Carbapenem Resistance in Gram-Negative Bacteria: The Not-So-Little Problem in the Little Red Dot
Abstract
:1. Introduction
2. Epidemiology
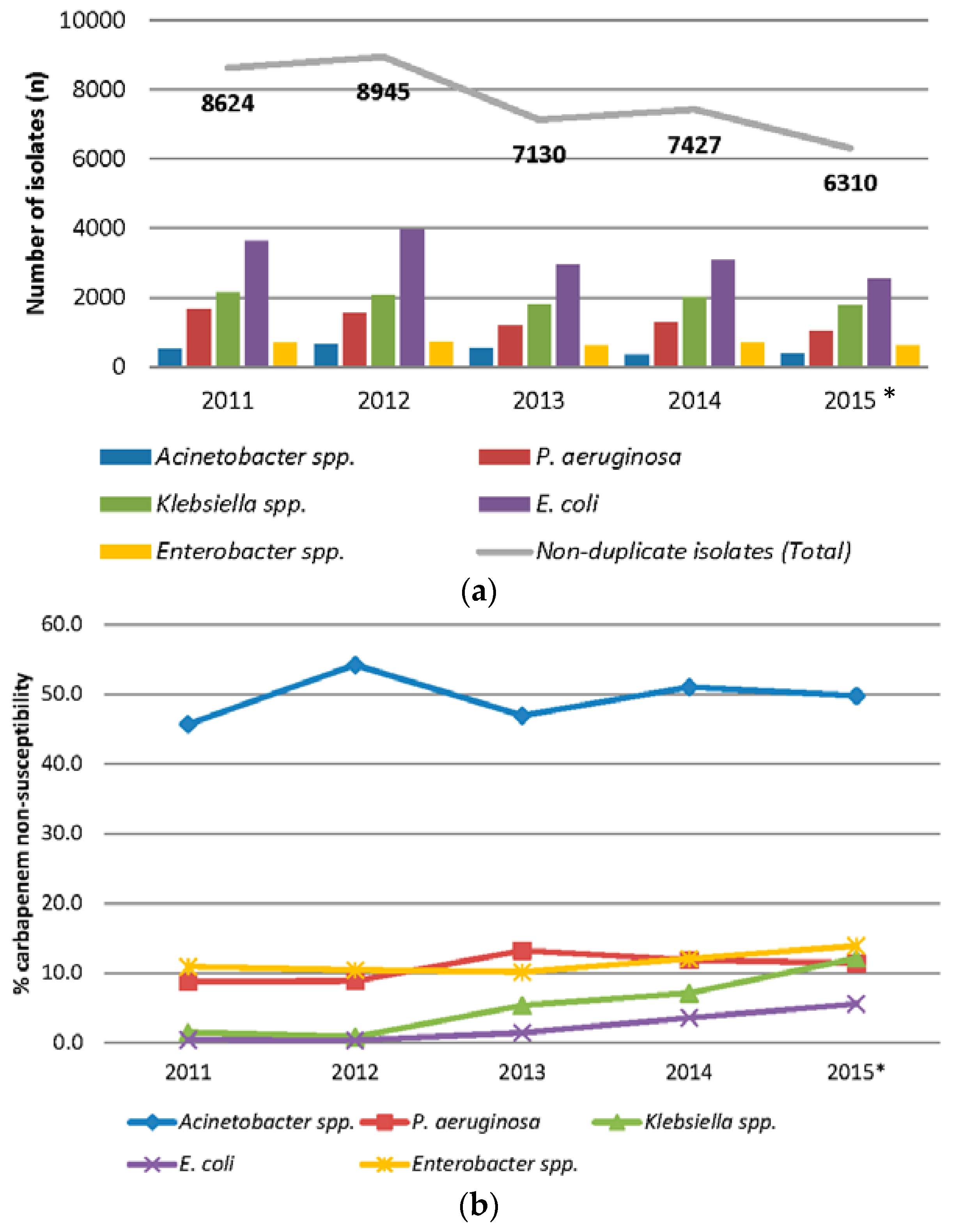
2.1. Prevalence
| Antibiotic | MIC50 (µg/mL) | MIC90 (µg /mL) | Range (µg/mL) | %S |
|---|---|---|---|---|
| Ertapenem | ≥32 | ≥32 | 1 - ≥32 | 0 |
| Imipenem | 16 | ≥32 | 0.5 - ≥32 | 8.1 |
| Meropenem | ≥32 | ≥32 | 0.5 - ≥32 | 6.6 |
| Doripenem | 16 | ≥32 | 0.5 - ≥32 | 5.1 |
| Amikacin | 4 | ≥128 | ≤1 - ≥128 | 75.7 |
| Aztreonam | ≥128 | ≥128 | ≤0.5 - ≥128 | 2.9 |
| Cefepime | ≥64 | ≥64 | ≤1 - ≥64 | 2.2 |
| Levofloxacin | 16 | ≥64 | ≤ 0.25 - ≥64 | 34.6 |
| Piperacillin/tazobactam | ≥256 | ≥256 | 8 - ≥256 | 1.5 |
| Polymyxin B | 1 | 16 | ≤0.25 - ≥32 | 83.1 |
| Tigecycline | 1 | 4 | ≤0.25 - 16 | 82.4 |
2.2. Mechanisms of Resistance
3. Control and Prevention
3.1. Infection Control and Active Surveillance
3.2. Antimicrobial Stewardship Programs (ASP) in Singapore
4. Treatment
Use of Antibiotic Combination Therapy against XDR-GNB
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Paterson, D.L.; Doi, Y. A step closer to extreme drug resistance (XDR) in Gram-negative bacilli. Clin. Infect. Dis. Off. Pub. Infect. Dis. Soc. Am. 2007, 45, 1179–1181. [Google Scholar] [CrossRef] [PubMed]
- Jean, S.S.; Hsueh, P.R. High burden of antimicrobial resistance in Asia. Int. J. Antimicrob. Agents 2011, 37, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Hawkey, P.M. Multidrug-resistant Gram-negative bacteria: A product of globalization. J. Hosp. Infect. 2015, 89, 241–247. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance. Available online: http://apps.who.int/iris/bitstream/10665/112642/1/9789241564748_eng.pdf (accessed on 30 November 2015).
- Molton, J.S.; Tambyah, P.A.; Ang, B.S.; Ling, M.L.; Fisher, D.A. The global spread of healthcare-associated multidrug-resistant bacteria: A perspective from Asia. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2013, 56, 1310–1318. [Google Scholar]
- Hsu, L.Y.; Tan, T.Y.; Tam, V.H.; Kwa, A.; Fisher, D.A.; Koh, T.H. Network for Antimicrobial Resistance Surveillance (Singapore). Surveillance and correlation of antibiotic prescription and resistance of Gram-negative bacteria in Singaporean hospitals. Antimicrob. Agents Chemother. 2010, 54, 1173–1178. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.Y.; Hsu, L.Y.; Koh, T.H.; Ng, L.S.; Tee, N.W.; Krishnan, P.; Lin, R.T.; Jureen, R. Antibiotic resistance in Gram-negative bacilli: A Singapore perspective. Ann. Acad Med. Singap. 2008, 37, 819–825. [Google Scholar] [PubMed]
- Koh, T.H.; Cao, D.; Shan, Q.Y.; Bacon, A.; Hsu, L.Y.; Ooi, E.E. Acquired carbapenemases in enterobactericeae in Singapore, 1996–2012. Pathology 2013, 45, 600–603. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.; Lim, T.P.; Cai, Y.; Koh, T.H.; Tan, T.T.; Koh, T.H.; Kwa, A.L. Molecular epidemiology of carbapenem-resistant Gram negative bacteria in Singapore. Antimicrob. Agents Chemother. 2016. submitted. [Google Scholar]
- Krishnan, P.U.; La, M.V.; Jureen, R.; Koh, M.; Teo, J.; Lin, R. Changing trends in carbapenemase-producing Enterobacteriaceae in Singapore from 2010 to 2014 (August). In Proceedings of the 25th European Congress of Clinical Microbiology and Infectious Diseases, Copenhagen, Denmark, 25–28 April 2015.
- Koh, T.H.; Ko, K.; Jureen, R.; Deepak, R.N.; Tee, N.W.; Tan, T.Y.; Tay, M.R.; Lee, V.J.; Barkham, T.M. High counts of carbapenemase-producing Enterobacteriaceae in hospital sewage. Infect. Control. Hosp. Epidemiol. 2015, 36, 619–621. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.; Lee, S.; Leck, H.; Chia, S.; Wang, R.; Cai, Y.; Lim, T.P.; Koh, T.H.; Tan, T.T.; Kwa, A.L. Molecular characterization of carbapenem-resistant Klebsiella species in Singapore. In Proceedings of the 25th European Congress of Clinical Microbiology and Infectious Diseases, Copenhagen, Denmark, 25–28 April 2015.
- Alekshun, M.N.; Levy, S.B. Molecular mechanisms of antibacterial multidrug resistance. Cell 2007, 128, 1037–1050. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P.; Poirel, L. The difficult-to-control spread of carbapenemase producers among Enterobacteriaceae worldwide. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2014, 20, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P.; Naas, T.; Poirel, L. Global spread of carbapenemase-producing Enterobacteriaceae. Emerg. Infect. Dis. 2011, 17, 1791–1798. [Google Scholar] [CrossRef] [PubMed]
- Balm, M.N.; Ngan, G.; Jureen, R.; Lin, R.T.; Teo, J.W. Oxa-181-producing Klebsiella pneumoniae establishing in Singapore. BMC Infect. Dis. 2013. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.H.; Babini, G.S.; Woodford, N.; Sng, L.H.; Hall, L.M.; Livermore, D.M. Carbapenem-hydrolysing IMP-1 beta-lactamase in Klebsiella pneumoniae from Singapore. Lancet 1999. [Google Scholar] [CrossRef]
- Koh, T.H.; Khoo, C.T.; Wijaya, L.; Leong, H.N.; Lo, Y.L.; Lim, L.C.; Koh, T.Y. Global spread of New Delhi metallo-beta-lactamase 1. Lancet Infect. Dis. 2010. [Google Scholar] [CrossRef]
- Balm, M.N.; Ngan, G.; Jureen, R.; Lin, R.T.; Teo, J. Molecular characterization of newly emerged blaKPC-2-producing Klebsiella pneumoniae in Singapore. J. Clin. Microbiol. 2012, 50, 475–476. [Google Scholar] [CrossRef] [PubMed]
- Marimuthu, K.; Teo, J.W.; Fong, P.B.; Chin, J.O.; Qi, K.J.; Boon, D.L.; Ping, A.C.; Krishnan, P.; Peng, B.A. First report of emergence of OXA-48 carbapenemase-producing Enterobacteriaceae in Singapore: Proactive or reactive infection control strategy? Am. J. Infect. Control. 2014, 42, 577–578. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.W.; La, M.V.; Krishnan, P.; Ang, B.; Jureen, R.; Lin, R.T. Enterobacter cloacae producing an uncommon class a carbapenemase, IMI-1, from Singapore. J. Med. Microbiol. 2013, 62, 1086–1088. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.W.; Kurup, A.; Lin, R.T.; Hsien, K.T. Emergence of clinical Klebsiella pneumoniae producing OXA-232 carbapenemase in Singapore. New Microbes New Infect. 2013, 1, 13–15. [Google Scholar] [CrossRef] [PubMed]
- Balm, M.N.; La, M.V.; Krishnan, P.; Jureen, R.; Lin, R.T.; Teo, J.W. Emergence of Klebsiella pneumoniae co-producing NDM-type and OXA-181 carbapenemases. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2013, 19, E421–E423. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.H.; Sng, L.H.; Wang, G.C.; Hsu, L.Y.; Zhao, Y. IMP-4 and oxa beta-lactamases in Acinetobacter baumannii from Singapore. J. Antimicrob. Chemother. 2007, 59, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.H.; Sng, L.H.; Wang, G.C.; Hsu, L.Y.; Zhao, Y. Carbapenemase and efflux pump genes in acinetobacter calcoaceticus Acinetobacter baumannii complex strains from Singapore. J. Antimicrob. Chemother. 2007, 60, 1173–1174. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.H.; Wang, G.C.; Sng, L.H. Clonal spread of IMP-1-producing Pseudomonas aeruginosa in two hospitals in Singapore. J. Clin. Microbiol. 2004, 42, 5378–5380. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.H.; Khoo, C.T.; Tan, T.T.; Arshad, M.A.; Ang, L.P.; Lau, L.J.; Hsu, L.Y.; Ooi, E.E. Multilocus sequence types of carbapenem-resistant Pseudomonas aeruginosa in Singapore carrying metallo-beta-lactamase genes, including the novel blaIMP-26 gene. J. Clin. Microbiol. 2010, 48, 2563–2564. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.H.; Wang, G.C.; Sng, L.H. IMP-1 and a novel metallo-beta-lactamase, VIM-6, in fluorescent pseudomonads isolated in Singapore. Antimicrob. Agents Chemother. 2004, 48, 2334–2336. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.H. Acquired Carbapenemases in Gram-Negative Bacilli in Singapore. Ph.D. Thesis, National University of Singapore, Singapore, 2013. [Google Scholar]
- Park, Y.K.; Lee, G.H.; Baek, J.Y.; Chung, D.R.; Peck, K.R.; Song, J.H.; Ko, K.S. A single clone of Acinetobacter baumannii, ST22, is responsible for high antimicrobial resistance rates of Acinetobacter spp. Isolates that cause bacteremia and urinary tract infections in Korea. Microb. Drug Resist. 2010, 16, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Samuelsen, O.; Toleman, M.A.; Sundsfjord, A.; Rydberg, J.; Leegaard, T.M.; Walder, M.; Lia, A.; Ranheim, T.E.; Rajendra, Y.; Hermansen, N.O.; et al. Molecular epidemiology of metallo-beta-lactamase-producing Pseudomonas aeruginosa isolates from norway and Sweden shows import of international clones and local clonal expansion. Antimicrob. Agents Chemother. 2010, 54, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.; Ngan, G.; Balm, M.; Jureen, R.; Krishnan, P.; Lin, R. Molecular characterization of NDM-1 producing Enterobacteriaceae isolates in Singapore hospitals. West. Pac. Surveill. Response J. 2012, 3, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Abate, G.; Koh, T.H.; Gardner, M.; Siu, L.K. Clinical and bacteriological characteristics of Klebsiella pneumoniae causing liver abscess with less frequently observed multi-locus sequences type, ST163, from Singapore and Missouri, US. J. Microbiol. Immunol. Infect. 2012, 45, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Wang, Y.; Walsh, T.R.; Yi, L.X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2015. [Google Scholar] [CrossRef]
- Lim, T.P.; Ong, R.T.; Hon, P.Y.; Hawkey, J.; Holt, K.E.; Koh, T.H.; Leong, M.L.; Teo, J.Q.; Tan, T.Y.; Ng, M.M.; et al. Multiple genetic mutations associated with polymyxin resistance in Acinetobacter baumannii. Antimicrob. Agents Chemother. 2015, 59, 7899–7902. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.; Ong Rick, T.; Koh, T.H.; Lim, T.P.; Kwa, A.L. Multidrug-resistant Escherichia coli co-harbouring blaKPC-2 and MCR-1 in Singapore. Lancet 2016. under review. [Google Scholar]
- Ministry of Health. Guidelines for Control and Prevention of Multi-Drug Resistant Organisms (MDROs) in Healthcare Facilities. Available online: https://www.moh.gov.sg/content/dam/moh_web/Publications/Guidelines/Infection%20Control%20guidelines/GUIDELINES%20FOR%20CONTROL%20AND%20PREVENTION%20OF%20MULTI-DRUG%20RESISTANT%20ORGANISMS%20(MDROS)%20IN%20HEALTHCARE%20FACILITIES%20-%20Nov%202013.pdf (accessed on 30 November 2015).
- Nordmann, P.; Poirel, L. Strategies for identification of carbapenemase-producing Enterobacteriaceae. J. Antimicrob. Chemother. 2013, 68, 487–489. [Google Scholar] [CrossRef] [PubMed]
- Hammoudi, D.; Moubareck, C.A.; Sarkis, D.K. How to detect carbapenemase producers? A literature review of phenotypic and molecular methods. J. Microbiol. Methods 2014, 107, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P.; Gniadkowski, M.; Giske, C.G.; Poirel, L.; Woodford, N.; Miriagou, V.; European Network on Carbapenemases. Identification and screening of carbapenemase-producing Enterobacteriaceae. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2012, 18, 432–438. [Google Scholar]
- Hrabak, J.; Chudackova, E.; Papagiannitsis, C.C. Detection of carbapenemases in Enterobacteriaceae: A challenge for diagnostic microbiological laboratories. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2014, 20, 839–853. [Google Scholar] [CrossRef] [PubMed]
- Nordmann, P.; Poirel, L.; Dortet, L. Rapid detection of carbapenemase-producing Enterobacteriaceae. Emerg. Infect. Dis. 2012, 18, 1503–1507. [Google Scholar] [CrossRef] [PubMed]
- Tijet, N.; Boyd, D.; Patel, S.N.; Mulvey, M.R.; Melano, R.G. Evaluation of the carba np test for rapid detection of carbapenemase-producing Enterobacteriaceae and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2013, 57, 4578–4580. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Testing: Seventeenth Informational Supplement M100-S20; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2010; Volume 30. [Google Scholar]
- Girlich, D.; Poirel, L.; Nordmann, P. Value of the modified hodge test for detection of emerging carbapenemases in Enterobacteriaceae. J. Clin. Microbiol. 2012, 50, 477–479. [Google Scholar] [CrossRef] [PubMed]
- Miriagou, V.; Cornaglia, G.; Edelstein, M.; Galani, I.; Giske, C.G.; Gniadkowski, M.; Malamou-Lada, E.; Martinez-Martinez, L.; Navarro, F.; Nordmann, P.; et al. Acquired carbapenemases in gram-negative bacterial pathogens: Detection and surveillance issues. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2010, 16, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Pasteran, F.; Mendez, T.; Guerriero, L.; Rapoport, M.; Corso, A. Sensitive screening tests for suspected class a carbapenemase production in species of Enterobacteriaceae. J. Clin. Microbiol. 2009, 47, 1631–1639. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.; Kwa, A.L.; Loh, J.; Chlebicki, M.P.; Lee, W. The effect of a whole-system approach in an antimicrobial stewardship programme at the Singapore general hospital. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Lew, K.Y.; Ng, T.M.; Tan, M.; Tan, S.H.; Lew, E.L.; Ling, L.M.; Ang, B.; Lye, D.; Teng, C.B. Safety and clinical outcomes of carbapenem de-escalation as part of an antimicrobial stewardship programme in an esbl-endemic setting. J. Antimicrob. Chemother. 2015, 70, 1219–1225. [Google Scholar] [CrossRef] [PubMed]
- Yeo, C.L.; Wu, J.E.; Chung, G.W.; Chan, D.S.; Chen, H.H.; Hsu, L.Y. Antimicrobial stewardship auditing of patients reviewed by infectious diseases physicians in a tertiary university hospital. Antimicrob. Resist. Infect. Control. 2013. [Google Scholar] [CrossRef] [PubMed]
- Seah, X.F.; Ong, Y.L.; Tan, S.W.; Krishnaswamy, G.; Chong, C.Y.; Tan, N.W.; Thoon, K.C. Impact of an antimicrobial stewardship program on the use of carbapenems in a tertiary women's and children's hospital, Singapore. Pharmacotherapy 2014, 34, 1141–1150. [Google Scholar] [CrossRef] [PubMed]
- Chow, A.L.; Lye, D.C.; Arah, O.A. Patient and physician predictors of patient receipt of therapies recommended by a computerized decision support system when initially prescribed broad-spectrum antibiotics: A cohort study. J. Am. Med. Inform. Assoc. 2015. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.B.; Ng, T.M.; Tan, M.W.; Tan, S.H.; Tay, M.; Lim, S.F.; Ling, L.M.; Ang, B.S.; Lye, D.C. Safety and effectiveness of improving carbapenem use via prospective review and feedback in a multidisciplinary antimicrobial stewardship programme. Ann. Acad. Med. Singap. 2015, 44, 19–25. [Google Scholar] [PubMed]
- Liew, Y.X.; Lee, W.; Loh, J.C.; Cai, Y.; Tang, S.S.; Lim, C.L.; Teo, J.; Ong, R.W.; Kwa, A.L.; Chlebicki, M.P. Impact of an antimicrobial stewardship programme on patient safety in Singapore general hospital. Int. J. Antimicrob. Agents 2012, 40, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Liew, Y.X.; Lee, W.; Tay, D.; Tang, S.S.; Chua, N.G.; Zhou, Y.; Kwa, A.L.; Chlebicki, M.P. Prospective audit and feedback in antimicrobial stewardship: Is there value in early reviewing within 48 h of antibiotic prescription? Int. J. Antimicrob. Agents 2015, 45, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Yeo, C.L.; Chan, D.S.; Earnest, A.; Wu, T.S.; Yeoh, S.F.; Lim, R.; Jureen, R.; Fisher, D.; Hsu, L.Y. Prospective audit and feedback on antibiotic prescription in an adult hematology-oncology unit in Singapore. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Lee, W.; Kwa, A.L. Polymyxin b versus colistin: An update. Expert Rev. Anti Infect. Ther. 2015, 13, 1481–1497. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Rayner, C.R.; Nation, R.L.; Owen, R.J.; Spelman, D.; Tan, K.E.; Liolios, L. Heteroresistance to colistin in multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2006, 50, 2946–2950. [Google Scholar] [CrossRef] [PubMed]
- Zavascki, A.P.; Bulitta, J.B.; Landersdorfer, C.B. Combination therapy for carbapenem-resistant gram-negative bacteria. Expert Rev. Anti Infect. Ther. 2013, 11, 1333–1353. [Google Scholar] [CrossRef] [PubMed]
- Morrill, H.J.; Pogue, J.M.; Kaye, K.S.; LaPlante, K.L. Treatment options for carbapenem-resistant Enterobacteriaceae infections. Open Forum Infect. Dis. 2015. [Google Scholar] [CrossRef] [PubMed]
- Falagas, M.E.; Lourida, P.; Poulikakos, P.; Rafailidis, P.I.; Tansarli, G.S. Antibiotic treatment of infections due to carbapenem-resistant Enterobacteriaceae: Systematic evaluation of the available evidence. Antimicrob. Agents Chemother. 2014, 58, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Durante-Mangoni, E.; Signoriello, G.; Andini, R.; Mattei, A.; de Cristoforo, M.; Murino, P.; Bassetti, M.; Malacarne, P.; Petrosillo, N.; Galdieri, N.; et al. Colistin and rifampicin compared with colistin alone for the treatment of serious infections due to extensively drug-resistant Acinetobacter baumannii: A multicenter, randomized clinical trial. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2013, 57, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.P.; Tan, T.Y.; Lee, W.; Sasikala, S.; Tan, T.T.; Hsu, L.Y.; Kwa, A.L. In vitro activity of various combinations of antimicrobials against carbapenem-resistant Acinetobacter species in Singapore. J. Antibiot. (Tokyo) 2009, 62, 675–679. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.P.; Lee, W.; Tan, T.Y.; Sasikala, S.; Teo, J.; Hsu, L.Y.; Tan, T.T.; Syahidah, N.; Kwa, A.L. Effective antibiotics in combination against extreme drug-resistant Pseudomonas aeruginosa with decreased susceptibility to polymyxin b. PLoS ONE 2011, 6, e28177. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.P.; Tan, T.Y.; Lee, W.; Sasikala, S.; Tan, T.T.; Hsu, L.Y.; Kwa, A.L. In-vitro activity of polymyxin B, rifampicin, tigecycline alone and in combination against carbapenem-resistant Acinetobacter baumannii in Singapore. PLoS ONE 2011, 6, e18485. [Google Scholar] [CrossRef] [PubMed]
- Teo, J.; Lim, T.P.; Hsu, L.Y.; Tan, T.Y.; Sasikala, S.; Hon, P.Y.; Kwa, A.L.; Apisarnthanarak, A. Extensively drug-resistant Acinetobacter baumannii in a thai hospital: A molecular epidemiologic analysis and identification of bactericidal polymyxin B-based combinations. Antimicrob. Resist. Infect. Control. 2015. [Google Scholar] [CrossRef] [PubMed]
- Chua, N.G.; Lim, T.P.; Lee, W.; Liew, Y.; Teo, J.; Cai, Y.; Tan, T.T.; Kurup, A.; Kwa, A.L. Combination bactericidal testing-guided therapy (MCBT-GT) is effective in the treatment of extensively drug-resistant (XDR) Gram-negative bacteria (GNB) infections. In Proceedings of the Interscience Conference of Antimicrobial Agents and Chemotherapy, Washington, DC, USA, 5–9 September 2014.
- Lim, T.P.; Cai, Y.; Hong, Y.; Chan, E.C.; Suranthran, S.; Teo, J.Q.; Lee, W.H.; Tan, T.Y.; Hsu, L.Y.; Koh, T.H.; et al. In vitro pharmacodynamics of various antibiotics in combination against extensively drug-resistant Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2015, 59, 2515–2524. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Teo, J.; Lim, T.P.; Leck, H.; Lee, S.; Chia, S.; Lee, W.; Tan, T.T.; Koh, T.H.; Kwa, A.L. Correlating molecular mechanisms of resistance of extensively-drug resistant gram negative bacteria with in vitro activity of combination antibiotics & patient outcomes. In Proceedings of the Interscience Conference of Antimicrobial Agents and Chemotherapy/International Congress of Chemotherapy and Infection, San Diego, CA, USA, 17–21 September 2015.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teo, J.Q.M.; Cai, Y.; Lim, T.-P.; Tan, T.T.; Kwa, A.L.-H. Carbapenem Resistance in Gram-Negative Bacteria: The Not-So-Little Problem in the Little Red Dot. Microorganisms 2016, 4, 13. https://doi.org/10.3390/microorganisms4010013
Teo JQM, Cai Y, Lim T-P, Tan TT, Kwa AL-H. Carbapenem Resistance in Gram-Negative Bacteria: The Not-So-Little Problem in the Little Red Dot. Microorganisms. 2016; 4(1):13. https://doi.org/10.3390/microorganisms4010013
Chicago/Turabian StyleTeo, Jocelyn Qi Min, Yiying Cai, Tze-Peng Lim, Thuan Tong Tan, and Andrea Lay-Hoon Kwa. 2016. "Carbapenem Resistance in Gram-Negative Bacteria: The Not-So-Little Problem in the Little Red Dot" Microorganisms 4, no. 1: 13. https://doi.org/10.3390/microorganisms4010013
APA StyleTeo, J. Q. M., Cai, Y., Lim, T.-P., Tan, T. T., & Kwa, A. L.-H. (2016). Carbapenem Resistance in Gram-Negative Bacteria: The Not-So-Little Problem in the Little Red Dot. Microorganisms, 4(1), 13. https://doi.org/10.3390/microorganisms4010013






