Elucidation of the Initial Growth Process and the Infection Mechanism of Penicillium digitatum on Postharvest Citrus (Citrus reticulata Blanco)
Abstract
1. Introduction
2. Materials and Methods
2.1. Pathogen and Fruit
2.2. Infection of Citrus by P. digitatum
2.3. The Growth Process of P. digitatum in Vitro
2.4. The Process of P. digitatum Infection of Citrus Fruits
2.5. Enzyme Activity Assay
2.6. Effects of P. digitatum on Biochemical Changes of Citrus Wound Tissues
2.7. Transcriptome Assay
2.7.1. Sample Preparation
2.7.2. Total RNA Extraction
2.7.3. RNA-Seq Library Construction and Sequencing
2.7.4. De Novo Assembly and Bioinformatics Analysis of RNA-seq Data
2.8. Statistical Analysis
3. Results
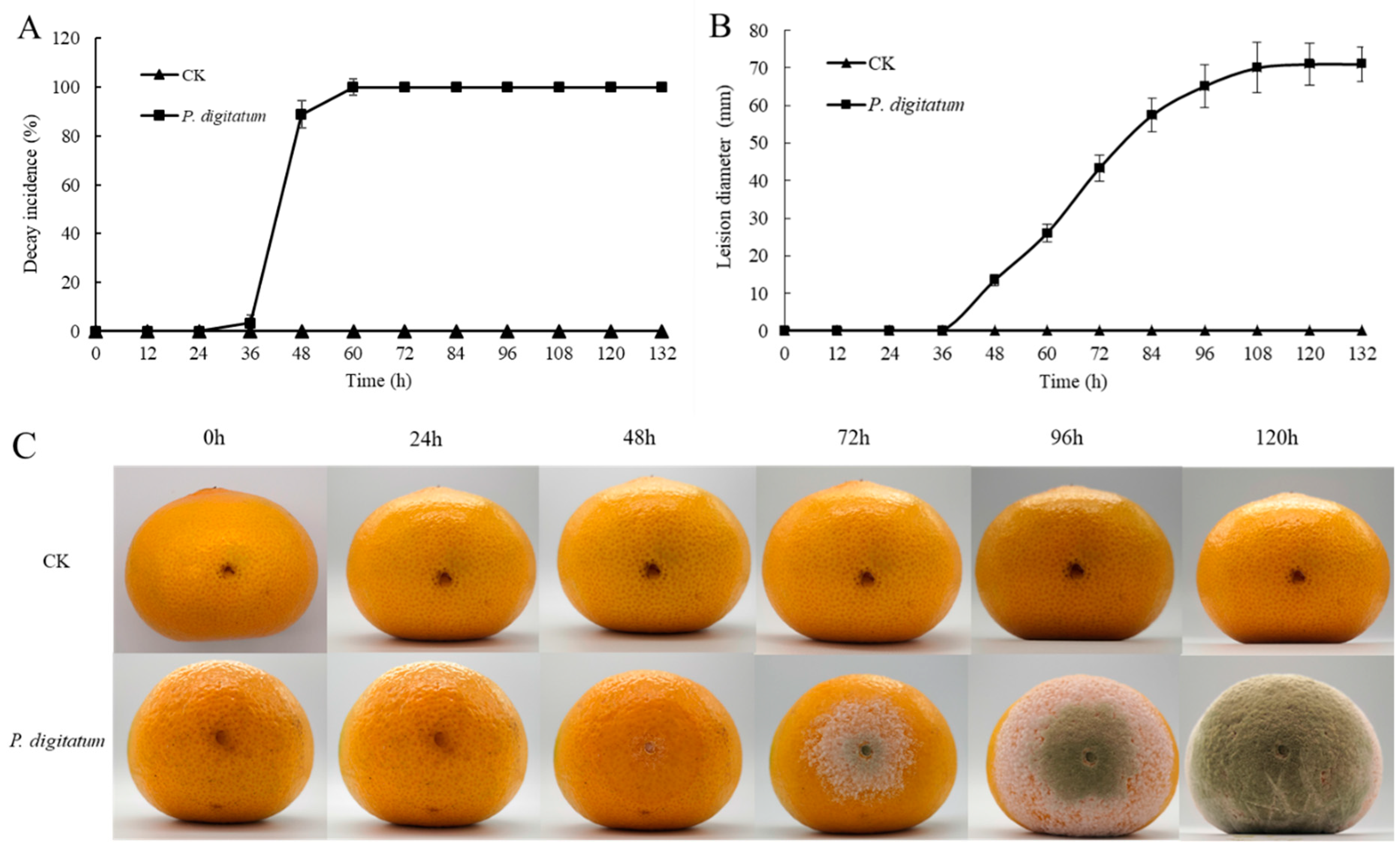
3.1. The Infection of Citrus by P. digitatum
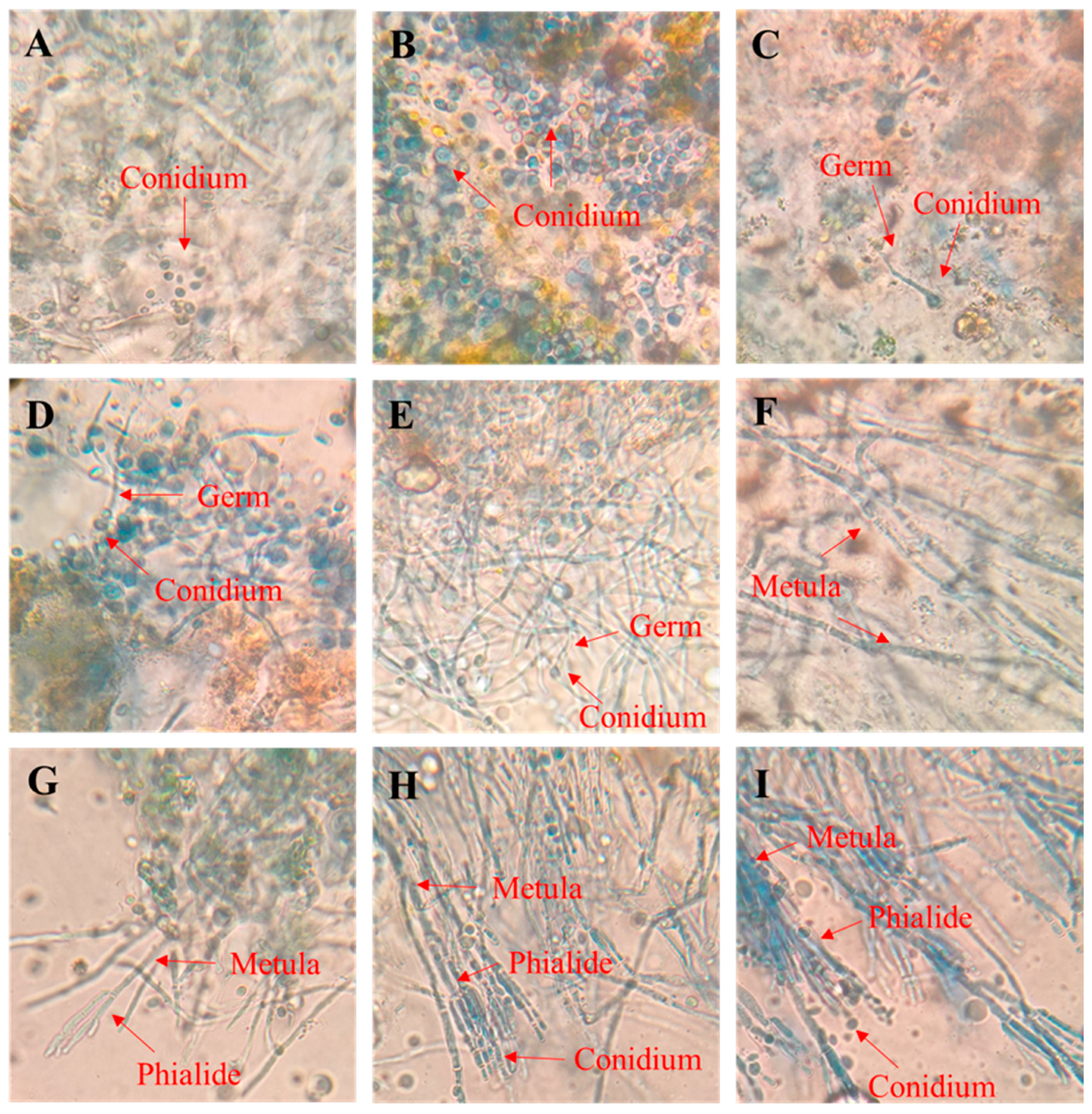
3.2. The Growth Process of P. digitatum on PDA
3.3. The Growth Process of P. digitatum in Citrus Wounds
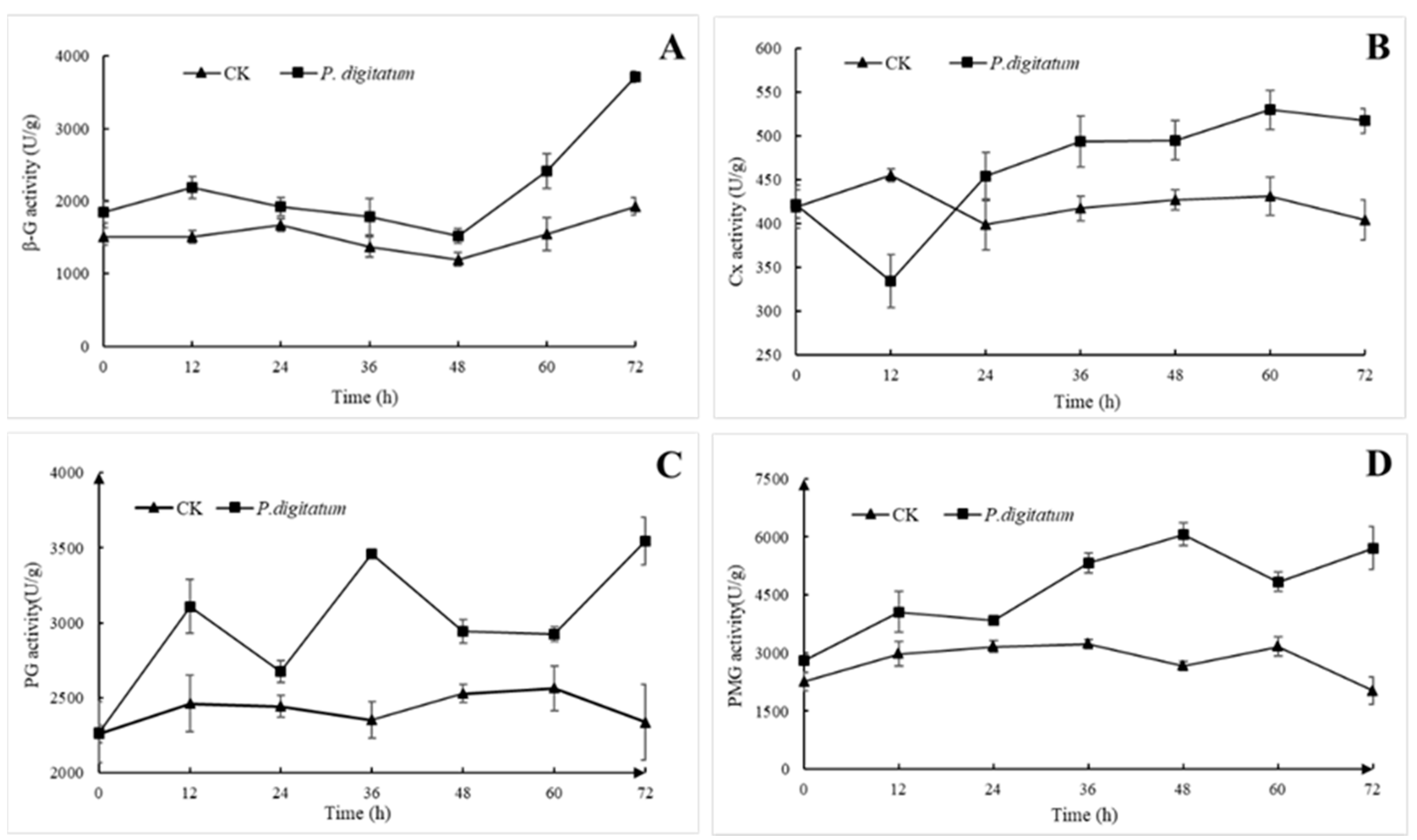
3.4. Effects of P. digitatum on Cell Wall Degrading Enzyme Activity of Citrus
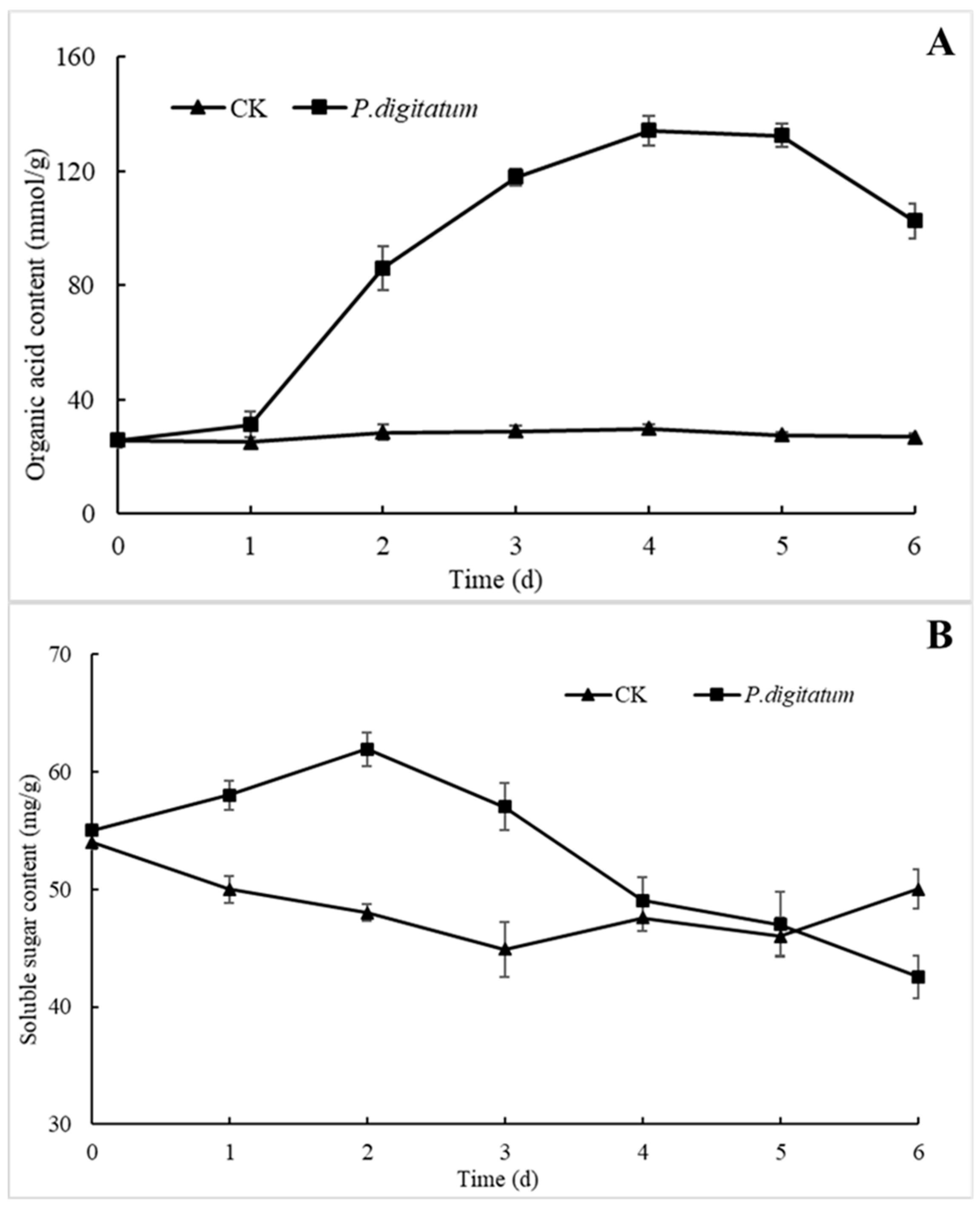
3.5. Effects of P. digitatum on Biochemical Changes of Citrus Wound Tissues
3.6. Transcriptomic Analysis of P.digitatum Before and After Infection of Citrus
3.7. Analysis of Cell Wall Degrading Enzymes-Related Pathways
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Liu, Y.; Heying, E.; Tanumihardjo, S.A. History, global distribution, and nutritional importance of citrus fruits. Compr. Rev. Food Sci. Food Saf. 2012, 11, 530–545. [Google Scholar] [CrossRef]
- Cerrillo, J.; Palomares, A.E.; Rey, F.; Valenciaa, S.; Palou, L.; Pérez-Gago, M.B. Ag-zeolites as fungicidal material: Control of citrus green mold caused by Penicillium digitatum. Micropor. Mesopor. Mat. 2017, 254, 69–76. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, Z.; Ma, Z.; Li, H. A molecular mechanism of azoxystrobin resistance in Penicillium digitatum UV mutants and a PCR-based assay for detection of azoxystrobin-resistant strains in packing- or store-house isolates. Int. J. Food. Microbiol. 2009, 131, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Kinay, P.; Mansour, M.F.; Gabler, F.M.; Margosan, D.A.; Smilanic, J.L. Characterization of fungicide-resistant isolates of Penicillium digitatum collected in California. Crop. Prot. 2007, 26, 647–656. [Google Scholar] [CrossRef]
- Lee, M.-H.; Pan, S.-M.; Ng, T.-W.; Chen, P.-S.; Wang, L.-Y.; Chung, K.-R. Mutations of β-tubulin codon 198 or 200 indicate thiabendazole resistance among isolates of Penicillium digitatum collected from citrus in Taiwan. Int. J. Food Microbiol. 2011, 150, 157–163. [Google Scholar] [CrossRef]
- Koeck, M.; Hardham, A.R.; Dodds, P.N. The role of effectors of biotrophic and hemibiotrophic fungi in infection. Cell Microbiol. 2011, 13, 1849–1857. [Google Scholar] [CrossRef]
- Yang, S.; Peng, L.; Cheng, Y.; Feng, C.; Pan, S. Control of citrus green and blue molds by Chinese propolis. Food Sci. Biotechnol. 2010, 19, 1303–1308. [Google Scholar] [CrossRef]
- Erasmus, A.; Lennox, C.L.; Korsten, L.; Lesar, K.; Fourie, P.H. Imazalil resistance in Penicillium digitatum and P. italicum causing citrus postharvest green and blue mould: Impact and options. Postharvest Biol. Tecnol. 2015, 107, 66–76. [Google Scholar] [CrossRef]
- Underwood, W. The plant cell wall: A dynamic barrier against pathogen invasion. Front Plant Sci. 2012, 3, 85. [Google Scholar] [CrossRef]
- Cosgrove, D.J. Growth of the plant cell wall. Nat. Rev. Mol. Cell. Biol. 2005, 6, 850–861. [Google Scholar] [CrossRef]
- Kubicek, C.P.; Starr, T.L.; Glass, N.L. Plant cell wall–degrading enzymes and their secretion in plant-pathogenic fungi. Annu. Rev. Phytopathol. 2014, 52, 427–451. [Google Scholar] [CrossRef] [PubMed]
- Carpita, N.C.; Gibeaut, D.M. Structural models of primary cell walls in flowering plants: Consistency of molecular structure with the physical properties of the walls during growth. Plant J. 1993, 3, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Miyara, I.; Shafran, H.; Kramer Haimovich, H.; Rollins, J.; Sherman, A.; Prusky, D. Multi-factor regulation of pectate lyase secretion by Colletotrichum gloeosporioides pathogenic on avocado fruits. Mol. Plant Pathol. 2008, 9, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.Y.; Chen, H.L.; Lu, M.Y.J.; Chen, Y.C.; Sung, H.M.; Mao, C.T.; Cho, H.Y.; Ke, H.M.; Hwa, T.Y.; Ruan, S.K.; et al. Functional characterization of cellulases identified from the cow rumen fungus Neocallimastix patriciarum W5 by transcriptomic and secretomic analyses. Biotechnol. Biofuels 2011, 4, 24. [Google Scholar] [CrossRef] [PubMed]
- Pardo, A.G.; Forchiassin, F. Influence of temperature and pH on cellulase activity and stability in Nectria catalinensis. Rev. Argent. Microbiol. 1999, 31, 31–35. [Google Scholar] [PubMed]
- Zhao, D.; Mao, T.; Zhao, X.; Liu, X.; Cai, L.; Tao, G. Microscopic observation of early infection of Colletotrichum fragariae on strawberry leaves. J. South. Agric. 2016, 47, 1140–1145. [Google Scholar]
- Yuan, H.; Ling, X.; Liu, T.; Chen, T.; Yang, Y.; Yao, S.; Zhang, B. Microscopic observations of strawberry plant colonization by a GFP-labelled strain of Fusarium oxysporumf. sp. fragariae. Can. J. Plant Pathol. 2014, 36, 501–508. [Google Scholar] [CrossRef]
- Visagie, C.M.; Houbraken, J.; Frisvad, J.C.; Hong, S.B.; Klaassen, C.H.W.; Perrone, G.; Seifert, K.A.; Varga, J.; Yaguchi, T.; Samson, R.A. Identification and nomenclature of the genus Penicillium. Stud. Mycol. 2014, 78, 343–371. [Google Scholar] [CrossRef]
- Tu, Q.; Chen, J.; Guo, J. Screening and identification of antagonistic bacteria with potential for biological control of Penicillium italicum of citrus fruits. Sci. Hortic. 2013, 150, 125–129. [Google Scholar] [CrossRef]
- Zhang, X.; Lin, Z.; Apaliya, M.T.; Gu, X.; Zheng, X.; Zhao, L.; Abdelhai, M.H.; Zhang, H.; Hu, W. The possible mechanisms involved in citrinin elimination by Cryptococcus podzolicus Y3 and the effects of extrinsic factors on the degradation of citrinin. J. Microbiol. Biotechnol. 2017, 27, 2119–2128. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, L.; Li, Z.; Li, C.; Li, B.; Gu, X.; Zhang, X.; Zhang, H. Screening and identification of an antagonistic yeast controlling postharvest blue mold decay of pears and the possible mechanisms involved. Biol. Control. 2019, 133, 26–33. [Google Scholar] [CrossRef]
- Kema, G.H.J.; van der Lee, T.A.J.; Mendes, O.; Verstappen, E.C.P.; Lankhorst, R.K.; Sandbrink, H.; van der Burgt, A.; Zwiers, L.-H.; Csukai, M.; Waalwijk, C. Large-Scale gene discovery in the septoria tritici blotch fungus Mycosphaerella graminicola with a focus on in planta expression. Mol. Plant Microbe Interact. 2008, 21, 1249–1260. [Google Scholar] [CrossRef] [PubMed]
- Zeng, K.; Cao, J.; Jiang, W. Enhancing disease resistance in harvested mango (Mangifera indica L. cv. ‘Matisu’) fruit by salicylic acid. J. Sci. Food Agric. 2006, 85, 694–698. [Google Scholar] [CrossRef]
- Hsu, P.D.; A Scott, D.; A Weinstein, J.; Ran, F.A.; Konermann, S.; Agarwala, V.; Li, Y.; Fine, E.J.; Wu, X.; Shalem, O.; et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 2013, 31, 827–832. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, X.; Zhao, L.; Apaliya, M.T.; Yang, Q.; Sun, W.; Zhang, X.; Zhang, H. Screening of deoxynivalenol producing strains and elucidation of possible toxigenic molecular mechanism. Toxins 2017, 9, 184. [Google Scholar] [CrossRef]
- Ma, H.; Sun, X.; Wang, M.; Gai, Y.; Chung, K.; Li, H. The citrus postharvest pathogen Penicillium digitatum depends on the PdMpkB kinase for developmental and virulence functions. Int. J. Food Microbiol. 2016, 236, 167–176. [Google Scholar] [CrossRef]
- Vilanova, L.; Teixidó, N.; Torres, R.; Usall, J.; Viñas, I.; Sánchez-Torres, P. Relevance of the transcription factor PdSte12 in Penicillium digitatum conidiation and virulence during citrus fruit infection. Int. J. Food Microbiol. 2016, 235, 93–102. [Google Scholar] [CrossRef]
- Sánchez-Torres, P.; Vilanova, L.; Ballester, A.R.; López-Pérez, M.; Teixidó, N.; Viñas, I.; Usall, J.; González-Candelas, L.; Torres, R. Unravelling the contribution of the Penicillium expansum PeSte12 transcription factor to virulence during apple fruit infection. Food Microbiol. 2018, 69, 123–135. [Google Scholar] [CrossRef]
- Zhou, T.; Wang, X.; Luo, J.; Ye, B.; Zhou, Y.; Zhou, L.; Lai, T. Identification of differentially expressed genes involved in spore germination of Penicillium expansum by comparative transcriptome and proteome approaches. MicrobiologyOpen 2017, 7, e00562. [Google Scholar] [CrossRef]
- Teeri, T.T.; Koivula, A.; Linder, M.; Wohlfahrt, G.; Divne, C.; A Jones, T. Trichoderma reesei cellobiohydrolases: Why so efficient on crystalline cellulose? Biochem. Soc. Trans. 1998, 26, 173–178. [Google Scholar] [CrossRef]
- Abouna, S.; Legendre, L.; Manteau, S.; Lambert, B. Differential regulation by ambient pH of putative virulence factor secretion by the phytopathogenic fungus Botrytis cinerea. FEMS Microbiol. Ecol. 2003, 43, 359–366. [Google Scholar]
- López-Pérez, M.; Ballester, A.R.; González-Candelas, L. Identification and functional analysis of Penicillium digitatum genes putatively involved in virulence towards citrus fruit. Mol. Plant. Pathol. 2015, 16, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Juturu, V.; Wu, J.C. Insight into microbial hemicellulases other than xylanases: A review. J. Chem. Technol. Biotechnol. 2013, 88, 353–363. [Google Scholar] [CrossRef]
- Da Silva, G.P.; Mack, M.; Contiero, J. Glycerol: A promising and abundant carbon source for industrial microbiology. Biotechnol. Adv. 2009, 27, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Li, J.; Shin, H.-D.; Du, G.; Liu, L.; Chen, J. Metabolic engineering in the biotechnological production of organic acids in the tricarboxylic acid cycle of microorganisms: Advances and prospects. Biotechnol. Adv. 2015, 33, 830–841. [Google Scholar] [CrossRef] [PubMed]

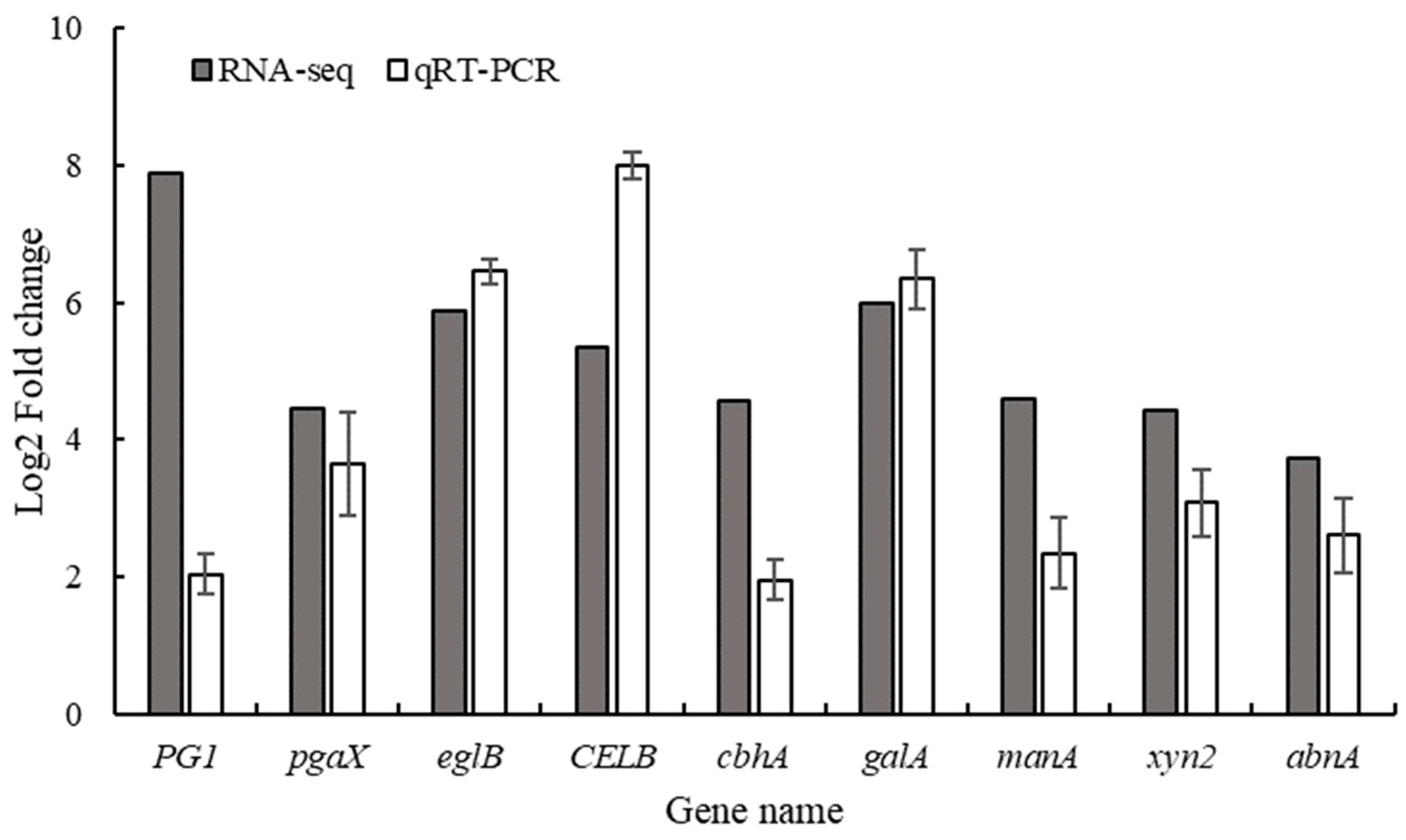
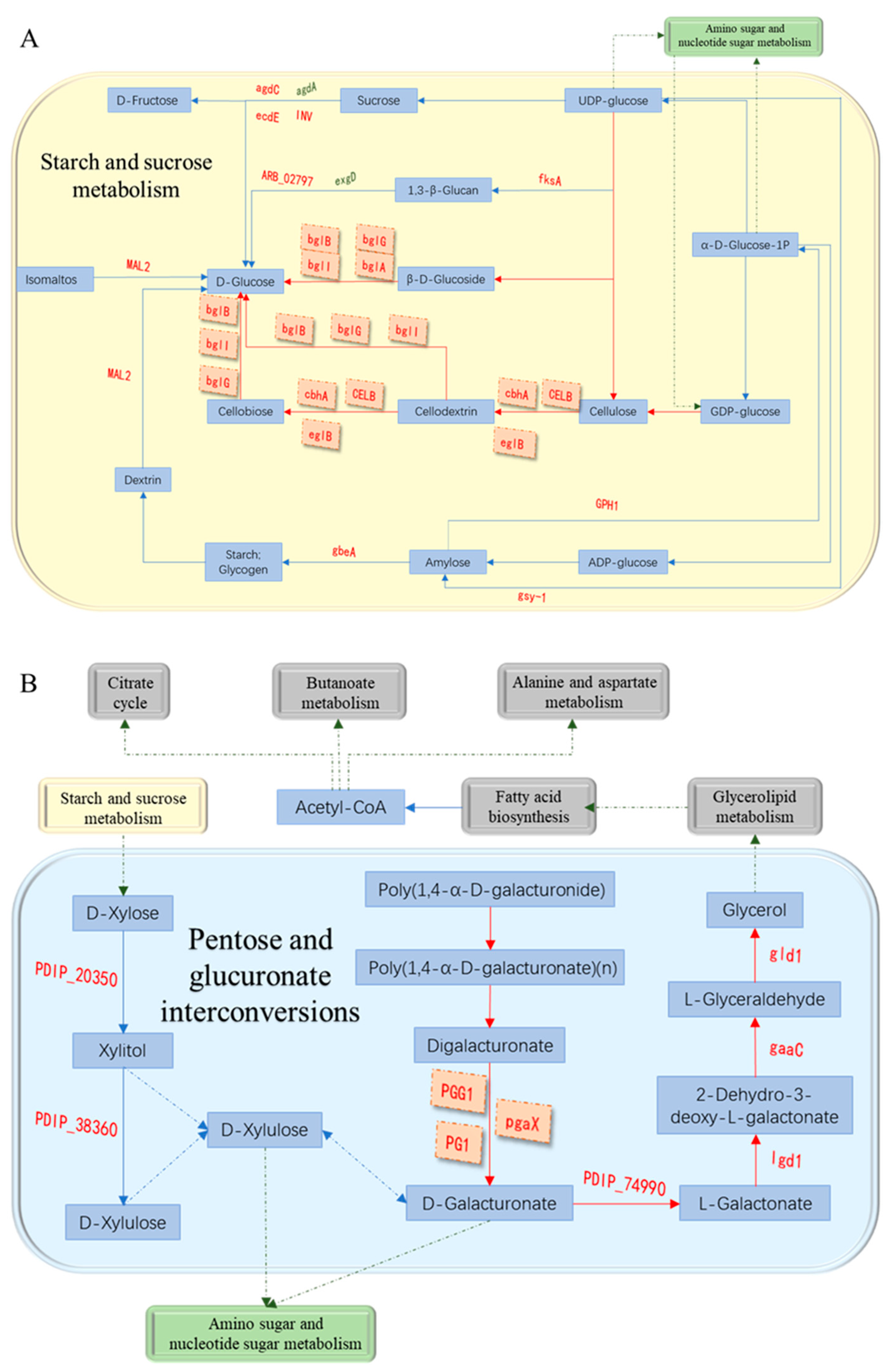
| #id | log2 Fold change | Cellular Component (GOa) | Swissport annotation (GNb) | KEGGc pathway |
|---|---|---|---|---|
| PDIP_64460 | 7.89 | extracellular region | Polygalacturonase (PG1) | Pentose and glucuronate interconversions |
| PDIP_62540 | 4.46 | extracellular region | exopolygalacturonase X (pgaX) | Pentose and glucuronate interconversions |
| PDIP_19910 | 3.56 | extracellular region | Polygalacturonase (PGG1) | Pentose and glucuronate interconversions |
| PDIP_65210 | 5.87 | extracellular region | endo-beta-1,4-glucanase B (eglB) | Starch and sucrose metabolism |
| PDIP_47720 | 5.35 | extracellular region | Endoglucanase B (CELB) | Starch and sucrose metabolism |
| PDIP_71000 | 4.56 | extracellular region | 1,4-beta-D-glucan cellobiohydrolase A (cbhA) | Starch and sucrose metabolism |
| PDIP_17570 | 2.72 | extracellular region | Beta-glucosidase B (bglB) | Biosynthesis of secondary metabolites |
| PDIP_68890 | 6.00 | extracellular region | arabinogalactan endo-beta-1,4-galactanase (galA) | |
| PDIP_55030 | 4.58 | extracellular region | mannan endo-1,4-beta-mannosidase A (manA) | |
| PDIP_02920 | 4.42 | extracellular region | Endo-1,4-beta-xylanase 2 (xyn2) | |
| MSTRG.2424 | 3.86 | extracellular region | Alpha-xylosidase A (axlA) | |
| PDIP_24590 | 3.73 | extracellular region | arabinan endo-1,5-alpha-L-arabinosidase A (abnA) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qian, X.; Yang, Q.; Zhang, Q.; Abdelhai, M.H.; Dhanasekaran, S.; Serwah, B.N.A.; Gu, N.; Zhang, H. Elucidation of the Initial Growth Process and the Infection Mechanism of Penicillium digitatum on Postharvest Citrus (Citrus reticulata Blanco). Microorganisms 2019, 7, 485. https://doi.org/10.3390/microorganisms7110485
Qian X, Yang Q, Zhang Q, Abdelhai MH, Dhanasekaran S, Serwah BNA, Gu N, Zhang H. Elucidation of the Initial Growth Process and the Infection Mechanism of Penicillium digitatum on Postharvest Citrus (Citrus reticulata Blanco). Microorganisms. 2019; 7(11):485. https://doi.org/10.3390/microorganisms7110485
Chicago/Turabian StyleQian, Xin, Qiya Yang, Qidi Zhang, Mandour H. Abdelhai, Solairaj Dhanasekaran, Boateng Nana Adwoa Serwah, Ning Gu, and Hongyin Zhang. 2019. "Elucidation of the Initial Growth Process and the Infection Mechanism of Penicillium digitatum on Postharvest Citrus (Citrus reticulata Blanco)" Microorganisms 7, no. 11: 485. https://doi.org/10.3390/microorganisms7110485
APA StyleQian, X., Yang, Q., Zhang, Q., Abdelhai, M. H., Dhanasekaran, S., Serwah, B. N. A., Gu, N., & Zhang, H. (2019). Elucidation of the Initial Growth Process and the Infection Mechanism of Penicillium digitatum on Postharvest Citrus (Citrus reticulata Blanco). Microorganisms, 7(11), 485. https://doi.org/10.3390/microorganisms7110485






