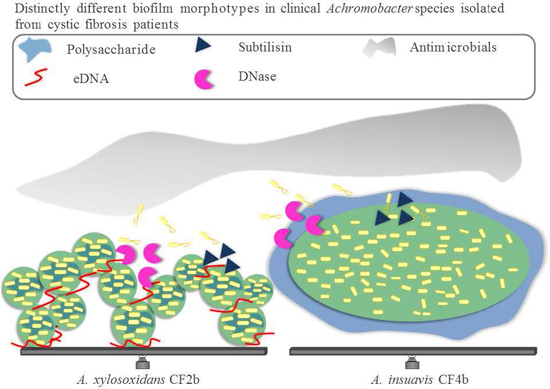
Achromobacter Species Isolated from Cystic Fibrosis Patients Reveal Distinctly Different Biofilm Morphotypes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Fast Screening of Biofilm Formation
2.3. Characterisation of Biofilms Grown under Continuous Flow
2.4. Characterisation of Statically Grown Biofilms
2.5. Detection of Achromobacter Aggregates in Sputum Samples from Cystic Fibrosis Patients Using Peptide Nucleic Acid (PNA) Fluorescent in Situ Hybridization (FISH)
2.6. Probe Validation on Pure Cultures
2.7. Detection and Distribution of Achromobacter in Sputum Samples Obtained from CF Patients
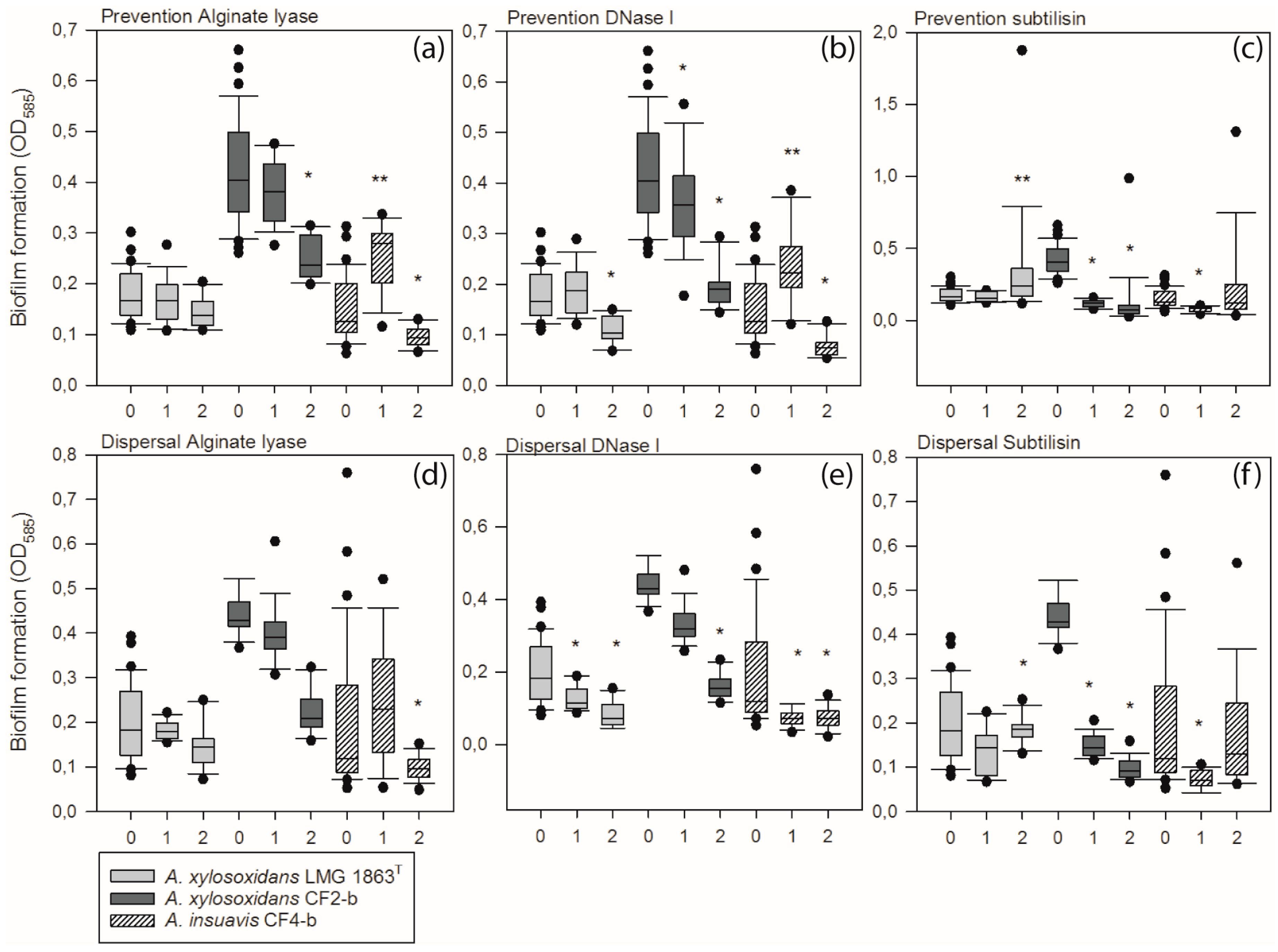
2.8. Enzyme Treatment to Prevent Biofilm Formation
2.9. Enzyme Treatment of Mature Biofilms
2.10. Antimicrobial Susceptibility Testing
2.11. Statistical Analysis
3. Results
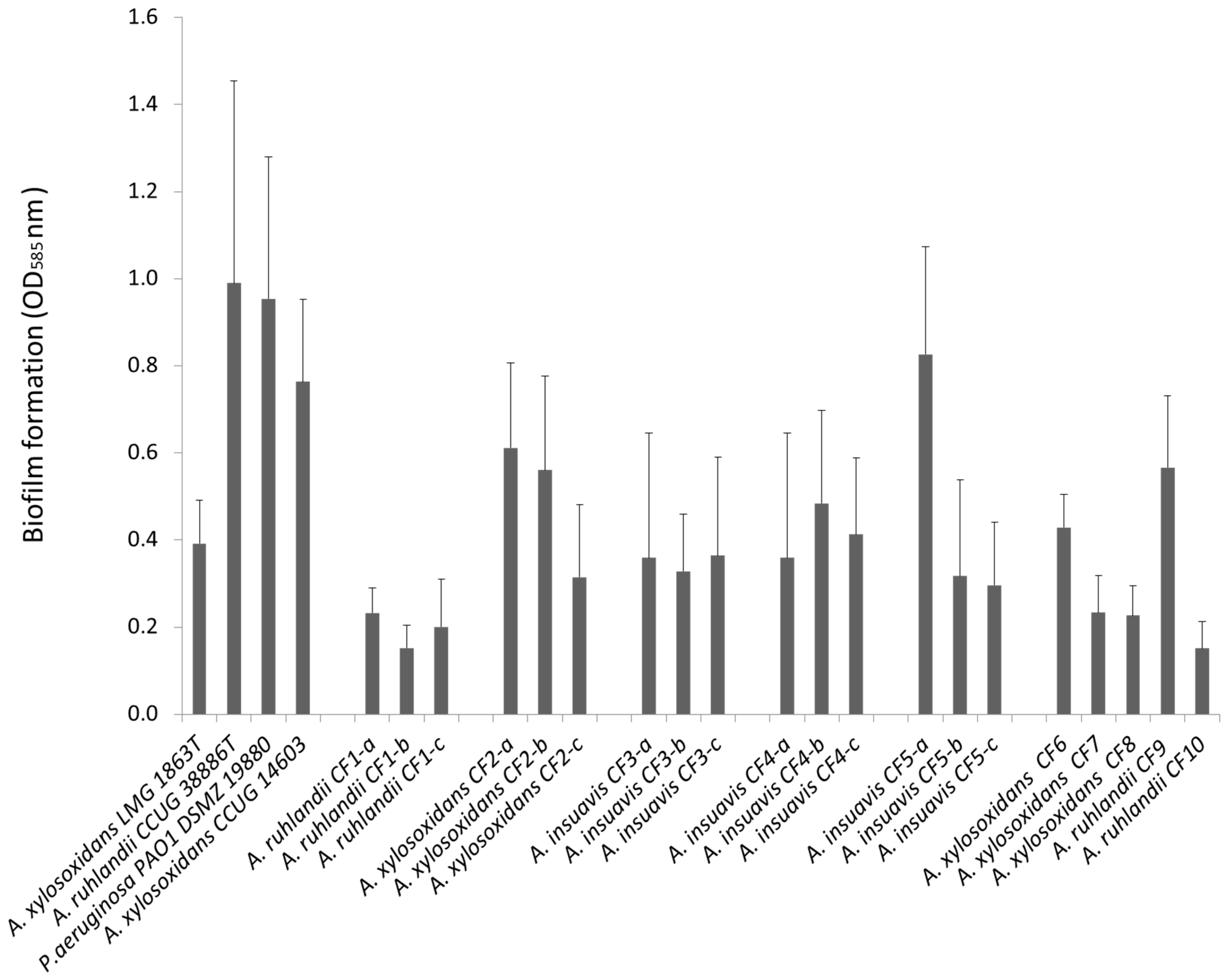
3.1. Clinical Achromobacter Isolates form Biofilm in Vitro
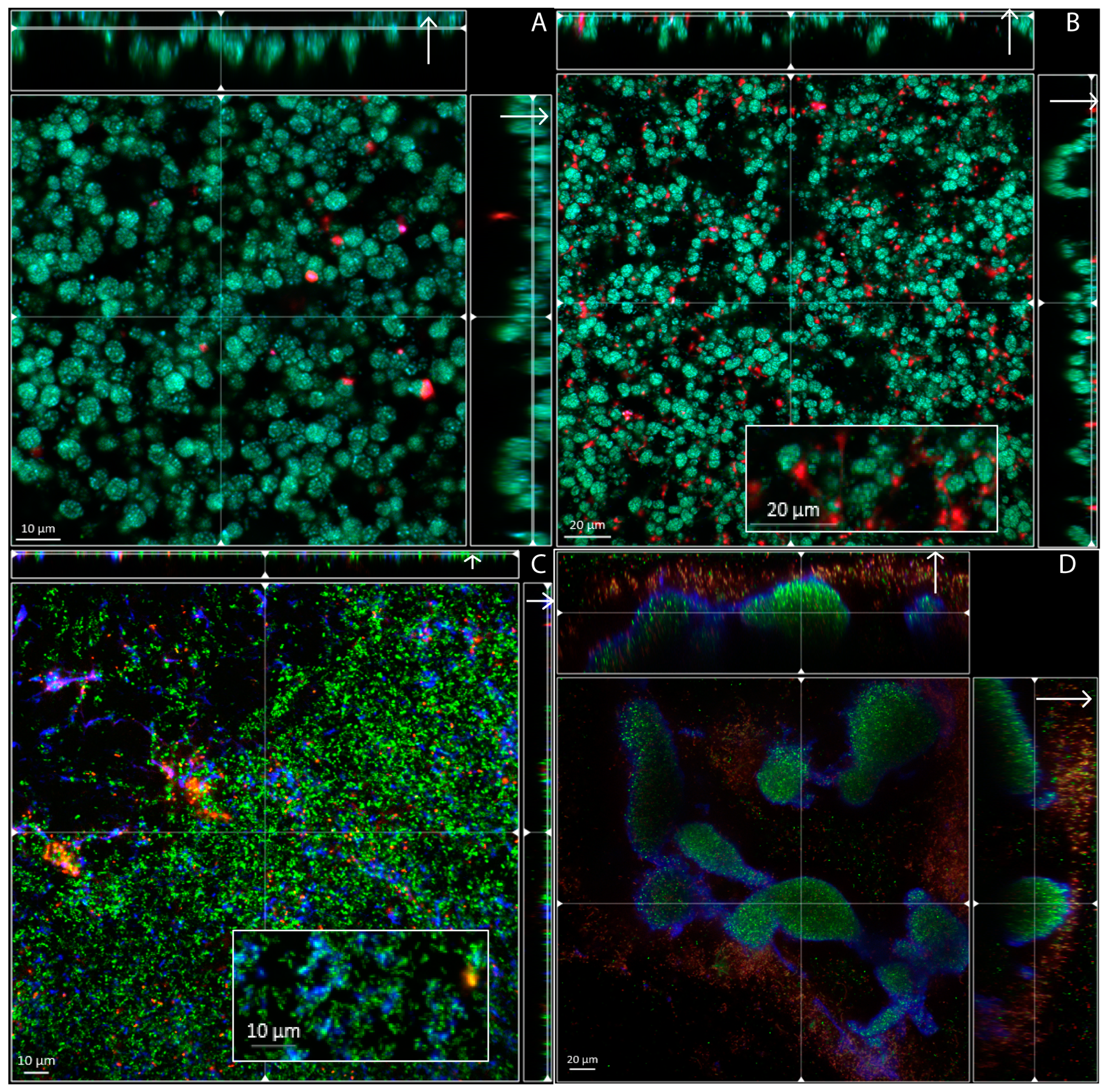
3.2. Biofilm Structure, Morphology and Extracellular Matrix Composition
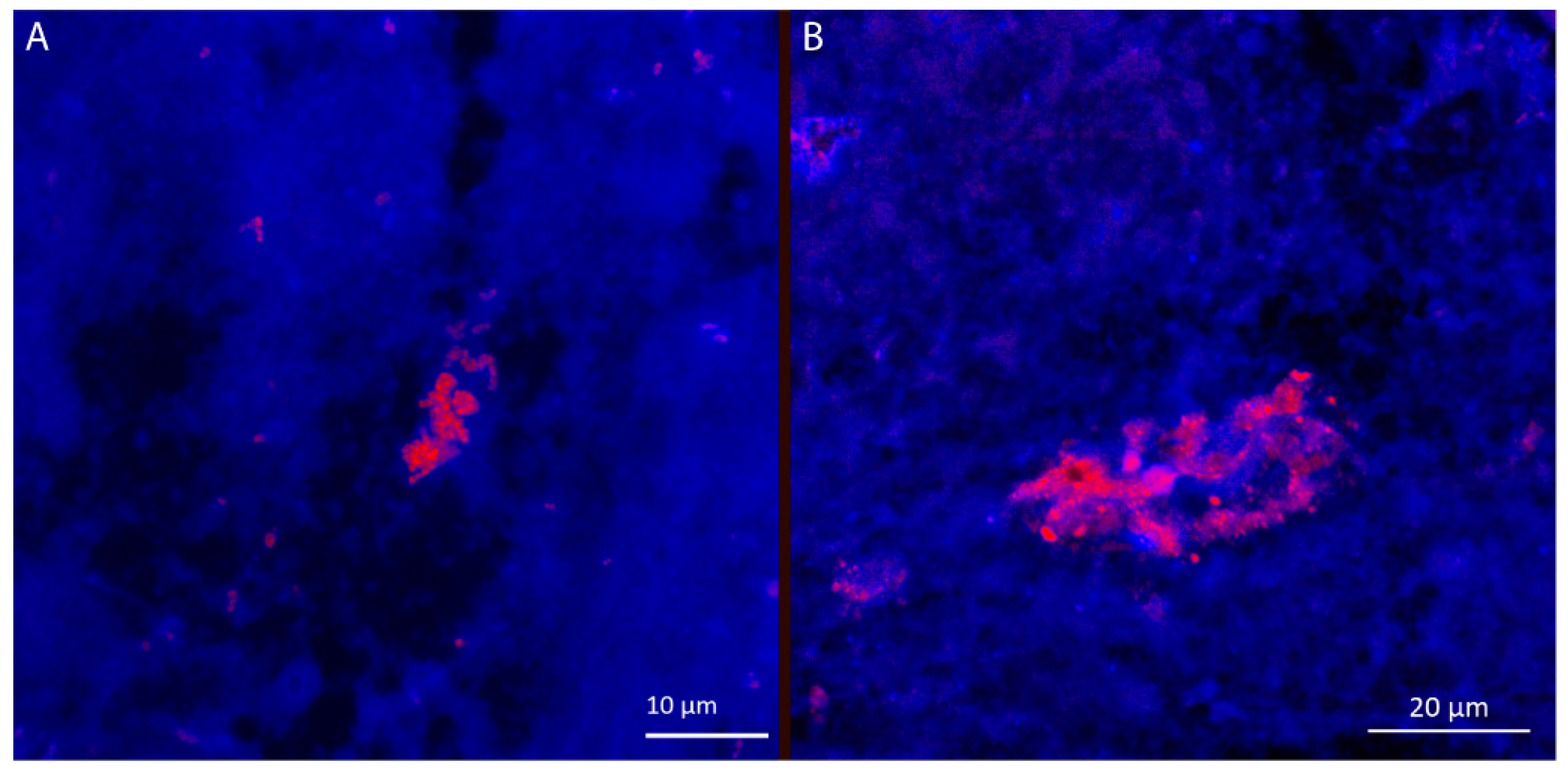
3.3. Visualization of Achromobacter in Sputum Samples from Cystic Fibrosis Patients, Exhibit Resemblance with Biofilm Aggregates
3.4. Extracellular Matrix Components Are Important for Formation and Stability of Achromobacter Biofilm
3.5. Increased Antimicrobial Tolerance of Bacteria Embedded in Biofilm
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Koch, C.; Hoiby, N. Diagnosis and treatment of cystic fibrosis. Respiration 2000, 67, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Boucher, R.C. Evidence for airway surface dehydration as the initiating event in CF airway disease. J. Intern. Med. 2007, 261, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Callaghan, M.; McClean, S. Bacterial host interactions in cystic fibrosis. Curr. Opin. Microbiol. 2012, 15, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Rolston, K.V.; Messer, M. The in vitro susceptibility of Alcaligenes denitrificans subsp. xylosoxidans to 40 antimicrobial agents. J. Antimicrob. Chemother. 1990, 26, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Raso, T.; Bianco, O.; Grosso, B.; Zucca, M.; Savoia, D. Achromobacter xylosoxidans respiratory tract infections in cystic fibrosis patients. APMIS 2008, 116, 837–841. [Google Scholar] [CrossRef] [PubMed]
- Almuzara, M.; Limansky, A.; Ballerini, V.; Galanternik, L.; Famiglietti, A.; Vay, C. In vitro susceptibility of Achromobacter spp. isolates: Comparison of disk diffusion, Etest and agar dilution methods. Int. J. Antimicrob. Agents 2010, 35, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Amoureux, L.; Bador, J.; Siebor, E.; Taillefumier, N.; Fanton, A.; Neuwirth, C. Epidemiology and resistance of Achromobacter xylosoxidans from cystic fibrosis patients in Dijon, Burgundy: First French data. J. Cyst. Fibros. 2013, 12, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.C.; Rubin, B.K. Emerging and unusual gram-negative infections in cystic fibrosis. Semin. Respir. Crit. Care Med. 2007, 28, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Coward, A.; Kenna, D.T.; Perry, C.; Martin, K.; Doumith, M.; Turton, J.F. Use of nrdA gene sequence clustering to estimate the prevalence of different Achromobacter species among Cystic Fibrosis patients in the UK. J. Cyst. Fibros. 2016, 15, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Spilker, T.; Vandamme, P.; Lipuma, J.J. Identification and distribution of Achromobacter species in cystic fibrosis. J. Cyst. Fibros. 2013, 12, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, T.H.; Hansen, M.A.; Jensen, P.O.; Hansen, L.; Riber, L.; Cockburn, A.; Kolpen, M.; Hansen, C.R.; Ridderberg, W.; Eickhardt, S.; et al. Complete genome sequence of the cystic fibrosis pathogen Achromobacter xylosoxidans NH44784-1996 complies with important pathogenic phenotypes. PLoS ONE 2013, 8, e68484. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.R.; Pressler, T.; Nielsen, K.G.; Jensen, P.O.; Bjarnsholt, T.; Hoiby, N. Inflammation in Achromobacter xylosoxidans infected cystic fibrosis patients. J. Cyst. Fibros. 2010, 9, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- Alhede, M.; Kragh, K.N.; Qvortrup, K.; Allesen-Holm, M.; van Gennip, M.; Christensen, L.D.; Jensen, P.Ø.; Nielsen, A.K.; Parsek, M.; Wozniak, D.; et al. Phenotypes of non-attached Pseudomonas aeruginosa aggregates resemble surface attached biofilm. PLoS ONE 2011, 6, e27943. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C.; Moritz, R.; Kirschner, C.; Borchard, W.; Maibaum, R.; Wingender, J.; Flemming, H.C. The role of intermolecular interactions: Studies on model systems for bacterial biofilms. Int. J. Biol. Macromol. 1999, 26, 3–16. [Google Scholar] [CrossRef]
- Hentzer, M.; Teitzel, G.M.; Balzer, G.J.; Heydorn, A.; Molin, S.; Givskov, M.; Parsek, M.R. Alginate overproduction affects Pseudomonas aeruginosa biofilm structure and function. J. Bacteriol. 2001, 183, 5395–5401. [Google Scholar] [CrossRef] [PubMed]
- Parsek, M.R.; Tolker-Nielsen, T. Pattern formation in Pseudomonas aeruginosa biofilms. Curr. Opin. Microbiol. 2008, 11, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.G.; O’Toole, G.A. Innate and induced resistance mechanisms of bacterial biofilms. Curr. Top. Microbiol. Immunol. 2008, 322, 85–105. [Google Scholar] [PubMed]
- Tom, S.K.; Yau, Y.C.; Beaudoin, T.; LiPuma, J.J.; Waters, V. Effect of high-dose antimicrobials on biofilm growth of Achromobacter species from cystic fibrosis patients. Antimicrob. Agents Chemother. 2015, 60, 650–652. [Google Scholar] [CrossRef] [PubMed]
- Ridderberg, W.; Wang, M.; Norskov-Lauritsen, N. Multilocus Sequence Analysis of Isolates of Achromobacter from Patients with Cystic Fibrosis Reveals Infecting Species Other than Achromobacter xylosoxidans. J. Clin. Microbiol. 2012, 50, 2688–2694. [Google Scholar] [CrossRef] [PubMed]
- Ridderberg, W.; Nielsen, S.M.; Norskov-Lauritsen, N. Genetic Adaptation of Achromobacter sp. during Persistence in the Lungs of Cystic Fibrosis Patients. PLoS ONE 2015, 10, e0136790. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.R.; Pressler, T.; Hoiby, N.; Gormsen, M. Chronic infection with Achromobacter xylosoxidans in cystic fibrosis patients; a retrospective case control study. J. Cyst. Fibros. 2006, 5, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Ridderberg, W.; Bendstrup, K.E.; Olesen, H.V.; Jensen-Fangel, S.; Norskov-Lauritsen, N. Marked increase in incidence of Achromobacter xylosoxidans infections caused by sporadic acquisition from the environment. J. Cyst. Fibros. 2011, 10, 466–469. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, G.A. Microtiter dish biofilm formation assay. J. Vis. Exp. 2011. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.J.; Stremick, C.A.; Turner, R.J.; Allan, N.D.; Olson, M.E.; Ceri, H. Microtiter susceptibility testing of microbes growing on peg lids: A miniaturized biofilm model for high-throughput screening. Nat. Protoc. 2010, 5, 1236–1254. [Google Scholar] [CrossRef] [PubMed]
- Tolker-Nielsen, T.; Sternberg, C. Growing and analyzing biofilms in flow chambers. Curr. Protoc. Microbiol. 2011. [Google Scholar] [CrossRef]
- Allesen-Holm, M.; Barken, K.B.; Yang, L.; Klausen, M.; Webb, J.S.; Kjelleberg, S.; Molin, S.; Givskov, M.; Tolker-Nielsen, T. A characterization of DNA release in Pseudomonas aeruginosa cultures and biofilms. Mol. Microbiol. 2006, 59, 1114–1128. [Google Scholar] [CrossRef] [PubMed]
- Wellinghausen, N.; Wirths, B.; Poppert, S. Fluorescence in situ hybridization for rapid identification of Achromobacter xylosoxidans and Alcaligenes faecalis recovered from cystic fibrosis patients. J. Clin. Microbiol. 2006, 44, 3415–3417. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.; Almeida, C.; Salgueiro, D.; Henriques, A.; Vaneechoutte, M.; Haesebrouck, F.; Vieira, M.J.; Rodrigues, L.; Azevedo, N.F.; Cerca, N. Fluorescence in situ Hybridization method using Peptide Nucleic Acid probes for rapid detection of Lactobacillus and Gardnerella spp. BMC Microbiol. 2013, 13, 82. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Fazli, M.; Bjarnsholt, T.; Hoiby, N.; Givskov, M.; Tolker-Nielsen, T. PNA-based fluorescence in situ hybridization for identification of bacteria in clinical samples. Methods Mol. Biol. 2014, 1211, 261–271. [Google Scholar] [PubMed]
- Yang, L.; Haagensen, J.A.; Jelsbak, L.; Johansen, H.K.; Sternberg, C.; Hoiby, N.; Molin, S. In situ growth rates and biofilm development of Pseudomonas aeruginosa populations in chronic lung infections. J. Bacteriol. 2008, 190, 2767–2776. [Google Scholar] [CrossRef] [PubMed]
- Feliziani, S.; Marvig, R.L.; Lujan, A.M.; Moyano, A.J.; di Rienzo, J.A.; Krogh Johansen, H.; Molin, S.; Smania, A.M. Coexistence and within-host evolution of diversified lineages of hypermutable Pseudomonas aeruginosa in long-term cystic fibrosis infections. PLoS Genet. 2014, 10, e1004651. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, K.; Christophersen, L.; Bjarnsholt, T.; Jensen, P.O.; Moser, C.; Hoiby, N. Anti-Pseudomonas aeruginosa IgY Antibodies Induce Specific Bacterial Aggregation and Internalization in Human Polymorphonuclear Neutrophils. Infect. Immun. 2015, 83, 2686–2693. [Google Scholar] [CrossRef] [PubMed]
- Caceres, S.M.; Malcolm, K.C.; Taylor-Cousar, J.L.; Nichols, D.P.; Saavedra, M.T.; Bratton, D.L.; Moskowitz, S.M.; Burns, J.L.; Nicka, J.A. Enhanced in vitro formation and antibiotic resistance of nonattached Pseudomonas aeruginosa aggregates through incorporation of neutrophil products. Antimicrob. Agents Chemother. 2014, 58, 6851–6860. [Google Scholar] [CrossRef] [PubMed]
- Haaber, J.; Cohn, M.T.; Frees, D.; Andersen, T.J.; Ingmer, H. Planktonic aggregates of Staphylococcus aureus protect against common antibiotics. PLoS ONE 2012, 7, e41075. [Google Scholar] [CrossRef]
- Van Gennip, M.; Christensen, L.D.; Alhede, M.; Qvortrup, K.; Jensen, P.O.; Hoiby, N.; Givskov, M.; Bjarnsholt, T. Interactions between polymorphonuclear leukocytes and Pseudomonas aeruginosa biofilms on silicone implants in vivo. Infect. Immun. 2012, 80, 2601–2607. [Google Scholar] [CrossRef] [PubMed]
- Bjarnsholt, T.; Alhede, M.; Eickhardt-Sorensen, S.R.; Moser, C.; Kuhl, M.; Jensen, P.Ø.; Høiby, N. The in vivo biofilm. Trends Microbiol. 2013, 21, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Haagensen, J.A.; Klausen, M.; Ernst, R.K.; Miller, S.I.; Folkesson, A.; Tolker-Nielsen, T.; Molin, S. Differentiation and distribution of colistin- and sodium dodecyl sulfate-tolerant cells in Pseudomonas aeruginosa biofilms. J. Bacteriol. 2007, 189, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Chmiel, J.F.; Aksamit, T.R.; Chotirmall, S.H.; Dasenbrook, E.C.; Elborn, J.S.; LiPuma, J.J.; Ranganathan, S.C.; Waters, V.J.; Ratjen, F.A. Antibiotic management of lung infections in cystic fibrosis. I. The microbiome, methicillin-resistant Staphylococcus aureus, gram-negative bacteria, and multiple infections. Ann. Am. Thorac. Soc. 2014, 11, 1120–1129. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Ridderberg, W.; Hansen, C.R.; Hoiby, N.; Jensen-Fangel, S.; Olesen, H.V.; Skov, M.; Lemming, L.E.; Pressler, T.; Johansen, H.K.; et al. Early treatment with inhaled antibiotics postpones next occurrence of Achromobacter in cystic fibrosis. J. Cyst. Fibros. 2013, 12, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Hoiby, N.; Frederiksen, B.; Pressler, T. Eradication of early Pseudomonas aeruginosa infection. J. Cyst. Fibros. 2005, 4 (Suppl. 2), 49–54. [Google Scholar] [CrossRef] [PubMed]
- Thomson, A.H. Human recombinant DNase in cystic fibrosis. J. Royal Soc. Med. 1995, 88 (Suppl. 25), 24–29. [Google Scholar]
- Shak, S.; Capon, D.J.; Hellmiss, R.; Marsters, S.A.; Baker, C.L. Recombinant human DNase I reduces the viscosity of cystic fibrosis sputum. Proc. Natl. Acad. Sci. USA 1990, 87, 9188–9192. [Google Scholar] [CrossRef] [PubMed]
- Chiang, W.C.; Nilsson, M.; Jensen, P.O.; Hoiby, N.; Nielsen, T.E.; Givskov, M.; Tolker-Nielsen, T. Extracellular DNA shields against aminoglycosides in Pseudomonas aeruginosa biofilms. Antimicrob. Agents Chemother. 2013, 57, 2352–2361. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, H.J.; Borowitz, D.S.; Christiansen, D.H.; Morris, E.M.; Nash, M.L.; Ramsey, B.W.; Rosenstein, B.J.; Smith, A.I.; Wohl, M.E. Effect of aerosolized recombinant human DNase on exacerbations of respiratory symptoms and on pulmonary function in patients with cystic fibrosis. N. Engl. J. Med. 1994, 331, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, B.; Pressler, T.; Hansen, A.; Koch, C.; Hoiby, N. Effect of aerosolized rhDNase (Pulmozyme) on pulmonary colonization in patients with cystic fibrosis. Acta Paediatr. 2006, 95, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Sehar, S.; Manefield, M. The roles of extracellular DNA in the structural integrity of extracellular polymeric substance and bacterial biofilm development. Environ. Microbiol. Rep. 2013, 5, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Okshevsky, M.; Meyer, R.L. The role of extracellular DNA in the establishment, maintenance and perpetuation of bacterial biofilms. Crit. Rev. Microbiol. 2015, 41, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Whitchurch, C.B.; Tolker-Nielsen, T.; Ragas, P.C.; Mattick, J.S. Extracellular DNA required forbacterial biofilm formation. Science 2002, 295, 1487. [Google Scholar] [CrossRef] [PubMed]
- Okshevsky, M.; Regina, V.R.; Meyer, R.L. Extracellular DNA as a target for biofilm control. Curr. Opin. Biotechnol. 2015, 33, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.; Chakrabarty, A.M. Role of alginate lyase in cell detachment of Pseudomonas aeruginosa. Appl. Environ. Microbiol. 1994, 60, 2355–2359. [Google Scholar] [PubMed]
- Nguyen, U.T.; Burrows, L.L. DNase I and proteinase K impair Listeria monocytogenes biofilmformation and induce dispersal of pre-existing biofilms. Int. J. Food Microbiol. 2014, 187, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Davies, E.V.; James, C.E.; Williams, D.; O’Brien, S.; Fothergill, J.L.; Haldenby, S.; Paterson, S.; Winstanley, C.; Brockhurst, M.A. Temperate phages both mediate and drive adaptive evolution in pathogen biofilms. Proc. Natl. Acad. Sci. USA 2016, 113, 8266–8271. [Google Scholar] [CrossRef] [PubMed]
| Species | Isolate | Strain Identification | Origin | Source |
|---|---|---|---|---|
| A. xylosoxidans | Type strain | LMG 1863T | Osaka, Japan | Ear |
| A. ruhlandii | Type strain | CCUG 38886T | Soil | |
| P. aeruginosa | PAO1 | DSMZ 19880 | ||
| A. xylosoxidans | Environmental isolate | CCUG 14603 | Lyon, France | Water |
| A. ruhlandii | CF1-a | J9557-07 | Aarhus, Denmark | CF clinical isolate |
| A. ruhlandii | CF1-b | J4616-09 | Aarhus, Denmark | CF clinical isolate |
| A. ruhlandii | CF1-c | J4951-12 | Aarhus, Denmark | CF clinical isolate |
| A. xylosoxidans | CF2-a | J19840-07 | Aarhus, Denmark | CF clinical isolate |
| A. xylosoxidans | CF2-b | J19373-08 | Aarhus, Denmark | CF clinical isolate |
| A. xylosoxidans | CF2-c | J8663-11 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF3-a | J10719-08 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF3-b | J13140-09 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF3-c | J13665-10 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF4-a | J14174-08 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF4-b | J10317-09 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF4-c | J1036-12 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF5-a | J15059-09 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF5-b | J23156-10 | Aarhus, Denmark | CF clinical isolate |
| A. insuavis | CF5-c | J5762-12 | Aarhus, Denmark | CF clinical isolate |
| A. xylosoxidans | CF6 | J20454-08 | Aarhus, Denmark | CF clinical isolate |
| A. xylosoxidans | CF7 | J15887-08 | Aarhus, Denmark | CF clinical isolate |
| A. xylosoxidans | CF8 | J15976-09 | Aarhus, Denmark | CF clinical isolate |
| A. ruhlandii | CF9 | J18469-02 | Aarhus, Denmark | CF clinical isolate |
| A. ruhlandii | CF10 | J10633-12 | Aarhus, Denmark | CF clinical isolate |
| A. xylosoxidans LMG 1863T | A. xylosoxidans CF2-b | A. insuavis CF4-b | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Antibiotic (µg/mL) | MIC | MBC | MBEC | MIC | MBC | MBEC | MIC | MBC | MBEC |
| Piperacillin/tazobactam | 1 | 8 | 1024 | 1 | 8 | 2048 | 1 | 2 | 2048 |
| + DNase I (200 µg/mL) | - | - | 1024 | - | - | 2048 | - | - | 512 |
| + Subtilisin (200 µg/mL) | - | - | 1024 | - | - | 2048 | - | - | 512 |
| + Alginate lyase (200 µg/mL) | - | - | 1024 | - | - | 2048 | - | - | 2048 |
| Ceftazidime | 8 | 16 | 2048 | 8 | 8 | 2048 | 4 | 8 | 512 |
| + DNase I (200 µg/mL) | - | - | 2048 | - | - | 2048 | - | - | 512 |
| + Subtilisin (200 µg/mL) | - | - | 2048 | - | - | 2048 | - | - | 512 |
| + Alginate lyase (200 µg/mL) | - | - | 2048 | - | - | 2048 | - | - | 512 |
| Tobramycin | >256 | 1024 | 1024 | 48 | 128 | 1024 | 24 | 64 | 1024 |
| + DNase I (200 µg/mL) | - | - | 1024 | - | - | 1024 | - | - | 1024 |
| + Subtilisin (200 µg/mL) | - | - | 1024 | - | - | 1024 | - | - | 1024 |
| + Alginate lyase (200 µg/mL) | - | - | 1024 | - | - | 1024 | - | - | 1024 |
| Colistin | 4 | 8 | 128 | 4 | 8 | 128 | 4 | 16 | 128 |
| + DNase I (200 µg/mL) | - | - | 128 | - | - | 32 | - | - | 64 |
| + Subtilisin (200 µg/mL) | - | - | 64 | - | - | 32 | - | - | 64 |
| + Alginate lyase (200 µg/mL) | - | - | 128 | - | - | 128 | - | - | 128 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nielsen, S.M.; Nørskov-Lauritsen, N.; Bjarnsholt, T.; Meyer, R.L. Achromobacter Species Isolated from Cystic Fibrosis Patients Reveal Distinctly Different Biofilm Morphotypes. Microorganisms 2016, 4, 33. https://doi.org/10.3390/microorganisms4030033
Nielsen SM, Nørskov-Lauritsen N, Bjarnsholt T, Meyer RL. Achromobacter Species Isolated from Cystic Fibrosis Patients Reveal Distinctly Different Biofilm Morphotypes. Microorganisms. 2016; 4(3):33. https://doi.org/10.3390/microorganisms4030033
Chicago/Turabian StyleNielsen, Signe M., Niels Nørskov-Lauritsen, Thomas Bjarnsholt, and Rikke L. Meyer. 2016. "Achromobacter Species Isolated from Cystic Fibrosis Patients Reveal Distinctly Different Biofilm Morphotypes" Microorganisms 4, no. 3: 33. https://doi.org/10.3390/microorganisms4030033
APA StyleNielsen, S. M., Nørskov-Lauritsen, N., Bjarnsholt, T., & Meyer, R. L. (2016). Achromobacter Species Isolated from Cystic Fibrosis Patients Reveal Distinctly Different Biofilm Morphotypes. Microorganisms, 4(3), 33. https://doi.org/10.3390/microorganisms4030033