Microbiological Testing for the Proper Assessment of the Hygiene Status of Beef Carcasses
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of the Slaughterhouses
2.2. Sampling
2.3. Enumeration of Hygiene Indicator Microorganisms
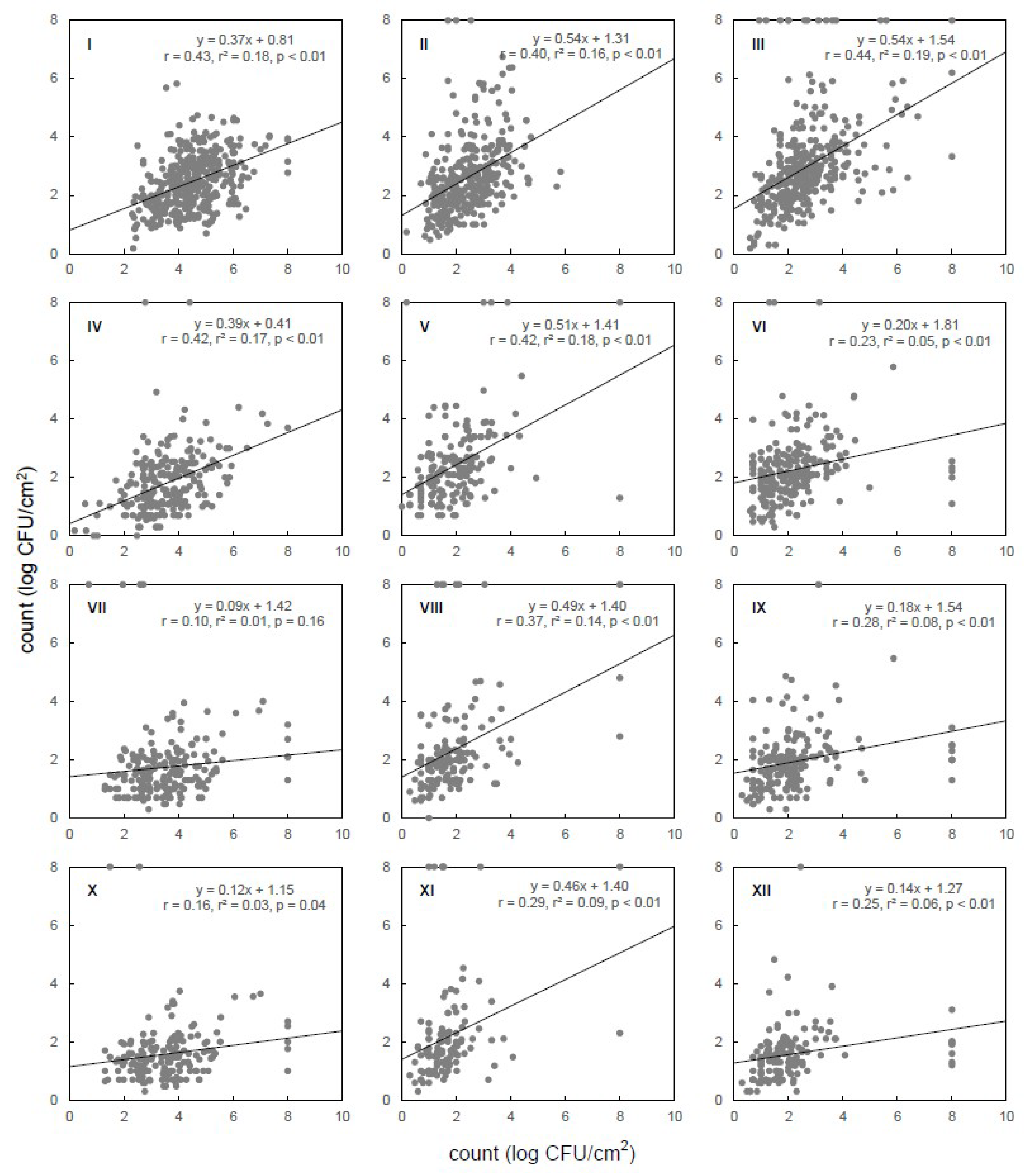
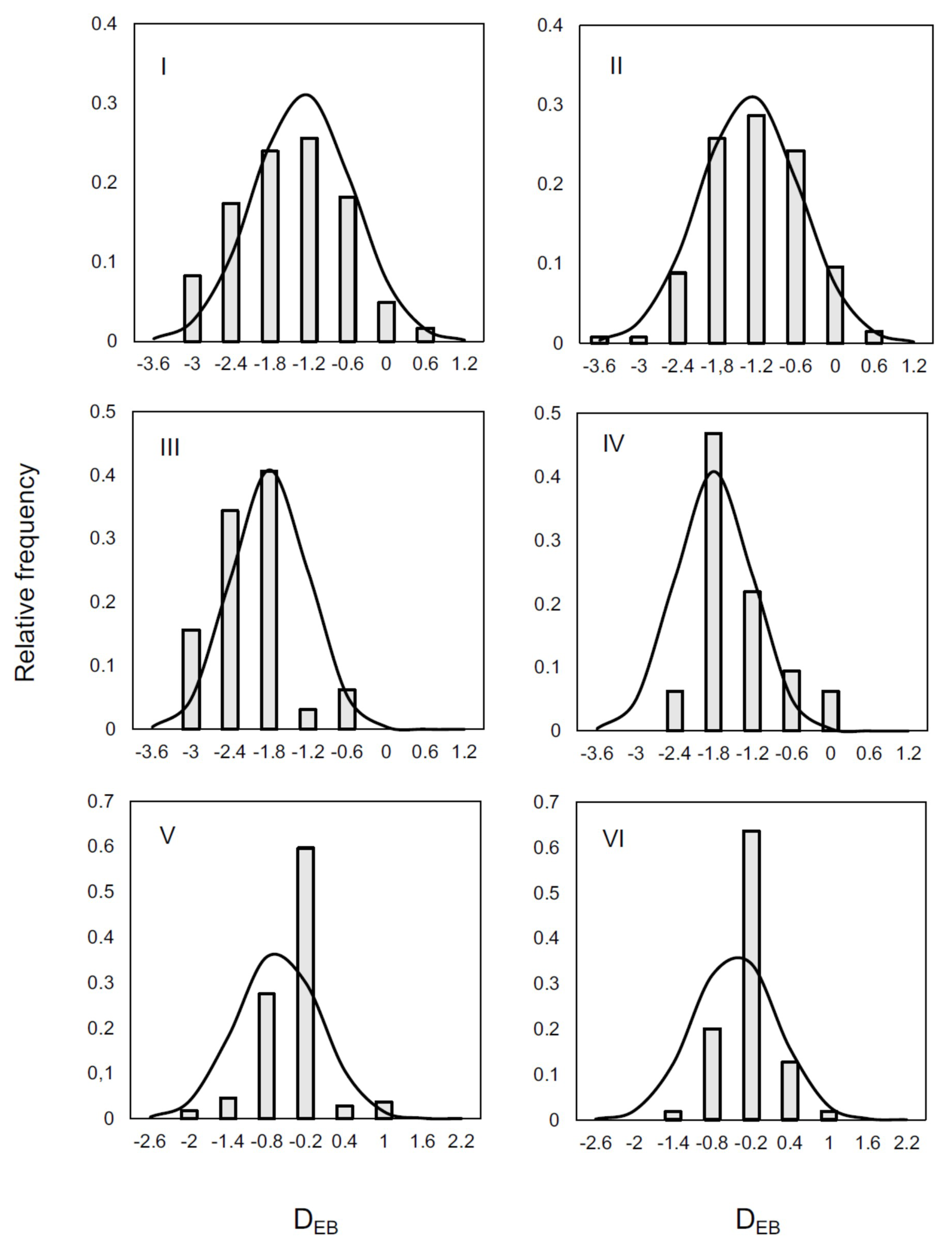
2.4. Statistical Analysis
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- ABIEC. Perfil da pecuária no Brasil—Profile of Brazilian Cattle Production. Available online: http://abiec.siteoficial.ws/images/upload/sumario-pt-010217.pdf (accessed on 11 January 2019).
- Brasil. Ministério da Indústria, Comércio Exterior e Serviços. Available online: http://www.mdic.gov.br/ (accessed on 11 January 2019).
- Neeliah, S.A.; Neeliah, H.; Goburdhun, D. Assessing the relevance of EU SPS measures to the food export sector: Evidence from a developing agro-food exporting country. Food Policy 2013, 41, 53–62. [Google Scholar] [CrossRef]
- Zach, L.; Doyle, M.E.; Bier, V.; Czuprynski, C. Systems and governance in food import safety: A U.S. perspective. Food Control 2012, 27, 153–162. [Google Scholar] [CrossRef]
- Lafisca, A.; Almeida, M.V.; Pinto, P.S.A.; Nero, L.A. European food safety requirements leading to the development of Brazilian cattle sanity and beef safety. Eur. Food Feed Law Rev. 2013, 8, 259–269. [Google Scholar]
- Peran i Sala, R.M.; Cedeño de Balabarca, V.; Etoundi, J.M.; Odame-Darkwah, J.; Oppong-Otoo, J.; Tossougbo Hinson, D.C.; Wouafo, M. Establishment of good hygiene practice-based microbiological criteria in food industries: Guidelines using an example for meat preparations. Food Control 2015, 58, 7–11. [Google Scholar] [CrossRef]
- Kafetzopoulos, D.P.; Psomas, E.L.; Kafetzopoulos, P.D. Measuring the effectiveness of the HACCP Food Safety Management System. Food Control 2013, 33, 505–513. [Google Scholar] [CrossRef]
- Alvseike, O.; Røssvoll, E.; Røtterud, O.-J.; Nesbakken, T.; Skjerve, E.; Prieto, M.; Sandberg, M.; Johannessen, G.; Økland, M.; Urdahl, A.M.; et al. Slaughter hygiene in European cattle and sheep abattoirs assessed by microbiological testing and Hygiene Performance Rating. Food Control 2019, 101, 233–240. [Google Scholar] [CrossRef]
- ICMSF. Microorganisms in Foods 8: Use of Data for Assessing Process Control and Product Acceptance; International Commission on Microbiological Specifications for Foods: New York, NY, USA, 2011; Volume 8, p. 400. [Google Scholar]
- Ray, B.; Bhunia, A. Fundamental Food Microbiology; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Luning, P.A.; Jacxsens, L.; Rovira, J.; Osés, S.M.; Uyttendaele, M.; Marcelis, W.J. A concurrent diagnosis of microbiological food safety output and food safety management system performance: Cases from meat processing industries. Food Control 2011, 22, 555–565. [Google Scholar] [CrossRef]
- Buncic, S.; Nychas, G.J.; Lee, M.R.F.; Koutsoumanis, K.; Hébraud, M.; Desvaux, M.; Chorianopoulos, N.; Bolton, D.; Blagojevic, B.; Antic, D. Microbial pathogen control in the beef chain: Recent research advances. Meat Sci. 2014, 97, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Barco, L.; Belluco, S.; Roccato, A.; Ricci, A. A systematic review of studies on Escherichia coli and Enterobacteriaceae on beef carcasses at the slaughterhouse. Int. J. Food Microbiol. 2015, 207, 30–39. [Google Scholar] [CrossRef]
- Brown, M.H.; Gill, C.O.; Hollingsworth, J.; Nickelson, R.; Seward, S.; Sheridan, J.J.; Stevenson, T.; Sumner, J.L.; Theno, D.M.; Usborne, W.R.; et al. The role of microbiological testing in systems for assuring the safety of beef. Int. J. Food Microbiol. 2000, 62, 7–16. [Google Scholar] [CrossRef]
- Raspor, P. Total food chain safety: How good practices can contribute? Trends Food Sci. Technol. 2008, 19, 405–412. [Google Scholar] [CrossRef]
- Brasil. Circular Nº 175/2005/CGPE/DIPOA—Procedimentos de Verificação dos Programas de Autocontrole. In MAPA; MAPA: Brasília, Brazil, 2005. [Google Scholar]
- Wilhelm, B.; Rajić, A.; Greig, J.D.; Waddell, L.; Harris, J. The effect of hazard analysis critical control point programs on microbial contamination of carcasses in abattoirs: A systematic review of published data. Foodborne Pathog. Dis. 2011, 8, 949–960. [Google Scholar] [CrossRef] [PubMed]
- EU. Commission Regulation (EC) No 1441/2007 amending Regulation (EC) No 2073/2005 on microbiological criteria for foodstuffs. Eur. Union Off. J. Eur. Union 2007, 332, 12–29. [Google Scholar]
- Camargo, A.C.; Lafisca, A.; Cossi, M.V.C.; Lanna, F.G.P.A.; Dias, M.R.; Pinto, P.S.A.; Nero, L.A. Low occurrence of Listeria monocytogenes on bovine hides and carcasses in Minas Gerais state, Brazil: Molecular characterization and antimicrobial resistance. J. Food Prot. 2014, 77, 1148–1152. [Google Scholar] [CrossRef] [PubMed]
- Cossi, M.V.C.; Burin, R.C.K.; Camargo, A.C.; Dias, M.R.; Lanna, F.G.P.A.; Pinto, P.S.A.; Nero, L.A. Low occurrence of Salmonella in the beef processing chain from Minas Gerais state, Brazil: From bovine hides to end cuts. Food Control 2014, 40, 320–323. [Google Scholar] [CrossRef]
- Iglesias, M.A.; Kroning, I.S.; Decol, L.T.; Franco, B.D.G.M.; Silva, W.P. Occurrence and phenotypic and molecular characterization of Listeria monocytogenes and Salmonella spp. in slaughterhouses in southern Brazil. Food Res. Int. 2017, 100, 96–101. [Google Scholar] [CrossRef]
- Loiko, M.R.; Paula, C.M.D.; Langone, A.C.J.; Rodrigues, R.Q.; Cibulski, S.; Rodrigues, R.O.; Camargo, A.C.; Nero, L.A.; Mayer, F.Q.; Tondo, E.C. Genotypic and antimicrobial characterization of pathogenic bacteria at different stages of cattle slaughtering in southern Brazil. Meat Sci. 2016, 116, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Farah, S.M.S.S.; Souza, E.M.; Pedrosa, F.O.; Irino, K.; Silva, L.R.; Rigo, L.U.; Steffens, M.B.R.; Pigatto, C.P.; Fadel-Picheth, C.M.T. Phenotypic and genotypic traits of Shiga toxin-producing Escherichia coli strains isolated from beef cattle from Paraná State, southern Brazil. Lett. Appl. Microbiol. 2007, 44, 607–612. [Google Scholar] [CrossRef]
- ISO. ISO 17604—Microbiology of the Food Chain—Carcass Sampling for Microbiological Analysis; International Organization for Standardization: Geneva, Switzerland, 2015. [Google Scholar]
- Matos, A.V.R.; Nunes, L.B.S.; Vianna, C.; Spina, T.L.B.; Zuim, C.V.; Possebon, F.S.; Xavier, D.M.; Ferraz, M.C.; Pinto, J.P.A.N. Listeria monocytogenes, E. coli O157, Salmonella spp. e microrganismos indicadores em carcaças bovinas para exportação. Arquivo Brasileiro de Medicina Veterinária e Zootecnia 2013, 65, 981–988. [Google Scholar] [CrossRef]
- Zweifel, C.; Capek, M.; Stephan, R. Microbiological contamination of cattle carcasses at different stages of slaughter in two abattoirs. Meat Sci. 2014, 98, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Antic, D.; Blagojevic, B.; Ducic, M.; Nastasijevic, I.; Mitrovic, R.; Buncic, S. Distribution of microflora on cattle hides and its transmission to meat via direct contact. Food Control 2010, 21, 1025–1029. [Google Scholar] [CrossRef]
- McEvoy, J.M.; Doherty, A.M.; Finnerty, M.; Sheridan, J.J.; McGuire, L.; Blair, I.S.; McDowell, D.A.; Harrington, D. The relationship between hide cleanliness and bacterial numbers on beef carcasses at a commercial abattoir. Lett. Appl. Microbiol. 2000, 30, 390–395. [Google Scholar] [CrossRef]
- Blagojevic, B.; Antic, D.; Ducic, M.; Buncic, S. Ratio between carcass-and skin-microflora as an abattoir process hygiene indicator. Food Control 2011, 22, 186–190. [Google Scholar] [CrossRef]
- Hauge, S.J.; Nafstad, O.; Røtterud, O.-J.; Nesbakken, T. The hygienic impact of categorisation of cattle by hide cleanliness in the abattoir. Food Control 2012, 27, 100–107. [Google Scholar] [CrossRef]
- Hauge, S.J.; Nesbakken, T.; Moen, B.; Røtterud, O.-J.; Dommersnes, S.; Nesteng, O.; Østensvik, Ø.; Alvseike, O. The significance of clean and dirty animals for bacterial dynamics along the beef chain. Int. J. Food Microbiol. 2015, 214, 70–76. [Google Scholar] [CrossRef]
- Serraino, A.; Bardasi, L.; Riu, R.; Pizzamiglio, V.; Liuzzo, G.; Galletti, G.; Giacometti, F.; Merialdi, G. Visual evaluation of cattle cleanliness and correlation to carcass microbial contamination during slaughtering. Meat Sci. 2012, 90, 502–506. [Google Scholar] [CrossRef]
- Tergney, A.; Bolton, D.J. Validation studies on an online monitoring system for reducing faecal and microbial contamination on beef carcasses. Food Control 2006, 17, 378–382. [Google Scholar] [CrossRef]
- Brasil. Resolução-RDC 12 de 2 de janeiro: Regulamento técnico sobre os Padrões microbiológicos para alimentos. Diário Oficial da União 2001. Available online: http://portal.anvisa.gov.br/documents/33880/2568070/RDC_12_2001.pdf/15ffddf6-3767-4527-bfac-740a0400829b (accessed on 16 March 2019).
- FSIS (Ed.) Guidance for Minimizing the Risk of Escherichia coli O157:H7 and Salmonella in Beef Slaughter Operations; USDA: Washington, DC, USA, 2002; p. 32.
- Bell, R.G. Distribution and sources of microbial contamination on beef carcasses. J. Appl. Microbiol. 1997, 82, 292–300. [Google Scholar] [CrossRef]
- Loretz, M.; Stephan, R.; Zweifel, C. Antibacterial activity of decontamination treatments for cattle hides and beef carcasses. Food Control 2011, 22, 347–359. [Google Scholar] [CrossRef]
- Koohmaraie, M.; Arthur, T.M.; Bosilevac, J.M.; Guerini, M.; Shackelford, S.D.; Wheeler, T.L. Post-harvest interventions to reduce/eliminate pathogens in beef. Meat Sci. 2005, 71, 79–91. [Google Scholar] [CrossRef]
- Burin, R.C.K.; Silva, A.; Nero, L.A. Influence of lactic acid and acetic acid on Salmonella spp. growth and expression of acid tolerance-related genes. Food Res. Int. 2014, 64, 726–732. [Google Scholar] [CrossRef] [PubMed]
- Røssvoll, E.; Hauge, S.J.; Skjerve, E.; Johannessen, G.; Økland, M.; Røtterud, O.-J.; Nesbakken, T.; Alvseike, O. Experimental evaluation of performance of sampling techniques for microbiological quantification on carcass surfaces. Food Prot. Trends 2017, 37, 419–429. [Google Scholar]
- Milios, K.T.; Drosinos, E.H.; Zoiopoulos, P.E. Food safety management system validation and verification in meat industry: Carcass sampling methods for microbiological hygiene criteria—A review. Food Control 2014, 43, 74–81. [Google Scholar] [CrossRef]
| Slaughterhouse | Location (State) 1 | Slaughter Rate | Employees | Export |
|---|---|---|---|---|
| Sl 01 | MG | 150–180/day | 50 | Yes |
| Sl 02 | MG | 90–100/day2 | 25 | No |
| Sl 03 | MG | 130–150 /day | 50 | Yes |
| Sl 04 | PR | 600–650/day3 | 40 | No |
| Sl 05 | PR | 400–450/day | 50 | No |
| Sl 06 | PR | 200–250/day | 50 | No |
| Sl 07 | RS | 130–150/day | 140 | No |
| Sl 08 | RS | 600–650/day | 700 | Yes |
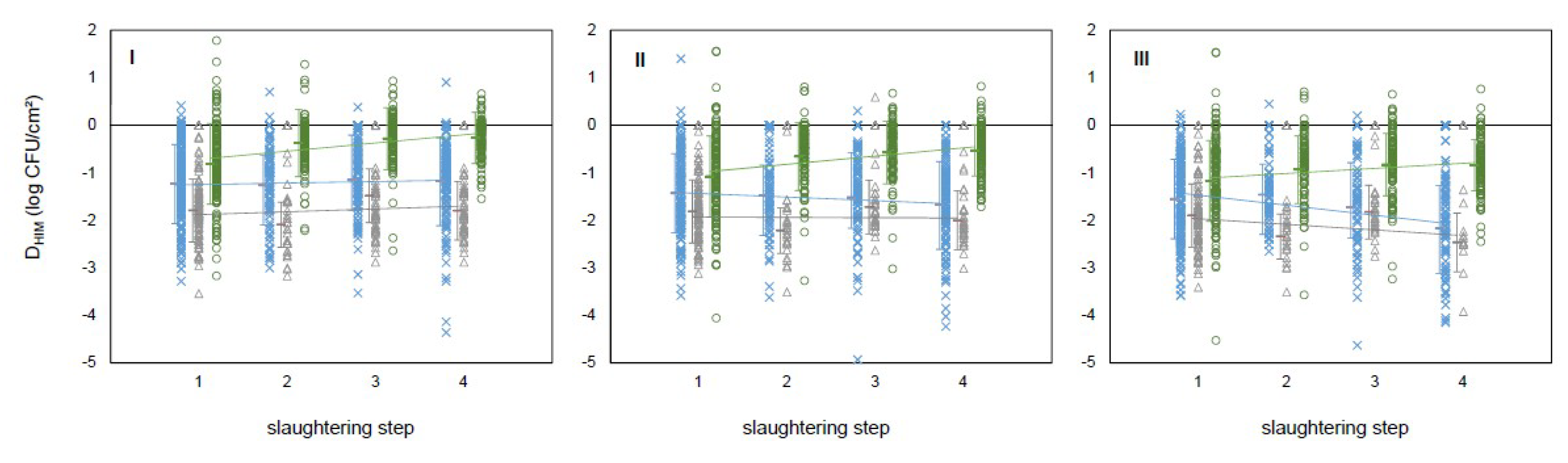
| HIM | Slaughtering Step | |||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
| MA | 4.51 ± 0.06 a | 2.47 ± 0.06 c | 2.64 ± 0.06 c | 2.93 ± 0.06 b |
| EB | 3.27 ± 0.06 a | 1.79 ± 0.08 c | 2.20 ± 0.07 b | 2.11 ± 0.07 b |
| TC | 3.20 ± 0.06 a | 1.73 ± 0.09 c | 2.05 ± 0.08 b | 1.81 ± 0.07 bc |
| EC | 3.06 ± 0.06 a | 1.58 ± 0.09 bc | 1.89 ± 0.08 b | 1.53 ± 0.08 c |
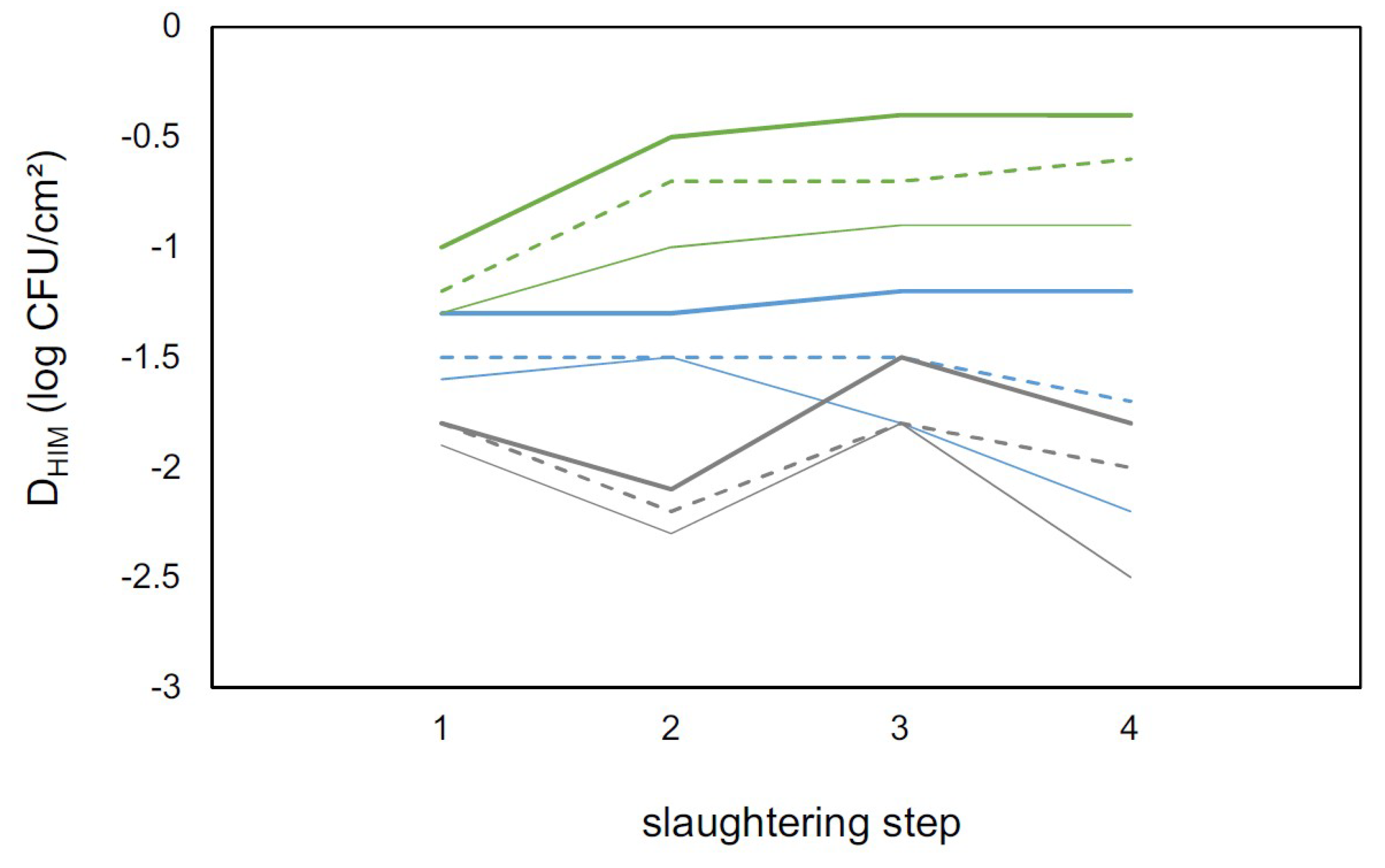
| HIM | State | Slaughtering Step | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| MA | MG | 4.66 ± 0.08 Aa | 2.85 ± 0.07 Ad | 3.30 ± 0.09 Ac | 3.8 ± 0.09 Ab |
| PR | 4.34 ± 0.11 Ba | 2.43 ± 0.09 Bb | 2.03 ± 0.12 Bc | 2.46 ± 0.12 Bb | |
| RS | 4.42 ± 0.09 ABa | 2.14 ± 0.07 Cb | 2.37 ± 0.09 Bb | 2.21 ± 0.1 Bb | |
| EB | MG | 3.37 ± 0.08 Aa | 1.89 ± 0.1 Ac | 2.24 ± 0.11 Abc | 2.46 ± 0.08 Ab |
| PR | 2.56 ± 0.11 Ba | 0.84 ± 0.17 Bc | 1.45 ± 0.23 Bb | 1.02 ± 0.15 Cbc | |
| RS | 3.61 ± 0.09 Aa | 2.01 ± 0.1 Ac | 2.37 ± 0.12 Ab | 2.09 ± 0.1 Bbc | |
| TC | MG | 3.25 ± 0.1 Aa | 1.77 ± 0.13 ABb | 2.05 ± 0.13 Ab | 2.0 ± 0.7 Ab |
| PR | 2.63 ± 0.13 Ba | 1.22 ± 0.22 Bb | 0.95 ± 0.25 Bb | 0.85 ± 0.15 Bb | |
| RS | 3.45 ± 0.1 Aa | 1.84 ± 0.12 Ac | 2.34 ± 0.13 Ab | 1.86 ± 0.08 Ac | |
| EC | MG | 3.14 ± 0.1 Aa | 1.72 ± 0.12 Ab | 1.91 ± 0.15 Ab | 1.66 ± 0.1 Ab |
| PR | 2.46 ± 0.13 Ba | 0.93 ± 0.2 Bb | 0.86 ± 0.28 Bb | 0.52 ± 0.24 Bb | |
| RS | 3.36 ± 0.1 Aa | 1.66 ± 0.1 Ac | 2.12 ± 0.13 Ab | 1.55 ± 0.08 Ac | |
| State | n | Mesophilic Aerobes | Enterobacteriaceae |
|---|---|---|---|
| MG | 209 | 30 (14.4%) | 61 (29.2%) |
| PR | 105 | 2 (1.9%) | 1 (1.0%) |
| RS | 150 | 0 (0.0%) | 9 (6.0%) |
| Total | 464 | 32 (6.9%) | 71 (15.3%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camargo, A.C.; Cossi, M.V.C.; Silva, W.P.d.; Bersot, L.d.S.; Landgraf, M.; Baranyi, J.; Franco, B.D.G.d.M.; Luís Augusto, N. Microbiological Testing for the Proper Assessment of the Hygiene Status of Beef Carcasses. Microorganisms 2019, 7, 86. https://doi.org/10.3390/microorganisms7030086
Camargo AC, Cossi MVC, Silva WPd, Bersot LdS, Landgraf M, Baranyi J, Franco BDGdM, Luís Augusto N. Microbiological Testing for the Proper Assessment of the Hygiene Status of Beef Carcasses. Microorganisms. 2019; 7(3):86. https://doi.org/10.3390/microorganisms7030086
Chicago/Turabian StyleCamargo, Anderson Carlos, Marcus Vinícius Coutinho Cossi, Wladimir Padilha da Silva, Luciano dos Santos Bersot, Mariza Landgraf, József Baranyi, Bernadette Dora Gombossy de Melo Franco, and Nero Luís Augusto. 2019. "Microbiological Testing for the Proper Assessment of the Hygiene Status of Beef Carcasses" Microorganisms 7, no. 3: 86. https://doi.org/10.3390/microorganisms7030086
APA StyleCamargo, A. C., Cossi, M. V. C., Silva, W. P. d., Bersot, L. d. S., Landgraf, M., Baranyi, J., Franco, B. D. G. d. M., & Luís Augusto, N. (2019). Microbiological Testing for the Proper Assessment of the Hygiene Status of Beef Carcasses. Microorganisms, 7(3), 86. https://doi.org/10.3390/microorganisms7030086






