Duration of Prebiotic Intake Is a Key-Factor for Diet-Induced Modulation of Immunity and Fecal Fermentation Products in Dogs
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Location, and Experimental Design
2.2. Feces Analysis (FS, pH, SCFAs, BCFAs, NH3, Lactic Acid, and IgA)
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, X.; Cao, S.; Zhang, X. Modulation of gut microbiota-brain axis by probiotcs, prebiotics and diet. J. Agric. Food Chem. 2015, 63, 7885–7895. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Loo, J.V.; Vancraeynest, D. Prebiotics and animal nutrition; CRC Press: Boca Raton, FL, USA, 2008; pp. 421–436. [Google Scholar]
- Ghosh, S.; Mehla, R.K. Influence of dietary supplementation of prebiotics (mannanoligosaccharide) on the performance of crossbred calves. Trop. Anim. Health Prod. 2012, 44, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Grand, E.; Respondek, F.; Martineau, C.; Detilleux, J.; Bertrand, G. Effects of short-chain fructooligosaccharides on growth performance of preruminant veal calves. J. Dairy Sci. 2013, 96, 1094–1101. [Google Scholar] [CrossRef] [PubMed]
- Kara, C.; Cihan, H.; Temizel, M.; Catik, S.; Meral, Y.; Orman, A.; Yibar, A.; Gencoglu, H. Effects of Supplemental Mannanoligosaccharides on Growth Performance, Faecal Characteristics and Health in Dairy Calves. Asian Australs. J. Anim. Sci. 2015, 28, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Middelbos, I.S.; Godoy, M.R.; Fastinger, N.D.; Fahey, G.C., Jr. A dose-response evaluation of spray-dried yeast cell wall supplementation of diets fed to adult dogs: Effects on nutrient digestibility, immune indices, and fecal microbial populations. J. Anim. Sci. 2007, 85, 3022–3032. [Google Scholar] [CrossRef] [PubMed]
- Vetvicka, V.; Oliveira, C. β (1-3)(1-6)-D-glucans modulate imune status and blood glucose levels in dogs. J. Pharm. Res. Int. 2014, 4, 981–991. [Google Scholar]
- De Oliveira, C.A.F.; Vetvicka, V.; Zanuzzo, F.S. β-glucan successfully stimulated the immune system in different jawed vertebrate species. Comp. Immun, Microb. Infect. Dis. 2019, 62, 1–6. [Google Scholar] [CrossRef]
- Zentek, J.; Marquat, B.; Pietrzak, T. Intestinal effects of mannanoligosaccharides, transgalactooligosaccharides, lactose and lactulose in dogs. J. Nutr. 2002, 132, 1682–1684. [Google Scholar] [CrossRef]
- Swanson, K.; Grieshop, C.; Flickinger, E.; Healy, H.P.; Dawson, K.A.; Merchen, N.R.; Merchen, N.R.; Fahey, G.C., Jr. Effects of supplemental fructooligosaccharides plus mannanoligosaccharides on immune function and ileal and fecal microbial populations in adult dogs. Arch. Anim. Nutr. 2002, 56, 309–318. [Google Scholar] [CrossRef]
- Swanson, K.S.; Grieshop, C.M.; Flickinger, E.A.; Bauer, L.L.; Healy, H.P.; Dawson, K.A.; Merchen, N.R.; Fahey, G.C., Jr. Supplemental fructooligosaccharides and mannanoligosaccharides influence immune function, ileal and total tract nutrient digestibilities, microbial populations and concentrations of protein catabolites in the large bowel of dogs. J. Nutri. 2002, 132, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Middelbos, I.S.; Fastinger, N.D.; Fahey, G.C., Jr. Evaluation of fermentable oligosaccharides in diets fed to dogs in comparison to fiber standards. J. Anim. Sci. 2007, 85, 3033–3044. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, J.P.S.; He, F.; Mangian, H.F.; Oba, P.M.; Godoy, M.R.C. Dietary supplementation of a fiber-prebiotic and saccharin-eugenol blend in extruded diets fed to dogs. J. Anim. Sci. 2019, 97, 4519–4531. [Google Scholar] [CrossRef] [PubMed]
- Biagi, G.; Cipollini, I.; Grandi, M.; Zaghini, G. Influence of some potential prebiotics and fibre-rich foodstuffs on composition and activity of canine intestinal microbiota. Ani. Feed Sci. Tech. 2010, 159, 50–58. [Google Scholar] [CrossRef]
- Grieshop, C.; Flickinger, E.A.; Bruce, K.J.; Patil, A.R.; Czarnecki-Maulden, G.L.; Fahey, G.C., Jr. Gastrointestinal and immunological responses of senior dogs to chicory and mannan-oligosaccharides. Arch. Anim. Nutr. 2004, 58, 483–494. [Google Scholar] [CrossRef]
- National Research Council. Nutrient Requirements of Dogs and Cats; National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Alexander, C.; Cross, T.W.L.; Devendran, S.; Neumer, F.; Theis, S.; Ridlon, J.M.; Suchodolski, J.S.; De Godoy, M.R.C.; Swanson, K.S. Effects of prebiotic inulin-type fructans on blood metabolite and hormone concentrations and faecal microbiota and metabolites in overweight dogs. Br. J. Nut. 2018, 120, 711–720. [Google Scholar] [CrossRef]
- Twomey, L.N.; Pluske, J.R.; Rowe, J.B.; Choct, M.; Brown, W.; Pethick, D.W. The effects of added fructooligosaccharide (Raftilose® P95) and inulinase on faecal quality and digestibility in dogs. Anim. Feed Sci. Tech. 2003, 108, 83–93. [Google Scholar] [CrossRef]
- Faber, T.A.; Hopkins, A.C.; Middelbos, I.S.; Price, N.P.; Fahey, G.C., Jr. Galactoglucomannan oligosaccharide supplementation affects nutrients digestibility fermentation end-product production, and large bowel microbiota of the dog. J. Ani. Sci. 2011, 89, 103–112. [Google Scholar] [CrossRef]
- Strickling, J.A.; Harmon, D.L.; Dawson, K.A.; Gross, K.L. Evaluation of oligosaccharide addition to dog diets: Influences on nutrient digestion and microbial populations. Anim. Feed Sci. Tech. 2000, 86, 205–219. [Google Scholar] [CrossRef]
- Swanson, K.S.; Grieshop, C.M.; Flickinger, E.A.; Bauer, L.L.; Chow, J.; Wolf, B.W.; Garleb, K.A.; Fahey, G.C., Jr. Fructooligosaccharides and Lactobacillus acidophilus modify gut microbial populations, total tract nutrient digestibilities and fecal protein catabolite concentrations in healthy adult dogs. The J. Nutri. 2002, 132, 3721–3731. [Google Scholar] [CrossRef]
- Flickinger, E.A.; Screijen, E.M.W.C.; Patil, A.R.; Hussein, H.S.; Grieshop, C.M.; Merchen, N.R.; Fahey, G.C., Jr. Nutrient digestibilities, microbial populations, and protein catabolites as affected by fructan supplementation of dog diets. J. Anim. Sci. 2003, 81, 2008–2018. [Google Scholar] [CrossRef] [PubMed]
- Beloshapka, N.A.; Dowd, S.E.; Suchodolski, J.S.; Steiner, J.M.; Duclos, L.; Swanson, K.S. Fecal microbial communities of healthy adult dogs fed raw meat-based diets with or without inulin or yeast cell wall extracts as assessed by 454 pyrosequencing. FEMS Microbiol. Ecol. 2013, 84, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Alexander, C.; Steelman, A.J.; Warzecha, C.M.; De Godoy, M.R.C.; Swanson, K.S. Effects of a Saccharomyces cerevisiae fermentation product on fecal characteristics, nutrient digestibility, fecal fermentative end-products, fecal microbial populations, immune function, and diet palatability in adult dogs. J. Anim. Sci. 2019, 97, 1586–1599. [Google Scholar] [CrossRef] [PubMed]
- Pinna, C.; Vecchiato, C.G.; Bolduan, C. Influence of dietary protein and fructooligosaccharides on fecal fermentative end-products, fecal bacterial populations and apparent total tract digestibility in dogs. BMC Vet. Res. 2018, 14, 106. [Google Scholar] [CrossRef]
- Hesta, M.; Roosen, W.; Janssens, G.P.J.; Millet, S.; De Wilde, R. Prebiotics affect nutrient digestibility but not faecal ammonia in dogs fed increased dietary protein levels. British J. Nutri. 2003, 90, 1007–1014. [Google Scholar]
- Santos, J.P.F.; Aquino, A.A.; Glória, M.B.A.; Avila-Campos, M.J.; Oba, P.M.; Santos, K.D.M.; Vendramini, T.H.A.; Carciofi, A.C.; Junior, A.R.; Brunetto, M.A. Effects of dietary yeast cell wall on faecal bacteria and fermentation products in adult cats. J. Anim. Physiol. An. N. 2018, 102, 1091–1101. [Google Scholar] [CrossRef]
- Theodoro, S.D.S.; Putarov, T.C.; Volpe, L.M. Effects of the solutibility of yeast cell wall preparations on their potential prebiotic properties in dogs. PLoS ONE 2019, 14, e0225659. [Google Scholar] [CrossRef]
- Diez, M.; Hornick, J.L.; Baldwin, P.; Eenaeme, C.V.; Istasse, L. The influence of sugar-beet fibre, guar gum and inulin on nutrient digestibility, water consumption and plasma metabolites in healthy Beagle dogs. Res. Vet. Sci. 1998, 64, 91–96. [Google Scholar] [CrossRef]
- Willard, M.D.; Simpson, R.B.; Cohen, N.D.; Clancy, J.S. Effects of dietary fructooligosaccharide on selected bacterial populations in feces of dogs. Am. J. Vet. Res. 2000, 61, 820–825. [Google Scholar] [CrossRef]
- Pawar, M.M.; Pattanaik, A.K.; Sinha, D.K.; Goswami, T.K.; Sharma, K. Effect of dietary mannanoligosaccharide supplementation on nutrient digestibility, hindgut fermentation, imune response and antioxidant indices in dogs. J. Anim. Sci. Tech. 2017, 59, 11. [Google Scholar] [CrossRef]
- Propst, E.L.; Flickinger, E.A.; Bauer, L.L.; Merchen, N.R.; Fahey, G.C., Jr. A dose-response experiment evaluating the effects of oligofructose and inulin on nutrient digestibility, stool quality, and fecal protein catabolites in healthy adult dogs. J. Anim. Sci. 2003, 81, 3057–3066. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi-Hayakawa, H.; Kimura, M.; Watanuki, M. Adaptation of rate of organic acid production of hindgut bacteria to chronic intake of galactooligosaccharide in the rat. J. Nutri. Sci. Vitam. 1997, 43, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Laflamme, D. Development and validation of a body condition score system for dogs: A clinical tool. Can. Pract. 1997, 22, 10–15. [Google Scholar]
- FEDIAF. Nutritional Guidelines for Complete and Complementary Pet. Food for Cats and Dogs. Available online: file:///C:/Users/MDPI/AppData/Local/Temp/FEDIAF_Nutritional_Guidelines_2017.pdf (accessed on 20 May 2017).
- Moxham, G. Waltham feces scoring system-A tool for veterinarians and pet owners: How does your pet rate. Wealth Focus. 2001, 11, 24–25. [Google Scholar]
- Walter, M.; Silva, L.P.; Perdomo, D.M.X. Biological response of rats to resistant starch. Rev. Inst. Adolfo Lutz. 2005, 64, 252–257. [Google Scholar]
- Erwin, E.S.; Marco, G.J.; Emery, E.M. Volatile fatty acid analyses of blood and rumen fluid by gas chromatography. J. Dairy Sci. 1961, 44, 1768–1771. [Google Scholar] [CrossRef]
- Sá, F.C.; Vasconcellos, R.S.; Brunetto, M.A.; Filho, F.O.R.; Gomes, M.O.S.; Carciofi, A.C. Enzyme use in kibble diets formulated with wheat bran for dogs: Effects on processing and digestibility. J. Ani. Physio. Ani. Nutri. 2013, 97, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Pryce, J.D. A modification of the Barker-Summerson method for the determination of latic acid. Analyst 1969, 94, 1121–1151. [Google Scholar] [CrossRef]
- Peters, I.R.; Calvert, E.L.; Hall, E.J.; Day, M.J. Measurement of immunoglobulin concentrations in the feces of healthy dogs. Clin. Diagn Lab. Immunol. 2004, 11, 841–848. [Google Scholar] [CrossRef]
- Silvio, J.; Harmon, D.L.; Gross, K.L.; McLeod, K.R. Influence of fiber fermentability on nutrient digestion in the dog. Nutrition 2000, 16, 289–295. [Google Scholar] [CrossRef]
- Gullón, P.; González-Muñoz, M.J.; Parajó, J.C. Manufacture and prebiotic potential of oligosaccharides derived from industrial solid wastes. Bioresour. Technol. 2011, 102, 6112–6119. [Google Scholar] [CrossRef] [PubMed]
- Nurmi, J.; Puolakkainen, P.; Rautonen, N. Bifidobacterium lactis sp. 420 up-regulates cylooxygenase (Cox) 1 and down-regulates COX-2 gene expression in a Caco-2 cell culture model. Nutr. Can. 2005, 51, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Blair, J.B.; Cook, D.E.; Lardy, H.A. Interaction of propionate and lactate in the perfused rat liver. J. Biol. Chem. 1973, 248, 3608–3614. [Google Scholar] [PubMed]
- Anderson, J.W.; Bridges, S.R. Short chain fatty acid fermentation products of plant fiber affect glucose metabolism of isolated rat hepatocytes. Proc. Soc. Exp. Biol. Med. 1984, 177, 372–376. [Google Scholar] [CrossRef]
- Verbrugghe, A.; Hexta, M.; Gommeren, K.; Daminet, S.; Wuyts, B.; Buyse, J.; Janssens, G.P.J. Oligofructose and inulin modulate glucose and amino acid metabolism through propionate production in normal-weight and obese cats. Br. J. Nutri. 2009, 102, 694–702. [Google Scholar] [CrossRef]
- Topping, D.L. Short-chain fatty acids produced by intestinal bacteria. Asia Pacific J. Clin. Nutr. 1996, 5, 15–19. [Google Scholar]
- Vulevic, J.; Juric, A.; Walton, G.E.; Claus, S.P. Influence of galacto-oligosaccharide mixture (B-GOS) on gut microbiota, immune parameters and metabonomics in elderly persons. Br. J. Nutri. 2015, 114, 586–595. [Google Scholar] [CrossRef]
- Levy-Pereira, N.; Yasui, G.S.; Cardozo, M.V. Immunostimulation and increase of intestinal lactic acid bacteria with dietary mannan-oligosaccharide in Nile tilapia juveniles. R. Bras. Zootec. 2018, 47, e20170006. [Google Scholar] [CrossRef]
- Momeni-Moghaddam, P.; Keyvanshokooh, S.; Ziaedi-Nejad, S.; Salati, A.P.; Pasha-Zanoosi, H. Effects of mannan oligosaccharide supplementation on growth, some imune responses and gut lactic acid bacteria of common carp (Cyprinus Carpio) fingerlings. Vet. Res. Forum 2015, 6, 239–244. [Google Scholar]
- Slavin, J. Fiber and Prebiotics: Mechanisms and Health Benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef]
- Hosono, A.; Ozawa, A.; Kato, R.; Ohnishi, Y.; Nakanishi, Y.; Kimura, T.; Nakamura, R. Dietary fructooligosaccharides induce immunoregulation of intestinal IgA secretion by murine Peyer’s patch cells. Biosci. Biotech. Bioch. 2003, 67, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Schley, P.D.; Field, C.J. The immune-enhancing effects of dietary fibres and prebiotics. Br. J. Nutr. 2002, 87, S221–S230. [Google Scholar] [CrossRef] [PubMed]
- Kroll, F.S.A.; Putarov, T.C.; Zaine, L.; Venturi, K.S.; Aoki, C.G.; Santos, J.P.F.; Pedrinelli, V.; Vendramini, T.H.A.; Brunetto, M.A.; Carciofi, A.C. Active fractions of mannoproteins derived from yeast cell wall stimulate innate and acquired immunity of adult and elderly dogs. Anim. Feed Sci. Tech. 2020, 261, 114392. [Google Scholar] [CrossRef]
- Gomes, M.O.S.; Beraldo, M.C.; Putarov, T.C.; Brunetto, M.A.B.; Zaine, L.; Glória, M.B.A.; Carciofi, A.C. Old beagle dogs have lower fecal concentrations of some fermentation products and lower peripheral lymphocyte counts than young adult beagles. Br. J. Nutr. 2011, 106, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Le Bourgot, C.; Ferret-Bernard, S.; Le Normand, L.; Savary, G.; Menendez-Aparicio, E.; Blat, S.; Appert-Bossard, E.; Respondek, F.; Huerou-Luron, I. Material short-chain fructooligosaccharide supplementation influences intestinal immune system maturation in piglets. PLoS ONE 2014, 9, e107508. [Google Scholar] [CrossRef] [PubMed]
- Pierre, F.; Perrin, P.; Bassonga, E.; Bornet, F.; Meflah, K.; Menanteau, J. T cell status influences colon tumor occurrence in mice fed short chain fructo-oligosaccharides as a diet supplement. Carcinogenesis 1999, 20, 1953–1956. [Google Scholar] [CrossRef]
- Manhart, N.; Spittler, A.; Bergmeister, H.; Mittlbock, M.; Roth, E. Influence of fructooligosaccharides on Peyer’s patch lymphocyte numbers in healthy and endotoxemic mice. Nutrition 2003, 19, 657–660. [Google Scholar] [CrossRef]
- Sangwan, V.; Tomar, S.K.; Ali, B.; Singh, R.R.B.; Singh, A.K. Galatooligosaccharides reduce infestion caused by Listeria monocytogenes and modulate IgG and IgA levels in mice. Inter. Dairy J. 2015, 41, 58–63. [Google Scholar] [CrossRef]
- Bakker-Zierikzee, A.M.; Tol, E.A.; Kroes, H.; Alles, M.S.; Kok, F.J.; Bindels, J.G. Fecal IgA secretion in infants fed on pre- or probiotic infant formula. Pediatric Allergy Immu. 2006, 17, 134–140. [Google Scholar] [CrossRef]
- Brown, G.D.; Gordon, S. Fungal beta-glucans and mammalian immunity. Immunity 2003, 19, 311–315. [Google Scholar] [CrossRef]
- Barry, K.A.; Wojcicki, B.J.; Middelbos, I.S.; Vester, B.M.; Swanson, K.S.; Fahey, G.C., Jr. Dietary cellulose, fructooligosaccharides, and pectin modify fecal protein catabolites and microbial populations in adult cats. J. Ani. Sci. 2010, 88, 2978–2987. [Google Scholar] [CrossRef] [PubMed]
- Sako, T.; Matsumoto, K.; Tanaka, R. Recent progress on research and application of non-digestible galacto-oligosaccharides. Internat. Dairy J. 1999, 9, 69–80. [Google Scholar] [CrossRef]
- Hoseinifar, S.H.; Khalili, M.; Rostami, H.K.; Esteban, M.A. Dietary galactooligosaccharide affects intestinal microbiota, stress resistance, and performance of Caspian roach (Rutilus rutilus) fry. Fish Shellfish Immun. 2013, 35, 1416–1420. [Google Scholar] [CrossRef] [PubMed]
- Matheus, L.F.O.; Ernandes, M.C.; Santos, J.P.F.; Santos, K.M.; Takeara, P.; Monti, M.; Pontieri, C.F.F.; Brunetto, M.A. Efeitos digestivos e fermentativos de leveduras (Saccharomyces cerevisiea) inativadas e enriquecidas em meio de cultura em dietas para gatos adultos. In Proceedings of the XV Congresso CBNA Pet, Campinas, Brazil, 18 May 2016. [Google Scholar]
- Adam, O. Dietary fatty acids and imune reactions in synovial tissue. Eur. J. Med. Res. 2003, 8, 381–387. [Google Scholar]
- Moro, G.E.; Mosca, F.; Miniello, V.; Fanaro, S.; Jelinek, J.; Stahl, B.; Boehm, G. Effects of a new mixture of prebiotics on faecal flora and stools in term infants. Acta Paediatr. 2003, 92, 77–79. [Google Scholar] [CrossRef]
- Santoso, B.; Mwenya, B.; Sar, Y.; Gamo, B.; Kobayashi, T.; Morikawa, R.; Kimura, K.; Mizukoshi, H.; Takahashi, J. Effects of supplementation galacto-oligosaccharides, Yucca schidigera or nisin on rumen methanogenesis, nitrogen and energy metabolism in sheep. Livest. Prod. Sci. 2004, 91, 209–217. [Google Scholar] [CrossRef]
| Item | Treatments | |||
|---|---|---|---|---|
| % | CO 1 | GOS 2 | B1 3 | B2 4 |
| Dry matter | 91.95 | 91.80 | 91.76 | 91.76 |
| Ash | 6.47 | 6.61 | 6.72 | 6.85 |
| Crude protein | 27.46 | 26.93 | 25.28 | 27.22 |
| Fat | 14.17 | 14.53 | 14.32 | 14.37 |
| Crude fiber | 6.30 | 6.40 | 5.97 | 5.83 |
| Nitrogen-free extract | 45.60 | 45.53 | 47.71 | 45.73 |
| Item | CO | GOS | B1 | B2 | MTE | p | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Averages | SD | Average | SD | Treat. | Time | Treat. × Time | ||||
| Latic acid (mMol/L in solution) | ||||||||||
| T30 | 2.13 | 2.15 | 2.12 | 2.14 | 2.13 B | |||||
| T60 | 2.16 | 2.19 | 2.16 | 2.18 | 0.007 | 2.17 A | 0.0037 | 0.0002 | <0.0001 | 0.7515 |
| MPE | 2.14 b | 2.17 a | 2.14 b | 2.16 a,b | 0.005 | |||||
| NH3 (mMol/kg in DM) | ||||||||||
| T30 | 119.87 | 133.58 | 157.58 | 140.7 | 137.93 | |||||
| T60 | 145.73 | 123.32 | 136.90 | 136.61 | 14.75 | 135.64 | 8.7738 | 0.5565 | 0.8144 | 0.3775 |
| MPE | 132.80 | 128.45 | 147.24 | 138.66 | 11.13 | |||||
| pH | ||||||||||
| T30 | 6.75 | 6.77 | 6.65 | 6.58 | 6.69 B | |||||
| T60 | 6.98 | 6.96 | 6.92 | 6.76 | 0.11 | 6.91 A | 0.0681 | 0.2004 | 0.0043 | 0.9696 |
| MPE | 6.87 | 6.87 | 6.78 | 6.67 | 0.08 | |||||
| Acetic acid (mMol/L in DM) | ||||||||||
| T30 | 22.50 | 19.98 | 19.15 | 22.23 | 22.23 | |||||
| T60 | 18.24 | 26.38 | 23.61 | 19.23 | 2.42 | 19.23 | 2.4202 | 0.6638 | 0.6029 | 0.0836 |
| MPE | 20.37 | 23.18 | 21.38 | 20.73 | 1.71 | |||||
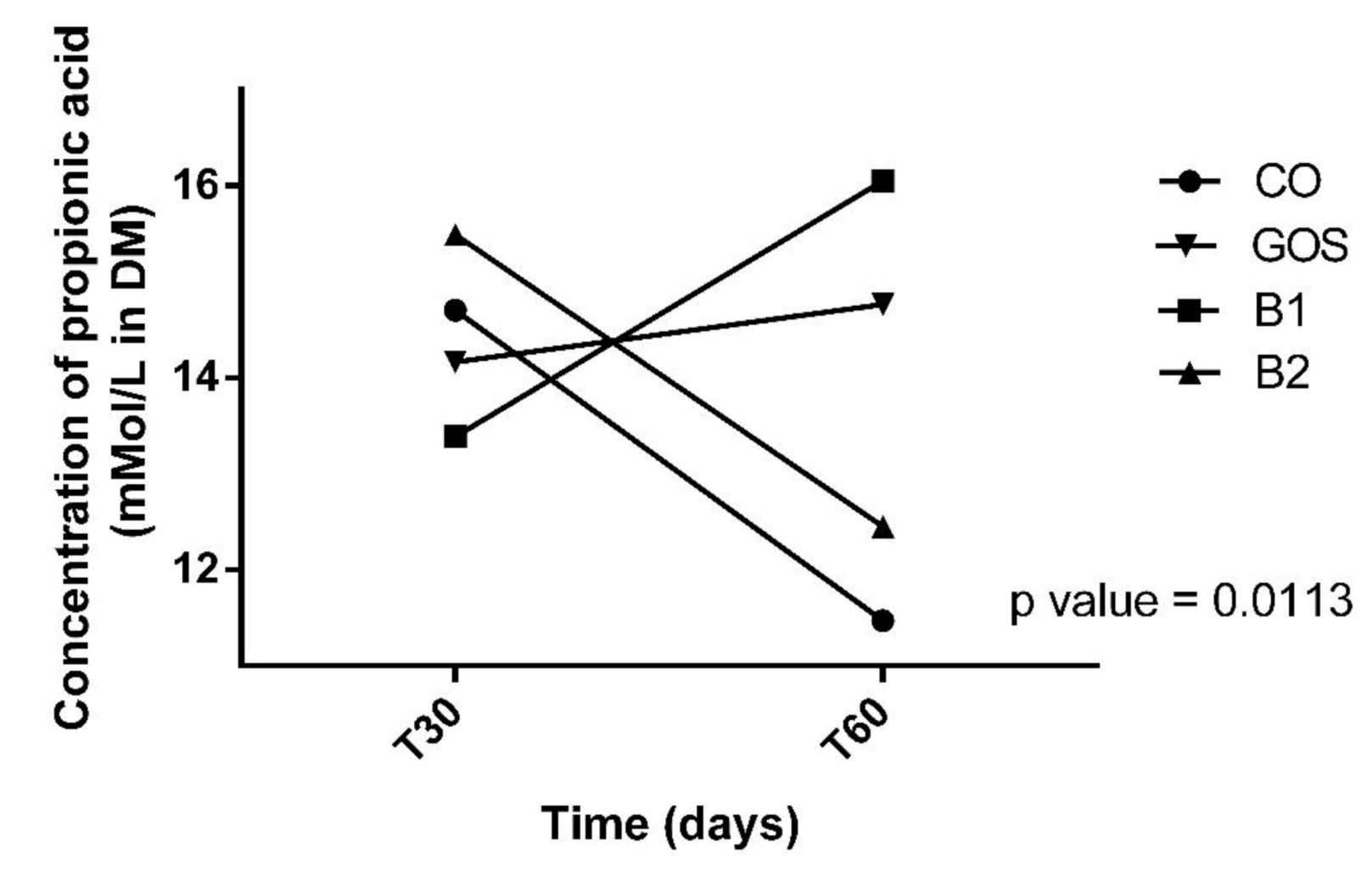
| Propionic acid (mMol/L in DM) | ||||||||||
| T30 | 14.70 | 14.16 | 13.38 | 15.49 A | 14.43 | |||||
| T60 | 11.47 | 14.76 | 16.05 | 12.45 B | 1.38 | 13.68 | 0.9051 | 0.7222 | 0.2664 | 0.0113 |
| MPE | 13.09 | 14.46 | 14.72 | 13.97 | 1.09 | |||||
| Butyric acid (mMol/L in DM) | ||||||||||
| T30 | 4.20 | 3.62 | 4.87 | 4.47 | 4.29 | |||||
| T60 | 4.04 | 4.46 | 4.06 | 4.63 | 0.50 | 4.30 | 0.25 | 0.6934 | 0.9847 | 0.4473 |
| MPE | 4.12 | 4.04 | 4.46 | 4.55 | 0.35 | |||||
| Total SCFA (mMol/L in DM) | ||||||||||
| T30 | 41.41 | 37.78 | 37.41 | 42.20 | 39.70 | |||||
| T60 | 33.76 | 45.61 | 43.72 | 36.32 | 3.95 | 39.85 | 2.11 | 0.7406 | 0.9555 | 0.1085 |
| MPE | 37.59 | 41.69 | 40.57 | 39.26 | 2.86 | |||||
| Iso-butyric (mMol/L in DM) | ||||||||||
| T30 | 0.76 | 0.79 | 0.85 | 0.82 | 0.81 | |||||
| T60 | 0.76 | 0.78 | 0.88 | 0.84 | 0.10 | 0.82 | 0.050 | 0.7104 | 0.9033 | 0.9958 |
| MPE | 0.76 | 0.79 | 0.87 | 0.83 | 0.07 | |||||
| Iso-valeric (mMol/L in DM) | ||||||||||
| T30 | 0.95 | 0.80 | 1.14 | 0.85 | 0.93 | |||||
| T60 | 1.12 | 0.85 | 0.96 | 0.94 | 0.10 | 0.97 | 0.052 | 0.0892 | 0.6463 | 0.3716 |
| MPE | 1.04 | 0.82 | 1.05 | 0.89 | 0.07 | |||||
| Valeric (mMol/L in DM) | ||||||||||
| T30 | 0.12 | 0.12 | 0.11 | 0.10 | 0.11 | |||||
| T60 | 0.14 | 0.13 | 0.11 | 0.09 | 0.01 | 0.12 | 0.008 | 0.0934 | 0.5187 | 0.6574 |
| MPE | 0.13 | 0.13 | 0.11 | 0.09 | 0.01 | |||||
| Total BCFA (mMol/L in DM) | ||||||||||
| T30 | 1.82 | 1.72 | 2.15 | 1.78 | 1.87 | |||||
| T60 | 2.00 | 1.75 | 2.01 | 1.84 | 0.19 | 1.90 | 0.090 | 0.3164 | 0.8144 | 0.8594 |
| MPE | 1.91 | 1.73 | 2.08 | 1.81 | 0.13 | |||||
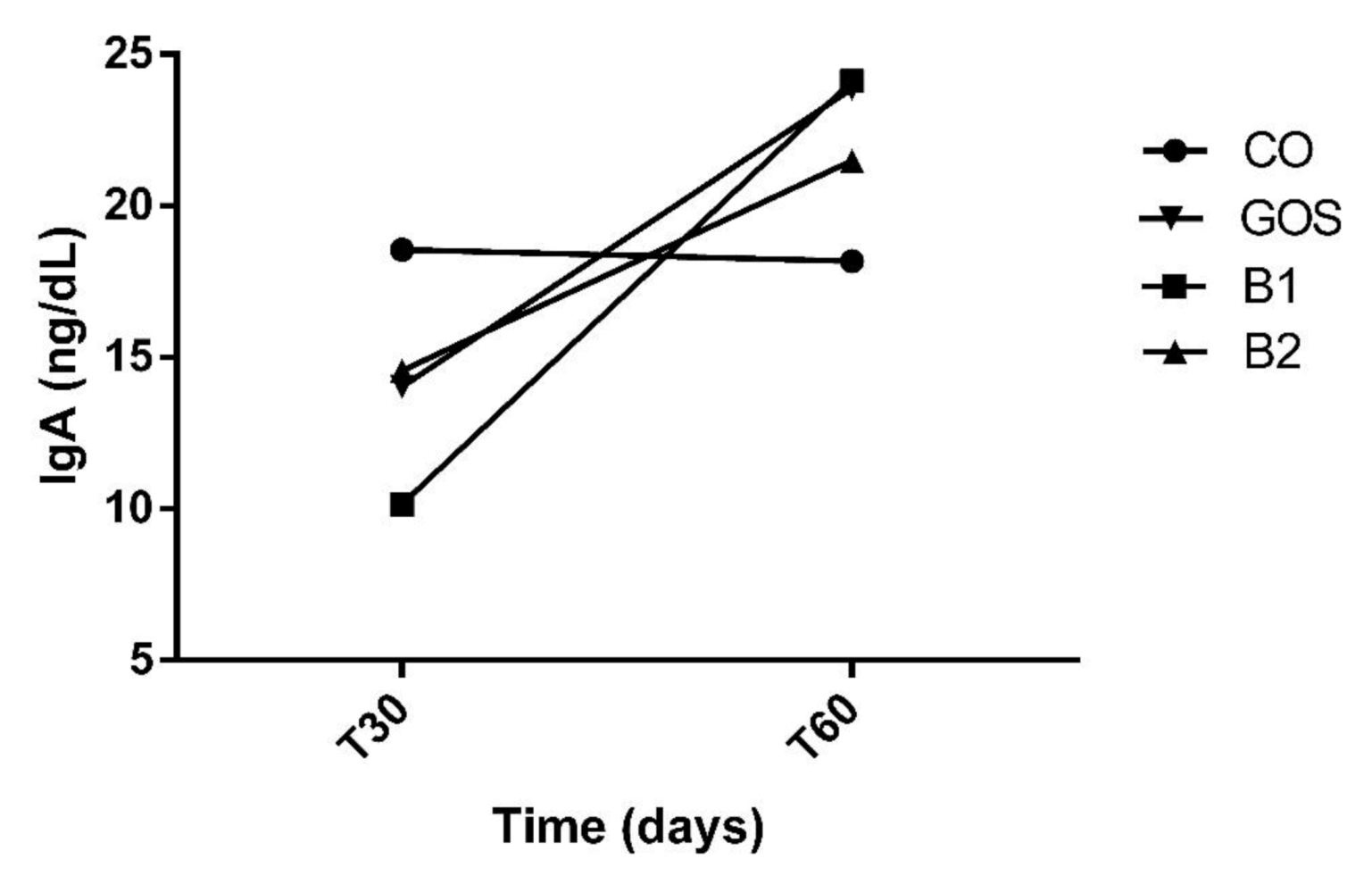
| IgA (ng/mL) | ||||||||||
| T30 | 14.03 | 10.15 | 14.57 | 18.55 | 14.33 B | |||||
| T60 | 23.87 | 24.11 | 21.49 | 18.18 | 4.80 | 21.91 A | 3.008 | 0.9796 | 0.0182 | 0.4167 |
| MPE | 18.95 | 17.13 | 18.03 | 18.37 | 3.70 | |||||
| Fecal score | ||||||||||
| T30 | 2.55 | 2.85 | 2.55 | 2.51 | 2.61 | |||||
| T60 | 2.50 | 2.78 | 2.50 | 2.51 | 0.10 | 2.57 | 0.057 | 0.0124 | 0.5664 | 0.9891 |
| MPE | 2.52 b | 2.81 a | 2.52 b | 2.51 b | 0.07 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perini, M.P.; Rentas, M.F.; Pedreira, R.; Amaral, A.R.; Zafalon, R.V.A.; Rodrigues, R.B.A.; Henríquez, L.B.F.; Zanini, L.; Vendramini, T.H.A.; Balieiro, J.C.C.; et al. Duration of Prebiotic Intake Is a Key-Factor for Diet-Induced Modulation of Immunity and Fecal Fermentation Products in Dogs. Microorganisms 2020, 8, 1916. https://doi.org/10.3390/microorganisms8121916
Perini MP, Rentas MF, Pedreira R, Amaral AR, Zafalon RVA, Rodrigues RBA, Henríquez LBF, Zanini L, Vendramini THA, Balieiro JCC, et al. Duration of Prebiotic Intake Is a Key-Factor for Diet-Induced Modulation of Immunity and Fecal Fermentation Products in Dogs. Microorganisms. 2020; 8(12):1916. https://doi.org/10.3390/microorganisms8121916
Chicago/Turabian StylePerini, Mariana P., Mariana F. Rentas, Raquel Pedreira, Andressa R. Amaral, Rafael V. A. Zafalon, Roberta B. A. Rodrigues, Lucas B. F. Henríquez, Lucca Zanini, Thiago H. A. Vendramini, Júlio C. C. Balieiro, and et al. 2020. "Duration of Prebiotic Intake Is a Key-Factor for Diet-Induced Modulation of Immunity and Fecal Fermentation Products in Dogs" Microorganisms 8, no. 12: 1916. https://doi.org/10.3390/microorganisms8121916
APA StylePerini, M. P., Rentas, M. F., Pedreira, R., Amaral, A. R., Zafalon, R. V. A., Rodrigues, R. B. A., Henríquez, L. B. F., Zanini, L., Vendramini, T. H. A., Balieiro, J. C. C., Pontieri, C. F. F., & Brunetto, M. A. (2020). Duration of Prebiotic Intake Is a Key-Factor for Diet-Induced Modulation of Immunity and Fecal Fermentation Products in Dogs. Microorganisms, 8(12), 1916. https://doi.org/10.3390/microorganisms8121916




