagius on the aerobic bacterial flora of turbot, Scophthalmus maximus (L.) larvae. J. Appl. Bacteriol. 1996, 81, 207–211. [Google Scholar] [CrossRef]
- Ringø, E. Intestinal microflora of fish larvae and fry. Aquac. Res. 1999, 30, 73. [Google Scholar] [CrossRef]
- Ringø, E.; Hoseinifar, S.H.; Ghosh, K.; van Doan, H.; Beck, B.R.; Song, S.K. Lactic Acid Bacteria in Finfish—An Update. Front. Microbiol. 2018, 9, 1818. [Google Scholar] [CrossRef]
- Ljungh Åsa and Wadström Torkel. Lactic Acid Bacteria as Probiotics. Curr. Issues Intest. Microbiol. 2018, 7, 73–90. [Google Scholar]
- Malinovskyi, O.; Veselý, L.; Blecha, M.; Křišťan, J.; Policar, T. The substrate selection and spawning behaviour of pikeperch Sander lucioperca L. broodstock under pond conditions. Aquac. Res. 2018, 49, 3541–3547. [Google Scholar] [CrossRef]
- Malinovskyi, O.; Kolářová, J.; Blecha, M.; Stará, A.; Velíšek, J.; Křišťan, J.; Policar, T. Behavior and physiological status of pond-cultured pikeperch (Sander lucioperca) broodstock effected by sexual interactions throughout semi-artificial reproduction. Aquac. Int. 2019, 27, 1093–1107. [Google Scholar] [CrossRef]
- Blecha, M.; Kristan, J.; Samarin, A.M.; Rodina, M.; Policar, T. Quality and quantity of pikeperch (Sander lucioperca) spermatozoa after varying cold water treatments. J. Appl. Ichthyol. 2015, 31, 75–78. [Google Scholar] [CrossRef]
- Blecha, M.; Samarin, A.M.; Křišťan, J.; Policar, T. Benefits of hormone treatment of both sexes in semi-artificial reproduction of pikeperch (Sander lucioperca L.). Czech J. Anim. Sci. 2016, 61, 203–208. [Google Scholar] [CrossRef]
- Samarin, M.A.; Miroslav, D.B.; Bytyutskyy; Policar, T. Post-Ovulatory Oocyte Ageing in Pikeperch (Sander lucioperca L.) and its Effect on Egg Viability Rates and the Occurrence of Larval Malformations and Ploidy Anomalies. Turkish J. Fish. Aquat. Sci. 2015, 15, 429–435. [Google Scholar]
- Křištan, T.; Alavi, J.; Stejskal, S.M.H.; Policar, V. Hormonal induction of ovulation in pikeperch (Sander lucioperca L.) using human chorionic gonadotropin (hCG) and mammalian GnRH analogue. Aquac. Int. 2013, 21, 811–818. [Google Scholar] [CrossRef]
- Křištan, J.; Stara, J.; Polgesek, A.; Drasovean, M.; Kolarova, A.; Priborsky, J.; Blecha, J.; Svacina, M.; Policar, P.; Velisek, T. Efficacy of different anaesthetics for pikeperch (Sander lucioperca L.) in relation to water temperature. Neuro. Endocrinol. Lett. 2014, 35, 81–85. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using Lme4; Journal of Statistical Software: Innsbruck, Austria, 2015; Volume 67. [Google Scholar]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, Second; Sage: Thousand Oaks, CA, USA, 2011. [Google Scholar]
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Nash, C.E.; Kuo, C.-M. Hypotheses for problems impeding the mass propagation of grey mullet and other finfish. Aquaculture 1975, 5, 119–133. [Google Scholar] [CrossRef]
- Howell, B.R. A re-appraisal of the potential of the sole, Solea solea (L.), for commercial cultivation. Aquaculture 1997, 155, 355–365. [Google Scholar] [CrossRef]
- Fuchs, J. Influence de la photoperiode sur la croissance et la survie de la larve et du juvenile de sole (Solea solea) en elevage. Aquaculture 1978, 15, 63–74. [Google Scholar] [CrossRef]
- Person-Le^Ruyet, J.; Verillaud, P. Techniques d’elevage intensif de la daurade doree (sparus aurata (L.)) de la naissance a l’age de deux mois. Aquaculture 1980, 20, 351–370. [Google Scholar] [CrossRef]
- Tandler, A.; Helps, S. The effects of photoperiod and water exchange rate on growth and survival of gilthead sea bream (Sparus aurata, Linnaeus; Sparidae) from hatching to metamorphosis in mass rearing systems. Aquaculture 1985, 48, 71–82. [Google Scholar] [CrossRef]
- Girin, M. Marine fish culture in France: Recent developments. Aquaculture 1975, 5, 113. [Google Scholar] [CrossRef]
- Nicolas, J.L.; Robic, E.; Ansquer, D. Bacterial flora associated with a trophic chain consisting of microalgae, rotifers and turbot larvae: Influence of bacteria on larval survival. Aquaculture 1989, 83, 237–248. [Google Scholar] [CrossRef]
- Munro, P.; Henderson, R.; Barbour, A.; Birkbeck, T. Partial decontamination of rotifers with ultraviolet radiation: The effect of changes in the bacterial load and flora of rotifers on mortalities in start-feeding larval turbot. Aquaculture 1999, 170, 229–244. [Google Scholar] [CrossRef]
- Skjermo, J.; Vadstein, O. Techniques for microbial control in the intensive rearing of marine larvae. Aquaculture 1999, 177, 333–343. [Google Scholar] [CrossRef]
- Hamza, A.; Fdhila, K.; Zouiten, D.; Masmoudi, A.S. Virgibacillus proomii and Bacillus mojavensis as probiotics in sea bass (Dicentrarchus labrax) larvae: Effects on growth performance and digestive enzyme activities. Fish Physiol. Biochem. 2016, 42, 495–507. [Google Scholar] [CrossRef]
- Dharmaraj, S.; Dhevendaran, K. Evaluation of Streptomyces as a Probiotic Feed for the Growth of Ornamental Fish Xiphophorus helleri. Food Technol. Biotechnol. 2010, 48, 497–504. [Google Scholar]
- Ghosh, S.; SinhaI, A.; Sahu, C. Dietary probiotic supplementation in growth and health of live-bearing ornamental fishes. Aquac. Nutr. 2008, 14, 289–299. [Google Scholar]
- Lara-Flores, M.; Olvera-Novoa, M.A.; Guzmán-Méndez, B.E.; López-Madrid, W. Use of the bacteria Streptococcus faecium and Lactobacillus acidophilus, and the yeast Saccharomyces cerevisiae as growth promoters in Nile tilapia (Oreochromis niloticus). Aquaculture 2003, 216, 193–201. [Google Scholar] [CrossRef]
- Carnevali, O.; Maradonna, F.; Gioacchini, G. Integrated control of fish metabolism, wellbeing and reproduction: The role of probiotic. Aquaculture 2017, 472, 144–155. [Google Scholar] [CrossRef]
- Carnevali, O.; Vivo, L.; Sulpizio, R.; Gioacchini, G.; Olivotto, I.; Silvi, S.; Cresci, A. Growth improvement by probiotic in European sea bass juveniles (Dicentrarchus labrax, L.), with particular attention to IGF-1, myostatin and cortisol gene expression. Aquaculture 2006, 258, 430–438. [Google Scholar] [CrossRef]
- Carnevali, O.; Claudia, M.; Sulpizio, Z.R.; Rollo, A.; Nardi, M.; Orpianesi, C.; Silvi, S.; Caggiano, M.; Polzonetti, A.M.; Cresci, A.; et al. Administration of Probiotic Strain to Improve Sea Bream Wellness during Development. Aquac. Int. 2004, 12, 377–386. [Google Scholar] [CrossRef]
- Avella, M.A.; Gioacchini, G.; Decamp, O.; Makridis, P.; Bracciatelli, C.; Carnevali, O. Application of multi-species of Bacillus in sea bream larviculture. Aquaculture 2010, 305, 12–19. [Google Scholar] [CrossRef]
- Avella, M.A.; Olivotto, I.; Silvi, S.; Ribecco, C.; Cresci, A.; Palermo, F.; Polzonetti, A.; Carnevali, O. Use of Enterococcus faecium to improve common sole (Solea solea) larviculture. Aquaculture 2011, 315, 384–393. [Google Scholar] [CrossRef]
- Carnevali, O.; Avella, M.A.; Gioacchini, G. Effects of probiotic administration on zebrafish development and reproduction. Gen. Comp. Endocrinol. 2013, 188, 297–302. [Google Scholar] [CrossRef]
- Vianello, S.; Brazzoduro, L.; Valle, L.D.; Belvedere, P.; Colombo, L. Myostatin expression during development and chronic stress in zebrafish (Danio rerio). J. Endocrinol. 2003, 176, 47–59. [Google Scholar] [CrossRef]
- Varela, J.L.; Ruiz-Jarabo, I.; Vargas-Chacoff, L.; Arijo, S.; León-Rubio, J.M.; García-Millán, I.; del Río, M.P.M.; Moriñigo, M.A.; Mancera, J.M. Dietary administration of probiotic Pdp11 promotes growth and improves stress tolerance to high stocking density in gilthead seabream Sparus auratus. Aquaculture 2010, 309, 265–271. [Google Scholar] [CrossRef]
- Tapia-Paniagua, S.T.; Díaz-Rosales, P.; León-Rubio, J.M.; de la Banda, I.G.; Lobo, C.; Alarcón, F.J.; Chabrillón, M.; Rosas-Ledesma, P.; Varela, J.L.; Ruiz-Jarabo, I.; et al. Use of the probiotic Shewanella putrefaciens Pdp11 on the culture of Senegalese sole (Solea senegalensis, Kaup 1858) and gilthead seabream (Sparus aurata L.). Aquac. Int. 2012, 20, 1025–1039. [Google Scholar] [CrossRef]
- Verschuere, L.; Rombaut, G.; Sorgeloos, P.; Verstraete, W. Probiotic bacteria as biological control agents in aquaculture. Microbiol. Mol. Biol. Rev. 2000, 64, 655–671. [Google Scholar] [CrossRef] [PubMed]



| DPH | Daily Feed:Rot-Art/mL | Flow (ml/min) |
|---|---|---|
| 3 | 10-0 | 100 |
| 4 | 10-0 | 100 |
| 5 | 10-0 | 100 |
| 6 | 10-0 | 100 |
| 7 | 10-0 | 100 |
| 8 | 14-0 | 160 |
| 9 | 14-0 | 160 |
| 10 | 14-0 | 160 |
| 11 | 14-0 | 160 |
| 12 | 14-2 | 200 |
| 13 | 10-3 | 200 |
| 14 | 8-4 | 200 |
| 15 | 0-7 | 250 |
| 16 | 0-7 | 250 |
| 17 | 0-8 | 250 |
| 18 | 0-8 | 250 |
| 19 | 0-8 | 250 |
| 20 | 0-8 | 250 |
| 21 | End of Trial | 250 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yanes-Roca, C.; Leclercq, E.; Vesely, L.; Malinovskyi, O.; Policar, T. Use of Lactic Acid Bacteria During Pikeperch (Sander lucioperca) Larval Rearing. Microorganisms 2020, 8, 238. https://doi.org/10.3390/microorganisms8020238
Yanes-Roca C, Leclercq E, Vesely L, Malinovskyi O, Policar T. Use of Lactic Acid Bacteria During Pikeperch (Sander lucioperca) Larval Rearing. Microorganisms. 2020; 8(2):238. https://doi.org/10.3390/microorganisms8020238
Chicago/Turabian StyleYanes-Roca, Carlos, Eric Leclercq, Lukas Vesely, Oleksandr Malinovskyi, and Tomas Policar. 2020. "Use of Lactic Acid Bacteria During Pikeperch (Sander lucioperca) Larval Rearing" Microorganisms 8, no. 2: 238. https://doi.org/10.3390/microorganisms8020238
APA StyleYanes-Roca, C., Leclercq, E., Vesely, L., Malinovskyi, O., & Policar, T. (2020). Use of Lactic Acid Bacteria During Pikeperch (Sander lucioperca) Larval Rearing. Microorganisms, 8(2), 238. https://doi.org/10.3390/microorganisms8020238






