Responses of Salt Marsh Plant Rhizosphere Diazotroph Assemblages to Drought
Abstract
1. Introduction
2. Materials and Methods
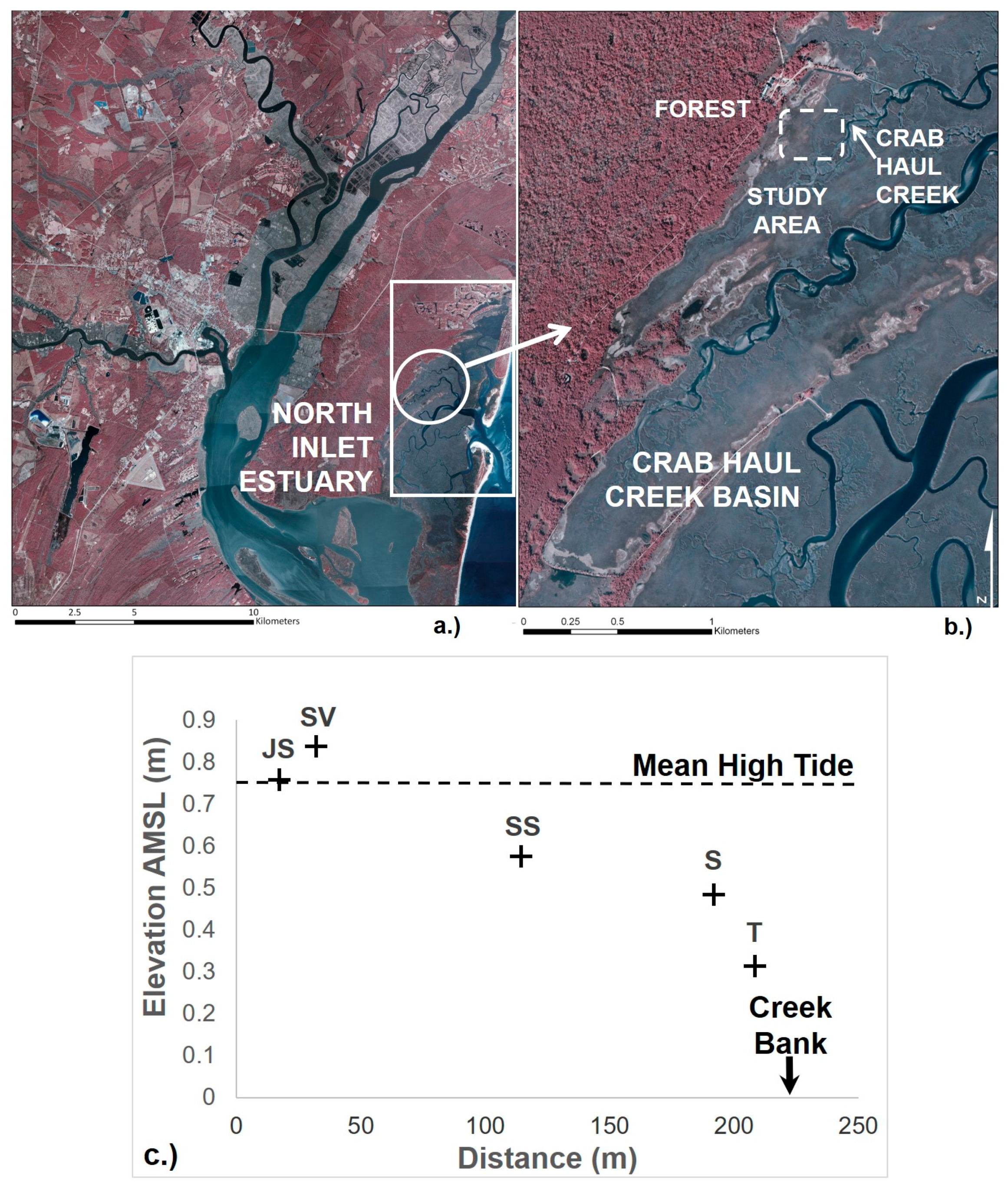
2.1. Study Area
2.2. Sample Collection
2.3. Sample Processing
2.3.1. DNA Purification
2.3.2. nifH Amplification
2.3.3. Denaturing Gradient Gel Electrophoresis
2.4. Statistical Analysis
2.4.1. Diazotroph Community Composition: DGGE Band Pattern Analysis
2.4.2. Environmental Conditions and Sediment Chemistry
2.4.3. Diazotroph Assemblages: Unconstrained Ordination Analysis
2.4.4. Drivers of Ordination and Diazotroph Assemblage Presence
3. Results
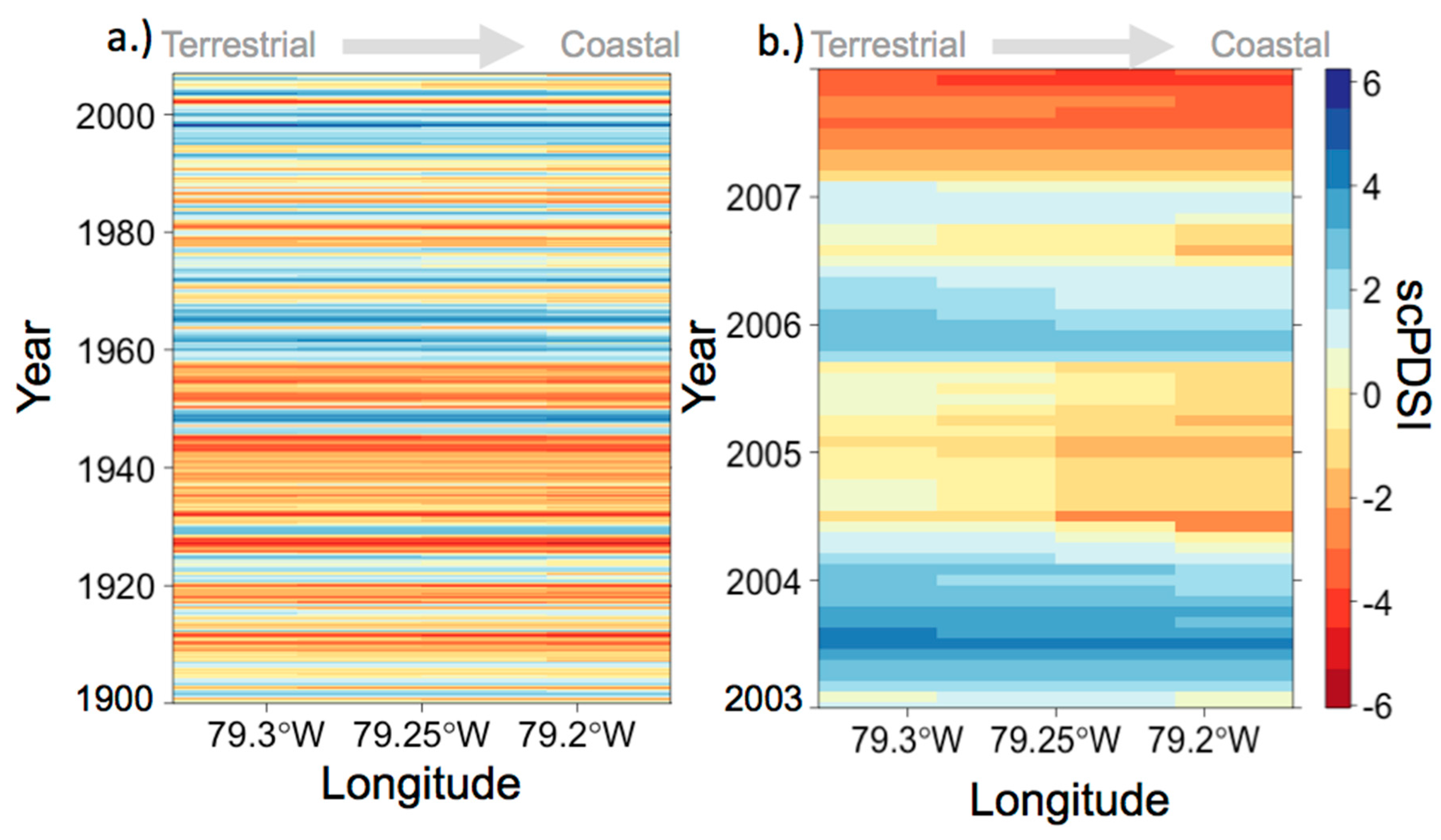
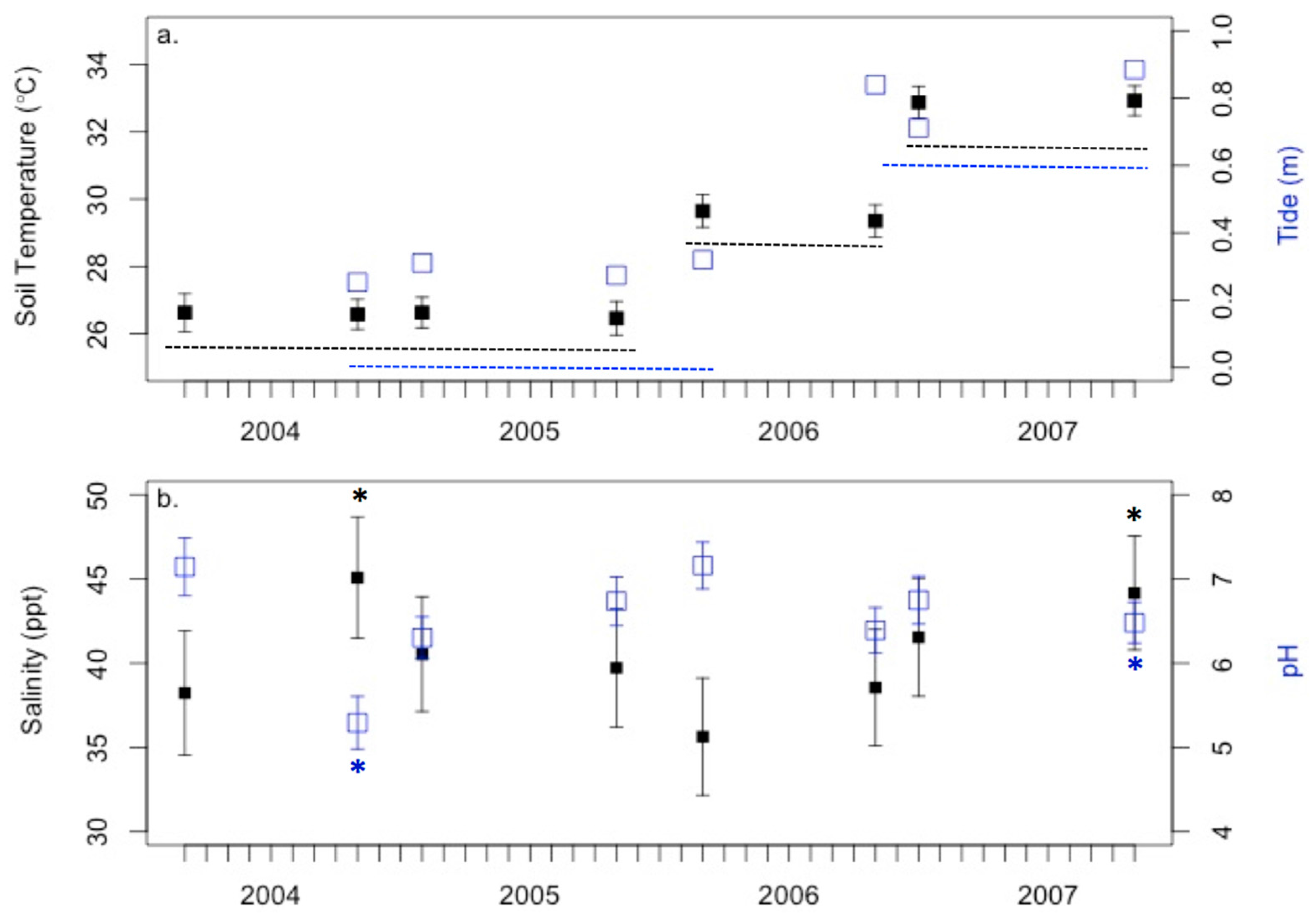
3.1. Anecdotal Evidence for 2004 Drought Event
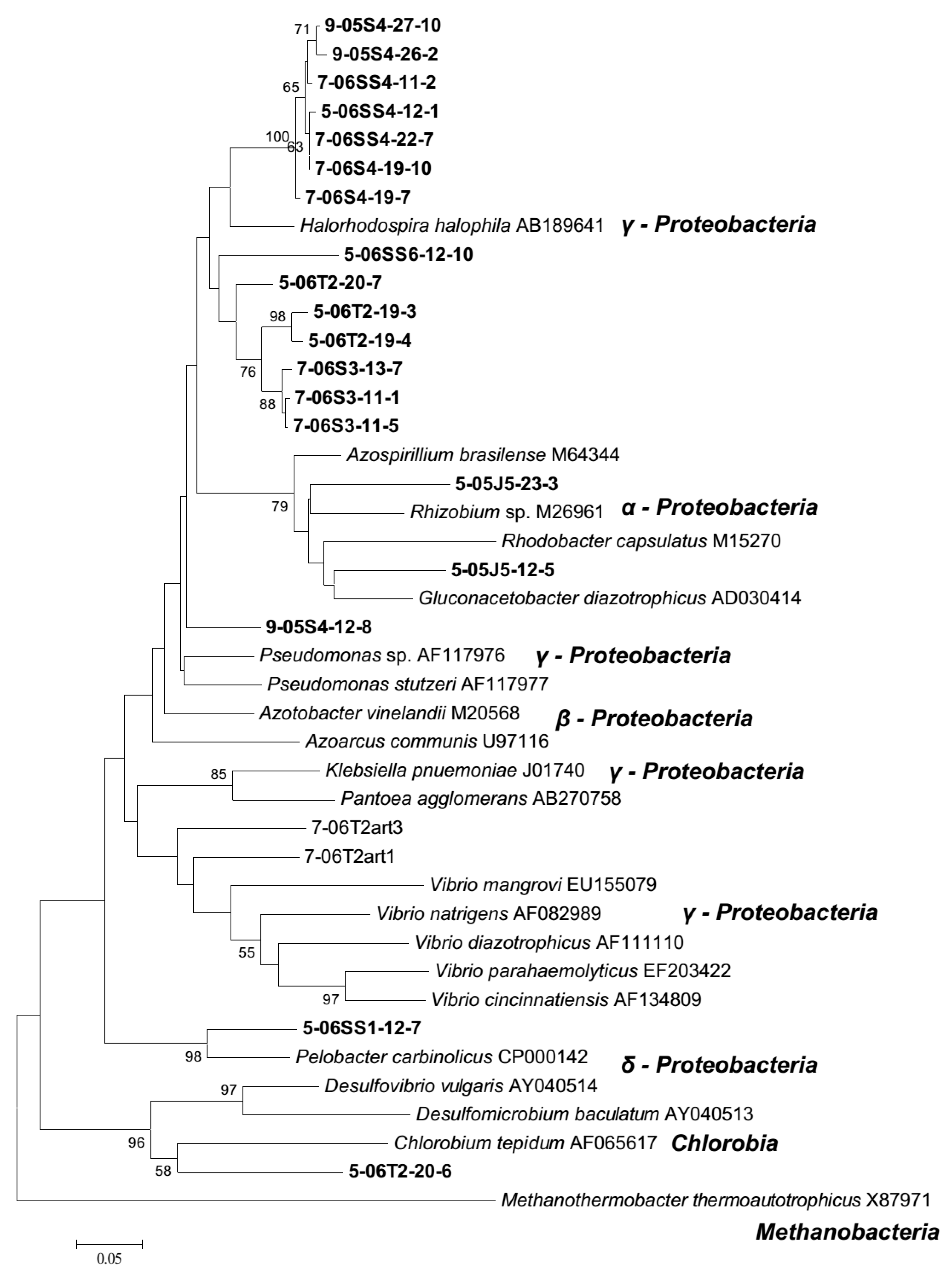
3.2. Diazotroph Community Structure and Composition
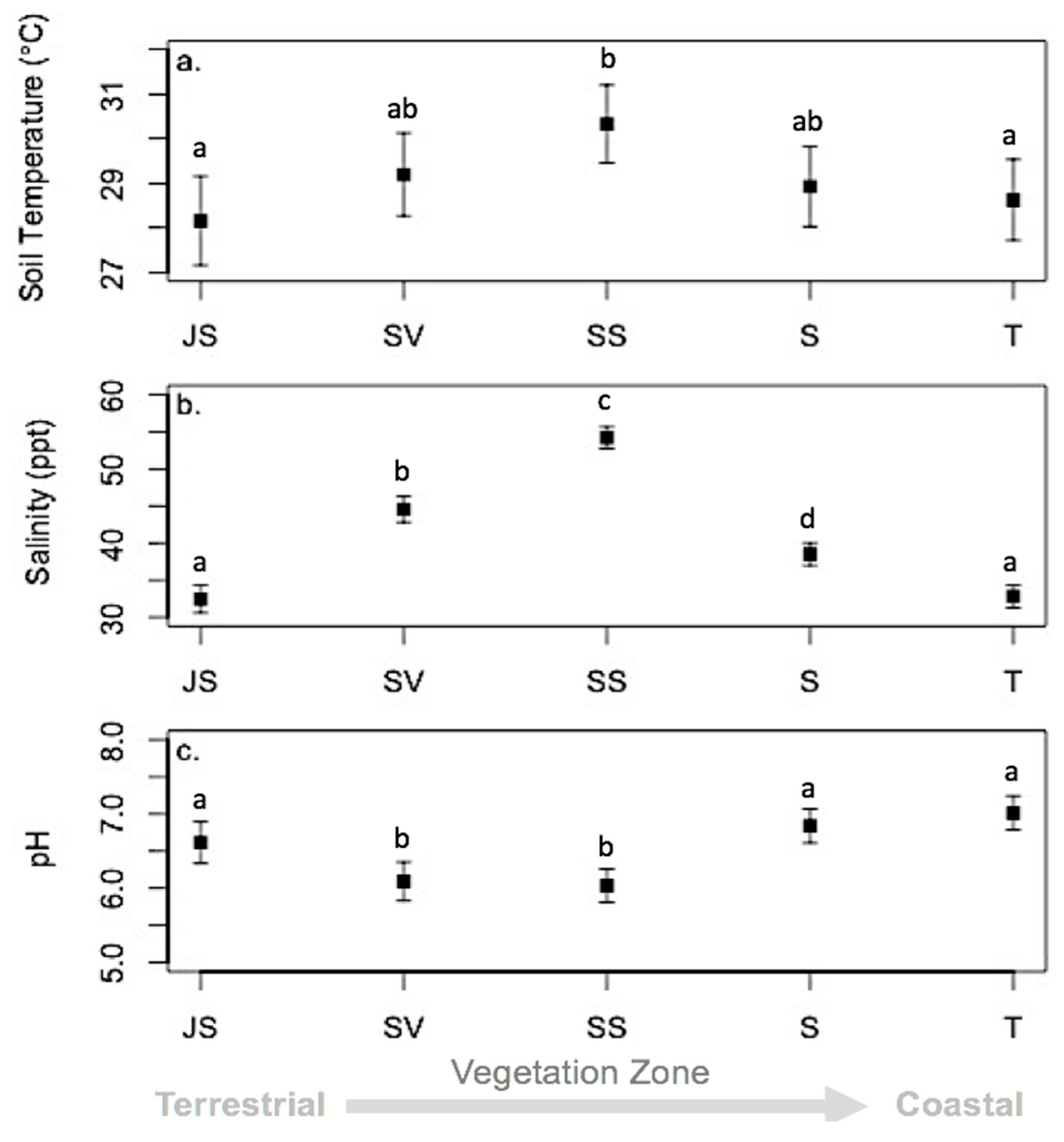
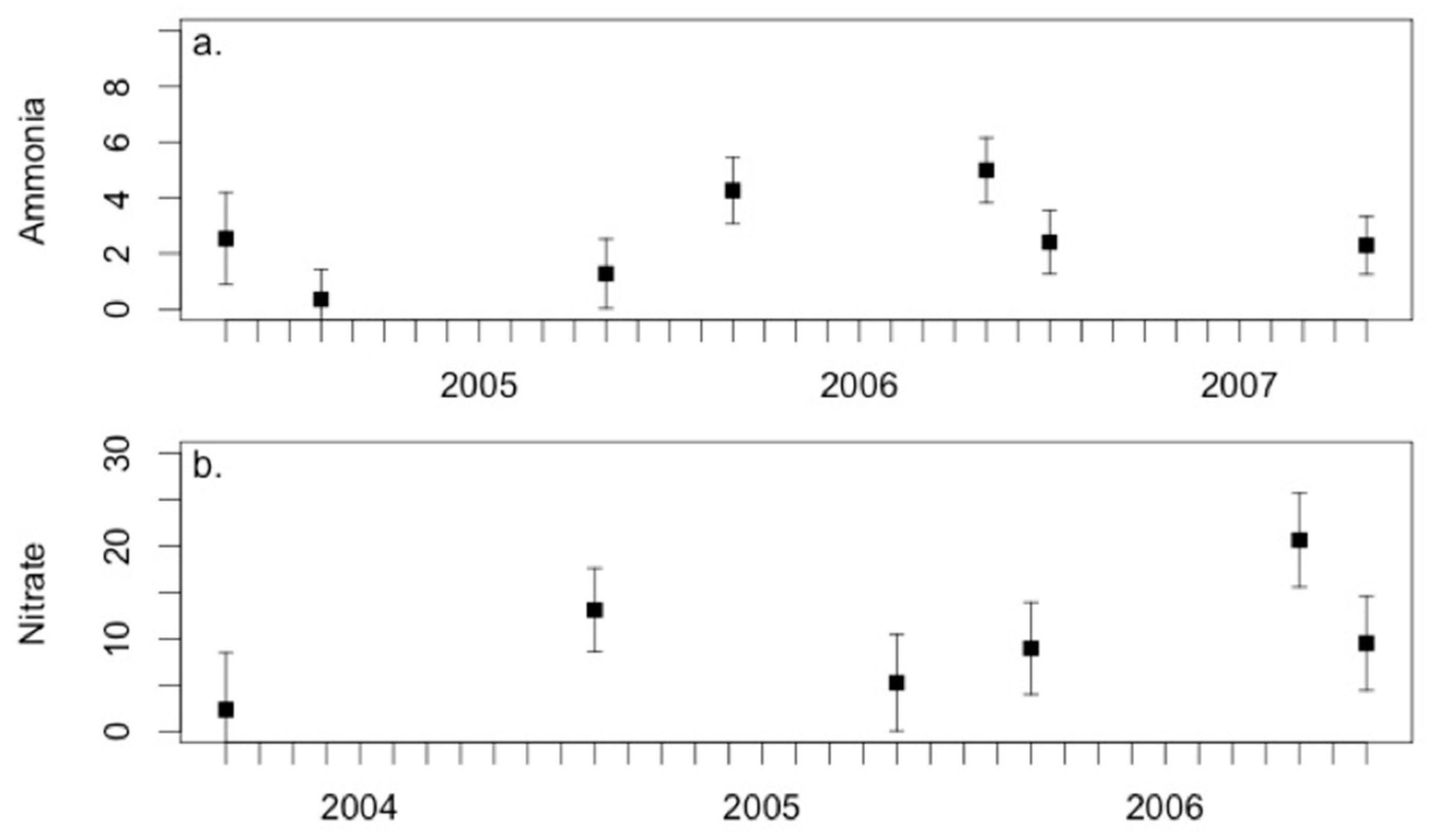
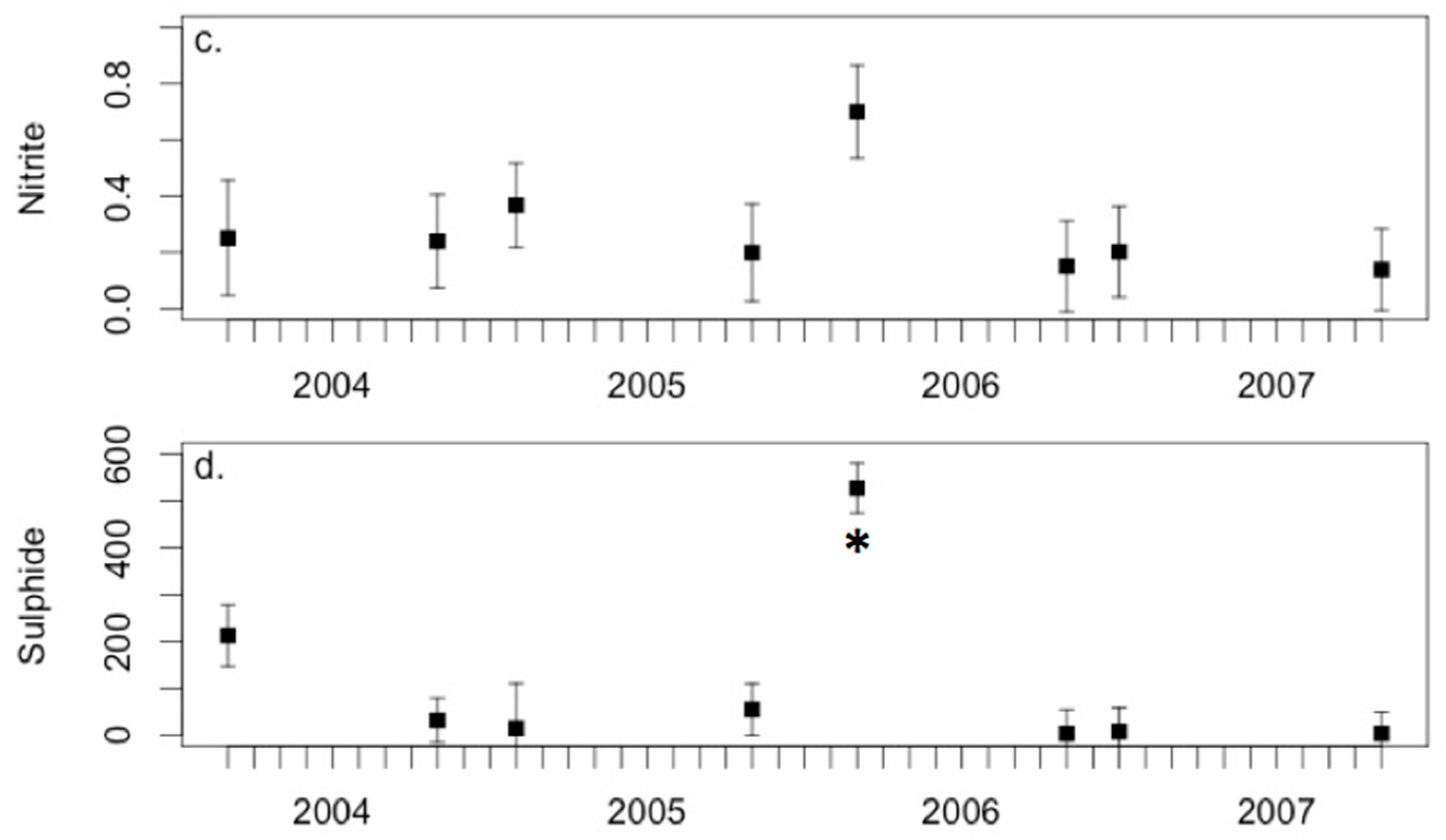
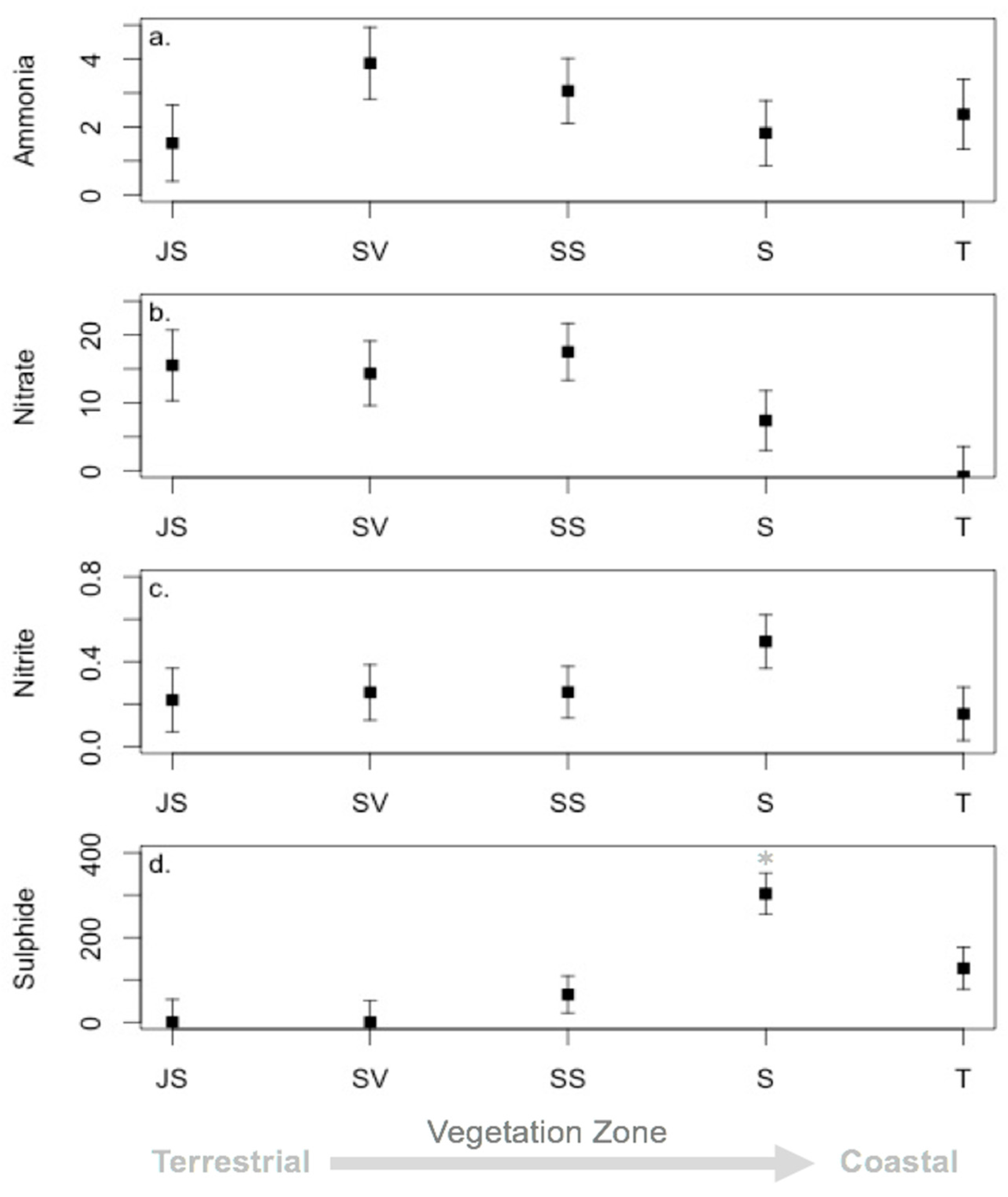
3.3. Environmental Conditions and Sediment Chemistry
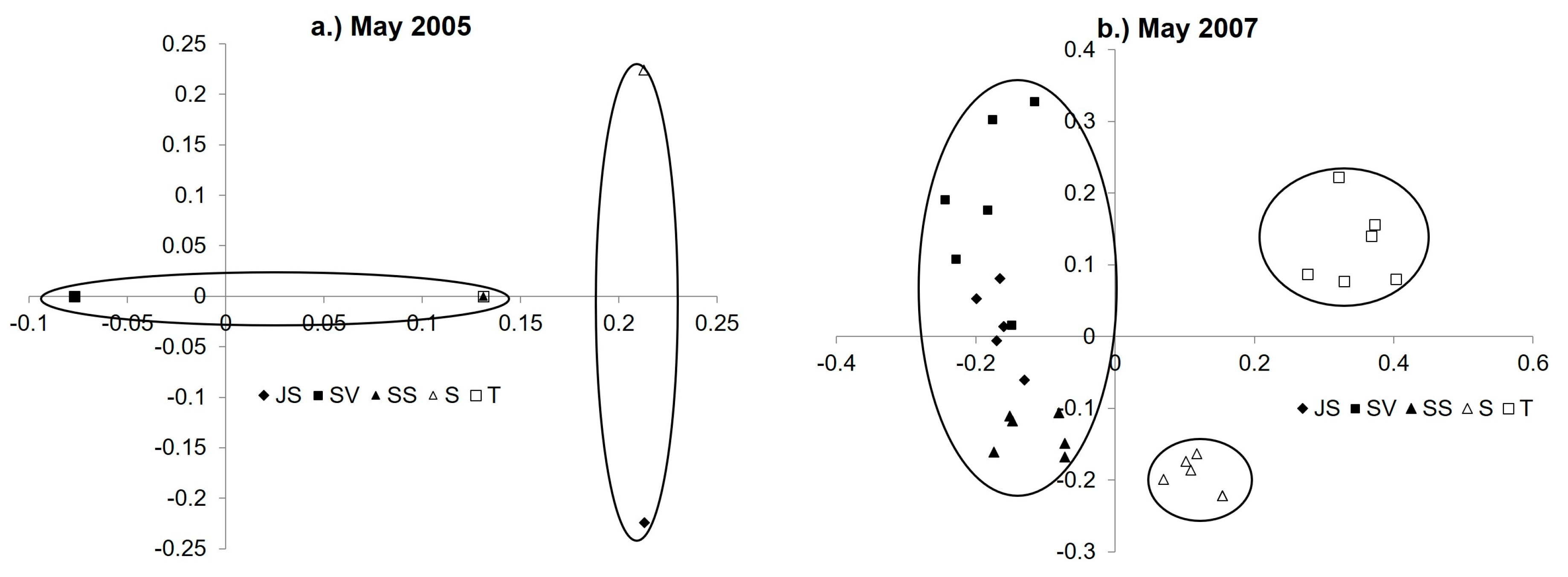
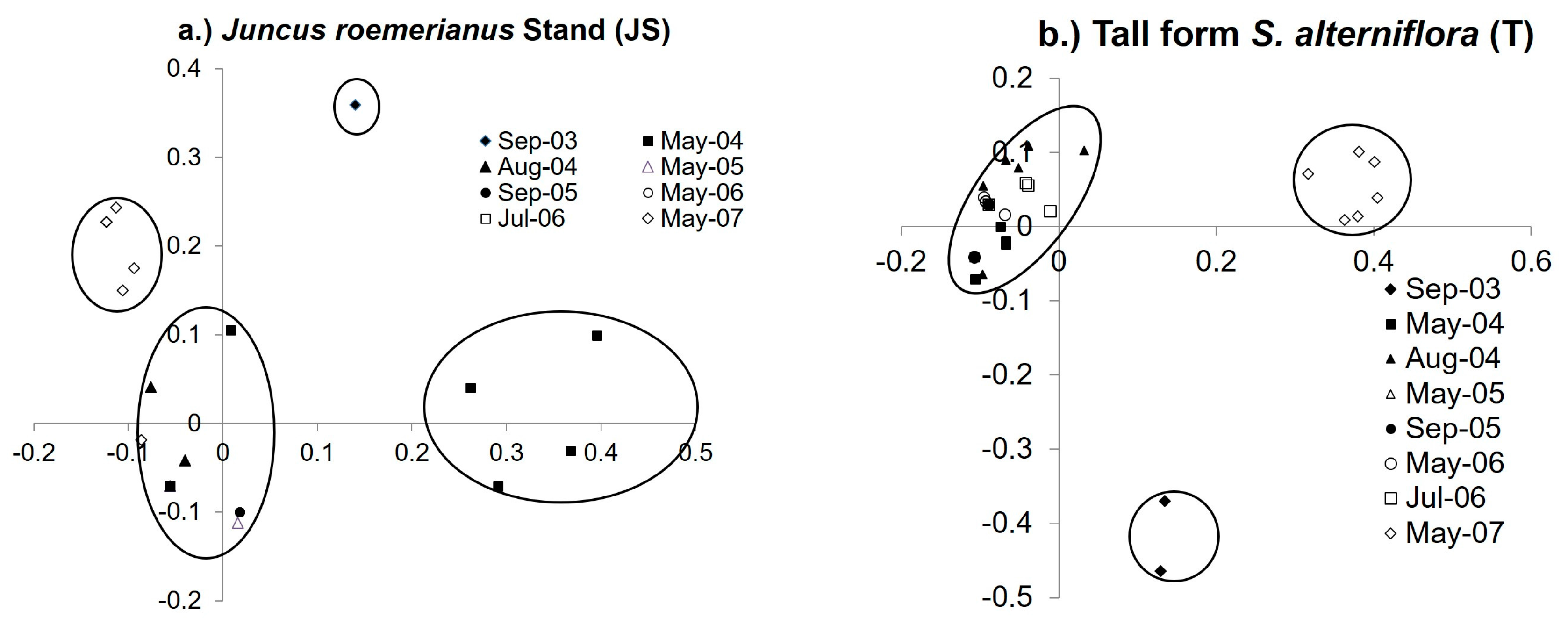
3.4. Diazotroph Assemblages
3.5. Drivers of Ordination and Diazotroph Assemblage Presence
4. Discussion
4.1. Sediment Chemistry is Somewhat Affected by Environmental Conditions
4.2. Diazotroph Assemblages
4.3. Persistent Diazotrophs Maintain Marsh Dynamics Post Drought Event
4.4. Implications and Future Studies
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Turner, R. Georgaphic variations in salt marsh macrophyte production: A review. Contrib. Mar. Sci. 1976, 20, 47–68. [Google Scholar]
- Adams, D. Factors influencing vascular plant zonation in North Carolina salt marshes. Ecology 1963, 44, 445–456. [Google Scholar] [CrossRef]
- Bertness, M.D.; Ellison, A.M. Determinants of pattern in a New England salt marsh plant community. Ecol. Monogr. 1987, 57, 129–147. [Google Scholar] [CrossRef]
- Pennings, S.C.; Callaway, R.M. Salt marsh plant zonation: The relative importance of competition and physical factors. Ecology 1992, 73, 681–690. [Google Scholar] [CrossRef]
- Adam, P. Saltmarsh Ecology; Cambridge University Press: Cambridge, UK, 1990. [Google Scholar]
- Lovell, C.R.; Davis, D.A. Specificity of salt marsh diazotrophs for vegetation zones and plant hosts: Results from a North American marsh. Front. Microbiol. 2012, 3, e84. [Google Scholar] [CrossRef] [PubMed]
- Gardner, L. The effect of hydrologic factors on the porewater chemistry of intertidal marsh sediments. Southeast. Geol. 1973, 15, 17–28. [Google Scholar]
- Hemond, H.F.; Fifield, J.L. Subsurface flow in salt marsh peat: A model and field study1: Flow in marsh peat. Limnol. Oceanogr. 1982, 27, 126–136. [Google Scholar] [CrossRef]
- Marinucci, A.C.; Hobbie, J.E.; Helfrich, J.V.K. Effect of litter nitrogen on decomposition and microbial biomass inSpartina alterniflora. Microb. Ecol. 1983, 9, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Mendelssohn, I.A. Nitrogen metabolism in the height Forms of Spartina alterniflora in North Carolina. Ecology 1979, 60, 574–584. [Google Scholar] [CrossRef]
- Valiela, I.; Teal, J.M.; Volkmann, S.; Shafer, D.; Carpenter, E.J. Nutrient and particulate fluxes in a salt marsh ecosystem: Tidal exchanges and inputs by precipitation and groundwater 1: Salt marsh nutrient exchange. Limnol. Oceanogr. 1978, 23, 798–812. [Google Scholar] [CrossRef]
- Dame, R.F.; Charzanowski, T.; Bildstein, K.; Kjerfve, B.; McKellar, H.; Nelson, D.; Spurrier, J.; Stancyk, S.; Stevenson, H.; Vernberg, J.; et al. The outwelling hypothesis and North Inlet, South Carolina. Mar. Ecol. Prog. Ser. 1986, 33, 217–229. [Google Scholar] [CrossRef]
- Morris, J.T. Effects of nitrogen loading on wetland ecosystems with particular reference to atmospheric deposition. Annu. Rev. Ecol. Syst. 1991, 22, 257–279. [Google Scholar] [CrossRef]
- Whiting, G.J.; Gandy, E.L.; Yoch, D.C. Tight coupling of root-associated nitrogen fixation and plant photosynthesis in the salt marsh grass Spartina alterniflora and carbon dioxide enhancement of nitrogenase activity. Appl. Environ. Microbiol. 1986, 52, 108–113. [Google Scholar] [PubMed]
- McClung, C.; van Berkum, P.; Davis, R.; Sloger, C. Enumeration and localization of N2-fixing bacteria associated with roots of Spartina alterniflora Loisel. Appl. Environ. Microbiol. 1983, 45, 1914–1920. [Google Scholar]
- Patriquin, D. Nitrogen fixation (acetylene reduction) associated with cord grass, Spartina alterniflora Loisel. Ecol. Bull. 1978, 26, 20–27. [Google Scholar]
- Yoch, D.C.; Whiting, G.J. Evidence for NH4+ switch-off regulation of nitrogenase activity by bacteria in salt marsh sediments and roots of the grass Spartina alterniflora. Appl. Environ. Microbiol. 1986, 51, 143–149. [Google Scholar] [PubMed]
- Gandy, E.L.; Yoch, D.C. Relationship between nitrogen-fixing sulfate reducers and fermenters in salt marsh sediments and roots of Spartina alterniflora. Appl. Environ. Microbiol. 1988, 54, 2031–2036. [Google Scholar] [PubMed]
- Bagwell, C.E.; Rocque, J.R.; Smith, G.W.; Polson, S.W.; Friez, M.J.; Longshore, J.W.; Lovell, C.R. Molecular diversity of diazotrophs in oligotrophic tropical seagrass bed communities. FEMS Microbiol. Ecol. 2002, 39, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Bagwell, C.; Dantzler, M.; Bergholz, P.; Lovell, C. Host-specific ecotype diversity of rhizoplane diazotrophs of the perennial glasswort Salicornia virginica and selected salt marsh grasses. Aquat. Microb. Ecol. 2001, 23, 293–300. [Google Scholar] [CrossRef]
- Bagwell, C.E.; Piceno, Y.M.; Ashburne-Lucas, A.; Lovell, C.R. Physiological diversity of the rhizosphere diazotroph assemblages of selected salt marsh grasses. Appl. Environ. Microbiol. 1998, 64, 4276–4282. [Google Scholar] [PubMed]
- Burke, D. Interactions between the salt marsh grass Spartina patens, arbuscular mycorrhizal fungi and sediment bacteria during the growing season. Soil Biol. Biochem. 2003, 35, 501–511. [Google Scholar] [CrossRef]
- Lovell, C.R.; Friez, M.J.; Longshore, J.W.; Bagwell, C.E. Recovery and phylogenetic analysis of nifH sequences from diazotrophic bacteria associated with dead aboveground biomass of Spartina alterniflora. Appl. Environ. Microbiol. 2001, 67, 5308–5314. [Google Scholar] [CrossRef] [PubMed]
- Lovell, C.R.; Piceno, Y.M.; Quattro, J.M.; Bagwell, C.E. Molecular analysis of diazotroph diversity in the rhizosphere of the smooth cordgrass, Spartina alterniflora. Appl. Environ. Microbiol. 2000, 66, 3814–3822. [Google Scholar] [CrossRef] [PubMed]
- Welsh, A.; Burke, D.J.; Hahn, D. Analysis of Nitrogen-Fixing Members of the subclass of Proteobacteria in salt marsh sediments. Appl. Environ. Microbiol. 2007, 73, 7747–7752. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Welsh, A.K.; Burke, D.J.; Hamerlynck, E.P.; Hahn, D. Seasonal analyses of arbuscular mycorrhizae, nitrogen-fixing bacteria and growth performance of the salt marsh grass Spartina patens. Plant Soil 2010, 330, 251–266. [Google Scholar] [CrossRef]
- Davis, D.A.; Gamble, M.D.; Bagwell, C.E.; Bergholz, P.W.; Lovell, C.R. Responses of salt marsh plant rhizosphere diazotroph assemblages to changes in marsh elevation, edaphic conditions and plant host species. Microb. Ecol. 2011, 61, 386–398. [Google Scholar] [CrossRef] [PubMed]
- Gamble, M.D.; Bagwell, C.E.; LaRocque, J.; Bergholz, P.W.; Lovell, C.R. Seasonal variability of diazotroph assemblages associated with the rhizosphere of the salt marsh cordgrass, Spartina alterniflora. Microb. Ecol. 2010, 59, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Bagwell, C.E.; Lovell, C.R. Persistence of selected Spartina alterniflora rhizoplane diazotrophs exposed to natural and manipulated environmental variability. Appl. Environ. Microbiol. 2000, 66, 4625–4633. [Google Scholar] [CrossRef] [PubMed]
- Piceno, Y.; Lovell, C.R. Stability in natural bacterial communities: I. Nutrient addition effects on rhizosphere diazotroph assemblage composition. Microb. Ecol. 2000, 39, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Piceno, Y.; Lovell, C.R. Stability in natural bacterial communities: II. Plant resource allocation effects on rhizosphere diazotroph assemblage composition. Microb. Ecol. 2000, 39, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Scavia, D.; Field, J.C.; Boesch, D.F.; Buddemeier, R.W.; Burkett, V.; Cayan, D.R.; Fogarty, M.; Harwell, M.A.; Howarth, R.W.; Mason, C.; et al. Climate change impacts on U.S. coastal and marine ecosystems. Estuaries 2002, 25, 149–164. [Google Scholar] [CrossRef]
- Silliman, B.R. Drought, snails, and large-scale die-off of southern U.S. salt marshes. Science 2005, 310, 1803–1806. [Google Scholar] [CrossRef] [PubMed]
- Visser, J.M.; Sasser, C.E.; Chabreck, R.H.; Linscombe, R.G. The impact of a severe drought on the vegetation of a subtropical estuary. Estuaries 2002, 25, 1184–1195. [Google Scholar] [CrossRef]
- Kirwan, M.L.; Guntenspergen, G.R.; Morris, J.T. Latitudinal trends in Spartina alterniflora productivity and the response of coastal marshes to global change. Glob. Chang. Biol. 2009, 15, 1982–1989. [Google Scholar] [CrossRef]
- Michener, W.K.; Blood, E.R.; Bildstein, K.L.; Brinson, M.M.; Gardner, L.R. Climate change, hurricanes and tropical storms, and rising sea level in coastal wetlands. Ecol. Appl. 1997, 7, 770–801. [Google Scholar] [CrossRef]
- Morris, J. Long-term Studies of Salt Marsh Primary Production. Available online: http://ww2.biol.sc.edu/~morris/annprod.html (accessed on 24 December 2017).
- Morris, J.T.; Kjerfve, B.; Dean, J.M. Dependence of estuarine productivity on anomalies in mean sea level. Limnol. Oceanogr. 1990, 35, 926–930. [Google Scholar] [CrossRef]
- Hughes, A.L.H.; Wilson, A.M.; Morris, J.T. Hydrologic variability in a salt marsh: Assessing the links between drought and acute marsh dieback. Estuar. Coast. Shelf Sci. 2012, 111, 95–106. [Google Scholar] [CrossRef]
- US Climate Data. Available online: https://www.usclimatedata.com/climate/georgetown/south-carolina/united-states/ussc0127 (accessed on 24 December 2017).
- Kjerfve, B. Circulation and salt flux in a well mixed estuary. In Physics of Shallow Estuaries and Bays; Van de Kreeke, J., Ed.; Springer: New York, NY, USA, 1986. [Google Scholar]
- Bertness, M.D. Zonation of Spartina patens and Spartina alterniflora in New England salt marsh. Ecology 1991, 72, 138–148. [Google Scholar] [CrossRef]
- Wells, N.; Goddard, S.; Hayes, M.J. A self-calibrating Palmer Drought Severity Index. J. Clim. 2004, 17, 2335–2351. [Google Scholar] [CrossRef]
- Malone, S.L.; Tulbure, M.G.; Pérez-Luque, A.J.; Assal, T.J.; Bremer, L.L.; Drucker, D.P.; Hillis, V.; Varela, S.; Goulden, M.L. Drought resistance across California ecosystems: Evaluating changes in carbon dynamics using satellite imagery. Ecosphere 2016, 7, e01561. [Google Scholar] [CrossRef]
- Malone, S.L. Monitoring changes in water use efficiency to understand drought induced tree mortality. Forests 2017, 8, 365. [Google Scholar] [CrossRef]
- Hanson, R.B. Nitrogen fixation activity (acetylene reduction) in the rhizosphere of salt marsh angiosperms, Georgia, U.S.A. Bot. Mar. 1983, 26, 49–60. [Google Scholar] [CrossRef]
- Bradley, P.M.; Morris, J.T. The influence of salinity on the kinetics of NH4+ uptake in Spartina alterniflora. Oecologia 1991, 85, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Trüper, H.G.; Schlegel, H.G. Sulphur metabolism in Thiorhodaceae I. Quantitative measurements on growing cells of Chromatium okenii. Antonie Van Leeuwenhoek 1964, 30, 225–238. [Google Scholar] [CrossRef]
- Mullen, J.B.; Riley, J.P. The spectrophotometric determination of nitrate in natural waters, with particular reference to sea-water. Anal. Chim. Acta 1955, 12, 464–480. [Google Scholar] [CrossRef]
- Hansen, H.; Grasshoff, K. Automated chemical analysis. In Methods of Seawater Analysis; Grasshoff, K., Ehrhardt, M., Kremling, K., Eds.; Chemie: Deerfield Beach, FL, USA, 1983; pp. 347–395. [Google Scholar]
- Nitrate-Nitrite in Water and Seawater; Technicon Industrial Systems. Technicon Industrial Methods No. 161-71W/B; Technicon Instruments Corporation: Tarrytown, New York, NY, USA, 1976.
- Tides and Currents. Available online: https://tidesandcurrents.noaa.gov/ (accessed on 1 June 2017).
- Lovell, C.R.; Piceno, Y. Purification of DNA from estuarine sediments. J. Microbiol. Methods 1994, 20, 161–174. [Google Scholar] [CrossRef]
- Piceno, Y.; Noble, P.; Lovell, C.R. Spatial and temporal assessment of diazotroph assemblage composition in vegetated salt marsh sediments using denaturing gradient gel electrophoresis analysis. Microb. Ecol. 1999, 38, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.K.; Brown, D.M. Synthesis and duplex stability of oligonucleotides containing cytosine-thymine analogues. Nucleic Acids Res. 1989, 17, 10373–10383. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Thoo Lin, P.K. Synthesis and duplex stability of oligonucleotides containing adenine-guanine analogues. Carbohydr. Res. 1992, 216, 129–139. [Google Scholar] [CrossRef]
- LaRocque, J.R.; Bergholz, P.W.; Bagwell, C.E.; Lovell, C.R. Influence of host plant-derived and abiotic environmental parameters on the composition of the diazotroph assemblage associated with roots of Juncus roemerianus. Antonie Van Leeuwenhoek 2004, 86, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Fromin, N.; Hamelin, J.; Tarnawski, S.; Roesti, D.; Jourdain-Miserez, K.; Forestier, N.; Teyssier-Cuvelle, S.; Gillet, F.; Aragno, M.; Rossi, P. Statistical analysis of denaturing gel electrophoresis (DGE) fingerprinting patterns: Use of statistics for the interpretation of DGE patterns. Environ. Microbiol. 2002, 4, 634–643. [Google Scholar] [CrossRef] [PubMed]
- Debra, D. Responses of Salt Marsh Plant Rhizosphere Diazotroph Assemblages to Drought. KNB 2018. [Google Scholar] [CrossRef]
- Lovell, C.R.; Decker, P.V.; Bagwell, C.E.; Thompson, S.; Matsui, G.Y. Analysis of a diverse assemblage of diazotrophic bacteria from Spartina alterniflora using DGGE and clone library screening. J. Microbiol. Methods 2008, 73, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K.; Kumar, S. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. The R Project for Statistical Computing. Available online: www.r-project.org (accessed on 23 June 2017).
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67. [Google Scholar] [CrossRef]
- Oksanen, J. Vegan: An Introduction to Ordination. Available online: https://cran.r-project.org/web/packages/vegan/vignettes/intro-vegan.pdf (accessed on 23 June 2017).
- Oksanen, J. Multivariate analysis of ecological communities in R: Vegan tutorial. Available online: http://cc.oulu.fi/~jarioksa/opetus/metodi/vegantutor.pdf (accessed on 23 June 2017).
- Oksanen, J.; Guillaume Blanchet, F.; Friendly, M.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Simpson, G.L.; Solymos, P.; Henry, M.; et al. Community Ecology Package. Available online: https://cran.ism.ac.jp/web/packages/vegan/vegan.pdf (accessed on 23 June 2017).
- Hamers, L.; Hemeryck, Y.; Herweyers, G.; Janssen, M.; Keters, H.; Rousseau, R.; Vanhoutte, A. Similarity measures in scientometric research: The Jaccard index versus Salton’s cosine formula. Inf. Process. Manag. 1989, 25, 315–318. [Google Scholar] [CrossRef]
- Bien, J.; Tibshirani, R. Hierarchical clustering with prototypes via minimax linkage. J. Am. Stat. Assoc. 2011, 106, 1075–1084. [Google Scholar] [CrossRef] [PubMed]
- Ramette, A. Multivariate analyses in microbial ecology. FEMS Microbiol. Ecol. 2007, 62, 142–160. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Eijkemans, M.J.C.; Wallinga, J.; Biesbroek, G.; Trzciński, K.; Sanders, E.A.M.; Bogaert, D. Multivariate approach for studying interactions between environmental variables and microbial communities. PLoS ONE 2012, 7, e50267. [Google Scholar] [CrossRef] [PubMed]
- Venter, J.C.; Remington, K.; Heidelberg, J.F.; Halpern, A.L.; Rusch, D.; Eisen, J.A.; Wu, D.; Paulsen, I.; Nelson, K.E.; Nelson, W.; et al. Environmental genome shotgun sequencing of the Sargasso Sea. Science 2004, 304, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Giblin, A.E.; Howarth, R.W. Porewater evidence for a dynamic sedimentary iron cycle in salt marshes1: Iron cycle in salt marshes. Limnol. Oceanogr. 1984, 29, 47–63. [Google Scholar] [CrossRef]
- Bradley, P.M.; Morris, J.T. Influence of oxygen and sulfide concentration on nitrogen uptake kinetics in Spartina alterniflora. Ecology 1990, 71, 282–287. [Google Scholar] [CrossRef]
- Benner, R.; Newell, S.; MacCubbin, A.; Hodson, R.E. Relative contributions of bacteria and fungi to rates of degradation of lignocellulosic detritus in salt-marsh sediments. Appl. Environ. Microbiol. 1984, 48, 36–40. [Google Scholar] [PubMed]
- McKee, K.L.; Mendelssohn, I.A.; Materne, M.D. Acute salt marsh dieback in the Mississippi River deltaic plain: A drought-induced phenomenon? Glob. Ecol. Biogeogr. 2004, 13, 65–73. [Google Scholar] [CrossRef]
- Thompson, S.P.; Paerl, H.W.; Go, M.C. Seasonal patterns of nitrification and denitrification in a natural and a restored salt marsh. Estuaries 1995, 18, 399–408. [Google Scholar] [CrossRef]
- Lovell, C. Stability of a rhizosphere microbial community exposed to natural and manipulated environmental variability. FEMS Microbiol. Ecol. 2001, 38, 69–76. [Google Scholar] [CrossRef][Green Version]
- Gillis, M.; Kersters, K.; Hoste, B.; Janssens, D.; Kroppenstedt, R.M.; Stephan, M.P.; Teixeira, K.R.S.; Dobereiner, J.; De Ley, J. Acetobacter diazotrophicus sp. nov., a nitrogen-fixing acetic acid bacterium associated with sugarcane. Int. J. Syst. Bacteriol. 1989, 39, 361–364. [Google Scholar] [CrossRef]
- Panikov, N.S. Understanding and prediction of soil microbial community dynamics under global change. Appl. Soil Ecol. 1999, 11, 161–176. [Google Scholar] [CrossRef]
- Schimel, J.; Balser, T.C.; Wallenstein, M. Microbial stress-response physiology and its implications for ecosystem function. Ecology 2007, 88, 1386–1394. [Google Scholar] [CrossRef] [PubMed]

| Parameters | PC1 | PC2 | R2 | p-Value | |
|---|---|---|---|---|---|
| Tide (m) | −0.8349 | 0.5504 | 0.01 | 0.894 | |
| Sediment Temperature (°C) | −0.4182 | 0.9084 | 0.01 | 0.931 | |
| Salinity (ppt) | −0.3844 | −0.9232 | 0.01 | 0.782 | |
| pH | 0.5676 | 0.8233 | 0.02 | 0.461 | |
| Ammonia | −0.4668 | −0.8844 | 0.01 | 0.698 | |
| Nitrate | −1.0000 | −0.0023 | 0.02 | 0.449 | |
| Nitrite | 0.8412 | −0.5408 | 0.02 | 0.326 | |
| Sulfide | 0.9900 | 0.1414 | 0.02 | 0.352 | |
| Group | 1 | −0.1079 | −0.1349 | 0.41 | 0.001 |
| 2 | 0.4435 | 0.1412 | |||
| 3 | 0.0883 | 0.6255 | |||
| 4 | 0.4232 | 0.4104 | |||
| 5 | −0.0277 | 0.3129 | |||
| Vegetation Zone | JS | 0.0159 | −0.1705 | 0.07 | 0.131 |
| SAL | −0.0235 | −0.0705 | |||
| SS | −0.0573 | −0.0658 | |||
| S | 0.0731 | 0.1548 | |||
| T | 0.0402 | 0.0563 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davis, D.A.; Malone, S.L.; Lovell, C.R. Responses of Salt Marsh Plant Rhizosphere Diazotroph Assemblages to Drought. Microorganisms 2018, 6, 27. https://doi.org/10.3390/microorganisms6010027
Davis DA, Malone SL, Lovell CR. Responses of Salt Marsh Plant Rhizosphere Diazotroph Assemblages to Drought. Microorganisms. 2018; 6(1):27. https://doi.org/10.3390/microorganisms6010027
Chicago/Turabian StyleDavis, Debra A., Sparkle L. Malone, and Charles R. Lovell. 2018. "Responses of Salt Marsh Plant Rhizosphere Diazotroph Assemblages to Drought" Microorganisms 6, no. 1: 27. https://doi.org/10.3390/microorganisms6010027
APA StyleDavis, D. A., Malone, S. L., & Lovell, C. R. (2018). Responses of Salt Marsh Plant Rhizosphere Diazotroph Assemblages to Drought. Microorganisms, 6(1), 27. https://doi.org/10.3390/microorganisms6010027





