Microbiological Quality of Fresh Nopal Juice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling of Fresh Juice
2.2. Microbiological Analysis
2.3. Biochemical Identification and Characterization
2.4. Serological and Molecular Confirmation
2.5. Statistical Analysis
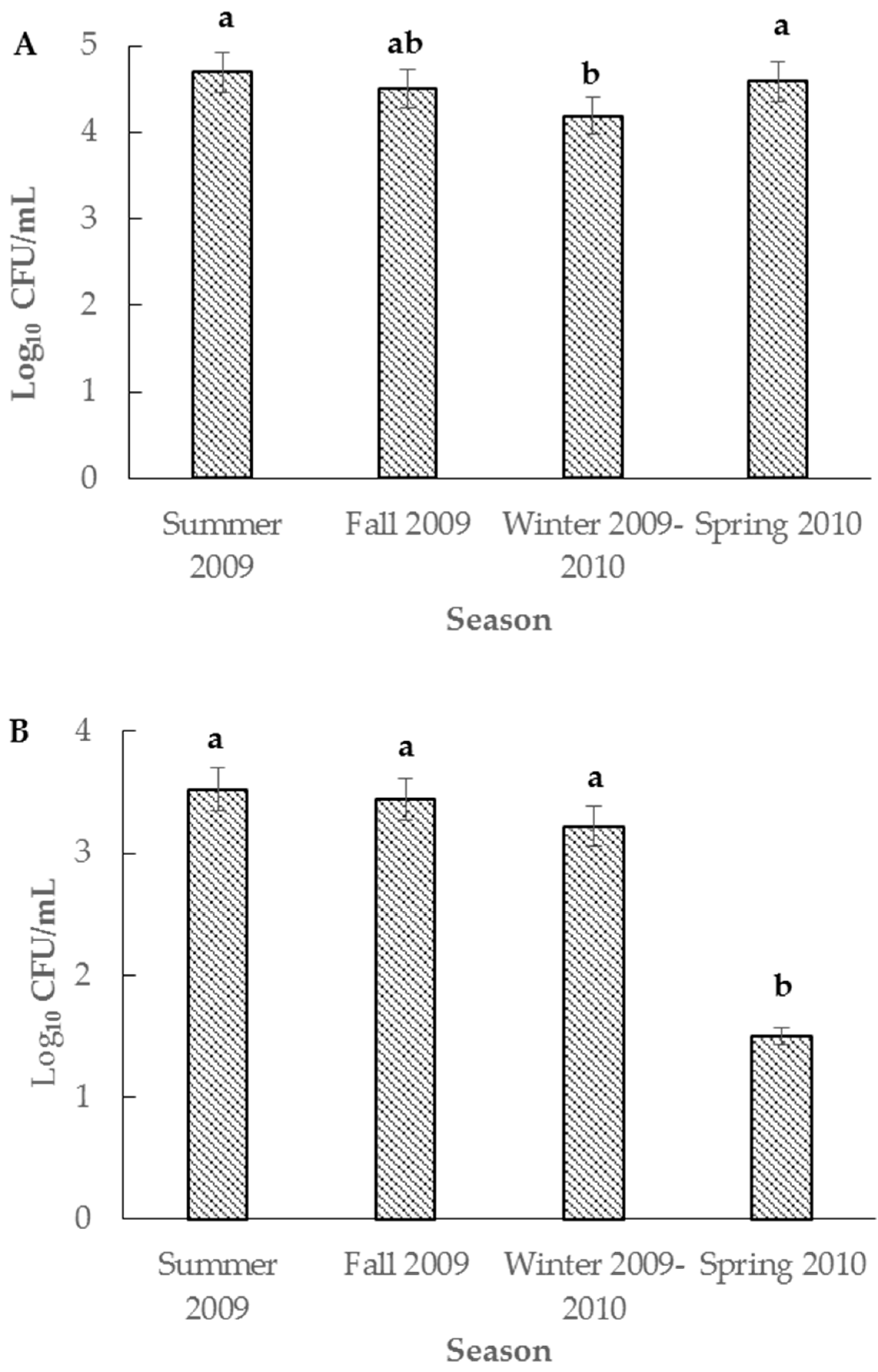
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Feugang, J.; Konarski, A.P.; Zou, D.; Stintzing, F.C.; Zou, C. Nutritional and medicinal use of cactus pear (Opuntia spp.) cladodes and fruits. Front. Biosci. 2006, 11, 2574–2589. [Google Scholar] [CrossRef] [PubMed]
- Galati, E.M.; Monforte, M.T.; Tripodo, M.M.; d’Aquino, A.; Mondello, M.R. Antiulcer activity of Opuntia ficus-indica (L.) Mill. (Cactaceae): Ultrastructural study. J. Ethnopharmacol. 2001, 76, 1–9. [Google Scholar] [CrossRef]
- Galati, E.M.; Monforte, M.T.; Miceli, N.; Mondello, M.R.; Taviano, M.R.; Galluzzo, M.; Tripodo, M.M. Opuntia ficus-indica (L.) Mill. Mucilages show cytoprotective effect on gastric mucosa in rat. Phytother. Res. 2007, 21, 344–346. [Google Scholar] [CrossRef] [PubMed]
- Galati, E.M.; Mondello, M.R.; Monforte, M.T.; Galluzzo, M.; Miceli, N.; Tripodo, M.M. Effect of Opuntia ficus-indica (L.) Mill. Cladodes in the wound-healing process. J. Prof. Assoc. Cactus 2003, 5, 1–16. [Google Scholar]
- Basurto, S.D.; Lorenzana, J.M.; Magos, G.A. Utilidad del nopal para el control de la glucosa en la diabetes mellitus tipo 2. Rev. Fac. Med. UNAM 2006, 49, 157–162. [Google Scholar]
- Hernández, A.A.M.; Landa, S.P.; Mora, A.G.; Eslava, C.C.A.; Call, J.E.; Porto-Fett, A.C.S.; Luchansky, J.B. Characterization of Salmonella spp. from nopal leaves and associated soil and water samples in Morelos, Mexico. In Proceedings of the International Association for Food Protection (IAFP) Annual Meeting, Grapevine, TX, USA, 3–6 August 2008; pp. 74–75.
- Hirotani, H.; Naranjo, J.; Motoyoqui, P.G.; Gerb, C.P. Demonstration of indicator of microorganisms on surface of vegetables on the market in the United States and Mexico. J. Food Sci. 2002, 67, 1847–1850. [Google Scholar] [CrossRef]
- Quiroz-Santiago, C.; Rodas-Súarez, O.R.; Vázquez, Q.C.R.; Fernández, F.J.; Quiñones-Ramírez, E.I.; Vázquez-Salinas, C. Prevalence of Salmonella in vegetables from México. J. Food Prot. 2009, 72, 1279–1282. [Google Scholar] [CrossRef] [PubMed]
- Castillo, A.; Villarruel-López, A.; Navarro-Hidalgo, V.; Martínez-González, N.E.; Torres-Vitela, M.R. Salmonella and Shigella in freshly squeezed orange juice fresh oranges, and wiping cloths collected from public markets and street booths in Guadalajara, México: Incidence and comparison and analytical routes. J. Food Prot. 2006, 69, 2595–2599. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.E.; Thompson, P.; Rao, B.N.; Kalavati, C.; Rajanna, B. Human bacteria in street vended fruit juices: A case study of Visakhapatnam city, India. Internet J. Food Saf. 2006, 8, 35–38. [Google Scholar]
- Parish, M.E. Public health and nonpasteurized fruit juices. Crit. Rev. Microbiol. 1997, 23, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Parish, M.E. Food safety issues and the microbiology of fruit beverages and bottled water. In Microbiologically Safe Foods; Heredia, N., Wesley, I., Garcia, S., Eds.; John Wiley and Son Inc.: Hoboken, NJ, USA, 2009; pp. 291–304. [Google Scholar]
- Norma Oficial Mexicana. NOM-113-SSA-1994. Bienes y Servicios. Método Para la Cuenta de Microorganismos Coliformes Totales en Placa. Diario Oficial de la Federación, México. Available online: http://www.salud.gob.mx/unidades/cdi/nom/113ssa14.html (accessed on July 2008).
- Norma Oficial Mexicana. NOM-114-SSA1-1994. Bienes y Servicios. Método Para la Determinación de Salmonella en Alimentos. Diario Oficial de la Federación, México. Available online: http://www.salud.gob.mx/unidades/cdi/nom/114ssa14.html (accessed on July 2008).
- Winn, W.C.; Allen, S.D.; Janda, W.M.; Koneman, E.W.; Procop, G.W.; Schreckenberger, P.C.; Woods, G.L. Diagnóstico Microbiológico, texto y atlas en Color, 6th ed.; Editorial Medica Panamericana: Buenos Aires, Argentina, 2008; p. 1475. [Google Scholar]
- Terragno, R.; Caffer, M.A.I.; Bruno, S.; Binsztein, N. Parte I: Aislamiento Identificación y Serotipificación de Salmonella, Manual de Procedimientos; Instituto Nacional de Enfermedades Infecciosas (INEI), Departamento de Bacteriología: Buenos Aires, Argentina, 2003; p. 56. [Google Scholar]
- Popoff, M.; Bockemuhl, J.; McWorther-Murlin, A. The Kauffmann-White scheme. Res. Microbiol. 1991, 34, 1029–1033. [Google Scholar] [CrossRef]
- Rahn, K.; De Grandis, S.A.; Clarke, R. Amplification of invA gene sequence of Salmonella typhimurium by polymerase chain reaction as a specific method of detection of Salmonella. Mol. Cell. Probes 1992, 6, 271–279. [Google Scholar] [CrossRef]
- Mexican Official Standards (NOM). Environmental Health: Water for Human Use and Consumption. Permissible Limits of Quality and Treatments Must Undergo for Water Purification. Available online: http://www.salud.gob.mx/unidades/cdi/nom/127ssa14.html (accessed on July 2008).
- Food Safety Modernitation Act (FSMA). Available online: http://www.gpo.gov/fdsys/pkg/PLAW-111publ353/pdf/PLAW-111publ353.pdf (accessed on 29 June 2016).
- Berger, C.N.; Sodha, S.V.; Shaw, R.K.; Griffin, P.M.; Pink, D.; Hand, P.; Frankel, G. Fresh fruit and vegetables as vehicles for the transmission of human pathogens. Environ. Microbiol. 2010, 12, 2385–2397. [Google Scholar] [CrossRef] [PubMed]
- Winfield, M.D.; Groisman, E.A. Role of nonhost environments in the lifestyles of Salmonella and Escherichia coli. Appl. Environ. Microb. 2003, 69, 3687–3694. [Google Scholar] [CrossRef]
- Changla, A.H.; Borczyk, A.A.; Aldom, J.E.; Dalla Rosa, S.; Cole, D.D. Evaluation of the L-pyrrolidonyl-beta-naphthylamide hydrolysis test for the differentiation of members of the families Enterobacteriaceae and Vibrionaceae. J. Clin. Microbiol. 1993, 31, 1946–1948. [Google Scholar]
- Reyes, R.E.; Ramírez-Saad, H.; Pérez, J.; Meléndez, E.; Navaroo, A.; Murata, C.; Cravioto, A. Intra-strain variation of lipopolysaccharides from Citrobacter freundii E97750 by in vitro passages. Afr. J. Microbiol. Res. 2010, 4, 1324–1331. [Google Scholar]
- O’Hara, C.; Roman, S.B.; Miller, J.M. Ability of commercial identification systems to identify newly recognized species of Citrobacter. J. Clin. Microbiol. 1995, 33, 242–245. [Google Scholar] [PubMed]
- Ronald, A. The etiology of urinary tract infection: Traditional and emerging pathogens. Dis. Mon. 2003, 49, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Doran, T.I. The role of Citrobacter in clinical disease of children: Review. Clin. Infect. Dis. 1999, 28, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Jackson, B.R.; Griffin, P.M.; Cole, D.; Walsh, K.A.; Chai, S.J. Outbreak-Associated Salmonella enterica Serotypes and Food Commodities, United States, 1998–2008. Emerg. Infect. Dis. 2013, 19, 1239–1244. Available online: www.cdc.gov/eid (accessed on 1 March 2016). [Google Scholar] [CrossRef] [PubMed]
- Cook, K.A.; Dobbs, T.E.; Hlady, W.G.; Wells, J.G.; Barrett, T.J.; Puhr, N.D.; Lancette, G.A.; Bodager, D.W.; Toth, B.L.; Genese, C.A.; et al. Outbreak of Salmonella serotype Hartford infections associated with unpasteurized orange juice. JAMA 1998, 280, 1504–1509. [Google Scholar] [CrossRef] [PubMed]
- Krause, G.; Terzagian, R.; Hammond, R. Outbreak of Salmonella serotype Anatum infection associated with unpasteurized orange juice. South. Med. J. 2001, 94, 1168–1172. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Bidol, S.A.; Austion, J.L.; Berl, E.; Elson, F.; Williams, M.L.; Deasy, M.; Moll, M.E.; Rea, V.; et al. Multistate Outbreak of Salmonella Typhimurium and Saintpaul Infections Associated with Unpasteurized Orange Juice—United States, 2005. Clin. Infect. Dis. 2009, 48, 1065–1071. [Google Scholar] [CrossRef] [PubMed]

| Stand | Juice Composition | Recommendation * |
|---|---|---|
| In building (A) | ||
| A | Nopal, natural water | |
| A | Nopal, celery, orange, lemon, pineapple, and purslane | Cholesterol |
| A | Nopal, oats, orange and apple | Cholesterol |
| A | Nopal, broccoli, cabbage, asparagus and grapefruit | Glucose |
| A | Nopal, aloe vera, grapefruit and xoconostle | Diabetes |
| A | Nopal, garlic, lemon, parsley and pineapple | Reumas |
| A | Nopal, celery, sugar beet and pineapple | Slim down |
| A | Nopal, celery, lemon, parsley and carrot | Clean stomach |
| A | Nopal, squash, orange, cucumber and pineapple | Kidneys |
| A | Nopal, natural water, aloe vera and xoconostle | Matrix and cysts |
| A | Nopal, celery, parsley and pineapple | Diet |
| A | Nopal, prunes, flaxseed, papaya and orange | Constipation |
| A | Nopal, natural water, parsley and sugar beet | High triglycerides |
| A | Nopal, celery, honey, orange and pineapple | Diet |
| A | Nopal, oats, apple, honey and orange | Cholesterol 1 |
| A | Nopal, alfalfa, lemon, honey, orange and pineapple | Cholesterol 2 |
| A | Nopal, natural water, aloe vera and xoconostle | Diabetes |
| A | Nopal, olive oil, sugar beet, flax, papaya, honey and orange | Laxative |
| A | Nopal, aloe vera, grape fruit and xoconostle | Cysts |
| A | Nopal, prunes, flaxseed, honey, papaya and orange | Constipation |
| In street booth (B) | ||
| B | Nopal, natural water | |
| B | Nopal, celery, orange, parsley and pineapple | Diet |
| Stand-Juice Composition | pH | TC Log10 | E. coli Log10 | Citrobacter | Proteus | Salmonella |
|---|---|---|---|---|---|---|
| In building | ||||||
| Nopal, natural water | 4.9 abcd | 4.3 cdef | 2.7 bc | 7 | ||
| Nopal, celery, orange, lemon, pineapple, and purslane | 4.5 defg | 4.8 abcd | 3.6 ab | 1 | ||
| Nopal, oats, orange and apple | 4.5 cdefg | 4.4 cdef | 3.1 bc | |||
| Nopal, broccoli, cabbage, asparagus and grapefruit | 4.4 defg | 4.2 def | 3.3 bc | |||
| Nopal, aloe vera, grapefruit and xoconostle | 4.3 efg | 4.2 cdef | 3.4 b | |||
| Nopal, garlic, lemon, parsley and pineapple | 4.3 efg | 3.3 g | 2.1 cd | |||
| Nopal, celery, sugar beet and pineapple | 4.5 cdefg | 4.9 abcd | 3.8 ab | 7 | ||
| Nopal, celery, lemon, parsley and carrot | 5.1 ab | 5.6 a | 4.8 a | 10 | ||
| Nopal, squash, orange, cucumber and pineapple | 4.4 defg | 4.7 bcde | 3.4 b | |||
| Nopal, natural water, aloe vera and xoconostle | 4.6 bcdefg | 4.4 cdef | 3.1 bc | 18 | ||
| Nopal, celery, parsley and pineapple | 4.5 cdefg | 5.1 abc | 3.8 ab | 2 | ||
| Nopal, prunes, flaxseed, papaya and orange | 4.7 bcdef | 4.3 cdef | 3.2 bc | 2 | ||
| Nopal, natural water, parsley and sugar beet | 4.7 bcdefg | 4.7 abcd | 3.4 bc | |||
| Nopal, celery, honey, orange and pineapple | 4.1 g | 5.3 ab | 1.3 de | |||
| Nopal, oats, apple, honey and orange | 4.5 cdefg | 4.6 bcde | 1.2 de | |||
| Nopal, alfalfa, lemon, honey, orange and pineapple | 4.2 fg | 4.9 abcd | 1.0 e | |||
| Nopal, natural water, aloe vera and xoconostle | 4.3 efg | 4.1 defg | 1.0 de | |||
| Nopal, olive oil, sugar beet, flax, papaya, honey, orange | 5.0 abc | 4.8 bcd | 0.9 e | |||
| Nopal, aloe vera, grape fruit and xoconostle | 4.6 bcdefg | 4.7 bcde | 1.3 g | 3 | ||
| Nopal, prunes, flaxseed, honey, papaya and orange | 4.8 abcd | 3.9 efg | 1.0 e | |||
| In street booth | ||||||
| Nopal, natural water | 4.9 abcd | 4.5 cdef | 3.1 bc | 5 | 5 | 1 |
| Nopal, celery, orange, parsley and pineapple | 4.5 defg | 3.7 fg | 2.89 bc |
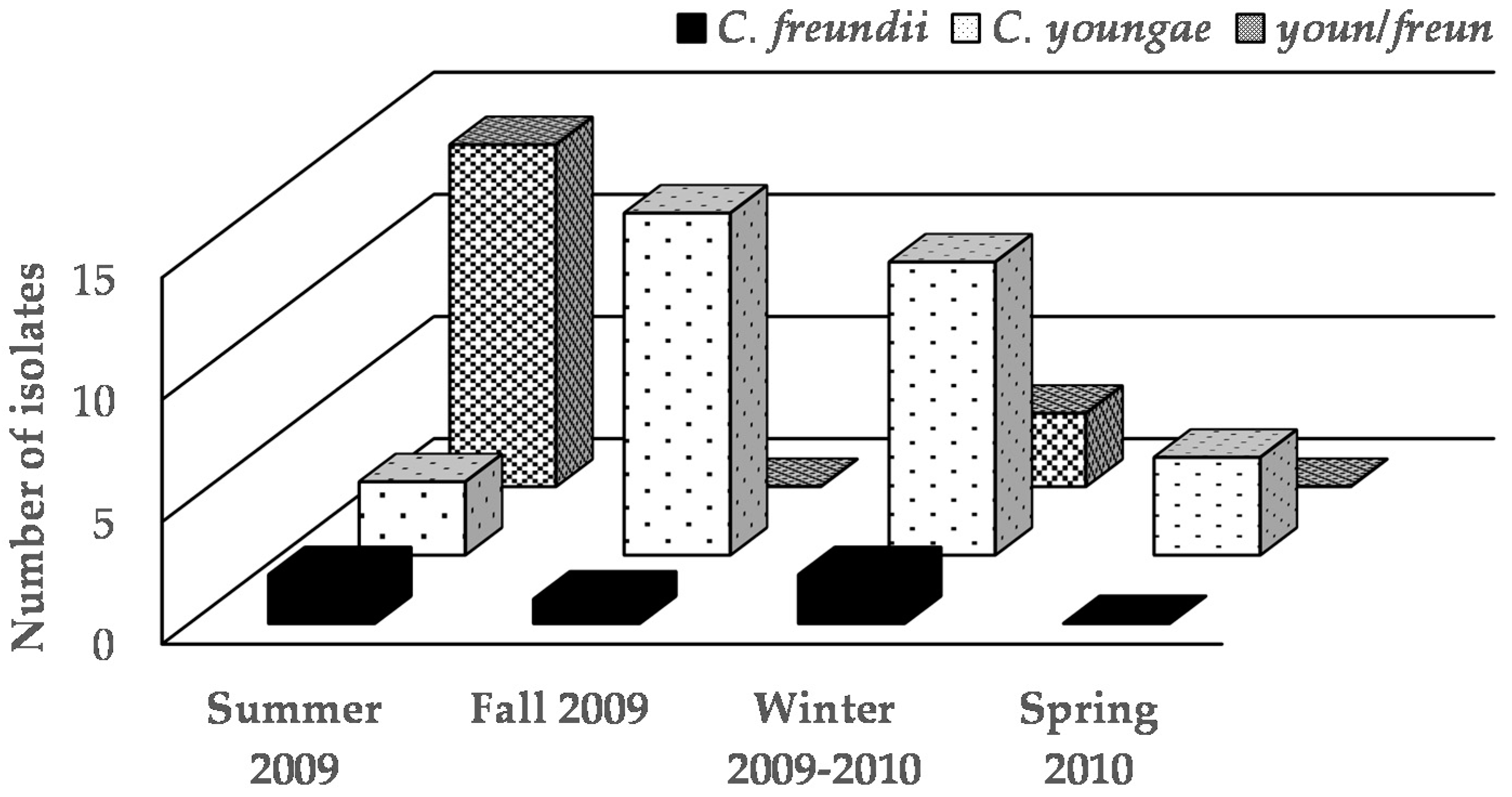
| Month | Stand-Juice | Number of Strains | Characterization | |
|---|---|---|---|---|
| Biochemical | VITEK 2 | |||
| June | A-Nopal, natural water, aloe vera and xoconostle | 4 | C. freundii (4) ¥ | C. youngae/C. freundii (3) C. youngae (1) |
| July | A-Nopal, celery, lemon, parsley and carrot | 2 | C. freundii (2) | C. youngae/C. freundii (2) |
| A-Nopal, natural water, aloe vera and xoconostle | 3 | C. freundii (3) | Unidentified (1) C. youngae/C. freundii (2) | |
| August | A-Nopal, celery, orange, lemon, pineapple, and purslane | 1 | C. freundii (1) | C. freundii (1) |
| A-Nopal, celery, sugar beet and pineapple | 5 | C. freundii (5) | C. youngae/C. freundii (4) | |
| C. freundii (1) | ||||
| A-Nopal, celery, lemon, parsley and carrot | 1 | C. freundii (1) | C. youngae (1) | |
| A-Nopal, prunes, flaxseed, papaya and orange | 2 | C. freundii (2) | C. youngae/C. freundii (1) | |
| C. youngae (1) | ||||
| B-Nopal, natural water | 3 | C. freundii (3) | Salmonella (1) | |
| C. youngae/C. freundii (2) | ||||
| September | A-Nopal, celery, lemon, parsley and carrot | 7 | C. freundii (7) | C. youngae (7) |
| A-Nopal, natural water, aloe vera and xoconostle | 1 | C. freundii (1) | C. freundii (1) | |
| October | A-Nopal, celery, sugar beet and pineapple | 2 | C. freundii (2) | C. youngae (2) |
| A-Nopal, celery, lemon, parsley and carrot | 2 | C. freundii (2) | C. youngae (2) | |
| B-Nopal, natural water | 1 | C. freundii (1) | C. youngae (1) | |
| November | A-Nopal, celery, parsley and pineapple | 2 | C. freundii (2) | C. youngae (2) |
| December | A-Nopal, natural water | 5 | C. freundii (5) | C. youngae (5) |
| A-Nopal, natural water, aloe vera and xoconostle | 5 | C. freundii (5) | C. freundii (2) C. youngae/C. freundii (3) | |
| B-Nopal, natural water | 2 | C. freundii (2) | C. youngae (2) | |
| January | A-Nopal, natural water, aloe vera and xoconostle | 2 | C. freundii (2) | C. youngae (2) |
| February | A-Nopal, natural water | 2 | C. freundii (2) | C. youngae (2) |
| March | A-Nopal, celery, lemon, parsley and carrot | 1 | C. freundii (1) | C. youngae (1) |
| April | A-Nopal, natural water, aloe vera and xoconostle | 1 | C. freundii (1) | C. youngae (1) |
| May | A-Nopal, aloe vera, grape fruit and xoconostle | 3 | C. freundii (3) | C. youngae (3) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Anguiano, A.M.; Landa-Salgado, P.; Eslava-Campos, C.A.; Vargas-Hernández, M.; Patel, J. Microbiological Quality of Fresh Nopal Juice. Microorganisms 2016, 4, 46. https://doi.org/10.3390/microorganisms4040046
Hernández-Anguiano AM, Landa-Salgado P, Eslava-Campos CA, Vargas-Hernández M, Patel J. Microbiological Quality of Fresh Nopal Juice. Microorganisms. 2016; 4(4):46. https://doi.org/10.3390/microorganisms4040046
Chicago/Turabian StyleHernández-Anguiano, Ana María, Patricia Landa-Salgado, Carlos Alberto Eslava-Campos, Mateo Vargas-Hernández, and Jitendra Patel. 2016. "Microbiological Quality of Fresh Nopal Juice" Microorganisms 4, no. 4: 46. https://doi.org/10.3390/microorganisms4040046






