Adhesion Properties of Lactic Acid Bacteria on Intestinal Mucin
Abstract
:1. Introduction
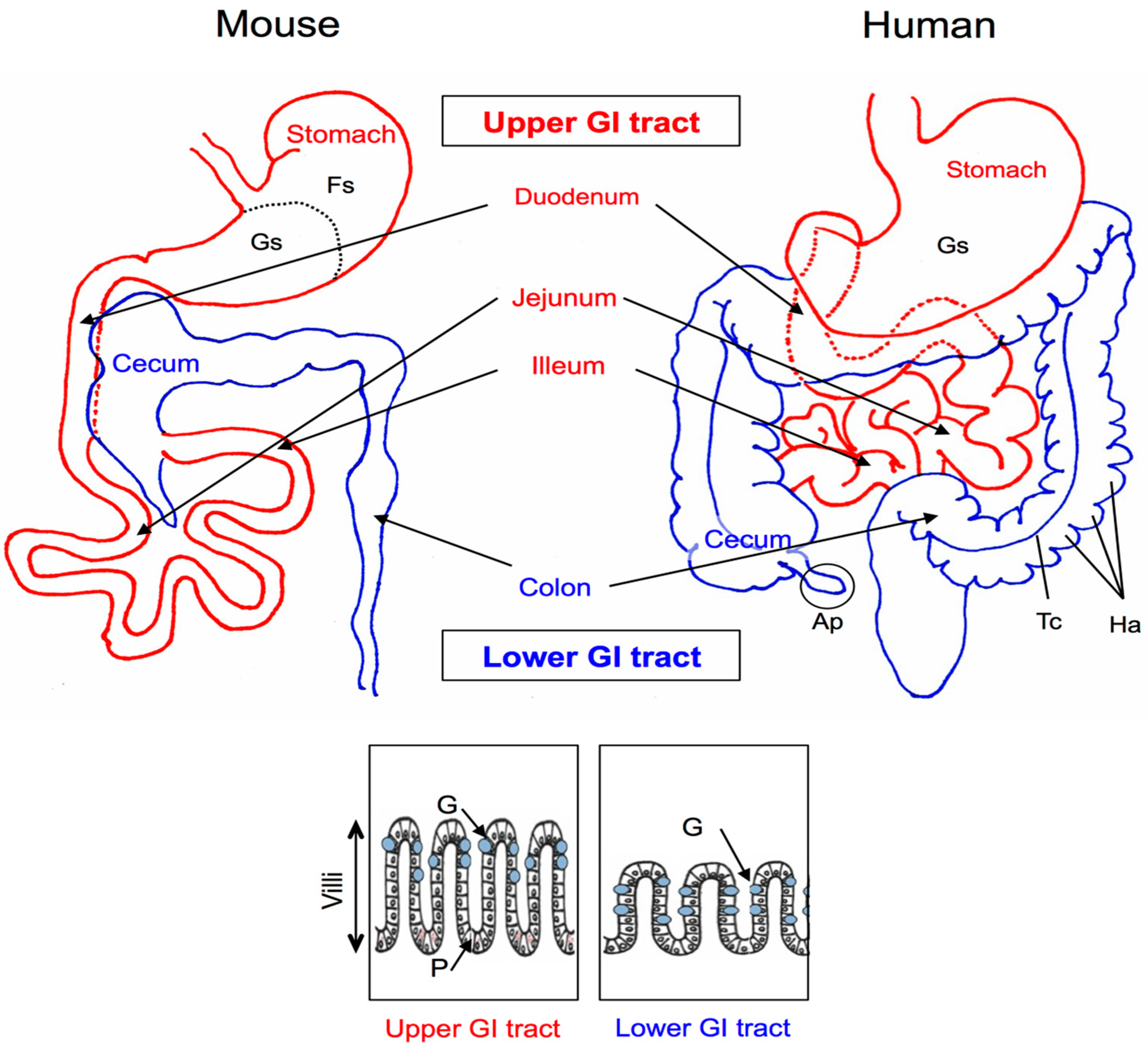
2. Anatomy and Histology of the Mouse and Human GI Tracts
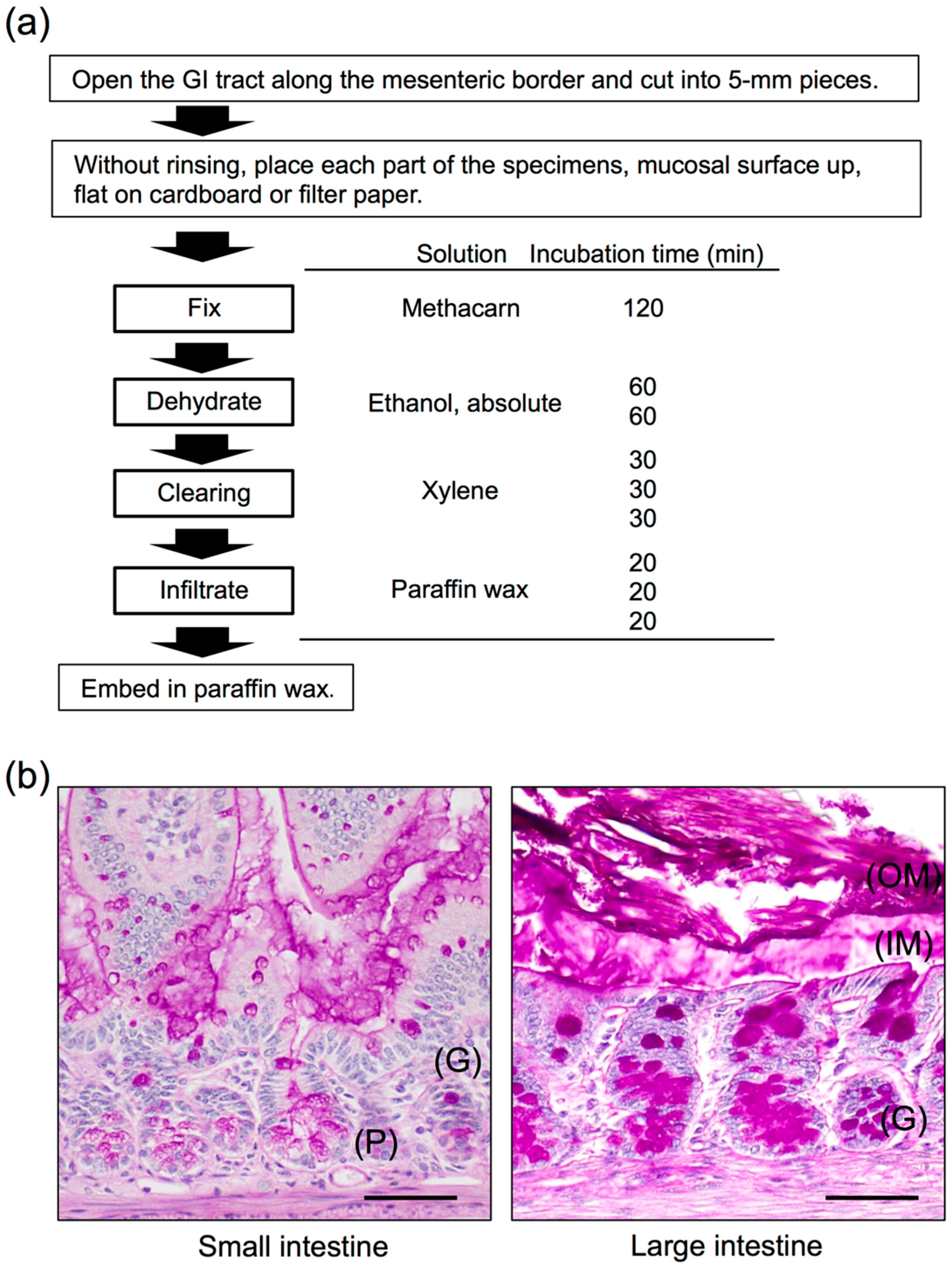
3. Histochemical Studies of the GI Mucus
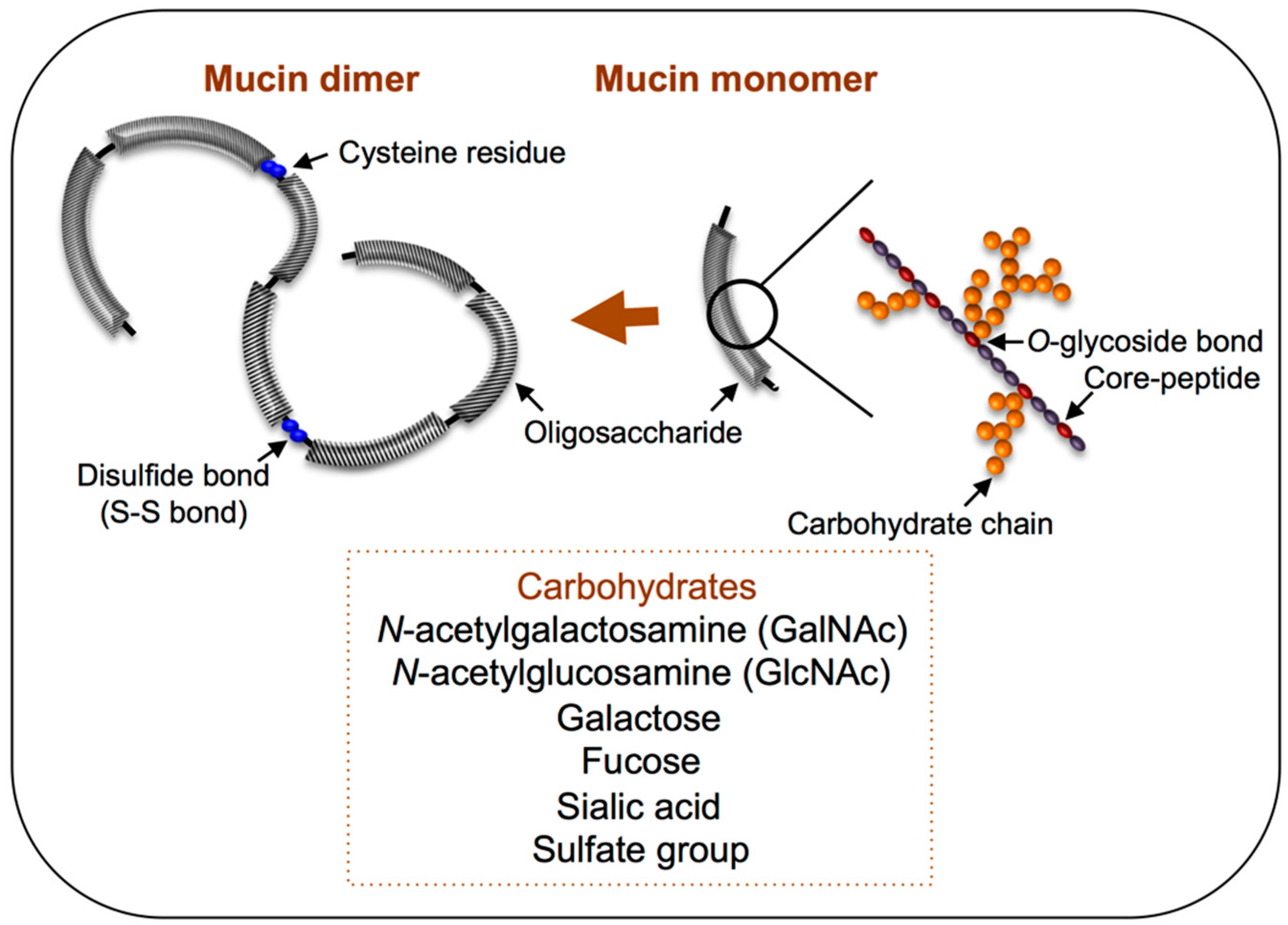
4. Mucin Biosynthesis
5. Interactions of Lactobacillus with Intestinal Glycoconjugates
6. Mucin Adhesion Factors and Adhesion Mechanisms in Lactobacillus
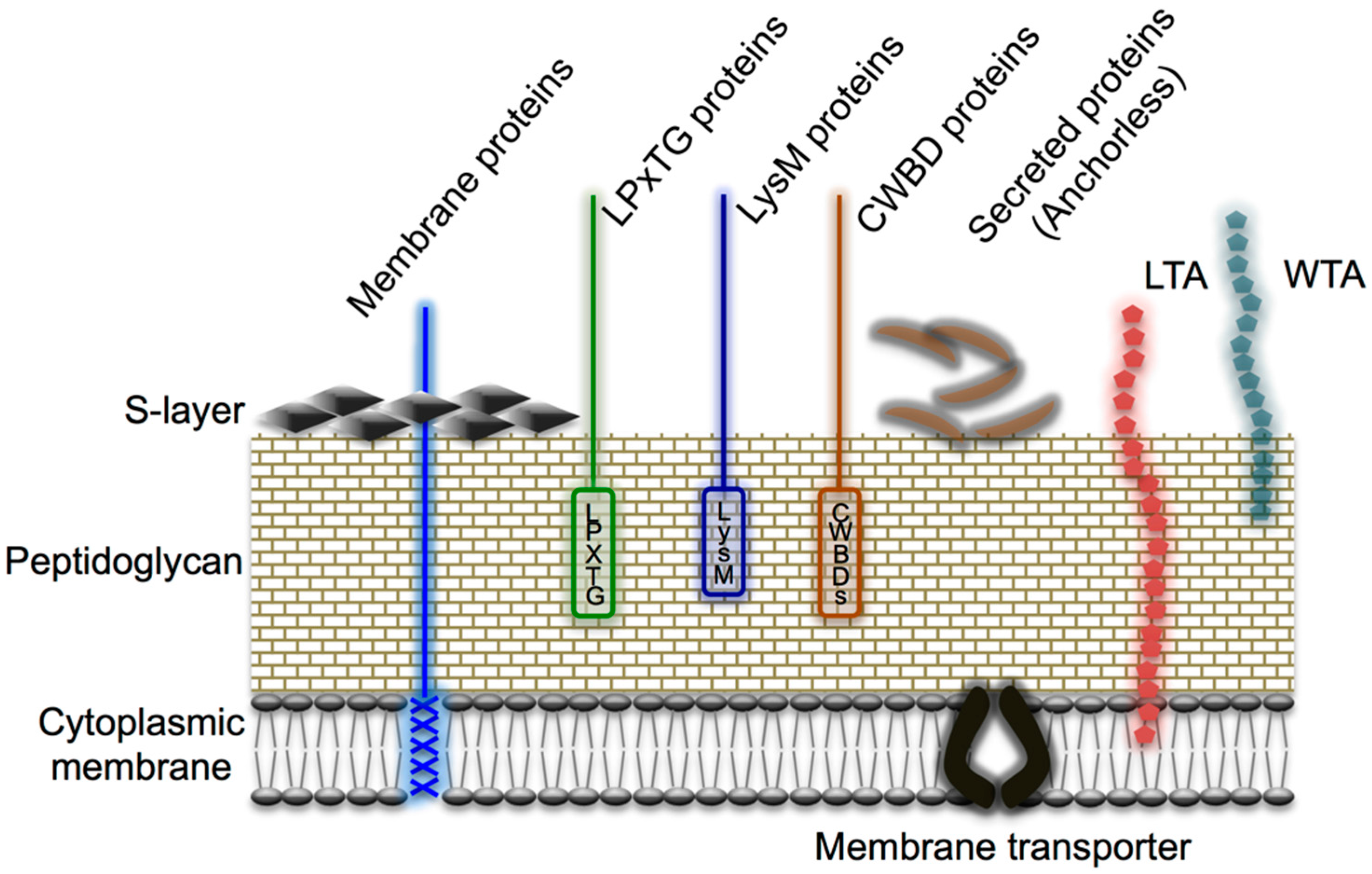
6.1. Cell Wall-Anchored Proteins
6.2. Moonlighting Proteins
7. Conclusions
Author Contributions
Conflicts of Interest
Ethical Statements
References
- Dethlefsen, L.; McFall-Ngai, M.; Relman, D.A. An ecological and evolutionary perspective on human–microbe mutualism and disease. Nature 2007, 449, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Rajilić-Stojanović, M.; de Vos, W.M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 2014, 38, 996–1047. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, A.M.; Shanahan, F.; Abreu, M.; Vora, P.; Faure, E.; Thomas, L.; Arnold, E.; Arditi, M.; Abreu, M.; Fukata, M.; et al. The gut flora as a forgotten organ. EMBO Rep. 2006, 7, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Purchiaroni, F.; Tortora, A.; Gabrielli, M.; Bertucci, F.; Gigante, G.; Ianiro, G.; Ojetti, V.; Scarpellini, E.; Gasbarrini, A. The role of intestinal microbiota and the immune system. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 323–333. [Google Scholar] [PubMed]
- Blum, S.; Schiffrin, E.J. Intestinal microflora and homeostasis of the mucosal immune response: Implications for probiotic bacteria? Curr. Issues Intest. Microbiol. 2003, 4, 53–60. [Google Scholar] [PubMed]
- Smith, M.I.; Yatsunenko, T.; Manary, M.J.; Trehan, I.; Mkakosya, R.; Cheng, J.; Kau, A.L.; Rich, S.S.; Concannon, P.; Mychaleckyj, J.C.; et al. Gut microbiomes of Malawian twin pairs discordant for kwashiorkor. Science 2013, 339, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Donaldson, G.P.; Mikulski, Z.; Boyajian, S.; Ley, K.; Mazmanian, S.K. Bacterial colonization factors control specificity and stability of the gut microbiota. Nature 2013, 501, 426–429. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The international scientific association for probiotics and prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Claesson, M.J.; van Sinderen, D.; O’Toole, P.W. The genus Lactobacillus—A genomic basis for understanding its diversity. FEMS Microbiol. Lett. 2007, 269, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Claesson, M.J.; van Sinderen, D.; O’Toole, P.W. Lactobacillus phylogenomics—Towards a reclassification of the genus. Int. J. Syst. Evol. Microbiol. 2008, 58, 2945–2954. [Google Scholar] [CrossRef] [PubMed]
- Mitsuoka, T. The human gastrointestinal tract. In the Lactic Acid Bacteria in Health and Disease; Springer US: New York, NY, USA, 1992; pp. 69–114. [Google Scholar]
- Reuter, G. The Lactobacillus and Bifidobacterium microflora of the human intestine: Composition and succession. Curr. Issues. Intest. Microbiol. 2001, 2, 43–53. [Google Scholar] [PubMed]
- Hill, G.B.; Eschenbach, D.A.; Holmes, K.K. Bacteriology of the vagina. Scand. J. Urol. Nephrol. Suppl. 1984, 86, 23–39. [Google Scholar] [PubMed]
- Zhou, X.; Brown, C.J.; Abdo, Z.; Davis, C.C.; Hansmann, M.A.; Joyce, P.; Foster, J.A.; Forney, L.J. Differences in the composition of vaginal microbial communities found in healthy Caucasian and black women. ISME J. 2007, 2, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Tannock, G.W. Lactic microbiota of pigs, mice and rats. In the Lactic Acid Bacteria in Health and Disease; Springer US: New York, NY, USA, 1992; pp. 21–48. [Google Scholar]
- Johansson, M.E.V.; Phillipson, M.; Petersson, J.; Velcich, A.; Holm, L.; Hansson, G.C. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl. Acad. Sci. USA 2008, 105, 15064–15069. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.V.; Larsson, J.M.H.; Hansson, G.C. The two mucus layers of colon are organized by the Muc2 mucin, whereas the outer layer is a legislator of host-microbial interactions. Proc. Natl. Acad. Sci. USA 2011, 108, 4659–4665. [Google Scholar] [CrossRef] [PubMed]
- Walter, J. Ecological role of lactobacilli in the gastrointestinal tract: Implications for fundamental and biomedical research. Appl. Environ. Microbiol. 2008, 74, 4985–4996. [Google Scholar] [CrossRef] [PubMed]
- Lebeer, S.; Vanderleyden, J.; de Keersmaecker, S.C.J. Genes and molecules of lactobacilli supporting probiotic action. Microbiol. Mol. Biol. Rev. 2008, 72, 728–764. [Google Scholar] [CrossRef] [PubMed]
- Frese, S.A.; Mackenzie, D.A.; Peterson, D.A.; Schmaltz, R.; Fangman, T.; Zhou, Y.; Zhang, C.; Benson, A.K.; Cody, L.A.; Mulholland, F.; et al. Molecular characterization of host-specific biofilm formation in a vertebrate gut symbiont. PLoS Genet. 2013, 9, e1004057. [Google Scholar] [CrossRef] [PubMed]
- Van Tassell, M.L.; Miller, M.J. Lactobacillus adhesion to mucus. Nutrients 2011, 3, 613–636. [Google Scholar] [CrossRef] [PubMed]
- Juge, N. Microbial adhesins to gastrointestinal mucus. Trends. Microbiol. 2012, 20, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Etzold, S.; Juge, N. Structural insights into bacterial recognition of intestinal mucins. Curr. Opin. Struct. Biol. 2014, 28, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Van Zyl, W.F.; Deane, S.M.; Dicks, L.M.T. Use of the mCherry fluorescent protein to study intestinal colonization by Enterococcus mundtii ST4SA and Lactobacillus plantarum 423 in mice. Appl. Environ. Microbiol. 2015, 81, 5993–6002. [Google Scholar] [CrossRef] [PubMed]
- Ardita, C.S.; Mercante, J.W.; Kwon, Y.M.; Luo, L.; Crawford, M.E.; Powell, D.N.; Jones, R.M.; Neish, A.S. Epithelial adhesion mediated by pilin SpaC is required for Lactobacillus rhamnosus GG-induced cellular responses. Appl. Environ. Microbiol. 2014, 80, 5068–5077. [Google Scholar] [CrossRef] [PubMed]
- O’Connell Motherway, M.; Zomer, A.; Leahy, S.C.; Reunanen, J.; Bottacini, F.; Claesson, M.J.; O’Brien, F.; Flynn, K.; Casey, P.G.; Munoz, J.A.M.; et al. Functional genome analysis of Bifidobacterium breve UCC2003 reveals type IVb tight adherence (Tad) pili as an essential and conserved host-colonization factor. Proc. Natl. Acad. Sci. USA 2011, 108, 11217–11222. [Google Scholar] [CrossRef] [PubMed]
- Sheahan, D.G.; Jervis, H.R. Comparative histochemistry of gastrointestinal mucosubstances. Am. J. Anat. 1976, 146, 103–131. [Google Scholar] [CrossRef] [PubMed]
- Treuting, P.M.; Valasek, M.A.; Dintzis, S.M. Upper gastrointestinal tract. In Comparative Anatomy and Histology—A Mouse and Human Atlas, 1st ed.; Dintzis, S.M., Treuting, P.M., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2012; pp. 155–175. [Google Scholar]
- Treuting, P.M.; Dintzis, S.M. Upper gastrointestinal tract. In Comparative Anatomy and Histology–A Mouse and Human Atlas, 1st ed.; Dintzis, S.M., Treuting, P.M., Eds.; Elsevier Inc.: Amsterdam, the Netherlands, 2012; pp. 177–192. [Google Scholar]
- Caballero, S.; Pamer, E.G. Microbiota-mediated inflammation and antimicrobial defense in the intestine. Annu. Rev. Immunol. 2015, 33, 227–256. [Google Scholar] [CrossRef] [PubMed]
- El Aidy, S.; van den Bogert, B.; Kleerebezem, M. The small intestine microbiota, nutritional modulation and relevance for health. Curr. Opin. Biotechnol. 2015, 32, 14–20. [Google Scholar] [CrossRef] [PubMed]
- McGuckin, M.; Lindén, S.K.; Sutton, P.; Florin, T.H. Mucin dynamics and enteric pathogens. Nat. Rev. Microbiol. 2011, 9, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Podolsky, D.K.; Fournier, D.A.; Lynch, K.E. Human colonic goblet cells. Demonstration of distinct subpopulations defined by mucin-specific monoclonal antibodies. J. Clin. Invest. 1986, 77, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Vertty, M.A.; Mellinkoff, S.M.; Frankland, M.; Greipel, M. Serotonin content and argentaffin and Paneth cell changes in ulcerative colitis. Gastroenterology 1962, 43, 24–31. [Google Scholar]
- Wong, W.M.; Poulsom, R.; Wright, N.A. Trefoil peptides. Gut 1999, 44, 890–895. [Google Scholar] [CrossRef] [PubMed]
- Howat, W.J.; Wilson, B.A. Tissue fixation and the effect of molecular fixatives on downstream staining procedures. Methods 2014, 70, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Dapson, R.W. Macromolecular changes caused by formalin fixation and antigen retrieval. Biotech. Histochem. 2007, 82, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Puchtler, H.; Waldrop, F.S.; Conner, H.M.; Terry, M.S. Carnoy fixation: Practical and theoretical considerations. Histochemie 1968, 16, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Puchtler, H.; Waldrop, F.S.; Melan, S.N.; Terry, M.S.; Conner, H.M. Methacarn (methanol-Carnoy) fixation. Practical and theoretical considerations. Histochemie 1970, 21, 97–116. [Google Scholar] [CrossRef] [PubMed]
- Ota, H.; Katsuyama, T. Alternating laminated array of two types of mucin in the human gastric surface mucous layer. Histochem. J. 1992, 24, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Ermund, A.; Schütte, A.; Johansson, M.E.V.; Gustafsson, J.K.; Hansson, G.C. Studies of mucus in mouse stomach, small intestine, and colon. I. Gastrointestinal mucus layers have different properties depending on location as well as over the Peyer’s patches. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, G341–G347. [Google Scholar] [CrossRef] [PubMed]
- Pelaseyed, T.; Bergström, J.H.; Gustafsson, J.K.; Ermund, A.; Birchenough, G.M.H.; Schütte, A.; van der Post, S.; Svensson, F.; Rodríguez-Piñeiro, A.M.; Nyström, E.E.L.; et al. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol. Rev. 2014, 260, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Lesuffleur, T.; Zweibaum, A.; Real, F.X. Mucins in normal and neoplastic human gastrointestinal tissues. Crit. Rev. Oncol. Hematol. 1994, 17, 153–180. [Google Scholar] [CrossRef]
- Larsson, J.M.; Karlsson, H.; Sjovall, H.; Hansson, G.C. A complex, but uniform O-glycosylation of the human MUC2 mucin from colonic biopsies analyzed by nanoLC/MSn. Glycobiology 2009, 19, 756–766. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, K.; Ueno, S.; Sugiyama, M.; Yamamoto, Y.; Mukai, T. Lactobacillus rhamnosus GG SpaC pilin subunit binds to the carbohydrate moieties of intestinal glycoconjugates. Anim. Sci. J. 2016, 87, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Brockhausen, I. Sulphotransferases acting on mucin-type oligosaccharides. Biochem. Soc. Trans. 2003, 31, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Corfield, A.P.; Myerscough, N.; Longman, R.; Sylvester, P.; Arul, S.; Pignatelli, M. Mucins and mucosal protection in the gastrointestinal tract: New prospects for mucins in the pathology of gastrointestinal disease. Gut 2000, 47, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Croix, J.A.; Carbonero, F.; Nava, G.M.; Russell, M.; Greenberg, E.; Gaskins, H.R. On the relationship between sialomucin and sulfomucin expression and hydrogenotrophic microbes in the human colonic mucosa. PLoS One 2011, 6, e24447. [Google Scholar] [CrossRef] [PubMed]
- Sakata, T.; von Engelhardt, W. Luminal mucin in the large intestine of mice, rats and guinea pigs. Cell Tissue Res. 1981, 219, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Corfield, A.P.; Myerscough, N.; Bradfield, N.; Corfield, C.A.; Gough, M.; Clamp, J.R.; Durdey, P.; Warren, B.F.; Bartolo, D.C.; King, K.R.; et al. Colonic mucins in ulcerative colitis: Evidence for loss of sulfation. Glycoconj. J. 1996, 13, 809–822. [Google Scholar] [CrossRef] [PubMed]
- Agawa, S.; Muto, T.; Morioka, Y. Mucin abnormality of colonic mucosa in ulcerative colitis associated with carcinoma and/or dysplasia. Dis. Colon Rectum 1988, 31, 387–389. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, L.; Zhou, R.; Wang, X.; Song, L.; Huang, S.; Wang, G.; Xia, B. Increased proportions of Bifidobacterium and the Lactobacillus group and loss of butyrate-producing bacteria in inflammatory bowel disease. J. Clin. Microbiol. 2014, 52, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Constable, F.L. Fimbriae and haemagglutinating activity in strains of Bacterium cloacae. J. Pathol. Bacteriol. 1956, 72, 133–136. [Google Scholar] [CrossRef] [PubMed]
- Duguid, J.P.; Old, D.C. Adhesive properties of enterobacteriaceae. In Bacterial Adherence; Springer Netherlands: Dordrecht, the Netherlands, 1980; pp. 185–217. [Google Scholar]
- Huesca, M.; Goodwin, A.; Bhagwansingh, A.; Hoffman, P.; Lingwood, C.A. Characterization of an acidic-pH-inducible stress protein (hsp70), a putative sulfatide binding adhesin, from Helicobacter pylori. Infect. Immun. 1998, 66, 4061–4067. [Google Scholar] [PubMed]
- Aspholm-Hurtig, M.; Dailide, G.; Lahmann, M.; Kalia, A.; Ilver, D.; Roche, N.; Vikström, S.; Sjöström, R.; Lindén, S.; Bäckström, A.; et al. Functional adaptation of BabA, the H. pylori ABO blood group antigen binding adhesin. Science 2004, 305, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Mukai, T.; Arihara, K.; Itoh, H. Lectin-like activity of Lactobacillus acidophilus strain JCM 1026. FEMS Microbiol. Lett. 1992, 98, 71–74. [Google Scholar] [CrossRef]
- Coconnier, M.H.; Klaenhammer, T.R.; Kernéis, S.; Bernet, M.F.; Servin, A.L. Protein-mediated adhesion of Lactobacillus acidophilus BG2FO4 on human enterocyte and mucus-secreting cell lines in culture. Appl. Environ. Microbiol. 1992, 58, 2034–2039. [Google Scholar] [PubMed]
- Yamamoto, K.; Miwa, T.; Taniguchi, H.; Nagano, T.; Shimamura, K.; Tanaka, T.; Kumagai, H. Binding specificity of Lactobacillus to glycolipids. Biochem. Biophys. Res. Commun. 1996, 228, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Mukai, T.; Kaneko, S.; Ohori, H. Haemagglutination and glycolipid-binding activities of Lactobacillus reuteri. Lett. Appl. Microbiol. 1998, 27, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Mukai, T.; Arihara, K. Presence of intestinal lectin-binding glycoproteins on the cell surface of Lactobacillus acidophilus. Biosci. Biotechnol. Biochem. 1994, 58, 1851–1854. [Google Scholar] [CrossRef]
- Uchida, H.; Kinoshita, H.; Kawai, Y.; Kitazawa, H.; Miura, K.; Shiiba, K.; Horii, A.; Kimura, K.; Taketomo, N.; Oda, M.; et al. Lactobacilli binding human A-antigen expressed in intestinal mucosa. Res. Microbiol. 2006, 157, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Uchida, H.; Kawai, Y.; Kinoshita, H.; Kitazawa, H.; Miura, K.; Shiiba, K.; Horii, A.; Kimura, K.; Taketomo, N.; Oda, M.; et al. Lactic acid bacteria (LAB) bind to human B- or H-antigens expressed on intestinal mucosa. Biosci. Biotechnol. Biochem. 2006, 70, 3073–3076. [Google Scholar] [CrossRef] [PubMed]
- Huang, I.N.; Okawara, T.; Watanabe, M.; Kawai, Y.; Kitazawa, H.; Ohnuma, S.; Shibata, C.; Horii, A.; Kimura, K.; Taketomo, N.; et al. New screening methods for probiotics with adhesion properties to sialic acid and sulphate residues in human colonic mucin using the Biacore assay. J. Appl. Microbiol. 2013, 114, 854–860. [Google Scholar] [CrossRef] [PubMed]
- Desvaux, M.; Dumas, E.; Chafsey, I.; Hébraud, M. Protein cell surface display in Gram-positive bacteria: From single protein to macromolecular protein structure. FEMS Microbiol. Lett. 2006, 256, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Roos, S.; Jonsson, H. A high-molecular-mass cell-surface protein from Lactobacillus reuteri 1063 adheres to mucus components. Microbiology 2002, 148, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Buck, B.L.; Altermann, E.; Svingerud, T.; Klaenhammer, T.R. Functional analysis of putative adhesion factors in Lactobacillus acidophilus NCFM. Appl. Environ. Microbiol. 2005, 71, 8344–8351. [Google Scholar] [CrossRef] [PubMed]
- Etzold, S.; Mackenzie, D.A.; Jeffers, F.; Walshaw, J.; Roos, S.; Hemmings, A.M.; Juge, N. Structural and molecular insights into novel surface-exposed mucus adhesins from Lactobacillus reuteri human strains. Mol. Microbiol. 2014, 92, 543–556. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, D.A.; Jeffers, F.; Parker, M.L.; Vibert-Vallet, A.; Bongaerts, R.J.; Roos, S.; Walter, J.; Juge, N. Strain-specific diversity of mucus-binding proteins in the adhesion and aggregation properties of Lactobacillus reuteri. Microbiology 2010, 156, 3368–3378. [Google Scholar] [CrossRef] [PubMed]
- Bierne, H.; Cossart, P. Listeria monocytogenes surface proteins: From genome predictions to function. Microbiol. Mol. Biol. Rev. 2007, 71, 377–397. [Google Scholar] [CrossRef] [PubMed]
- Boekhorst, J.; Helmer, Q.; Kleerebezem, M.; Siezen, R.J. Comparative analysis of proteins with a mucus-binding domain found exclusively in lactic acid bacteria. Microbiology 2006, 152, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Etzold, S.; Kober, O.I.; Mackenzie, D.A.; Tailford, L.E.; Gunning, A.P.; Walshaw, J.; Hemmings, A.M.; Juge, N. Structural basis for adaptation of lactobacilli to gastrointestinal mucus. Environ. Microbiol. 2014, 16, 888–903. [Google Scholar] [CrossRef] [PubMed]
- Coïc, Y.M.; Baleux, F.; Poyraz, Ö.; Thibeaux, R.; Labruyere, E.; Chretien, F.; Sobhani, I.; Lazure, T.; Wyplosz, B.; Schneider, G.; et al. Design of a specific colonic mucus marker using a human commensal bacterium cell surface domain. J. Biol. Chem. 2012, 287, 15916–15922. [Google Scholar] [CrossRef] [PubMed]
- Kankainen, M.; Paulin, L.; Tynkkynen, S.; von Ossowski, I.; Reunanen, J.; Partanen, P.; Satokari, R.; Vesterlund, S.; Hendrickx, A.P.A.; Lebeer, S.; et al. Comparative genomic analysis of Lactobacillus rhamnosus GG reveals pili containing a human-mucus binding protein. Proc. Natl. Acad. Sci. USA 2009, 106, 17193–17198. [Google Scholar] [CrossRef] [PubMed]
- Reunanen, J.; von Ossowski, I.; Hendrickx, A.P.A.; Palva, A.; de Vos, W.M. Characterization of the SpaCBA pilus fibers in the probiotic Lactobacillus rhamnosus GG. Appl. Environ. Microbiol. 2012, 78, 2337–2344. [Google Scholar] [CrossRef] [PubMed]
- Pretzer, G.; Snel, J.; Molenaar, D.; Wiersma, A.; Bron, P.A.; Lambert, J.; de Vos, W.M.; van der Meer, R.; Smits, M.A.; Kleerebezem, M. Biodiversity-based identification and functional characterization of the mannose-specific adhesin of Lactobacillus plantarum. J. Bacteriol. 2005, 187, 6128–6136. [Google Scholar] [CrossRef] [PubMed]
- Von Ossowski, I.; Satokari, R.; Reunanen, J.; Lebeer, S.; de Keersmaecker, S.C.J.; Vanderleyden, J.; de Vos, W.M.; Palva, A. Functional characterization of a mucus-specific LPXTG surface adhesin from probiotic Lactobacillus rhamnosus GG. Appl. Environ. Microbiol. 2011, 77, 4465–4472. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, K.; Nakamata, K.; Ueno, S.; Terao, A.; Aryantini, N.P.D.; Sujaya, I.N.; Fukuda, K.; Urashima, T.; Yamamoto, Y.; Mukai, T. Adhesion properties of Lactobacillus rhamnosus mucus-binding factor to mucin and extracellular matrix proteins. Biosci. Biotechnol. Biochem. 2015, 79, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Jensen, H.; Roos, S.; Jonsson, H.; Rud, I.; Grimmer, S.; van Pijkeren, J.P.; Britton, R.A.; Axelsson, L. Role of Lactobacillus reuteri cell and mucus-binding protein A (CmbA) in adhesion to intestinal epithelial cells and mucus in vitro. Microbiology 2014, 160, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Pridmore, R.D.; Berger, B.; Desiere, F.; Vilanova, D.; Barretto, C.; Pittet, A.C.; Zwahlen, M.C.; Rouvet, M.; Altermann, E.; Barrangou, R. The genome sequence of the probiotic intestinal bacterium Lactobacillus johnsonii NCC 533. Proc. Natl. Acad. Sci. USA 2004, 101, 2512–2517. [Google Scholar] [CrossRef] [PubMed]
- Von Ossowski, I.; Reunanen, J.; Satokari, R.; Vesterlund, S.; Kankainen, M.; Huhtinen, H.; Tynkkynen, S.; Salminen, S.; de Vos, W.M.; Palva, A. Mucosal adhesion properties of the probiotic Lactobacillus rhamnosus GG SpaCBA and SpaFED pilin subunits. Appl. Environ. Microbiol. 2010, 76, 2049–2057. [Google Scholar] [CrossRef] [PubMed]
- Douillard, F.P.; Ribbera, A.; Kant, R.; Pietilä, T.E.; Järvinen, H.M.; Messing, M.; Randazzo, C.L.; Paulin, L.; Laine, P.; Ritari, J.; et al. Comparative genomic and functional analysis of 100 Lactobacillus rhamnosus strains and their comparison with strain GG. PLoS Genet. 2013, 9, e1003683. [Google Scholar] [CrossRef] [PubMed]
- Rintahaka, J.; Yu, X.; Kant, R.; Palva, A.; von Ossowski, I. Phenotypical analysis of the Lactobacillus rhamnosus GG fimbrial spaFED operon: Surface expression and functional characterization of recombinant SpaFED pili in Lactococcus lactis. PLoS One 2014, 9, e113922. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, K.; Ochiai, A.; Tsubokawa, D.; Ishihara, K.; Yamamoto, Y.; Mukai, T. Identification and characterization of sulfated carbohydrate-binding protein from Lactobacillus reuteri. PLoS One 2013, 8, e83703. [Google Scholar] [CrossRef] [PubMed]
- Turroni, F.; Serafini, F.; Foroni, E.; Duranti, S.; O’Connell Motherway, M.; Taverniti, V.; Mangifesta, M.; Milani, C.; Viappiani, A.; Roversi, T.; et al. Role of sortase-dependent pili of Bifidobacterium bifidum PRL2010 in modulating bacterium-host interactions. Proc. Natl. Acad. Sci. USA 2013, 110, 11151–11156. [Google Scholar] [CrossRef] [PubMed]
- Schell, M.A.; Karmirantzou, M.; Snel, B.; Vilanova, D.; Berger, B.; Pessi, G.; Zwahlen, M.C.; Desiere, F.; Bork, P.; Delley, M.; et al. The genome sequence of Bifidobacterium longum reflects its adaptation to the human gastrointestinal tract. Proc. Natl. Acad. Sci. USA 2002, 99, 14422–14427. [Google Scholar] [CrossRef] [PubMed]
- Iguchi, A.; Umekawa, N.; Maegawa, T.; Tsuruta, H.; Odamaki, T.; Xiao, J.Z.; Osawa, R. Polymorphism and distribution of putative cell-surface adhesin-encoding ORFs among human fecal isolates of Bifidobacterium longum subsp. longum. Antonie Van Leeuwenhoek 2011, 99, 457–471. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Nishiyama, K.; Miyajima, H.; Osawa, R.; Yamamoto, Y.; Mukai, T. Adhesion properties of a putative polymorphic fimbrial subunit protein from Bifidobacterium longum subsp. longum. Biosci. Microbiota. Food Heal. 2016, 35, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, C.J. Moonlighting proteins—An update. Mol. Biosyst. 2009, 5, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Mukai, T.; Asasaka, T.; Sato, E.; Mori, K.; Matsumoto, M.; Ohori, H. Inhibition of binding of Helicobacter pylori to the glycolipid receptors by probiotic Lactobacillus reuteri. FEMS Immunol. Med. Microbiol. 2002, 32, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Granato, D.; Bergonzelli, G.E.; Pridmore, R.D.; Marvin, L.; Rouvet, M.; Corthésy-Theulaz, I.E. Cell surface-associated elongation factor Tu mediates the attachment of Lactobacillus johnsonii NCC533 (La1) to human intestinal cells and mucins. Infect. Immun. 2004, 72, 2160–2169. [Google Scholar] [CrossRef] [PubMed]
- Dhanani, A.S.; Bagchi, T. The expression of adhesin EF-Tu in response to mucin and its role in Lactobacillus adhesion and competitive inhibition of enteropathogens to mucin. J. Appl. Microbiol. 2013, 115, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, H.; Uchida, H.; Kawai, Y.; Kawasaki, T.; Wakahara, N.; Matsuo, H.; Watanabe, M.; Kitazawa, H.; Ohnuma, S.; Miura, K.; et al. Cell surface Lactobacillus plantarum LA 318 glyceraldehyde-3-phosphate dehydrogenase (GAPDH) adheres to human colonic mucin. J. Appl. Microbiol. 2008, 104, 1667–1674. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, H.; Wakahara, N.; Watanabe, M.; Kawasaki, T.; Matsuo, H.; Kawai, Y.; Kitazawa, H.; Ohnuma, S.; Miura, K.; Horii, A.; et al. Cell surface glyceraldehyde-3-phosphate dehydrogenase (GAPDH) of Lactobacillus plantarum LA 318 recognizes human A and B blood group antigens. Res. Microbiol. 2008, 159, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.K.; Shah, K.R.; Pappachan, A.; Gupta, S.; Singh, D.D. Cloning, expression and characterization of a mucin-binding GAPDH from Lactobacillus acidophilus. Int. J. Biol. Macromol. 2016, 91, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Bergonzelli, G.E.; Granato, D.; Pridmore, R.D.; Marvin-Guy, L.F.; Donnicola, D.; Corthésy-Theulaz, I.E. GroEL of Lactobacillus johnsonii La1 (NCC 533) is cell surface associated: potential role in interactions with the host and the gastric pathogen Helicobacter pylori. Infect. Immun. 2006, 74, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.; Ascencio, F.; Conway, P.L. Purification and characterization of a surface protein from Lactobacillus fermentum 104R that binds to porcine small intestinal mucus and gastric mucin. Appl. Environ. Microbiol. 2002, 68, 2330–2336. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, Y.; Okada, S.; Uchimura, T.; Satoh, E. A mucus adhesion promoting protein, MapA, mediates the adhesion of Lactobacillus reuteri to Caco-2 human intestinal epithelial cells. Biosci. Biotechnol. Biochem. 2006, 70, 1622–1628. [Google Scholar] [CrossRef] [PubMed]
- Macías-Rodríguez, M.E.; Zagorec, M.; Ascencio, F.; Vázquez-Juárez, R.; Rojas, M. Lactobacillus fermentum BCS87 expresses mucus- and mucin-binding proteins on the cell surface. J. Appl. Microbiol. 2009, 107, 1866–1874. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kinoshita, H.; Nitta, M.; Yukishita, R.; Kawai, Y.; Kimura, K.; Taketomo, N.; Yamazaki, Y.; Tateno, Y.; Miura, K.; et al. Identification of a new adhesin-like protein from Lactobacillus mucosae ME-340 with specific affinity to the human blood group A and B antigens. J. Appl. Microbiol. 2010, 109, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kinoshita, H.; Huang, I.N.; Eguchi, K.; Tsurumi, T.; Kawai, Y.; Kitazawa, H.; Kimura, K.; Taketomo, N.; Kikuchi, D.; et al. An adhesin-like protein, Lam29, from Lactobacillus mucosae ME-340 binds to histone H3 and blood group antigens in human colonic mucus. Biosci. Biotechnol. Biochem. 2012, 76, 1655–1660. [Google Scholar] [CrossRef] [PubMed]
- Antikainen, J.; Kuparinen, V.; Kupannen, V.; Lähteenmäki, K.; Korhonen, T.K. pH-dependent association of enolase and glyceraldehyde-3-phosphate dehydrogenase of Lactobacillus crispatus with the cell wall and lipoteichoic acids. J. Bacteriol. 2007, 189, 4539–4543. [Google Scholar] [CrossRef] [PubMed]
- Matsui, H.; Takahashi, T.; Øverby, A.; Murayama, S.Y.; Yoshida, H.; Yamamoto, Y.; Nishiyama, K.; Seto, Y.; Takahashi, T.; Mukai, T.; et al. Mouse models for assessing the protective efficacy of Lactobacillus gasseri SBT2055 against Helicobacter suis infection associated with the development of gastric mucosa-associated lymphoid tissue lymphoma. Helicobacter 2015, 20, 291–298. [Google Scholar] [CrossRef] [PubMed]
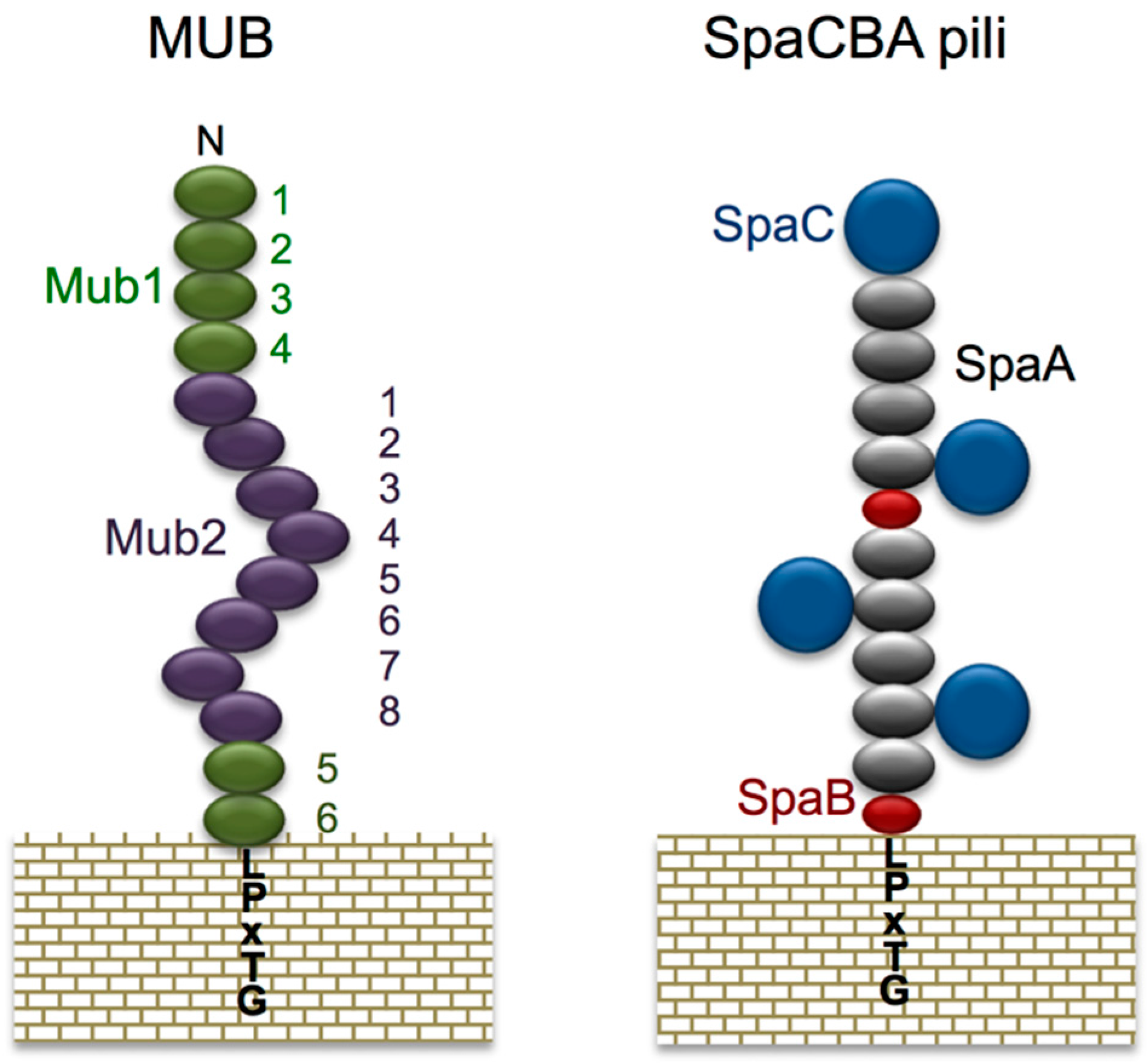
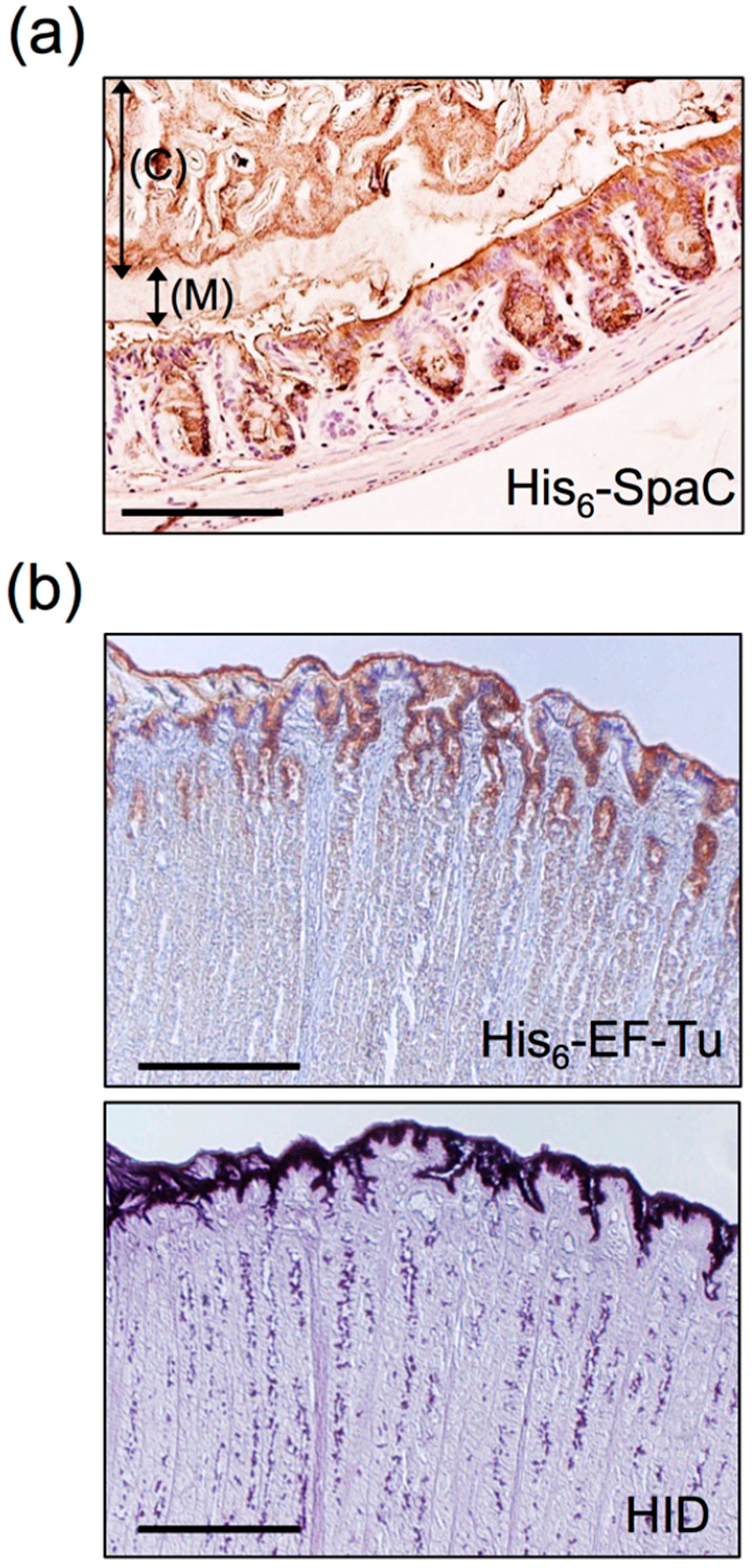
| Adhesion Factor | Species | Anchoring Type | Reference |
|---|---|---|---|
| CmbA/Lar_0958 | L. reuteri ATCC PTA 6475, JCM1112 | LPXTG | [70,81] |
| EF-Tu | L. johnsonii NCC533 | Anchorless | [86,92,93,94] |
| L. reuteri JCM1081 | |||
| L. plantarum strains | |||
| GAPDH | L. acidophilus La14 | Anchorless | [95,96,97] |
| L. plantarum La318 | |||
| GroEL | L. johnsonii NCC533 | Anchorless | [98] |
| Lam29 | L. mucosae ME-340 | Membrane bound | [102,103] |
| MapA | L. reuteri 104R | Membrane bound | [99,100] |
| MBF | L. rhamnosus GG, FSMM22 | LPXTG | [79,80] |
| Msa | L. plantarum WCFS-1 | LPXTG | [78] |
| Mub (Mub family) | L. acidophilus NCFM | LPXTG | [68,69,70,71,73,74] |
| L. reuteri strains | |||
| Pili | L. rhamnosus GG | LPXTG (Fimbriae type) | [47,76,77,83,85] |
| 32-Mmubp | L. fermentum BCS87 | Membrane bound | [101] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nishiyama, K.; Sugiyama, M.; Mukai, T. Adhesion Properties of Lactic Acid Bacteria on Intestinal Mucin. Microorganisms 2016, 4, 34. https://doi.org/10.3390/microorganisms4030034
Nishiyama K, Sugiyama M, Mukai T. Adhesion Properties of Lactic Acid Bacteria on Intestinal Mucin. Microorganisms. 2016; 4(3):34. https://doi.org/10.3390/microorganisms4030034
Chicago/Turabian StyleNishiyama, Keita, Makoto Sugiyama, and Takao Mukai. 2016. "Adhesion Properties of Lactic Acid Bacteria on Intestinal Mucin" Microorganisms 4, no. 3: 34. https://doi.org/10.3390/microorganisms4030034
APA StyleNishiyama, K., Sugiyama, M., & Mukai, T. (2016). Adhesion Properties of Lactic Acid Bacteria on Intestinal Mucin. Microorganisms, 4(3), 34. https://doi.org/10.3390/microorganisms4030034





