Comparative Genomics of Pathogenic Clavibacter michiganensis subsp. michiganensis Strains from Chile Reveals Potential Virulence Features for Tomato Plants
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
2.2. Pathogenicity Assays in Tomato Seedlings
2.3. Genome Sequencing, Assembly and Annotation
2.4. Pangenome Analysis of Chilean Strains
2.5. Phylogenomic Analysis of Strains of C. michiganensis
2.6. Identification of Virulence Factors, Pathogenicity Islands and Antibiotic Resistance Determinants
2.7. Phylogenetic Analysis of Pathogenicity Genes
2.8. Statistical Analysis
3. Results
3.1. Chilean Strains Induced Symptoms of Bacterial Canker in Tomato
3.2. Whole-Genome Sequence Analysis of Strains VL527, MSF322 and OP3
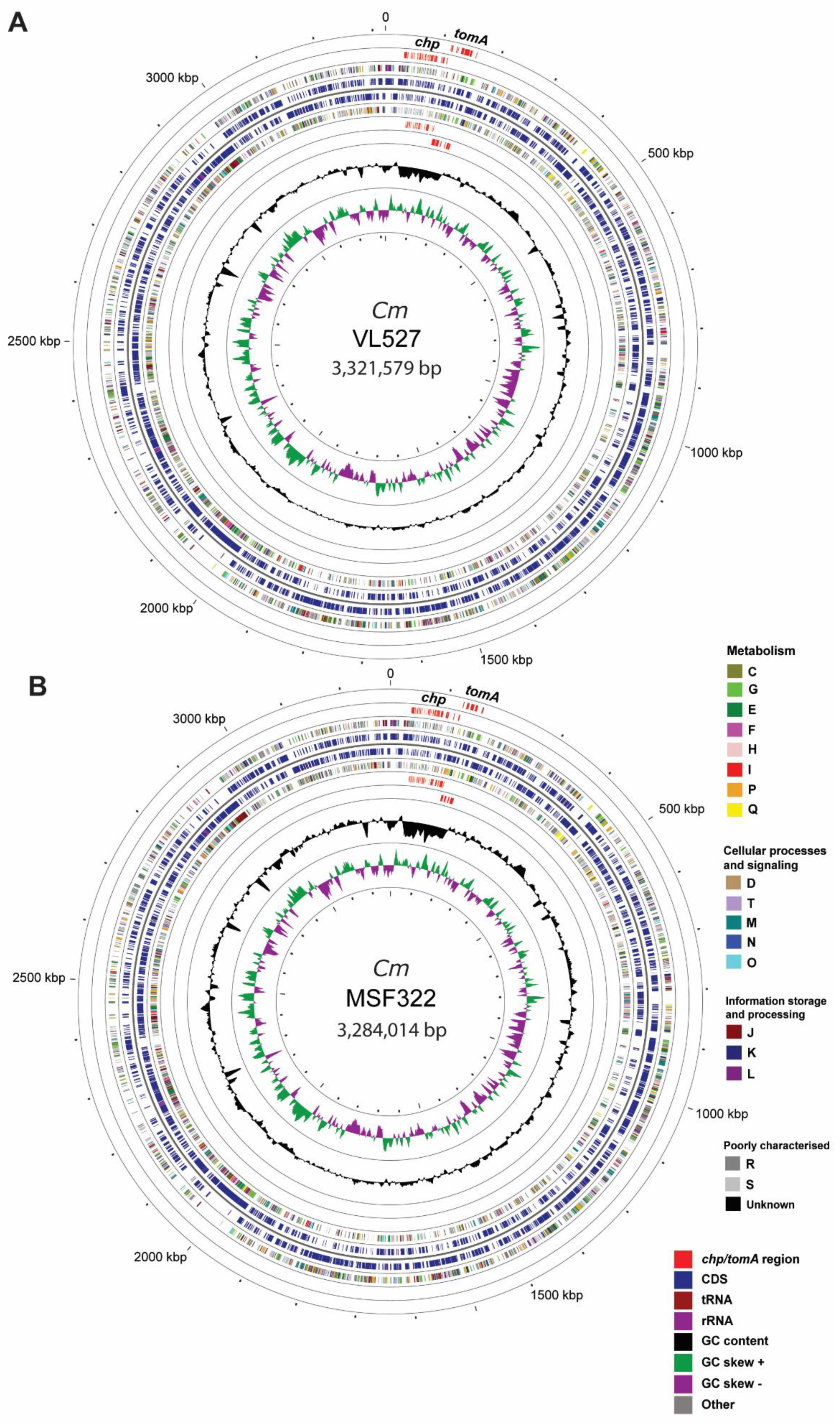
3.2.1. Assembly and Genome Features
3.2.2. Genome Annotation
3.2.3. Plasmid Annotation
3.2.4. Antibiotic Resistance Genes
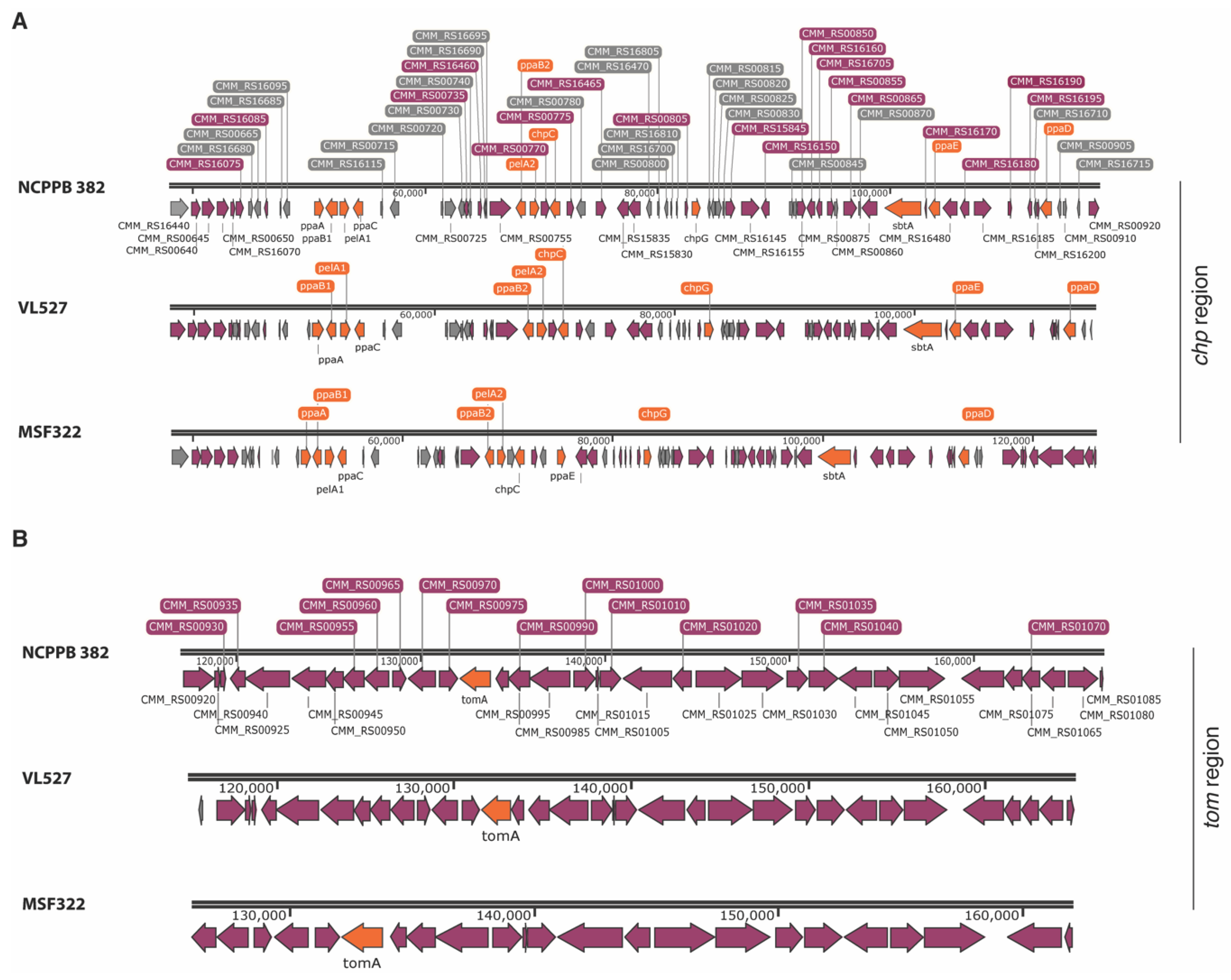
3.3. Pathogenicity Islands in Strains VL527, MSF322 and OP3
3.4. Pangenome Analysis of Chilean Strains
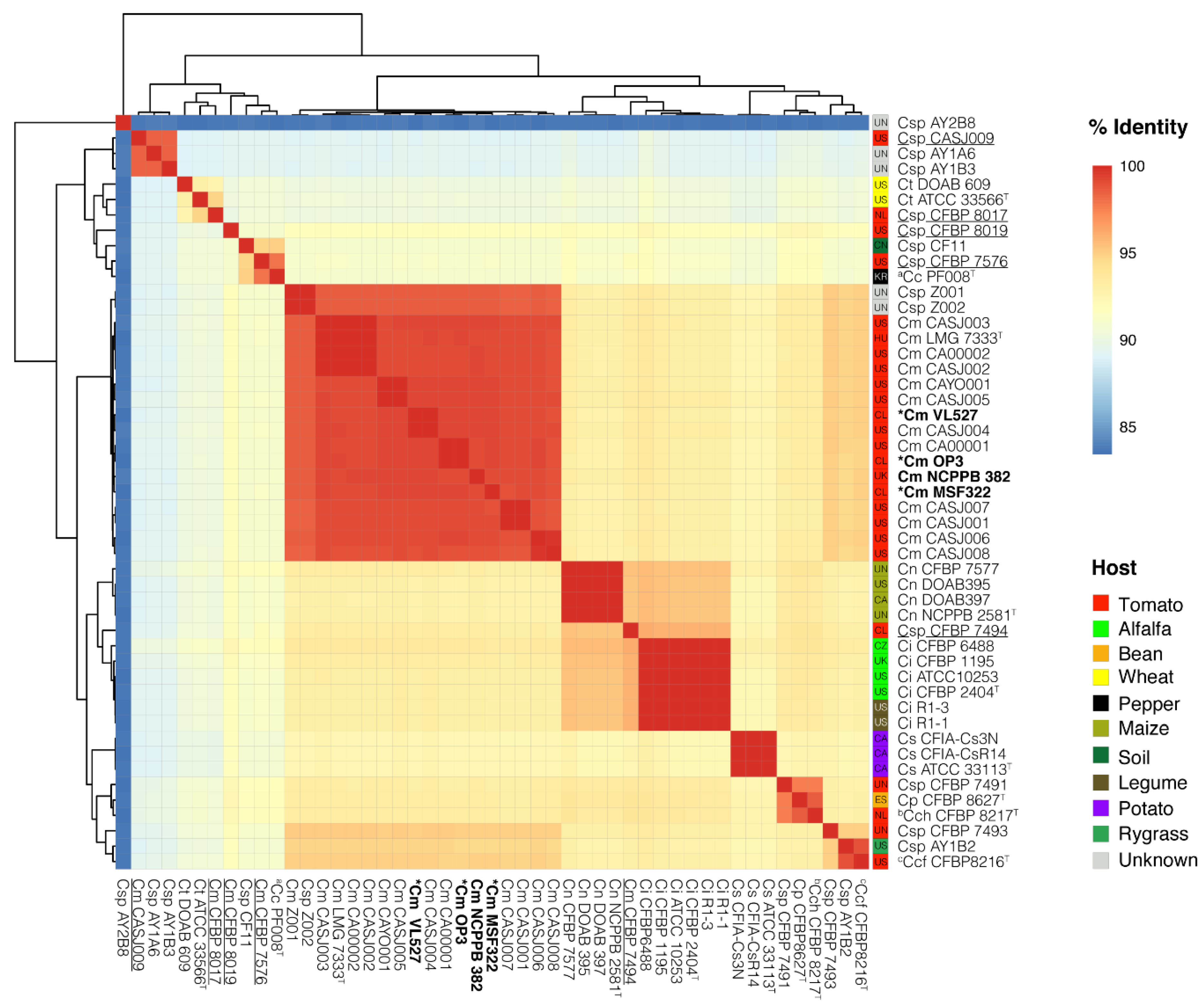
3.5. Phylogenomic Placement of Strains VL527, MSF322 and OP3
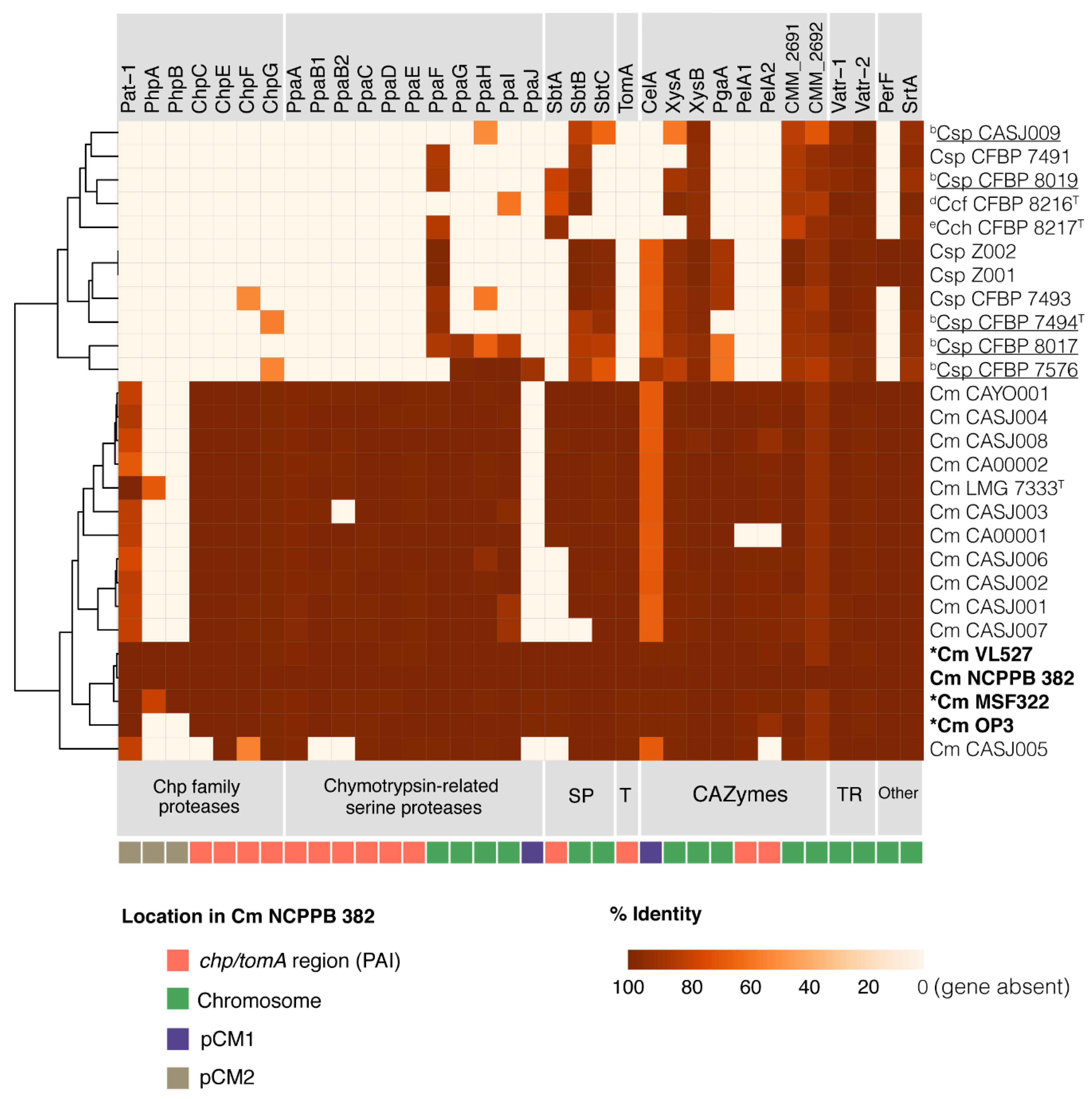
3.6. Search for Potential Virulence Markers in Strains VL527, MSF322 and OP3
3.6.1. Chp Family Proteases
3.6.2. Chymotrypsin-Related Serine Proteases
3.6.3. Subtilase Proteases and Tomatinase
3.6.4. CAZymes
3.6.5. Transcriptional Regulators, Perforine and Sortase
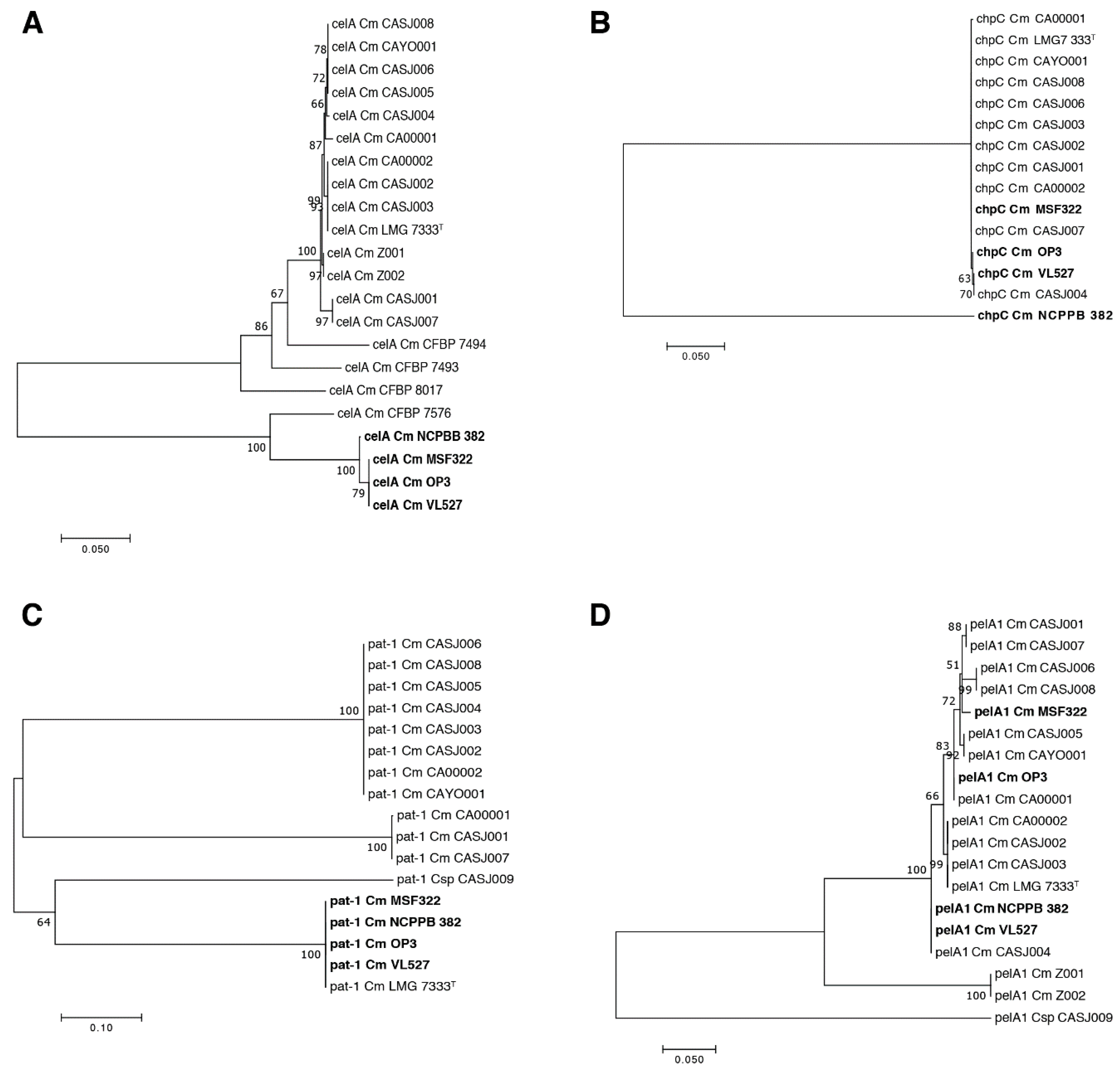
3.7. Genetic Relationships of Virulence Genes in C. michiganensis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Razifard, H.; Ramos, A.; Della-Valle, A.L.; Bodary, C.; Goetz, E.; Manser, E.J.; Li, X.; Zhang, L.; Visa, S.; Tieman, D.; et al. Genomic evidence for complex domestication history of the cultivated tomato in Latin America. Mol. Biol. Evol. 2020, 37, 1118–1132. [Google Scholar] [CrossRef] [PubMed]
- Şen, Y.; van der Wolf, J.; Visser, R.G.F.; van Heusden, S. Bacterial canker of tomato: Current knowledge of detection, management, resistance, and interactions. Plant Dis. 2015, 99, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Tambong, J.T. Comparative genomics of Clavibacter michiganensis subspecies, pathogens of important agricultural crops. PLoS ONE 2017, 12, e0172295. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Tambong, J.; Yuan, K.; Chen, W.; Xu, H.; Lévesque, C.A.; de Boer, S.H. Re-classification of Clavibacter michiganensis subspecies on the basis of whole-genome and multi-locus sequence analyses. Int. J. Syst. Evol. Microbiol. 2018, 68, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.J.; Gillaspie, A.G.; Vidaver, A.K.; Harris, R.W. Clavibacter: A new genus containing some phytopathogenic coryneform bacteria, including Clavibacter xyli subsp. xyli sp. nov., subsp. nov. and Clavibacter xyli subsp. cynodontis subsp. nov., pathogens that cause ratoon stunting disease of sugarcane and bermudagrass stunting disease. Int. J. Syst. Bacteriol. 1984, 34, 107–117. [Google Scholar] [CrossRef]
- González, A.J.; Trapiello, E. Clavibacter michiganensis subsp. phaseoli subsp. nov., pathogenic in bean. Int. J. Syst. Evol. Microbiol. 2014, 64, 1752–1755. [Google Scholar] [CrossRef]
- Oh, E.-J.; Bae, C.; Lee, H.-B.; Hwang, I.S.; Lee, H.-I.; Yea, M.C.; Yim, K.-O.; Lee, S.; Heu, S.; Cha, J.-S.; et al. Clavibacter michiganensis subsp. capsici subsp. nov., causing bacterial canker disease in pepper. Int. J. Syst. Evol. Microbiol. 2016, 66, 4065–4070. [Google Scholar] [CrossRef] [PubMed]
- Yasuhara-Bell, J.; Alvarez, A.M. Seed-associated subspecies of the genus Clavibacter are clearly distinguishable from Clavibacter michiganensis subsp. michiganensis. Int. J. Syst. Evol. Microbiol. 2015, 65, 811–826. [Google Scholar] [CrossRef]
- Nandi, M.; Macdonald, J.; Liu, P.; Weselowski, B.; Yuan, Z.-C. Clavibacter michiganensis ssp. michiganensis: Bacterial canker of tomato, molecular interactions and disease management. Mol. Plant Pathol. 2018, 19, 2036–2050. [Google Scholar] [CrossRef]
- Close, R.; Mulcock, A.P. Bacterial wilt Corynebacterium insidiosum (McCulloch, 1925) Jensen, 1934 of lucerne in New Zealand. New Zealand J. Agric. Res. 1972, 15, 141–148. [Google Scholar] [CrossRef]
- Vidaver, A.K.; Mandel, M. Corynebacterium nebraskense, a new, orange-pigmented phytopathogenic species. Int. J. Syst. Bacteriol. 1974, 24, 482–485. [Google Scholar] [CrossRef]
- Kaemmerer, D.; Seigner, L.; Poschenrieder, G.; Zellner, M.; Munzert, M. Epidemiology of bacterial ring rot of potato in plant and soil–consequences for disease management. J. Plant Dis. Prot. 2007, 114, 159–166. [Google Scholar] [CrossRef]
- Carlson, R.R. Bacterial mosaic, a new corynebacterial disease of wheat. Plant Dis. 1982, 66, 76. [Google Scholar] [CrossRef]
- Carlson, R.R.; Vidaver, A.K. Taxonomy of Corynebacterium plant pathogens, including a new pathogen of wheat, based on polyacrylamide gel electrophoresis of cellular proteins. Int. J. Syst. Bacteriol. 1982, 32, 315–326. [Google Scholar] [CrossRef]
- Lee, S.D.; Yoon, C.M.; Lee, Y.K.; Choi, Y.C.; Cho, Y.S. Occurrence and distribution of bacterial canker of red pepper caused by Clavibacter michiganensis subsp. michiganensis. Plant Dis. Agric. 1999, 5, 105–110. [Google Scholar]
- Yim, K.-O.; Lee, H.-I.; Kim, J.-H.; Lee, S.-D.; Cho, J.-H.; Cha, J.-S. Characterization of phenotypic variants of Clavibacter michiganensis subsp. michiganensis isolated from Capsicum annuum. Eur. J. Plant Pathol. 2011, 133, 559–575. [Google Scholar] [CrossRef][Green Version]
- Gartemann, K.-H.; Kirchner, O.; Engemann, J.; Gräfen, I.; Eichenlaub, R.; Burger, A. Clavibacter michiganensis subsp. michiganensis: First steps in the understanding of virulence of a Gram-positive phytopathogenic bacterium. J. Biotechnol. 2003, 106, 179–191. [Google Scholar] [CrossRef]
- Thapa, S.P.; Pattathil, S.; Hahn, M.G.; Jacques, M.-A.; Gilbertson, R.L.; Coaker, G. Genomic analysis of Clavibacter michiganensis reveals insight into virulence strategies and genetic diversity of a Gram-positive bacterial pathogen. Mol. Plant-Microbe Interact. 2017, 30, 786–802. [Google Scholar] [CrossRef]
- Zaluga, J.; Stragier, P.; Baeyen, S.; Haegeman, A.; van Vaerenbergh, J.; Maes, M.; de Vos, P. Comparative genome analysis of pathogenic and non-pathogenic Clavibacter strains reveals adaptations to their lifestyle. BMC Genom. 2014, 15, 1–14. [Google Scholar] [CrossRef]
- Eichenlaub, R.; Gartemann, K.-H. The Clavibacter michiganensis subspecies: Molecular investigation of Gram-positive bacterial plant pathogens. Annu. Rev. Phytopathol. 2011, 49, 445–464. [Google Scholar] [CrossRef]
- Valenzuela, M.; Besoain, X.; Durand, K.; Cesbron, S.; Fuentes, S.; Claverías, F.; Jacques, M.-A.; Seeger, M. Clavibacter michiganensis subsp. michiganensis strains from Central Chile exhibit low genetic diversity and sequence types match strains in other parts of the world. Plant Pathol. 2018, 67, 1944–1954. [Google Scholar] [CrossRef]
- Jacques, M.-A.; Durand, K.; Orgeur, G.; Balidas, S.; Fricot, C.; Bonneau, S.; Quillévéré, A.; Audusseau, C.; Olivier, V.; Grimault, V.; et al. phylogenetic analysis and polyphasic characterization of Clavibacter michiganensis strains isolated from tomato seeds reveal that nonpathogenic strains are distinct from C. michiganensis subsp. michiganensis. Appl. Environ. Microbiol. 2012, 78, 8388–8402. [Google Scholar] [CrossRef] [PubMed]
- Croce, V.; Pianzzola, M.J.; Durand, K.; González-Arcos, M.; Jacques, M.-A.; Siri, M.I. Multilocus Sequence Typing reveals high variability among Clavibacter michiganensis subsp. michiganensis strains affecting tomato crops in Uruguay. Eur. J. Plant Pathol. 2015, 144, 1–13. [Google Scholar] [CrossRef]
- Chalupowicz, L.; Barash, I.; Reuven, M.; Dror, O.; Sharabani, G.; Gartemann, K.-H.; Eichenlaub, R.; Sessa, G.; Manulis-Sasson, S. Differential contribution of Clavibacter michiganensis ssp. michiganensis virulence factors to systemic and local infection in tomato. Mol. Plant Pathol. 2016, 18, 336–346. [Google Scholar] [CrossRef] [PubMed]
- McKinney, H.H. Influence of soil temperature and moisture on infection of wheat seedlings by Helminthosporium sativum. J. Agric. Res. 1923, 26, 195–225. [Google Scholar]
- Marmur, J. A procedure for the isolation of deoxyribonucleic acid from micro-organisms. J. Mol. Biol. 1961, 3, 208. [Google Scholar] [CrossRef]
- Salvà-Serra, F.; Svensson-Stadler, L.; Busquets, A.; Jaén-Luchoro, D.; Karlsson, R.; Moore, E.R.B.; Gomila, M. A protocol for extraction and purification of high-quality and quantity bacterial DNA applicable for genome sequencing: A modified version of the Marmur procedure. Protoc. Exch. 2018. [Google Scholar] [CrossRef]
- Joshi, N.A.; Fass, J.N. Sickle: A Sliding-Window, Adaptive, Quality-Based Trimming Tool for FastQ Files (Version 1.33). 2011. Available online: https://github.com/najoshi/sickle (accessed on 19 August 2019).
- Wick, R.R.; Judd, L.M.; Gorrie, C.L.; Holt, K.E. Unicycler: Resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput. Biol. 2017, 13, e1005595. [Google Scholar] [CrossRef]
- Koren, S.; Walenz, B.P.; Berlin, K.; Miller, J.R.; Bergman, N.H.; Phillippy, A.M. Canu: Scalable and accurate long-read assembly via adaptive k-mer weighting and repeat separation. Genome Res. 2017, 2017, 1–36. [Google Scholar] [CrossRef]
- Kolmogorov, M.; Yuan, J.; Lin, Y.; Pevzner, P.A. Assembly of long, error-prone reads using repeat graphs. Nat. Biotechnol. 2019, 37, 540–546. [Google Scholar] [CrossRef]
- Gurevich, A.; Saveliev, V.; Vyahhi, N.; Tesler, G. QUAST: Quality assessment tool for genome assemblies. Bioinformatics 2013, 29, 1072–1075. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-H.; Ha, S.-M.; Kwon, S.; Lim, J.; Kim, Y.; Seo, H.; Chun, J. Introducing EzBioCloud: A taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int. J. Syst. Evol. Microbiol. 2017, 67, 1613–1617. [Google Scholar] [CrossRef] [PubMed]
- Tatusova, T.; di Cuccio, M.; Badretdin, A.; Chetvernin, V.; Nawrocki, E.P.; Zaslavsky, L.; Lomsadze, A.; Pruitt, K.D.; Borodovsky, M.; Ostell, J. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 2016, 44, 6614–6624. [Google Scholar] [CrossRef] [PubMed]
- Seemann, T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef]
- Gartemann, K.-H.; Abt, B.; Bekel, T.; Burger, A.; Engemann, J.; Flügel, M.; Gaigalat, L.; Goesmann, A.; Gräfen, I.; Kalinowski, J.; et al. The genome sequence of the tomato-pathogenic actinomycete Clavibacter michiganensis subsp. michiganensis NCPPB382 reveals a large island involved in pathogenicity. J. Bacteriol. 2008, 190, 2138–2149. [Google Scholar] [CrossRef]
- Stothard, P.; Wishart, D.S. Circular genome visualization and exploration using CGView. Bioinformatics 2004, 21, 537–539. [Google Scholar] [CrossRef]
- Galperin, M.Y.; Makarova, K.S.; Wolf, Y.I.; Koonin, E.V. Expanded microbial genome coverage and improved protein family annotation in the COG database. Nucleic Acids Res. 2014, 43, D261–D269. [Google Scholar] [CrossRef]
- Huerta-Cepas, J.; Szklarczyk, D.; Forslund, K.; Cook, H.; Heller, D.; Walter, M.C.; Rattei, T.; Mende, D.R.; Sunagawa, S.; Kuhn, M.; et al. eggNOG 4.5: A hierarchical orthology framework with improved functional annotations for eukaryotic, prokaryotic and viral sequences. Nucleic Acids Res. 2015, 44, D286–D293. [Google Scholar] [CrossRef]
- Huerta-Cepas, J.; Forslund, K.; Coelho, L.P.; Szklarczyk, D.; Jensen, L.J.; von Mering, C.; Bork, P. Fast Genome-wide functional annotation through orthology assignment by eggNOG-Mapper. Mol. Biol. Evol. 2017, 34, 2115–2122. [Google Scholar] [CrossRef]
- Darling, A.E.; Mau, B.; Perna, N.T. Progressive Mauve: Multiple genome alignment with gene gain, loss and rearrangement. PLoS ONE 2010, 5, e11147. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Contreras-Moreira, B.; Vinuesa, P. GET_HOMOLOGUES, a versatile software package for scalable and robust microbial pangenome analysis. Appl. Environ. Microbiol. 2013, 79, 7696–7701. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, D.M.; Kannan, L.; Coleman, M.K.; Wolf, Y.I.; Sorokin, A.; Koonin, E.V.; Mushegian, A. A low-polynomial algorithm for assembling clusters of orthologous groups from intergenomic symmetric best matches. Bioinformatics 2010, 26, 1481–1487. [Google Scholar] [CrossRef]
- Li, L.; Stoeckert, C.J.; Roos, D.S. OrthoMCL: Identification of ortholog groups for eukaryotic genomes. Genome Res. 2003, 13, 2178–2189. [Google Scholar] [CrossRef]
- Pearson, W.R. An introduction to sequence similarity (“homology”) searching. Curr. Protoc. Bioinform. 2013, 42, 3.1.1–3.1.8. [Google Scholar] [CrossRef]
- Durán, R.E.; Méndez, V.; Rodríguez-Castro, L.; Barra-Sanhueza, B.; Salvà-Serra, F.; Moore, E.R.B.; Castro-Nallar, E.; Seeger, M. Genomic and physiological traits of the marine bacterium Alcaligenes aquatilis QD168 Isolated From Quintero Bay, Central Chile, reveal a robust adaptive response to environmental stressors. Front. Microbiol. 2019, 10, 528. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, L.; Glover, R.H.; Humphris, S.; Elphinstone, J.G.; Toth, I.K. Genomics and taxonomy in diagnostics for food security: Soft-rotting enterobacterial plant pathogens. Anal. Methods 2016, 8, 12–24. [Google Scholar] [CrossRef]
- Kolde, R. Pheatmap: Pretty Heatmaps in R Package. R Package, Version 1.0.8. 2015. Available online: https://CRAN.R-project.org/package=pheatmap (accessed on 9 September 2019).
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.S.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 421. [Google Scholar] [CrossRef]
- Chen, C.; Huang, H.; Wu, C.H. Protein bioinformatics databases and resources. Adv. Struct. Safety Stud. 2017, 1558, 3–39. [Google Scholar] [CrossRef]
- El-Gebali, S.; Mistry, J.; Bateman, A.; Eddy, S.R.; Luciani, A.; Potter, S.C.; Qureshi, M.; Richardson, L.J.; A Salazar, G.; Smart, A.; et al. The Pfam protein family’s database in 2019. Nucleic Acids Res. 2018, 47, D427–D432. [Google Scholar] [CrossRef]
- Bertelli, C.; Laird, M.R.; Williams, K.P.; Lau, B.Y.; Hoad, G.; Winsor, G.L.; Brinkman, F.S.L. Simon Fraser University Research Computing Group IslandViewer 4: Expanded prediction of genomic islands for larger-scale datasets. Nucleic Acids Res. 2017, 45, W30–W35. [Google Scholar] [CrossRef] [PubMed]
- Waack, S.; Keller, O.; Asper, R.; Brodag, T.; Damm, C.; Fricke, W.F.; Surovcik, K.; Meinicke, P.; Merkl, R. Score-based prediction of genomic islands in prokaryotic genomes using hidden Markov models. BMC Bioinform. 2006, 7, 142. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, W.; Wan, I.; Jones, S.J.; Brinkman, F.S.L. IslandPath: Aiding detection of genomic islands in prokaryotes. Bioinformatics 2003, 19, 418–420. [Google Scholar] [CrossRef] [PubMed]
- Langille, M.G.I.; Hsiao, W.W.L.; Brinkman, F.S.L. Evaluation of genomic island predictors using a comparative genomics approach. BMC Bioinform. 2008, 9, 329. [Google Scholar] [CrossRef]
- Jia, B.; Raphenya, A.R.; Alcock, B.; Waglechner, N.; Guo, P.; Tsang, K.K.; Lago, B.A.; Dave, B.M.; Pereira, S.; Sharma, A.N.; et al. CARD 2017: Expansion and model-centric curation of the comprehensive antibiotic resistance database. Nucleic Acids Res. 2016, 45, D566–D573. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Tamura, K. Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G+C-content biases. Mol. Biol. Evol. 1992, 9, 678–687. [Google Scholar] [CrossRef]
- Valenzuela, M.; Méndez, V.; Montenegro, I.; Besoain, X.; Seeger, M. Streptomycin resistance in Clavibacter michiganensis subsp. michiganensis strains from Chile is related to a rpsL gene mutation. Plant Pathol. 2018, 68, 426–433. [Google Scholar] [CrossRef]
- Jahr, H.; Dreier, J.; Meletzus, D.; Bahro, R. The endo-beta-1,4-glucanase of Clavibacter michiganensis subsp. michiganensis is a pathogenicity determinant required for the induction of bacterial wilt of tomato. Mol. Plant Microbe Interact. 2000, 13, 703–714. [Google Scholar] [CrossRef]
- Dreier, J.; Meletzus, D.; Eichenlaub, R. Characterization of the plasmid encoded virulence region pat-1 of phytopathogenic Clavibacter michiganensis subsp. michiganensis. Mol. Plant-Microbe Interact. 1997, 10, 195–206. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meletzus, D.; Bermphol, A.; Dreier, J.; Eichenlaub, R. Evidence for plasmid-encoded virulence factors in the phytopathogenic bacterium Clavibacter michiganensis subsp. michiganensis NCPPB382. J. Bacteriol. 1993, 175, 2131–2136. [Google Scholar] [CrossRef] [PubMed]
- Flügel, M.; Becker, A.; Gartemann, K.-H.; Eichenlaub, R. Analysis of the interaction of Clavibacter michiganensis subsp. michiganensis with its host plant tomato by genome-wide expression profiling. J. Biotechnol. 2012, 160, 42–54. [Google Scholar] [CrossRef]
- Burger, A.; Gräfen, I.; Engemann, J.; Niermann, E.; Pieper, M.; Kirchner, O.; Gartemann, K.-H.; Eichenlaub, R. Identification of homologues to the pathogenicity factor Pat-1, a putative serine protease of Clavibacter michiganensis subsp. michiganensis. Microbiol. Res. 2005, 160, 417–427. [Google Scholar] [CrossRef]
- Savidor, A.; Teper, D.; Gartemann, K.-H.; Eichenlaub, R.; Chalupowicz, L.; Manulis-Sasson, S.; Barash, I.; Tews, H.; Mayer, K.; Giannone, R.J.; et al. The Clavibacter michiganensis subsp. michiganensis–tomato interactome reveals the perception of pathogen by the host and suggests mechanisms of infection. J. Proteome Res. 2011, 11, 736–750. [Google Scholar] [CrossRef]
- Kaup, O.; Gräfen, I.; Zellermann, E.-M.; Eichenlaub, R.; Gartemann, K.-H. Identification of a tomatinase in the tomato-pathogenic actinomycete Clavibacter michiganensis subsp. michiganensis NCPPB382. Mol. Plant-Microbe Interact. 2005, 18, 1090–1098. [Google Scholar] [CrossRef]
- Stork, I.; Gartemann, K.-H.; Burger, A.; Eichenlaub, R. A family of serine proteases of Clavibacter michiganensis subsp. michiganensis: ChpCplays a role in colonization of the host plant tomato. Mol. Plant Pathol. 2008, 9, 599–608. [Google Scholar] [CrossRef]
- Pareja-Jaime, Y.; Roncero, M.I.G.; Ruiz-Roldán, M.C. Tomatinase from Fusarium oxysporum f. sp. lycopersici is required for full virulence on tomato plants. Mol. Plant-Microbe Interact. 2008, 21, 728–736. [Google Scholar] [CrossRef]
- Chalupowicz, L.; Cohen-Kandli, M.; Dror, O.; Eichenlaub, R.; Gartemann, K.-H.; Sessa, G.; Barash, I.; Manulis-Sasson, S. Sequential expression of bacterial virulence and plant defense genes during infection of tomato with Clavibacter michiganensis subsp. michiganensis. Phytopathology 2010, 100, 252–261. [Google Scholar] [CrossRef]
- Richter, M.; Rosselló-Móra, R. Shifting the genomic gold standard for the prokaryotic species definition. Proc. Natl. Acad. Sci. USA 2009, 106, 19126–19131. [Google Scholar] [CrossRef]
- Osdaghi, E.; Rahimi, T.; Taghavi, S.M.; Ansari, M.; Zarei, S.; Portier, P.; Briand, M.; Jacques, M.-A. Comparative genomics and phylogenetic analyses suggest several novel species within the genus Clavibacter, including nonpathogenic tomato-associated strains. Appl. Environ. Microbiol. 2020, 86, 02873. [Google Scholar] [CrossRef] [PubMed]

| C. michiganensis Subspecies (Date) | Type Strain | Disease | Host | Reclassification (Date) | Reference | ||
|---|---|---|---|---|---|---|---|
| C. michiganensis subsp. michiganensis (1984) | LMG 7333T (=NCPPB 2979T) | Canker | Tomato (Solanum lycopersicum) | [2,5,9] | |||
| C. michiganensis subsp. insidiosus (1984) | CFBP 2404T (=LMG 3663 = NCPPB 1109T) | Wilt and stunting | Alfalfa/lucerne (Medicago sativa) | C. insidiosus (2018) | [4,5,10] | ||
| C. michiganensis subsp. nebraskensis (1984) | NCPPB 2581T | Wilt and leaf blight | Maize/corn (Zea mays) | C. nebraskensis (2018) | [4,5,11] | ||
| C. michiganensis subsp. sepedonicus (1984) | ATCC 33113T (=NCPPB 2137T) | Ring-rot | Potato (Solanum tuberosum) | C. seponicus (2018) | [4,5,12] | ||
| C. michiganensis subsp. tessellarius (1984) | ATCC 33566T (=NCPPB 3664T) | Mosaic disease | Wheat (Triticum aestivum) | C. tessellarius (2018) | [4,5,13,14] | ||
| C. michiganensis subsp. phaseoli (2014) | CFBP 8627T (=LMG 27667T) | Leaf yellowing | Bean (Phaseolus vulgaris) | C. phaseoli (2020) | [6], this study | ||
| C. michiganensis subsp. californiensis (2015) | CFBP 8216T | Asymptomatic | Tomato (Solanum lycopersicum) | C. californiensis (2020) | [8,15,16], this study | ||
| C. michiganensis subsp. chilensis (2015) | CFBP 8217T | Asymptomatic | Tomato (Solanum lycopersicum) | C. phaseoli (2020) | [8,15,16], this study | ||
| C. michiganensis subsp. capsici (2016) | PF 008T (=LMG 29047T) | Canker | Pepper (Capsicum spp.) | C. capsici (2018) | [4,7,15,16] | ||
| Inoculum | * Disease Index (DI) |
|---|---|
| strain VL527 | 65.33 ± 2.31 b |
| strain MSF322 | 58.67 ± 4.62 a,b |
| strain OP3 | 48.00 ± 8.00 a |
| Uninoculated (control) | 0 |
| a Features | VL527 | MSF322 | OP3 |
|---|---|---|---|
| GenBank Acc. No. | CP047054–CP047055 | CP047051–CP047053 | WTCS00000000 |
| Finishing quality | Closed genome | Closed genome | Draft |
| Sequencing platforms | Illumina + Oxford Nanopore | Illumina + Oxford Nanopore | Illumina + Oxford Nanopore |
| Assembler | Unicycler (v0.4.7) | Unicycler (v0.4.7) | Unicycler (v0.4.7) |
| Annotation pipeline | PGAP (v4.10) | PGAP (v4.10) | PGAP (v4.10) |
| N50 (bp) | 3,321,579 | 3,284,014 | 3,189,274 |
| Chromosome (bp) | 3,321,579 | 3,284,014 | 3,189,218 |
| Plasmids | 75,053 bp (pVL2) | 38,824 bp (pMSF1); 76,361 bp (pMSF2) | |
| Contigs (No., bp) | 5 contigs; Contig 1, 3,189,274 Contig 2, 131,602 Contig 3, 73,139 (pOP2) Contig 4, 38,824 (pOP3) Contig 5, 33,265 (pOP1) | ||
| Total length (bp) | 3,396,632 | 3,399,199 | 3,466,104 |
| G + C content (%) | 72.6 | 72.6 | 72.4 |
| CDS | 3,164 | 3,117 | 3,174 |
| Pseudogenes (total) | 41 | 45 | 55 |
| rRNA | 2 × 5S, 2 × 16S, 2 × 23S | 2 × 5S, 2 × 16S, 2 × 23S | 2 × 5S, 2 × 16S, 2 × 23S |
| tRNA | 46 | 45 | 45 |
| CRISPR arrays | 1 | 0 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Méndez, V.; Valenzuela, M.; Salvà-Serra, F.; Jaén-Luchoro, D.; Besoain, X.; Moore, E.R.B.; Seeger, M. Comparative Genomics of Pathogenic Clavibacter michiganensis subsp. michiganensis Strains from Chile Reveals Potential Virulence Features for Tomato Plants. Microorganisms 2020, 8, 1679. https://doi.org/10.3390/microorganisms8111679
Méndez V, Valenzuela M, Salvà-Serra F, Jaén-Luchoro D, Besoain X, Moore ERB, Seeger M. Comparative Genomics of Pathogenic Clavibacter michiganensis subsp. michiganensis Strains from Chile Reveals Potential Virulence Features for Tomato Plants. Microorganisms. 2020; 8(11):1679. https://doi.org/10.3390/microorganisms8111679
Chicago/Turabian StyleMéndez, Valentina, Miryam Valenzuela, Francisco Salvà-Serra, Daniel Jaén-Luchoro, Ximena Besoain, Edward R. B. Moore, and Michael Seeger. 2020. "Comparative Genomics of Pathogenic Clavibacter michiganensis subsp. michiganensis Strains from Chile Reveals Potential Virulence Features for Tomato Plants" Microorganisms 8, no. 11: 1679. https://doi.org/10.3390/microorganisms8111679
APA StyleMéndez, V., Valenzuela, M., Salvà-Serra, F., Jaén-Luchoro, D., Besoain, X., Moore, E. R. B., & Seeger, M. (2020). Comparative Genomics of Pathogenic Clavibacter michiganensis subsp. michiganensis Strains from Chile Reveals Potential Virulence Features for Tomato Plants. Microorganisms, 8(11), 1679. https://doi.org/10.3390/microorganisms8111679








