Evaluation of Physiological Effects Induced by Manuka Honey Upon Staphylococcus aureus and Escherichia coli
Abstract
1. Introduction
2. Results
2.1. Manuka Honey-Induced Alterations in S. aureus CECT 86 and E. coli CECT 515
2.1.1. Effects of MkH on Cell Viability
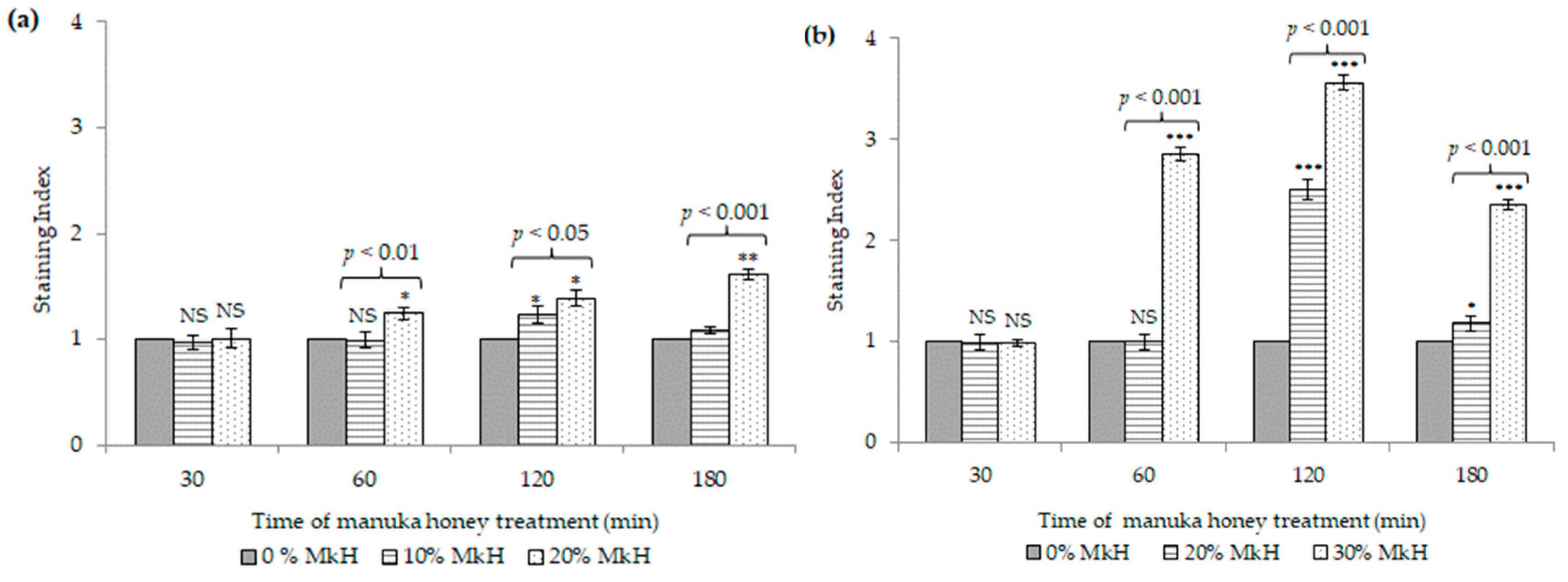
2.1.2. Effects of MkH on Cytoplasmic Membrane Potential
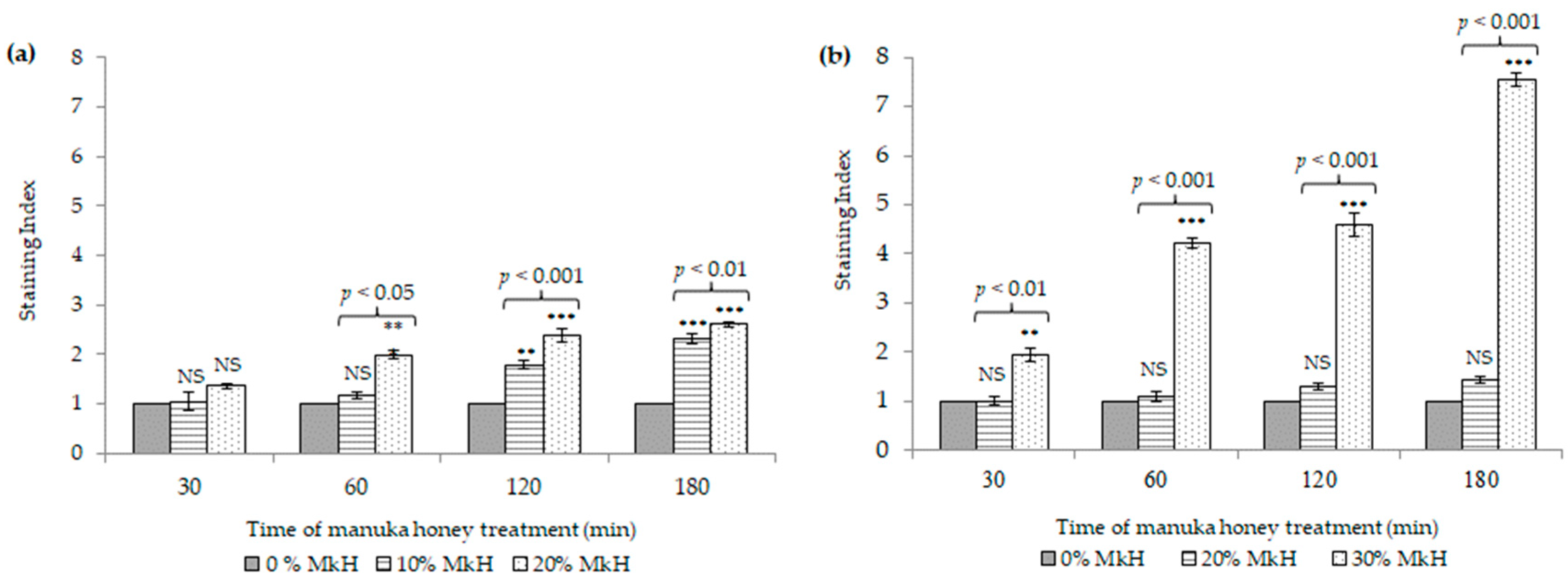
2.1.3. Effects of MkH on Membrane Integrity
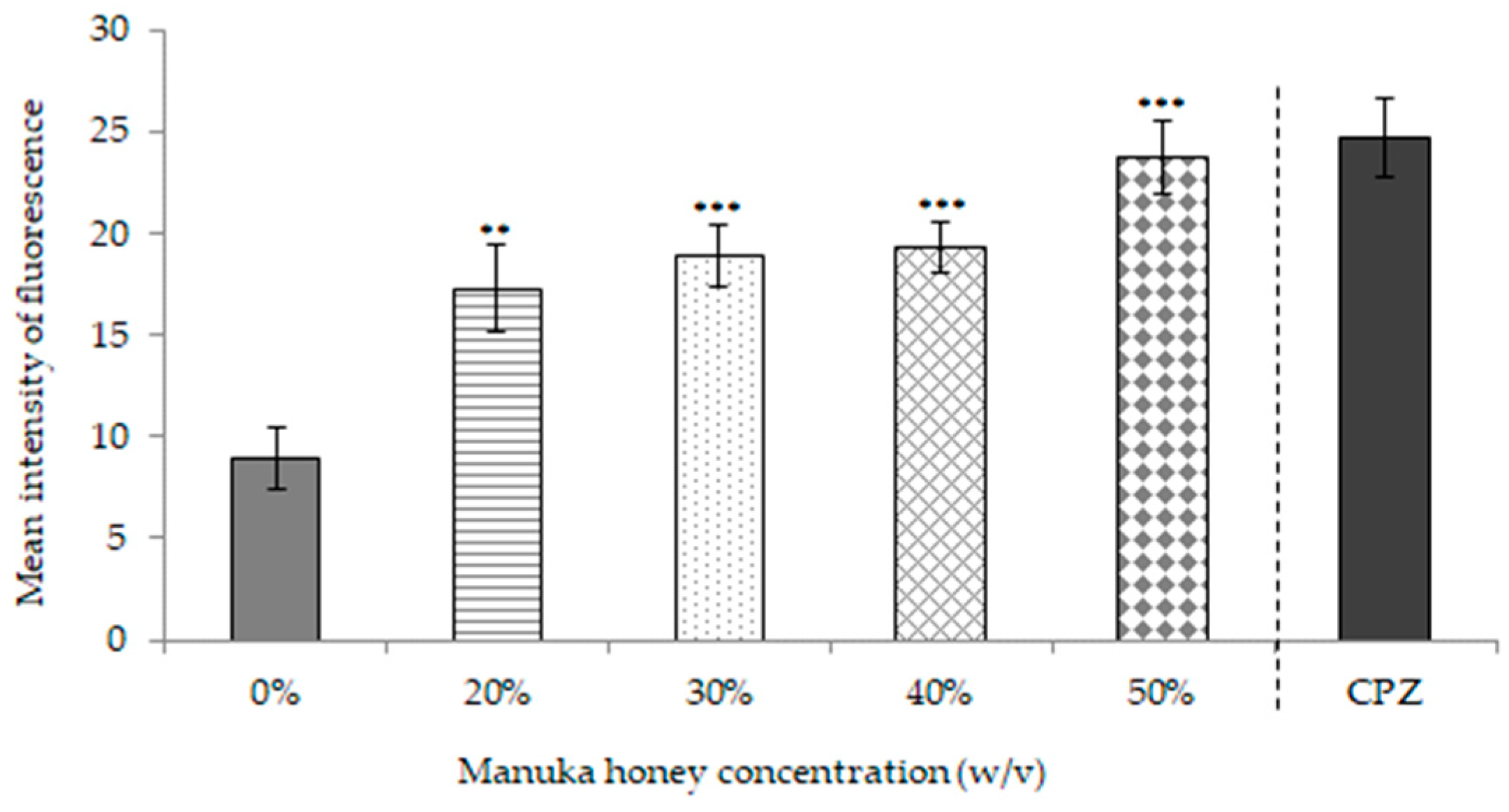
2.1.4. Effects of MkH on Metabolic Activity
2.2. Manuka Honey Effect on Efflux Pumps in E. coli AG100TET
3. Discussion
4. Conclusions
5. Materials and Methods
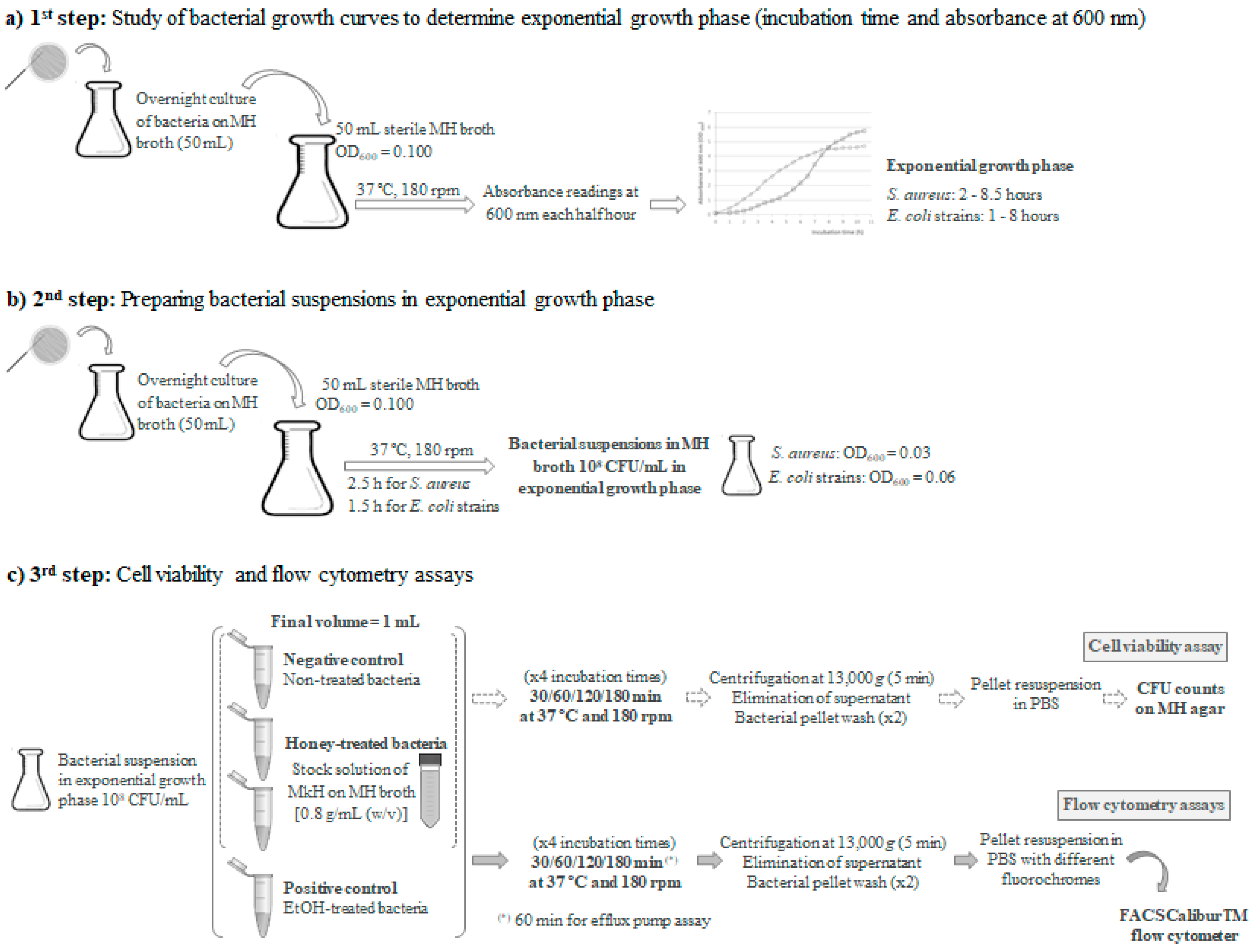
5.1. Bacterial Strains, Growth Conditions and Inoculated Broth Preparation
5.2. Bacterial Susceptibility to Manuka Honey
5.3. Evaluation of Cell Viability
5.4. Functional Characterization of Manuka Honey-Induced Action
5.4.1. Assessment of Membrane Potential
5.4.2. Assessment of Membrane Integrity
5.4.3. Assessment of Metabolic Activity
5.4.4. Assessment of Efflux Pumps
5.5. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Health 2015, 109, 309–318. [Google Scholar] [PubMed]
- Founou, L.L.; Founou, R.C.; Essack, S.Y. Antibiotic resistance in the food chain: A developing country-perspective. Front. Microbiol. 2016, 7, 1881. [Google Scholar]
- European Centre for Disease Prevention and Control (ECDC). Surveillance of antimicrobial resistance in Europe (2017). Annual report of the European Antimicrobial Resistance Surveillance Network (EARS-Net); ECDC: Stockholm, Sweden, 2018; ISBN 978-2-85653-642-1. [Google Scholar]
- Oniciuc, E.A.; Likotrafiti, E.; Alvarez-Molina, A.; Prieto, M.; López, M.; Alvarez-Ordóñez, A. Food processing as a risk factor for antimicrobial resistance spread along the food chain. Curr. Opin. Food Sci. 2019, 30, 21–26. [Google Scholar] [CrossRef]
- Blair, J.M.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J.V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2014, 13, 42–51. [Google Scholar] [PubMed]
- World Health Organization (WHO). 2017. Available online: https://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en/WHO (accessed on 13 August 2019).
- Lukasiewicz, M.; Kowalski, S.; Makarewicz, M. Antimicrobial an antioxidant activity of selected Polish herbhoneys. LWT-Food Sci. Technol. 2015, 64, 547–553. [Google Scholar] [CrossRef]
- Fyfe, L.; Okoro, P.; Paterson, E.; Coyle, S.; McDougall, G.J. Compositional analysis of Scottish honeys with antimicrobial activity against antibiotic-resistant bacteria reveals novel antimicrobial components. LWT-Food Sci. Technol. 2017, 79, 52–59. [Google Scholar] [CrossRef]
- Carter, D.A.; Blair, S.E.; Cokcetin, N.N.; Bouzo, D.; Brooks, P.; Schothauer, R.; Harry, E.J. Therapeutic Manuka Honey: No Longer So Alternative. Front. Microbiol. 2016, 7, 569. [Google Scholar] [PubMed]
- Jenkins, R.; Burton, N.; Cooper, R. Proteomic and genomic analysis of methicillin-resistant Staphylococcus aureus (MRSA) exposed to manuka honey in vitro demonstrated down-regulation of virulence markers. J. Antimicrob. Chemother. 2014, 69, 603–615. [Google Scholar]
- Packer, J.M.; Irish, J.; Herbert, B.R.; Hill, C.; Padula, M.; Blair, S.E.; Carter, D.A.; Harry, E.J. Specific non-peroxide antibacterial effect of manuka honey on the Staphylococcus aureus proteome. Int. J. Antimicrob. Agents 2012, 40, 43–50. [Google Scholar] [CrossRef]
- Henriques, A.F.; Jenkins, R.E.; Burton, N.F.; Cooper, R.A. The intracellular effects of manuka honey on Staphylococcus aureus. Eur. J. Clin. Microbiol. Infect. Dis. 2010, 29, 45–50. [Google Scholar]
- Liu, M.; Lu, J.; Müller, P.; Turnbull, L.; Burke, C.M.; Schlothauer, R.C.; Carter, D.A.; Whitchurch, C.B.; Harry, E.J. Antibiotic-specific differences in the response of Staphylococcus aureus to treatment with antimicrobials combined with manuka honey. Front. Microbiol. 2015, 5, 779. [Google Scholar] [PubMed]
- Teixeira-Santos, R.; Ricardo, E.; Guerreiro, S.G.; Costa-de-Oliveira, S.; Rodrigues, A.G.; Pina-Vaz, C. New insights regarding yeast survival following exposure to liposomal amphotericin B. Antimicrob. Agents Chemother. 2015, 59, 6181–6187. [Google Scholar] [CrossRef] [PubMed]
- Pina-Vaz, C.; Silva, A.P.; Faria-Ramos, I.; Teixeira-Santos, R.; Moura, D.; Vieira, T.F.; Sousa, S.F.; Costa-de-Oliveira, S.; Cantón, R.; Rodrigues, A.G. A Flow Cytometric and Computational Approaches to Carbapenems Affinity to the Different Types of Carbapenemases. Front. Microbiol. 2016, 7, 1259. [Google Scholar] [PubMed]
- Liu, F.; Wang, F.; Du, L.; Zhao, T.; Doyle, M.P.; Wang, D.; Zhang, X.; Sun, Z.; Xu, W. Antibacterial and antibiofilm activity of phenyllactic acid against Enterobacter cloacae. Food Control 2018, 84, 442–448. [Google Scholar]
- Kumar, G.; Degheidy, H.; Casey, B.J.; Goering, P.L. Flow cytometry evaluation of in vitro cellular necrosis and apoptosis induced by silver nanoparticles. Food Chem. Toxicol. 2015, 85, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Barrientos, A.; Arroyo, J.; Cantón, R.; Nombela, C.; Sánchez-Pérez, M. Applications of Flow Cytometry to Clinical Microbiology. Clin. Microbiol. Rev. 2000, 13, 167–195. [Google Scholar]
- Wilkinson, M.G. Flow cytometry as a potential method of measuring bacterial viability in probiotic products: A review. Trends Food Sci. Technol. 2018, 78, 1–10. [Google Scholar]
- Tracy, B.P.; Gaida, S.M.; Papoutsakis, E.T. Flow cytometry for bacteria: Enabling metabolic engineering, synthetic biology and the elucidation of complex phenotypes. Anal. Biotechnol. 2010, 21, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Faria-Ramos, I.; Costa-de-Oliveira, S.; Barbosa, J.; Cardoso, A.; Santos-Antunes, J.; Rodrigues, A.G.; Pina-Vaz, C. Detection of Legionella pneumophila on clinical samples and susceptibility assessment by flow cytometry. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 3351–3357. [Google Scholar] [PubMed]
- Sträuber, H.; Müller, S. Viability states of bacteria-Specific mechanisms of selected probes. Cytom. Part A 2010, 77A, 623–634. [Google Scholar]
- Blair, S.E.; Cokcetin, N.N.; Harry, E.J.; Carter, D.A. The unusual antibacterial activity of medical-grade Leptospermum honey: Antibacterial spectrum, resistance and transcriptome analysis. Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 1199–1208. [Google Scholar] [PubMed]
- Jenkins, R.; Burton, N.; Cooper, R. Effect of manuka honey on the expression of universal stress protein A in meticillin-resistant Staphylococcus aureus. Int. J. Antimicrob. Agents 2011, 37, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, R.; Burton, N.; Cooper, R. Manuka honey inhibits cell division in methicillin-resistant Staphylococcus aureus. J. Antimicrob. Chemother. 2011, 66, 2536–2542. [Google Scholar] [PubMed]
- Henriques, A.F.; Jenkins, R.E.; Burton, N.F.; Cooper, R.A. The effect of manuka honey on the structure of Pseudomonas aeruginosa. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.E.L.; Maddocks, S.E.; Cooper, R.A. Manuka honey is bactericidal against Pseudomonas aeruginosa and results in differential expression of oprF and algD. Microbiology 2012, 158, 3005–3013. [Google Scholar] [PubMed]
- Straatsma, T.P.; Soares, T.A. Characterization of the Outer Membrane Protein OprF of Pseudomonas aeruginosa in a Lipopolysaccharide Membrane by Computer Simulation. Proteins 2009, 74, 475–488. [Google Scholar] [PubMed]
- Comas-Riu, J.; Vives-Rego, J. Use of calcein and SYTO-13 to assess cell cycle phases and osmotic shock effects on E. coli and Staphylococcus aureus by flow cytometry. J. Microbiol. Methods 1999, 34, 215–221. [Google Scholar] [CrossRef]
- Kronda, J.M.; Cooper, R.A.; Maddocks, S.E. Manuka honey inhibits siderophore production in Pseudomonas aeruginosa. J. Appl. Microbiol. 2013, 115, 86–90. [Google Scholar] [CrossRef]
- Yilmaz, M.T.; Tatlisu, N.B.; Toker, O.S.; Karaman, S.; Dertli, E.; Sagdic, O.; Arici, M. Steady, dynamic and creep rheological analysis as a novel approach to detect honey adulteration by fructose and saccharose syrups: Correlations with HPLC-RID results. Food Res. Int. 2014, 64, 634–646. [Google Scholar]
- Andersen, J.L.; He, G.-X.; Kakarla, P.; Ranjana, K.C.; Kumar, S.; Lakra, W.S.; Mukherjee, M.M.; Ranaweera, I.; Shrestha, U.; Tran, T.; et al. Multidrug Efflux Pumps from Enterobacteriaceae, Vibrio cholerae and Staphylococcus aureus Bacterial Food Pathogens. Int. J. Environ. Res. Public Health 2015, 12, 1487–1547. [Google Scholar]
- Jenkins, R.; Cooper, R. Improving Antibiotic Activity against Wound Pathogens with Manuka Honey In Vitro. PLoS ONE 2012, 7, e45600. [Google Scholar]
- Müller, P.; Alber, D.G.; Turnbull, L.; Schlothauer, R.C.; Carter, D.A.; Whitchurch, C.B.; Harry, E.J. Synergism between Medihoney and Rifampicin against Methicillin-Resistant Staphylococcus aureus (MRSA). PLoS ONE 2013, 8, e57679. [Google Scholar]
- Liu, M.Y.; Cokcetin, N.N.; Lu, J.; Turnbull, L.; Carter, D.A.; Whitchurch, C.B.; Harry, E.J. Rifampicin-Manuka Honey Combinations Are Superior to Other Antibiotic-Manuka Honey Combinations in Eradicating Staphylococcus aureus Biofilms. Front. Microbiol. 2018, 8, 2653. [Google Scholar] [PubMed]
- Cooper, R.A.; Jenkins, L.; Henriques, A.F.M.; Duggan, R.S.; Burton, N.F. Absence of bacterial resistance to medical-grade manuka honey. Eur. J. Clin. Microbiol. Infect. Dis. 2010, 29, 1237–1241. [Google Scholar] [PubMed]
- Paixão, L.; Rodrigues, L.; Couto, I.; Martins, M.; Fernandes, P.; de Carvalho, C.C.C.R.; Monteiro, G.A.; Sansonetty, F.; Amaral, L.; Viveiros, M. Fluorometric determination of ethidium bromide efflux kinetics in Escherichia coli. J. Biol. Eng. 2009, 3, 18. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard—CLSI document M07-A9., 9th ed.; CLSI Wayne: West Valley, PA, USA, 2012. [Google Scholar]
- Combarros-Fuertes, P.; Estevinho, L.M.; Dias, L.G.; Castro, J.M.; Tomás-Barberán, F.A.; Tornadijo, M.E.; Fresno-Baro, J.M. Bioactive Components and Antioxidant and Antibacterial Activities of Different Varieties of Honey: A Screening Prior to Clinical Application. J. Agric. Food Chem. 2019, 67, 688–698. [Google Scholar] [PubMed]
- Teixeira-Santos, R.; Ricardo, E.; Branco, R.J.; Azevedo, M.M.; Rodrigues, A.G.; Pina-Vaz, C. Unveiling the Synergistic Interaction Between Liposomal Amphotericin B and Colistin. Front. Microbiol. 2016, 7, 1439. [Google Scholar] [PubMed]
- Faria-Ramos, I.; Espinar, M.J.; Rocha, R.; Santos-Antunes, J.; Rodrigues, A.G.; Canton, R.; Pina-Vaz, C. A novel flow cytometric assay for rapid detection of extended-spectrum beta-lactamases. Clin. Microbiol. Infect. 2013, 19, E8–E15. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Combarros-Fuertes, P.; Estevinho, L.M.; Teixeira-Santos, R.; Rodrigues, A.G.; Pina-Vaz, C.; Fresno, J.M.; Tornadijo, M.E. Evaluation of Physiological Effects Induced by Manuka Honey Upon Staphylococcus aureus and Escherichia coli. Microorganisms 2019, 7, 258. https://doi.org/10.3390/microorganisms7080258
Combarros-Fuertes P, Estevinho LM, Teixeira-Santos R, Rodrigues AG, Pina-Vaz C, Fresno JM, Tornadijo ME. Evaluation of Physiological Effects Induced by Manuka Honey Upon Staphylococcus aureus and Escherichia coli. Microorganisms. 2019; 7(8):258. https://doi.org/10.3390/microorganisms7080258
Chicago/Turabian StyleCombarros-Fuertes, Patricia, Leticia M. Estevinho, Rita Teixeira-Santos, Acácio G. Rodrigues, Cidália Pina-Vaz, Jose M. Fresno, and M. Eugenia Tornadijo. 2019. "Evaluation of Physiological Effects Induced by Manuka Honey Upon Staphylococcus aureus and Escherichia coli" Microorganisms 7, no. 8: 258. https://doi.org/10.3390/microorganisms7080258
APA StyleCombarros-Fuertes, P., Estevinho, L. M., Teixeira-Santos, R., Rodrigues, A. G., Pina-Vaz, C., Fresno, J. M., & Tornadijo, M. E. (2019). Evaluation of Physiological Effects Induced by Manuka Honey Upon Staphylococcus aureus and Escherichia coli. Microorganisms, 7(8), 258. https://doi.org/10.3390/microorganisms7080258






